Of Oestrogens and Sperm: A Review of the Roles of Oestrogens and Oestrogen Receptors in Male Reproduction
Abstract
:1. Introduction
2. Oestrogen Receptors
2.1. Signalling Pathways
2.1.1. Genomic Pathway
2.1.2. Non-Genomic Pathway
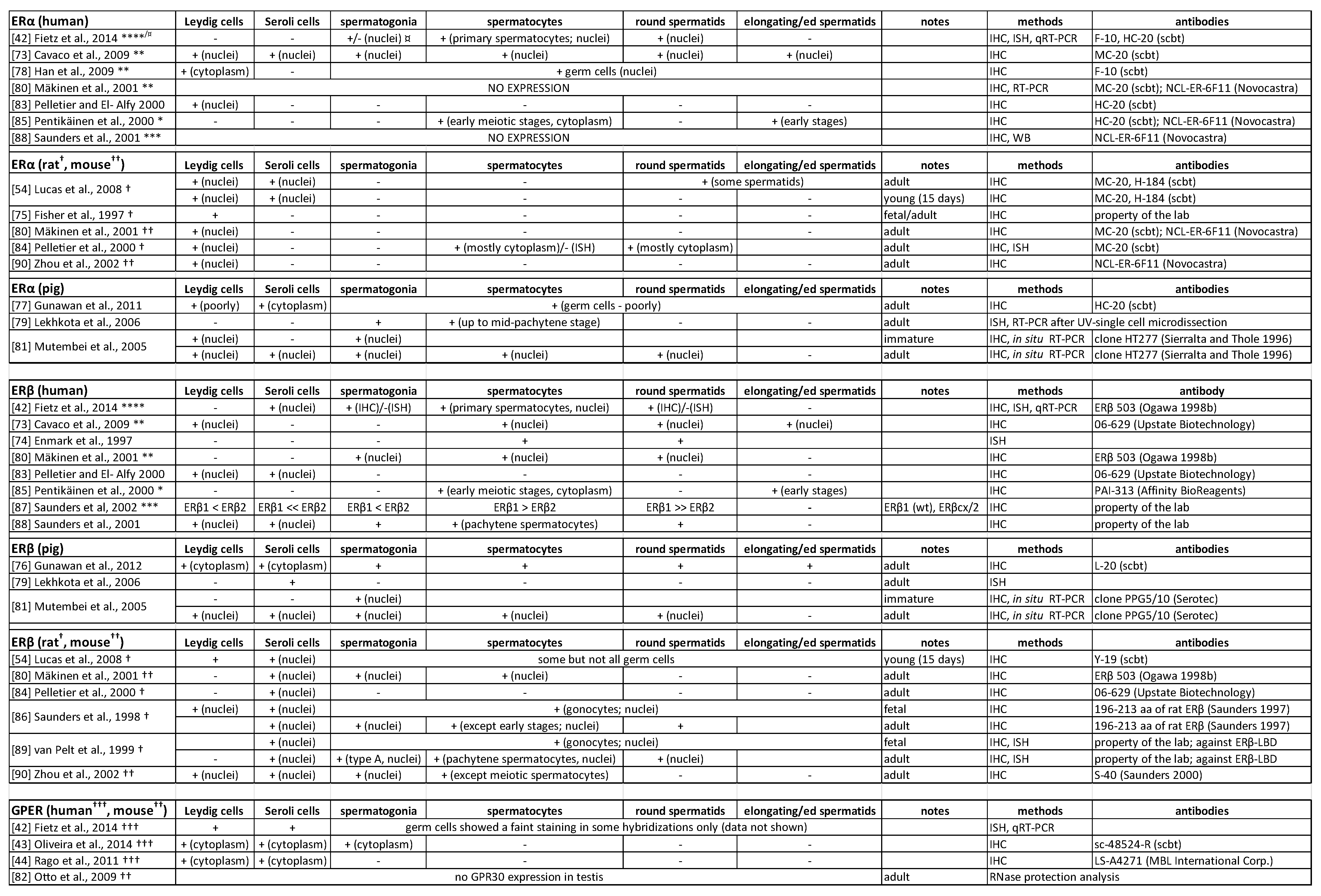
2.2. Oestrogen Receptors in the Testis
2.3. Oestrogen Receptors in Sperm
2.4. Genetically Manipulated Mouse Models
2.4.1. Aromatase Knockout
2.4.2. ERα Gene-Manipulated Models
2.4.3. ERβ Knockout
2.4.4. GPER Knockout
3. The Effect of Oestrogens on Spermatogenesis and Sperm
4. Interaction of Oestrogen-Like Compounds with ERs
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| ArKO | aromatase knockout |
| DES | diethylstilbestrol |
| E2 | 17β-estradiol |
| EDs | endocrine disruptors |
| ERE | oestrogen response element |
| ERs | oestrogen receptors |
| ERαKO | ER alpha knock-out |
| ERβKO | ER beta knock-out |
| ESR1/ERα | oestrogen receptor 1/alpha |
| ESR2/ERβ | oestrogen receptor 2/beta |
| GPER/GPR30 | G protein-coupled oestrogen receptor 1 |
| ZEA | Zearalenone |
References
- Hess, R.A. Estrogen in the adult male reproductive tract: A review. Reprod. Biol. Endocrinol. 2003, 1, 52. [Google Scholar] [CrossRef] [PubMed]
- Free, M.J.; Jaffe, R.A. Collection of rete testis fluid from rats without previous efferent duct ligation. Biol. Reprod. 1979, 20, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Hess, R.A.; Bunick, D.; Bahr, J.M. Sperm, a source of estrogen. Environ. Heal. Perspect. 1995, 103, 59–62. [Google Scholar] [CrossRef]
- Faccio, L.; Da Silva, A.S.; Tonin, A.A.; Franca, R.T.; Gressler, L.T.; Copetti, M.M.; Oliveira, C.B.; Sangoi, M.B.; Moresco, R.N.; Bottari, N.B.; et al. Serum levels of LH, FSH, estradiol and progesterone in female rats experimentally infected by trypanosoma evansi. Exp. Parasitol. 2013, 135, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Kelch, R.P.; Jenner, M.R.; Weinstein, R.; Kaplan, S.L.; Grumbach, M.M. Estradiol and testosterone secretion by human, simian, and canine testes, in males with hypogonadism and in male pseudohermaphrodites with the feminizing testes syndrome. J. Clin. Investig. 1972, 51, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Abraham, G.E.; Odell, W.D.; Swerdloff, R.S.; Hopper, K. Simultaneous radioimmunoassay of plasma FSH, LH, progesterone, 17-hydroxyprogesterone, and estradiol-17 β during the menstrual cycle. J. Clin. Endocrinol. Metab. 1972, 34, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Lubahn, D.B.; Moyer, J.S.; Golding, T.S.; Couse, J.F.; Korach, K.S.; Smithies, O. Alteration of reproductive function but not prenatal sexual development after insertional disruption of the mouse estrogen receptor gene. Proc. Natl. Acad. Sci. USA 1993, 90, 11162–11166. [Google Scholar] [CrossRef] [PubMed]
- Cooper, T.G.; Noonan, E.; von Eckardstein, S.; Auger, J.; Baker, H.W.; Behre, H.M.; Haugen, T.B.; Kruger, T.; Wang, C.; Mbizvo, M.T.; et al. World health organization reference values for human semen characteristics. Hum. Reprod. Update 2010, 16, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Pflieger-Bruss, S.; Schuppe, H.C.; Schill, W.B. The male reproductive system and its susceptibility to endocrine disrupting chemicals. Andrologia 2004, 36, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Schagdarsurengin, U.; Western, P.; Steger, K.; Meinhardt, A. Developmental origins of male subfertility: Role of infection, inflammation, and environmental factors. Semin. Immunopathol. 2016, 38, 765–781. [Google Scholar] [CrossRef] [PubMed]
- Contractor, R.G.; Foran, C.M.; Li, S.; Willett, K.L. Evidence of gender-and tissue-specific promoter methylation and the potential for ethinylestradiol-induced changes in japanese medaka (Oryzias latipes) estrogen receptor and aromatase genes. J. Toxicol. Environ. Health. Part A 2004, 67, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Mirbahai, L.; Chipman, J.K. Epigenetic memory of environmental organisms: A reflection of lifetime stressor exposures. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2014, 764–765, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Schagdarsurengin, U.; Steger, K. Epigenetics in male reproduction: Effect of paternal diet on sperm quality and offspring health. Nat. Rev. Urol. 2016, 13, 584–595. [Google Scholar] [CrossRef] [PubMed]
- Aschim, E.L.; Saether, T.; Wiger, R.; Grotmol, T.; Haugen, T.B. Differential distribution of splice variants of estrogen receptor β in human testicular cells suggests specific functions in spermatogenesis. J. Steroid Biochem. Mol. Biol. 2004, 92, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Flouriot, G.; Brand, H.; Denger, S.; Metivier, R.; Kos, M.; Reid, G.; Sonntag-Buck, V.; Gannon, F. Identification of a new isoform of the human estrogen receptor-α (HER-α) that is encoded by distinct transcripts and that is able to repress HER-α activation function 1. EMBO J. 2000, 19, 4688–4700. [Google Scholar] [CrossRef] [PubMed]
- Friend, K.E.; Resnick, E.M.; Ang, L.W.; Shupnik, M.A. Specific modulation of estrogen receptor mRNA isoforms in rat pituitary throughout the estrous cycle and in response to steroid hormones. Mol. Cell. Endocrinol. 1997, 131, 147–155. [Google Scholar] [CrossRef]
- Fuqua, S.A.; Fitzgerald, S.D.; Chamness, G.C.; Tandon, A.K.; McDonnell, D.P.; Nawaz, Z.; O’Malley, B.W.; McGuire, W.L. Variant human breast tumor estrogen receptor with constitutive transcriptional activity. Cancer Res. 1991, 51, 105–109. [Google Scholar] [PubMed]
- Hanstein, B.; Liu, H.; Yancisin, M.C.; Brown, M. Functional analysis of a novel estrogen receptor-β isoform. Mol. Endocrinol. 1999, 13, 129–137. [Google Scholar] [PubMed]
