Increased Force Variability Is Associated with Altered Modulation of the Motorneuron Pool Activity in Autism Spectrum Disorder (ASD)
Abstract
:1. Introduction
2. Results
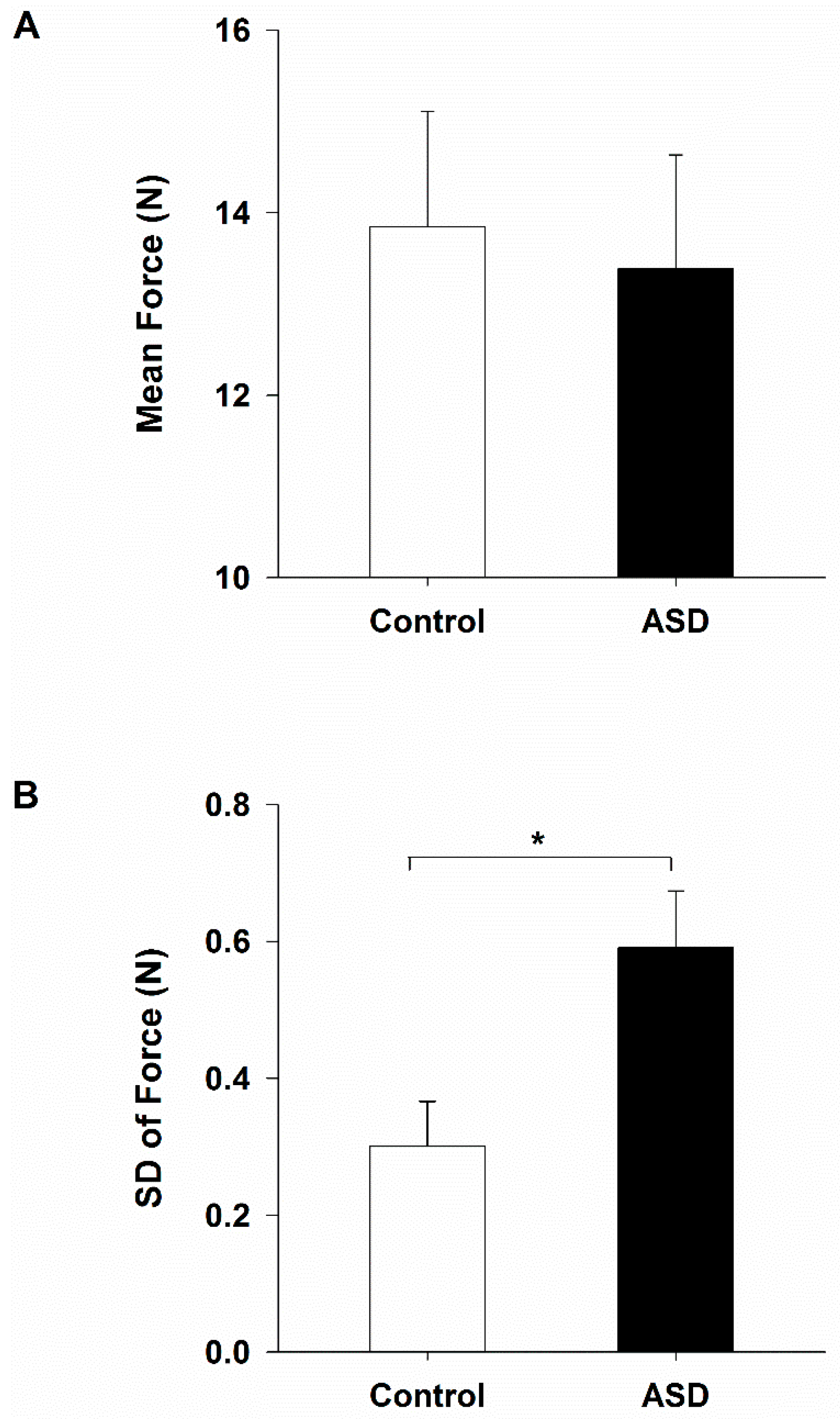