- Inoue, S.; Ogawa, S.; Horie, K.; Hoshino, S.; Goto, W.; Hosoi, T.; Tsutsumi, O.; Muramatsu, M.; Ouchi, Y. An estrogen receptor β isoform that lacks exon 5 has dominant negative activity on both ERα and ERβ. Biochem. Biophys. Res. Commun. 2000, 279, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Lambard, S.; Galeraud-Denis, I.; Saunders, P.T.; Carreau, S. Human immature germ cells and ejaculated spermatozoa contain aromatase and oestrogen receptors. J. Mol. Endocrinol. 2004, 32, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, S.; Kalita, K.; Kaczmarek, L. Estrogen receptor β. Potential functional significance of a variety of mRNA isoforms. FEBS Lett. 2002, 524, 1–5. [Google Scholar] [CrossRef]
- Lu, B.; Leygue, E.; Dotzlaw, H.; Murphy, L.J.; Murphy, L.C. Functional characteristics of a novel murine estrogen receptor-β isoform, estrogen receptor-β 2. J. Mol. Endocrinol. 2000, 25, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Leygue, E.; Dotzlaw, H.; Murphy, L.J.; Murphy, L.C.; Watson, P.H. Estrogen receptor-β mRNA variants in human and murine tissues. Mol. Cell. Endocrinol. 1998, 138, 199–203. [Google Scholar] [CrossRef]
- Ogawa, S.; Inoue, S.; Watanabe, T.; Orimo, A.; Hosoi, T.; Ouchi, Y.; Muramatsu, M. Molecular cloning and characterization of human estrogen receptor βCX: A potential inhibitor ofestrogen action in human. Nucleic Acids Res. 1998, 26, 3505–3512. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Lu, B.; Leygue, E.; Murphy, L.C. Putative functional characteristics of human estrogen receptor-β isoforms. J. Mol. Endocrinol. 2003, 30, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Petersen, D.N.; Tkalcevic, G.T.; Koza-Taylor, P.H.; Turi, T.G.; Brown, T.A. Identification of estrogen receptor β2, a functional variant of estrogen receptor β expressed in normal rat tissues. Endocrinology 1998, 139, 1082–1092. [Google Scholar] [CrossRef] [PubMed]
- Price, R.H., Jr.; Lorenzon, N.; Handa, R.J. Differential expression of estrogen receptor β splice variants in rat brain: Identification and characterization of a novel variant missing exon 4. Brain Res. Mol. Brain Res. 2000, 80, 260–268. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, X.; Shen, P.; Loggie, B.W.; Chang, Y.; Deuel, T.F. A variant of estrogen receptor-α, HER-α36: Transduction of estrogen- and antiestrogen-dependent membrane-initiated mitogenic signaling. Proc. Natl. Acad. Sci. USA 2006, 103, 9063–9068. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Brusco, L.; Fuqua, S.A. Estrogen receptor mutations and changes in downstream gene expression and signaling. Clin. Cancer Res. 2010, 16, 2702–2708. [Google Scholar] [CrossRef] [PubMed]
- Hirata, S.; Shoda, T.; Kato, J.; Hoshi, K. Isoform/variant mRNAs for sex steroid hormone receptors in humans. Trends. Endocrinol. Metab. 2003, 14, 124–129. [Google Scholar] [CrossRef]
- Luconi, M.; Forti, G.; Baldi, E. Genomic and nongenomic effects of estrogens: Molecular mechanisms of action and clinical implications for male reproduction. J. Steroid Biochem. Mol. Biol. 2002, 80, 369–381. [Google Scholar] [CrossRef]
- Nilsson, S.; Makela, S.; Treuter, E.; Tujague, M.; Thomsen, J.; Andersson, G.; Enmark, E.; Pettersson, K.; Warner, M.; Gustafsson, J.A. Mechanisms of estrogen action. Physiol. Rev. 2001, 81, 1535–1565. [Google Scholar] [PubMed]
- Paterni, I.; Granchi, C.; Katzenellenbogen, J.A.; Minutolo, F. Estrogen receptors α (ERα) and β (ERβ): Subtype-selective ligands and clinical potential. Steroids 2014, 90, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.; Quinn, J.; Pang, Y.; Graeber, C.; Shaw, S.; Dong, J.; Thomas, P. Activation of the novel estrogen receptor G protein-coupled receptor 30 (GPR30) at the plasma membrane. Endocrinology 2007, 148, 3236–3245. [Google Scholar] [CrossRef] [PubMed]
- Funakoshi, T.; Yanai, A.; Shinoda, K.; Kawano, M.M.; Mizukami, Y. G protein-coupled receptor 30 is an estrogen receptor in the plasma membrane. Biochem. Biophys. Res. Commun. 2006, 346, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Revankar, C.M.; Cimino, D.F.; Sklar, L.A.; Arterburn, J.B.; Prossnitz, E.R. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 2005, 307, 1625–1630. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, H.; Matsuda, K.; Hosokawa, K.; Nishi, M.; Morris, J.F.; Prossnitz, E.R.; Kawata, M. Expression of G protein-coupled receptor-30, a g protein-coupled membrane estrogen receptor, in oxytocin neurons of the rat paraventricular and supraoptic nuclei. Endocrinology 2007, 148, 5842–5850. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.J.; Graeber, C.T.; Quinn, J.A.; Resnick, M.B.; Giri, D.; DeLellis, R.A.; Steinhoff, M.M.; Sabo, E. Distribution of GPR30, a seven membrane-spanning estrogen receptor, in primary breast cancer and its association with clinicopathologic determinants of tumor progression. Clin. Cancer Res. 2006, 12, 6359–6366. [Google Scholar] [CrossRef] [PubMed]
- Hazell, G.G.; Yao, S.T.; Roper, J.A.; Prossnitz, E.R.; O’Carroll, A.M.; Lolait, S.J. Localisation of GPR30, a novel G protein-coupled oestrogen receptor, suggests multiple functions in rodent brain and peripheral tissues. J. Endocrinol. 2009, 202, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Plante, B.J.; Lessey, B.A.; Taylor, R.N.; Wang, W.; Bagchi, M.K.; Yuan, L.; Scotchie, J.; Fritz, M.A.; Young, S.L. G protein-coupled estrogen receptor (GPER) expression in normal and abnormal endometrium. Reprod. Sci. 2012, 19, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Prossnitz, E.R.; Roy, S.K. Expression of g protein-coupled receptor 30 in the hamster ovary: Differential regulation by gonadotropins and steroid hormones. Endocrinology 2007, 148, 4853–4864. [Google Scholar] [CrossRef] [PubMed]
- Fietz, D.; Ratzenbock, C.; Hartmann, K.; Raabe, O.; Kliesch, S.; Weidner, W.; Klug, J.; Bergmann, M. Expression pattern of estrogen receptors α and β and g-protein-coupled estrogen receptor 1 in the human testis. Histochem. Cell Biol. 2014, 142, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, P.F.; Alves, M.G.; Martins, A.D.; Correia, S.; Bernardino, R.L.; Silva, J.; Barros, A.; Sousa, M.; Cavaco, J.E.; Socorro, S. Expression pattern of G protein-coupled receptor 30 in human seminiferous tubular cells. Gen. Comp. Endocrinol. 2014, 201, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Rago, V.; Romeo, F.; Giordano, F.; Maggiolini, M.; Carpino, A. Identification of the estrogen receptor GPER in neoplastic and non-neoplastic human testes. Reprod. Biol. Endocrinol. 2011, 9, 135. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, S.C.; Deroo, B.J.; Korach, K.S. Signal transduction. A new mediator for an old hormone? Science 2005, 307, 1572–1573. [Google Scholar] [CrossRef] [PubMed]
- Sirianni, R.; Chimento, A.; Ruggiero, C.; De Luca, A.; Lappano, R.; Ando, S.; Maggiolini, M.; Pezzi, V. The novel estrogen receptor, G protein-coupled receptor 30, mediates the proliferative effects induced by 17β-estradiol on mouse spermatogonial GC-1 cell line. Endocrinology 2008, 149, 5043–5051. [Google Scholar] [CrossRef] [PubMed]
- Prossnitz, E.R.; Arterburn, J.B.; Smith, H.O.; Oprea, T.I.; Sklar, L.A.; Hathaway, H.J. Estrogen signaling through the transmembrane g protein-coupled receptor GPR30. Annu. Rev. Physiol. 2008, 70, 165–190. [Google Scholar] [CrossRef] [PubMed]
- Toran-Allerand, C.D.; Guan, X.; MacLusky, N.J.; Horvath, T.L.; Diano, S.; Singh, M.; Connolly, E.S.J.; Nethrapalli, I.S.; Tinnikov, A.A. Er-x: A novel, plasma membrane-associated, putative estrogen receptor that is regulated during development and after ischemic brain injury. J. Neurosci. 2002, 22, 8391–8401. [Google Scholar] [PubMed]
- Kampa, M.; Notas, G.; Pelekanou, V.; Troullinaki, M.; Andrianaki, M.; Azariadis, K.; Kampouri, E.; Lavrentaki, K.; Castanas, E. Early membrane initiated transcriptional effects of estrogens in breast cancer cells: First pharmacological evidence for a novel membrane estrogen receptor element (ERX). Steroids 2012, 77, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Bosch, M.A.; Tobias, S.C.; Grandy, D.K.; Scanlan, T.S.; Ronnekleiv, O.K.; Kelly, M.J. Rapid signaling of estrogen in hypothalamic neurons involves a novel G-protein-coupled estrogen receptor that activates protein kinase c. J. Neurosci. 2003, 23, 9529–9540. [Google Scholar] [PubMed]
- Micevych, P.E.; Dewing, P. Membrane-initiated estradiol signaling regulating sexual receptivity. Front. Endocrinol. 2011, 2, 26. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Chambliss, K.L.; Mineo, C.; Shaul, P.W. Recent insights into non-nuclear actions of estrogen receptor α. Steroids 2014, 81, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Marino, M.; Galluzzo, P.; Ascenzi, P. Estrogen signaling multiple pathways to impact gene transcription. Curr. Genom. 2006, 7, 497–508. [Google Scholar] [CrossRef]
- Lucas, T.F.; Siu, E.R.; Esteves, C.A.; Monteiro, H.P.; Oliveira, C.A.; Porto, C.S.; Lazari, M.F. 17β-estradiol induces the translocation of the estrogen receptors ESR1 and ESR2 to the cell membrane, MAPK3/1 phosphorylation and proliferation of cultured immature rat sertoli cells. Biol. Reprod. 2008, 78, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Cowley, S.M.; Hoare, S.; Mosselman, S.; Parker, M.G. Estrogen receptors α and β form heterodimers on DNA. J. Biol. Chem. 1997, 272, 19858–19862. [Google Scholar] [CrossRef] [PubMed]
- Hammes, S.R.; Levin, E.R. Extranuclear steroid receptors: Nature and actions. Endocr. Rev. 2007, 28, 726–741. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.J.; Liao, J.K. Nonnuclear actions of estrogen. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1952–1961. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.J.; Levin, E.R. Rapid actions of plasma membrane estrogen receptors. Trends Endocrinol. Metab. 2001, 12, 152–156. [Google Scholar] [CrossRef]
- Acconcia, F.; Ascenzi, P.; Bocedi, A.; Spisni, E.; Tomasi, V.; Trentalance, A.; Visca, P.; Marino, M. Palmitoylation-dependent estrogen receptor α membrane localization: Regulation by 17β-estradiol. Mol. Biol. Cell 2005, 16, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Haynes, M.P.; Bender, J.R. Plasma membrane localization and function of the estrogen receptor α variant (ER46) in human endothelial cells. Proc. Natl. Acad. Sci. USA 2003, 100, 4807–4812. [Google Scholar] [CrossRef] [PubMed]
- Marquez, D.C.; Chen, H.W.; Curran, E.M.; Welshons, W.V.; Pietras, R.J. Estrogen receptors in membrane lipid rafts and signal transduction in breast cancer. Mol. Cell. Endocrinol. 2006, 246, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Norfleet, A.M.; Thomas, M.L.; Gametchu, B.; Watson, C.S. Estrogen receptor-α detected on the plasma membrane of aldehyde-fixed GH3/B6/F10 rat pituitary tumor cells by enzyme-linked immunocytochemistry. Endocrinology 1999, 140, 3805–3814. [Google Scholar] [PubMed]
- Pappas, T.C.; Gametchu, B.; Watson, C.S. Membrane estrogen receptors identified by multiple antibody labeling and impeded-ligand binding. FASEB J. 1995, 9, 404–410. [Google Scholar] [PubMed]
- Pedram, A.; Razandi, M.; Levin, E.R. Nature of functional estrogen receptors at the plasma membrane. Mol. Endocrinol. 2006, 20, 1996–2009. [Google Scholar] [CrossRef] [PubMed]
- Razandi, M.; Alton, G.; Pedram, A.; Ghonshani, S.; Webb, P.; Levin, E.R. Identification of a structural determinant necessary for the localization and function of estrogen receptor α at the plasma membrane. Mol. Cell. Biol. 2003, 23, 1633–1646. [Google Scholar] [CrossRef] [PubMed]
- Razandi, M.; Pedram, A.; Greene, G.L.; Levin, E.R. Cell membrane and nuclear estrogen receptors (ERS) originate from a single transcript: Studies of ERα and ERβ expressed in chinese hamster ovary cells. Mol. Endocrinol. 1999, 13, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Razandi, M.; Pedram, A.; Merchenthaler, I.; Greene, G.L.; Levin, E.R. Plasma membrane estrogen receptors exist and functions as dimers. Mol. Endocrinol. 2004, 18, 2854–2865. [Google Scholar] [CrossRef] [PubMed]
- Pedram, A.; Razandi, M.; Sainson, R.C.; Kim, J.K.; Hughes, C.C.; Levin, E.R. A conserved mechanism for steroid receptor translocation to the plasma membrane. J. Biol. Chem. 2007, 282, 22278–22288. [Google Scholar] [CrossRef] [PubMed]
- Acconcia, F.; Ascenzi, P.; Fabozzi, G.; Visca, P.; Marino, M. S-palmitoylation modulates human estrogen receptor-α functions. Biochem. Biophys. Res. Commun. 2004, 316, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Galluzzo, P.; Caiazza, F.; Moreno, S.; Marino, M. Role of ERβ palmitoylation in the inhibition of human colon cancer cell proliferation. Endocr. Cancer 2007, 14, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, X.; Shen, P.; Loggie, B.W.; Chang, Y.; Deuel, T.F. Identification, cloning, and expression of human estrogen receptor-α36, a novel variant of human estrogen receptor-α66. Biochem. Biophys. Res. Commun. 2005, 336, 1023–1027. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, Y.; Cao, J.M. G protein-coupled receptors: Extranuclear mediators for the non-genomic actions of steroids. Int. J. Mol. Sci. 2014, 15, 15412–15425. [Google Scholar] [CrossRef] [PubMed]
- Cavaco, J.E.; Laurentino, S.S.; Barros, A.; Sousa, M.; Socorro, S. Estrogen receptors α and β in human testis: Both isoforms are expressed. Syst. Biol. Reprod. Med. 2009, 55, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Enmark, E.; Pelto-Huikko, M.; Grandien, K.; Lagercrantz, S.; Lagercrantz, J.; Fried, G.; Nordenskjold, M.; Gustafsson, J.A. Human estrogen receptor β-gene structure, chromosomal localization, and expression pattern. J.Clin. Endocrinol. Metab. 1997, 82, 4258–4265. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.S.; Millar, M.R.; Majdic, G.; Saunders, P.T.; Fraser, H.M.; Sharpe, R.M. Immunolocalisation of oestrogen receptor-α within the testis and excurrent ducts of the rat and marmoset monkey from perinatal life to adulthood. J. Endocrinol. 1997, 153, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Gunawan, A.; Cinar, M.U.; Uddin, M.J.; Kaewmala, K.; Tesfaye, D.; Phatsara, C.; Tholen, E.; Looft, C.; Schellander, K. Investigation on association and expression of ESR2 as a candidate gene for boar sperm quality and fertility. Reprod. Domest. Anim. 2012, 47, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Gunawan, A.; Kaewmala, K.; Uddin, M.J.; Cinar, M.U.; Tesfaye, D.; Phatsara, C.; Tholen, E.; Looft, C.; Schellander, K. Association study and expression analysis of porcine ESR1 as a candidate gene for boar fertility and sperm quality. Anim. Reprod. Sci. 2011, 128, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Feng, H.L.; Sandlow, J.I.; Haines, C.J. Comparing expression of progesterone and estrogen receptors in testicular tissue from men with obstructive and nonobstructive azoospermia. J. Androl. 2009, 30, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Lekhkota, O.; Brehm, R.; Claus, R.; Wagner, A.; Bohle, R.M.; Bergmann, M. Cellular localization of estrogen receptor-α (ERα) and -β (ERβ) mRNA in the boar testis. Histochem. Cell Biol. 2006, 125, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Makinen, S.; Makela, S.; Weihua, Z.; Warner, M.; Rosenlund, B.; Salmi, S.; Hovatta, O.; Gustafsson, J.A. Localization of oestrogen receptors α and β in human testis. Mol. Hum. Reprod. 2001, 7, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Mutembei, H.M.; Pesch, S.; Schuler, G.; Hoffmann, B. Expression of oestrogen receptors α and β and of aromatase in the testis of immature and mature boars. Reprod. Domest. Anim. 2005, 40, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.; Fuchs, I.; Kauselmann, G.; Kern, H.; Zevnik, B.; Andreasen, P.; Schwarz, G.; Altmann, H.; Klewer, M.; Schoor, M.; et al. Gpr30 does not mediate estrogenic responses in reproductive organs in mice. Biol. Reprod. 2009, 80, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, G.; El-Alfy, M. Immunocytochemical localization of estrogen receptors α and β in the human reproductive organs. J. Clin. Endocrinol. Metab. 2000, 85, 4835–4840. [Google Scholar] [PubMed]
- Pelletier, G.; Labrie, C.; Labrie, F. Localization of oestrogen receptor α, oestrogen receptor β and androgen receptors in the rat reproductive organs. J. Endocrinol. 2000, 165, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Pentikainen, V.; Erkkila, K.; Suomalainen, L.; Parvinen, M.; Dunkel, L. Estradiol acts as a germ cell survival factor in the human testis in vitro. J. Clin. Endocrinol. Metab. 2000, 85, 2057–2067. [Google Scholar] [CrossRef] [PubMed]
- Saunders, P.T.; Fisher, J.S.; Sharpe, R.M.; Millar, M.R. Expression of oestrogen receptor β (ERβ) occurs in multiple cell types, including some germ cells, in the rat testis. J. Endocrinol. 1998, 156, R13–R17. [Google Scholar] [CrossRef] [PubMed]
- Saunders, P.T.; Millar, M.R.; Macpherson, S.; Irvine, D.S.; Groome, N.P.; Evans, L.R.; Sharpe, R.M.; Scobie, G.A. ERβ1 and the ERβ2 splice variant (ERβcx/β2) are expressed in distinct cell populations in the adult human testis. J. Clin. Endocrinol. Metab. 2002, 87, 2706–2715. [Google Scholar] [CrossRef] [PubMed]
- Saunders, P.T.; Sharpe, R.M.; Williams, K.; Macpherson, S.; Urquart, H.; Irvine, D.S.; Millar, M.R. Differential expression of oestrogen receptor α and β proteins in the testes and male reproductive system of human and non-human primates. Mol. Hum. Reprod. 2001, 7, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Van Pelt, A.M.; de Rooij, D.G.; van der Burg, B.; van der Saag, P.T.; Gustafsson, J.A.; Kuiper, G.G. Ontogeny of estrogen receptor-β expression in rat testis. Endocrinology 1999, 140, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Nie, R.; Prins, G.S.; Saunders, P.T.; Katzenellenbogen, B.S.; Hess, R.A. Localization of androgen and estrogen receptors in adult male mouse reproductive tract. J. Androl. 2002, 23, 870–881. [Google Scholar] [PubMed]
- Snyder, M.A.; Smejkalova, T.; Forlano, P.M.; Woolley, C.S. Multiple ERβ antisera label in ERβ knockout and null mouse tissues. J. Neurosci. Methods 2010, 188, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Bois, C.; Delalande, C.; Nurmio, M.; Parvinen, M.; Zanatta, L.; Toppari, J.; Carreau, S. Age- and cell-related gene expression of aromatase and estrogen receptors in the rat testis. J. Mol. Endocrinol. 2010, 45, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Lazari, M.F.; Porto, C.S. Differential role of the estrogen receptors ESR1 and ESR2 on the regulation of proteins involved with proliferation and differentiation of sertoli cells from 15-day-old rats. Mol. Cell. Endocrinol. 2014, 382, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Chimento, A.; Sirianni, R.; Delalande, C.; Silandre, D.; Bois, C.; Ando, S.; Maggiolini, M.; Carreau, S.; Pezzi, V. 17 β-estradiol activates rapid signaling pathways involved in rat pachytene spermatocytes apoptosis through GPR30 and ERα. Mol. Cell. Endocrinol. 2010, 320, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Chimento, A.; Sirianni, R.; Zolea, F.; Bois, C.; Delalande, C.; Ando, S.; Maggiolini, M.; Aquila, S.; Carreau, S.; Pezzi, V. Gper and ESRS are expressed in rat round spermatids and mediate oestrogen-dependent rapid pathways modulating expression of cyclin B1 and Bax. Int. J. Androl. 2011, 34, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Pimenta, M.T.; Pisolato, R.; Lazari, M.F.; Porto, C.S. 17β-estradiol signaling and regulation of sertoli cell function. Spermatogenesis 2011, 1, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Chimento, A.; Sirianni, R.; Casaburi, I.; Ruggiero, C.; Maggiolini, M.; Ando, S.; Pezzi, V. 17β-estradiol activates GPER- and ESR1-dependent pathways inducing apoptosis in GC-2 cells, a mouse spermatocyte-derived cell line. Mol. Cell. Endocrinol. 2012, 355, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Royer, C.; Siu, E.R.; Lazari, M.F.; Porto, C.S. Expression and signaling of G protein-coupled estrogen receptor 1 (GPER) in rat sertoli cells. Biol. Reprod. 2010, 83, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Royer, C.; Lucas, T.F.; Lazari, M.F.; Porto, C.S. 17β-estradiol signaling and regulation of proliferation and apoptosis of rat sertoli cells. Biol. Reprod. 2012, 86, 108. [Google Scholar] [CrossRef] [PubMed]
- Prossnitz, E.R.; Hathaway, H.J. What have we learned about gper function in physiology and disease from knockout mice? J. Steroid Biochem. Mol. Biol. 2015, 153, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dumasia, K.; Deshpande, S.; Balasinor, N.H. Direct regulation of genes involved in sperm release by estrogen and androgen through their receptors and coregulators. J. Steroid Biochem. Mol. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dumasia, K.; Gaonkar, R.; Sonawane, S.; Kadam, L.; Balasinor, N.H. Estrogen and androgen regulate actin-remodeling and endocytosis-related genes during rat spermiation. Mol. Cell. Endocrinol. 2015, 404, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Qiu, Y.; DeMayo, F.J.; Tsai, S.Y.; Tsai, M.J.; O’Malley, B.W. Partial hormone resistance in mice with disruption of the steroid receptor coactivator-1 (SRC-1) gene. Science 1998, 279, 1922–1925. [Google Scholar] [CrossRef] [PubMed]
- Gehin, M.; Mark, M.; Dennefeld, C.; Dierich, A.; Gronemeyer, H.; Chambon, P. The function of TIF2/GRIP1 in mouse reproduction is distinct from those of SRC-1 and P/CIP. Mol. Cell. Biol. 2002, 22, 5923–5937. [Google Scholar] [CrossRef] [PubMed]
- Robyr, D.; Wolffe, A.P.; Wahli, W. Nuclear hormone receptor coregulators in action: Diversity for shared tasks. Mol. Endocrinol. 2000, 14, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Cravatt, B.F.; Demarest, K.; Patricelli, M.P.; Bracey, M.H.; Giang, D.K.; Martin, B.R.; Lichtman, A.H. Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proc. Natl. Acad. Sci. USA 2001, 98, 9371–9376. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, P.; Orlando, P.; Di Siena, S.; Lolicato, F.; Petrosino, S.; Bisogno, T.; Geremia, R.; De Petrocellis, L.; Di Marzo, V. The endocannabinoid system and pivotal role of the CB2 receptor in mouse spermatogenesis. Proc. Natl. Acad. Sci. USA 2009, 106, 11131–11136. [Google Scholar] [CrossRef] [PubMed]
- Maccarrone, M.; Barboni, B.; Paradisi, A.; Bernabo, N.; Gasperi, V.; Pistilli, M.G.; Fezza, F.; Lucidi, P.; Mattioli, M. Characterization of the endocannabinoid system in boar spermatozoa and implications for sperm capacitation and acrosome reaction. J. Cell Sci. 2005, 118, 4393–4404. [Google Scholar] [CrossRef] [PubMed]
- Rossato, M.; Ion Popa, F.; Ferigo, M.; Clari, G.; Foresta, C. Human sperm express cannabinoid receptor CB1, the activation of which inhibits motility, acrosome reaction, and mitochondrial function. J. Clin. Endocrinol. Metab. 2005, 90, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.; Gasperi, V.; Paro, R.; Barsacchi, D.; Cecconi, S.; Maccarrone, M. Follicle-stimulating hormone activates fatty acid amide hydrolase by protein kinase a and aromatase-dependent pathways in mouse primary sertoli cells. Endocrinology 2007, 148, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, P.; Pucci, M.; Di Siena, S.; Di Giacomo, D.; Pirazzi, V.; Geremia, R.; Maccarrone, M. The faah gene is the first direct target of estrogen in the testis: Role of histone demethylase LSD1. Cell. Mol. Life Sci. 2012, 69, 4177–4190. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Perez, O.; Ballesteros, L.M.; Rosado, A. Binding of 17β-estradiol to the outer surface and nucleus of human spermatozoa. Arch. Androl. 1979, 3, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Boettcher, B.; Rose, R.J.; Kay, D.J.; Tinneberg, H.R. The binding of sex steroids to human spermatozoa. An autoradiographic study. Int. J. Androl. 1981, 4, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Boettcher, B.; Rose, R.J. Lack of cytosol and nuclear estrogen receptors in human spermatozoa. Biochem. Biophys. Res. Commun. 1981, 100, 840–846. [Google Scholar] [CrossRef]
- Durkee, T.J.; Mueller, M.; Zinaman, M. Identification of estrogen receptor protein and messenger ribonucleic acid in human spermatozoa. Am. J. Obstet. Gynecol. 1998, 178, 1288–1297. [Google Scholar] [CrossRef]
- Rago, V.; Siciliano, L.; Aquila, S.; Carpino, A. Detection of estrogen receptors ER-α and ER-β in human ejaculated immature spermatozoa with excess residual cytoplasm. Reprod. Biol. Endocrinol. 2006, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Solakidi, S.; Psarra, A.M.; Nikolaropoulos, S.; Sekeris, C.E. Estrogen receptors α and β (ERα and ERβ) and androgen receptor (AR) in human sperm: Localization of ERβ and ar in mitochondria of the midpiece. Hum. Reprod. 2005, 20, 3481–3487. [Google Scholar] [CrossRef] [PubMed]
- Denger, S.; Reid, G.; Kos, M.; Flouriot, G.; Parsch, D.; Brand, H.; Korach, K.S.; Sonntag-Buck, V.; Gannon, F. ERα gene expression in human primary osteoblasts: Evidence for the expression of two receptor proteins. Mol. Endocrinol. 2001, 15, 2064–2077. [Google Scholar] [CrossRef] [PubMed]
- Aquila, S.; Sisci, D.; Gentile, M.; Middea, E.; Catalano, S.; Carpino, A.; Rago, V.; Ando, S. Estrogen receptor ERα and ERβ are both expressed in human ejaculated spermatozoa: Evidence of their direct interaction with phosphatidylinositol-3-OH kinase/AKT pathway. J. Clin. Endocrinol. Metab. 2004, 89, 1443–1451. [Google Scholar] [CrossRef] [PubMed]
- Rago, V.; Aquila, S.; Panza, R.; Carpino, A. Cytochrome p450arom, androgen and estrogen receptors in pig sperm. Reprod. Biol. Endocrinol. 2007, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Sebkova, N.; Cerna, M.; Ded, L.; Peknicova, J.; Dvorakova-Hortova, K. The slower the better: How sperm capacitation and acrosome reaction is modified in the presence of estrogens. Reproduction 2012, 143, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Luconi, M.; Muratori, M.; Forti, G.; Baldi, E. Identification and characterization of a novel functional estrogen receptor on human sperm membrane that interferes with progesterone effects. J. Clin. Endocrinol. Metab. 1999, 84, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Antal, M.C.; Krust, A.; Chambon, P.; Mark, M. Sterility and absence of histopathological defects in nonreproductive organs of a mouse ERβ-null mutant. Proc. Natl. Acad. Sci. USA 2008, 105, 2433–2438. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Krust, A.; Gansmuller, A.; Dierich, A.; Chambon, P.; Mark, M. Effect of single and compound knockouts of estrogen receptors α (ERα) and β (ERβ) on mouse reproductive phenotypes. Development 2000, 127, 4277–4291. [Google Scholar] [PubMed]
- Eddy, E.M.; Washburn, T.F.; Bunch, D.O.; Goulding, E.H.; Gladen, B.C.; Lubahn, D.B.; Korach, K.S. Targeted disruption of the estrogen receptor gene in male mice causes alteration of spermatogenesis and infertility. Endocrinology 1996, 137, 4796–4805. [Google Scholar] [PubMed]
- Krege, J.H.; Hodgin, J.B.; Couse, J.F.; Enmark, E.; Warner, M.; Mahler, J.F.; Sar, M.; Korach, K.S.; Gustafsson, J.A.; Smithies, O. Generation and reproductive phenotypes of mice lacking estrogen receptor β. Proc. Natl. Acad. Sci. USA 1998, 95, 15677–15682. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.R. Integration of the extranuclear and nuclear actions of estrogen. Mol. Endocrinol. 2005, 19, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.R.; Graves, K.H.; Parlow, A.F.; Simpson, E.R. Characterization of mice deficient in aromatase (ARKO) because of targeted disruption of the CYP19 gene. Proc. Natl. Acad. Sci. USA 1998, 95, 6965–6970. [Google Scholar] [CrossRef] [PubMed]
- Robertson, K.M.; O’Donnell, L.; Jones, M.E.; Meachem, S.J.; Boon, W.C.; Fisher, C.R.; Graves, K.H.; McLachlan, R.I.; Simpson, E.R. Impairment of spermatogenesis in mice lacking a functional aromatase (CYP 19) gene. Proc. Natl. Acad. Sci. USA 1999, 96, 7986–7991. [Google Scholar] [CrossRef] [PubMed]
- Dumasia, K.; Kumar, A.; Kadam, L.; Balasinor, N.H. Effect of estrogen receptor-subtype-specific ligands on fertility in adult male rats. J. Endocrinol. 2015, 225, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Honda, S.; Harada, N.; Ito, S.; Takagi, Y.; Maeda, S. Disruption of sexual behavior in male aromatase-deficient mice lacking exons 1 and 2 of the Cyp19 gene. Biochem. Biophys. Res. Commun. 1998, 252, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Robertson, K.M.; Simpson, E.R.; Lacham-Kaplan, O.; Jones, M.E. Characterization of the fertility of male aromatase knockout mice. J. Androl. 2001, 22, 825–830. [Google Scholar] [PubMed]
- Dudek, P.; Picard, D. Genomics of signaling crosstalk of estrogen receptor α in breast cancer cells. PLoS ONE 2008, 3, e1859. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Hess, R.A.; Bahr, J.M.; Lubahn, D.B.; Taylor, J.; Bunick, D. Estrogen receptor α has a functional role in the mouse rete testis and efferent ductules. Biol. Reprod. 2000, 63, 1873–1880. [Google Scholar] [CrossRef] [PubMed]
- Mahato, D.; Goulding, E.H.; Korach, K.S.; Eddy, E.M. Estrogen receptor-α is required by the supporting somatic cells for spermatogenesis. Mol. Cell. Endocrinol. 2001, 178, 57–63. [Google Scholar] [CrossRef]
- Joseph, A.; Hess, R.A.; Schaeffer, D.J.; Ko, C.; Hudgin-Spivey, S.; Chambon, P.; Shur, B.D. Absence of estrogen receptor α leads to physiological alterations in the mouse epididymis and consequent defects in sperm function. Biol. Reprod. 2010, 82, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Joseph, A.; Shur, B.D.; Ko, C.; Chambon, P.; Hess, R.A. Epididymal hypo-osmolality induces abnormal sperm morphology and function in the estrogen receptor α knockout mouse. Biol. Reprod. 2010, 82, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Nanjappa, M.K.; Hess, R.A.; Medrano, T.I.; Locker, S.H.; Levin, E.R.; Cooke, P.S. Membrane-localized estrogen receptor 1 is required for normal male reproductive development and function in mice. Endocrinology 2016, 157, 2909–2919. [Google Scholar] [CrossRef] [PubMed]
- Sinkevicius, K.W.; Laine, M.; Lotan, T.L.; Woloszyn, K.; Richburg, J.H.; Greene, G.L. Estrogen-dependent and -independent estrogen receptor-α signaling separately regulate male fertility. Endocrinology 2009, 150, 2898–2905. [Google Scholar] [CrossRef] [PubMed]
- Shaha, C. Estrogens and spermatogenesis. Adv. Exp. Med. Biol. 2008, 636, 42–64. [Google Scholar] [PubMed]
- Delbes, G.; Levacher, C.; Pairault, C.; Racine, C.; Duquenne, C.; Krust, A.; Habert, R. Estrogen receptor β-mediated inhibition of male germ cell line development in mice by endogenous estrogens during perinatal life. Endocrinology 2004, 145, 3395–3403. [Google Scholar] [CrossRef] [PubMed]
- Gould, M.L.; Hurst, P.R.; Nicholson, H.D. The effects of oestrogen receptors α and β on testicular cell number and steroidogenesis in mice. Reproduction 2007, 134, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Antal, M.C.; Petit-Demouliere, B.; Meziane, H.; Chambon, P.; Krust, A. Estrogen dependent activation function of ERβ is essential for the sexual behavior of mouse females. Proc. Natl. Acad. Sci. USA 2012, 109, 19822–19827. [Google Scholar] [CrossRef] [PubMed]
- Maneix, L.; Antonson, P.; Humire, P.; Rochel-Maia, S.; Castaneda, J.; Omoto, Y.; Kim, H.J.; Warner, M.; Gustafsson, J.A. Estrogen receptor β exon 3-deleted mouse: The importance of non-ere pathways in ERβ signaling. Proc. Natl. Acad. Sci. USA 2015, 112, 5135–5140. [Google Scholar] [CrossRef] [PubMed]
- Isensee, J.; Meoli, L.; Zazzu, V.; Nabzdyk, C.; Witt, H.; Soewarto, D.; Effertz, K.; Fuchs, H.; Gailus-Durner, V.; Busch, D.; et al. Expression pattern of G protein-coupled receptor 30 in lacz reporter mice. Endocrinology 2009, 150, 1722–1730. [Google Scholar] [CrossRef] [PubMed]
- Balasinor, N.H.; D'Souza, R.; Nanaware, P.; Idicula-Thomas, S.; Kedia-Mokashi, N.; He, Z.; Dym, M. Effect of high intratesticular estrogen on global gene expression and testicular cell number in rats. Reprod. Biol. Endocrinol. 2010, 8, 72. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, R.M.; Skakkebaek, N.E. Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract? Lancet 1993, 341, 1392–1395. [Google Scholar] [CrossRef]
- O’Donnell, L.; Robertson, K.M.; Jones, M.E.; Simpson, E.R. Estrogen and spermatogenesis. Endocr. Rev. 2001, 22, 289–318. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.P.; Boyd, J.; Frank, G.R.; Takahashi, H.; Cohen, R.M.; Specker, B.; Williams, T.C.; Lubahn, D.B.; Korach, K.S. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N. Engl. J. Med. 1994, 331, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Carani, C.; Qin, K.; Simoni, M.; Faustini-Fustini, M.; Serpente, S.; Boyd, J.; Korach, K.S.; Simpson, E.R. Effect of testosterone and estradiol in a man with aromatase deficiency. N. Engl. J. Med. 1997, 337, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, B.L.; Saller, B.; Janssen, O.E.; Gocke, P.; Bockisch, A.; Sperling, H.; Mann, K.; Broecker, M. Impact of estrogen replacement therapy in a male with congenital aromatase deficiency caused by a novel mutation in the Cyp19 gene. J. Clin. Endocrinol. Metab. 2002, 87, 5476–5484. [Google Scholar] [CrossRef] [PubMed]
- Prossnitz, E.R.; Barton, M. Signaling, physiological functions and clinical relevance of the G protein-coupled estrogen receptor gper. Prostaglandins Lipid Mediat. 2009, 89, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.; Pearl, C.A. Alterations in the estrogen environment of the testis contribute to declining sperm production in aging rats. Syst. Biol. Reprod. Med. 2014, 60, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Hamden, K.; Silandre, D.; Delalande, C.; Elfeki, A.; Carreau, S. Protective effects of estrogens and caloric restriction during aging on various rat testis parameters. Asian J. Androl. 2008, 10, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Colucci D’Amato, L.; Guarino, F.; Salvatore, G.; Angelini, F. 17 β-estradiol induces spermatogonial proliferation through mitogen-activated protein kinase (extracellular signal-regulated kinase 1/2) activity in the lizard (podarcis s. Sicula). Mol. Reprod. Dev. 2002, 61, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Colucci-D’Amato, G.L.; Staibano, S.; Franco, R.; Tramontano, D. Estradiol-induced mitogen-activated protein kinase (extracellular signal-regulated kinase 1 and 2) activity in the frog (rana esculenta) testis. J. Endocrinol. 2000, 167, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Dumasia, K.; Kumar, A.; Deshpande, S.; Sonawane, S.; Balasinor, N.H. Differential roles of estrogen receptors, ESR1 and ESR2, in adult rat spermatogenesis. Mol. Cell. Endocrinol. 2016, 428, 89–100. [Google Scholar] [CrossRef] [PubMed]
- D'Souza, R.; Pathak, S.; Upadhyay, R.; Gaonkar, R.; D’Souza, S.; Sonawane, S.; Gill-Sharma, M.; Balasinor, N.H. Disruption of tubulobulbar complex by high intratesticular estrogens leading to failed spermiation. Endocrinology 2009, 150, 1861–1869. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.D.; Kumar, A.V.; Ganeshan, M.; Balasinor, N.H. Tubulobulbar complex: Cytoskeletal remodeling to release spermatozoa. Reprod. Biol. Endocrinol. 2012, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Yanagimachi, R. Mammalian fertilization. In The Physiology of Reproduction; Knobil, E., Neill, J.D., Eds.; Raven Press: New York, NY, USA, 1994; pp. 189–317. [Google Scholar]
- Adeoya-Osiguwa, S.A.; Markoulaki, S.; Pocock, V.; Milligan, S.R.; Fraser, L.R. 17β-estradiol and environmental estrogens significantly affect mammalian sperm function. Hum. Reprod. 2003, 18, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Bathla, H.; Guraya, S.S.; Sangha, G.K. Role of estradiol in the capacitation and acrosome reaction of hamster epididymal spermatozoa in the isolated uterus of mice incubated in vitro. Indian J. Phys. Pharmacol. 1999, 43, 211–217. [Google Scholar]
- Ded, L.; Dostalova, P.; Dorosh, A.; Dvorakova-Hortova, K.; Peknicova, J. Effect of estrogens on boar sperm capacitation in vitro. Reprod. Biol. Endocrinol. 2010, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Fraser, L.R.; Beyret, E.; Milligan, S.R.; Adeoya-Osiguwa, S.A. Effects of estrogenic xenobiotics on human and mouse spermatozoa. Hum. Reprod. 2006, 21, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- He, Y.F.; Yue, L.M.; He, Y.P.; Zhang, J.H.; Zheng, J.; Gao, X.P. Effects of estrogen on acrosome reaction and intracellular calcium in human spermatozoa and the possible mechanism concerned. Sichuan Da Xue Xue Bao Yi Xue Ban 2005, 36, 500–502. [Google Scholar] [PubMed]
- Luconi, M.; Bonaccorsi, L.; Forti, G.; Baldi, E. Effects of estrogenic compounds on human spermatozoa: Evidence for interaction with a nongenomic receptor for estrogen on human sperm membrane. Mol. Cell. Endocrinol. 2001, 178, 39–45. [Google Scholar] [CrossRef]
- Mohamed el, S.A.; Park, Y.J.; Song, W.H.; Shin, D.H.; You, Y.A.; Ryu, B.Y.; Pang, M.G. Xenoestrogenic compounds promote capacitation and an acrosome reaction in porcine sperm. Theriogenology 2011, 75, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Vigil, P.; Toro, A.; Godoy, A. Physiological action of oestradiol on the acrosome reaction in human spermatozoa. Andrologia 2008, 40, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Ded, L.; Sebkova, N.; Cerna, M.; Elzeinova, F.; Dostalova, P.; Peknicova, J.; Dvorakova-Hortova, K. In vivo exposure to 17β-estradiol triggers premature sperm capacitation in cauda epididymis. Reproduction 2013, 145, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Shanle, E.K.; Xu, W. Endocrine disrupting chemicals targeting estrogen receptor signaling: Identification and mechanisms of action. Chem. Res. Toxicol. 2011, 24, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Brzozowski, A.M.; Pike, A.C.; Dauter, Z.; Hubbard, R.E.; Bonn, T.; Engstrom, O.; Ohman, L.; Greene, G.L.; Gustafsson, J.A.; Carlquist, M. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature 1997, 389, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Pike, A.C.; Brzozowski, A.M.; Hubbard, R.E.; Bonn, T.; Thorsell, A.G.; Engstrom, O.; Ljunggren, J.; Gustafsson, J.A.; Carlquist, M. Structure of the ligand-binding domain of oestrogen receptor β in the presence of a partial agonist and a full antagonist. EMBO J. 1999, 18, 4608–4618. [Google Scholar] [CrossRef] [PubMed]
- Troisi, R.; Hatch, E.E.; Titus-Ernstoff, L.; Hyer, M.; Palmer, J.R.; Robboy, S.J.; Strohsnitter, W.C.; Kaufman, R.; Herbst, A.L.; Hoover, R.N. Cancer risk in women prenatally exposed to diethylstilbestrol. Int. J. Cancer 2007, 121, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, A.J.; Baird, D.D.; Weinberg, C.R.; Hornsby, P.P.; Herbst, A.L. Fertility in men exposed prenatally to diethylstilbestrol. N. Engl. J. Med. 1995, 332, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R. Lessons learned from perinatal exposure to diethylstilbestrol. Toxicol. Appl. Pharmacol. 2004, 199, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R.; Hanson, R.B.; Jefferson, W.N.; Bullock, B.C.; Haseman, J.; McLachlan, J.A. Proliferative lesions and reproductive tract tumors in male descendants of mice exposed developmentally to diethylstilbestrol. Carcinogenesis 2000, 21, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Filipiak, E.; Walczak-Jedrzejowska, R.; Oszukowska, E.; Guminska, A.; Marchlewska, K.; Kula, K.; Slowikowska-Hilczer, J. Xenoestrogens diethylstilbestrol and zearalenone negatively influence pubertal rat’s testis. Folia Histochem. Cytobiol. 2009, 47, S113–S120. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C. Reproductive malformation of the male offspring following maternal exposure to estrogenic chemicals. Proc. Soc. Exp. Biol. Med. 2000, 224, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kyselova, V.; Peknicova, J.; Boubelik, M.; Buckiova, D. Body and organ weight, sperm acrosomal status and reproduction after genistein and diethylstilbestrol treatment of CD1 mice in a multigenerational study. Theriogenology 2004, 61, 1307–1325. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hamilton, K.J.; Lai, A.Y.; Burns, K.A.; Li, L.; Wade, P.A.; Korach, K.S. Diethylstilbestrol (DES)-stimulated hormonal toxicity is mediated by ERα alteration of target gene methylation patterns and epigenetic modifiers (DNMT3A, MBD2, AND HDAC2) in the mouse seminal vesicle. Environ. Health Perspect. 2014, 122, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Takemura, H.; Shim, J.Y.; Sayama, K.; Tsubura, A.; Zhu, B.T.; Shimoi, K. Characterization of the estrogenic activities of zearalenone and zeranol in vivo and in vitro. J. Steroid Biochem. Mol. Biol. 2007, 103, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Wang, G.X.; Liu, J.L.; Fan, J.J.; Cui, S. Toxic effects of zearalenone and its derivatives α-zearalenol on male reproductive system in mice. Reprod. Toxicol. 2007, 24, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Zatecka, E.; Ded, L.; Elzeinova, F.; Kubatova, A.; Dorosh, A.; Margaryan, H.; Dostalova, P.; Korenkova, V.; Hoskova, K.; Peknicova, J. Effect of zearalenone on reproductive parameters and expression of selected testicular genes in mice. Reprod. Toxicol. 2014, 45, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Adibnia, E.; Razi, M.; Malekinejad, H. Zearalenone and 17 β-estradiol induced damages in male rats reproduction potential; evidence for ERα and ERβ receptors expression and steroidogenesis. Toxicon 2016, 120, 133–146. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dostalova, P.; Zatecka, E.; Dvorakova-Hortova, K. Of Oestrogens and Sperm: A Review of the Roles of Oestrogens and Oestrogen Receptors in Male Reproduction. Int. J. Mol. Sci. 2017, 18, 904. https://doi.org/10.3390/ijms18050904
Dostalova P, Zatecka E, Dvorakova-Hortova K. Of Oestrogens and Sperm: A Review of the Roles of Oestrogens and Oestrogen Receptors in Male Reproduction. International Journal of Molecular Sciences. 2017; 18(5):904. https://doi.org/10.3390/ijms18050904
Chicago/Turabian StyleDostalova, Pavla, Eva Zatecka, and Katerina Dvorakova-Hortova. 2017. "Of Oestrogens and Sperm: A Review of the Roles of Oestrogens and Oestrogen Receptors in Male Reproduction" International Journal of Molecular Sciences 18, no. 5: 904. https://doi.org/10.3390/ijms18050904