2.1. Isometric Index Finger Abduction Force and Variability
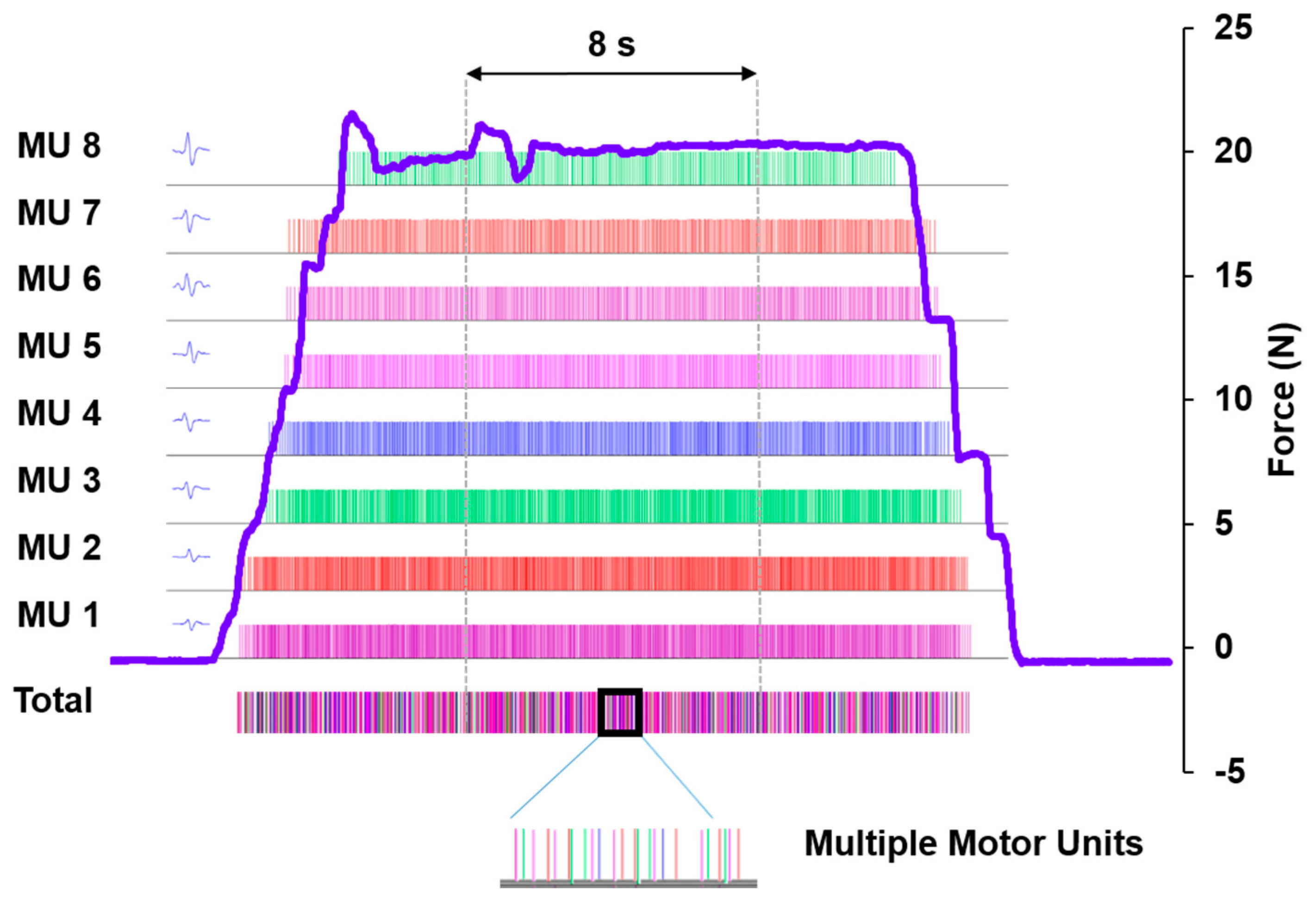
2.2. decomposition-Based Electromyography (dEMG) Assessments of Motorneuron pool Activituy of the First Dorsal Interosseous (FDI) Muscle
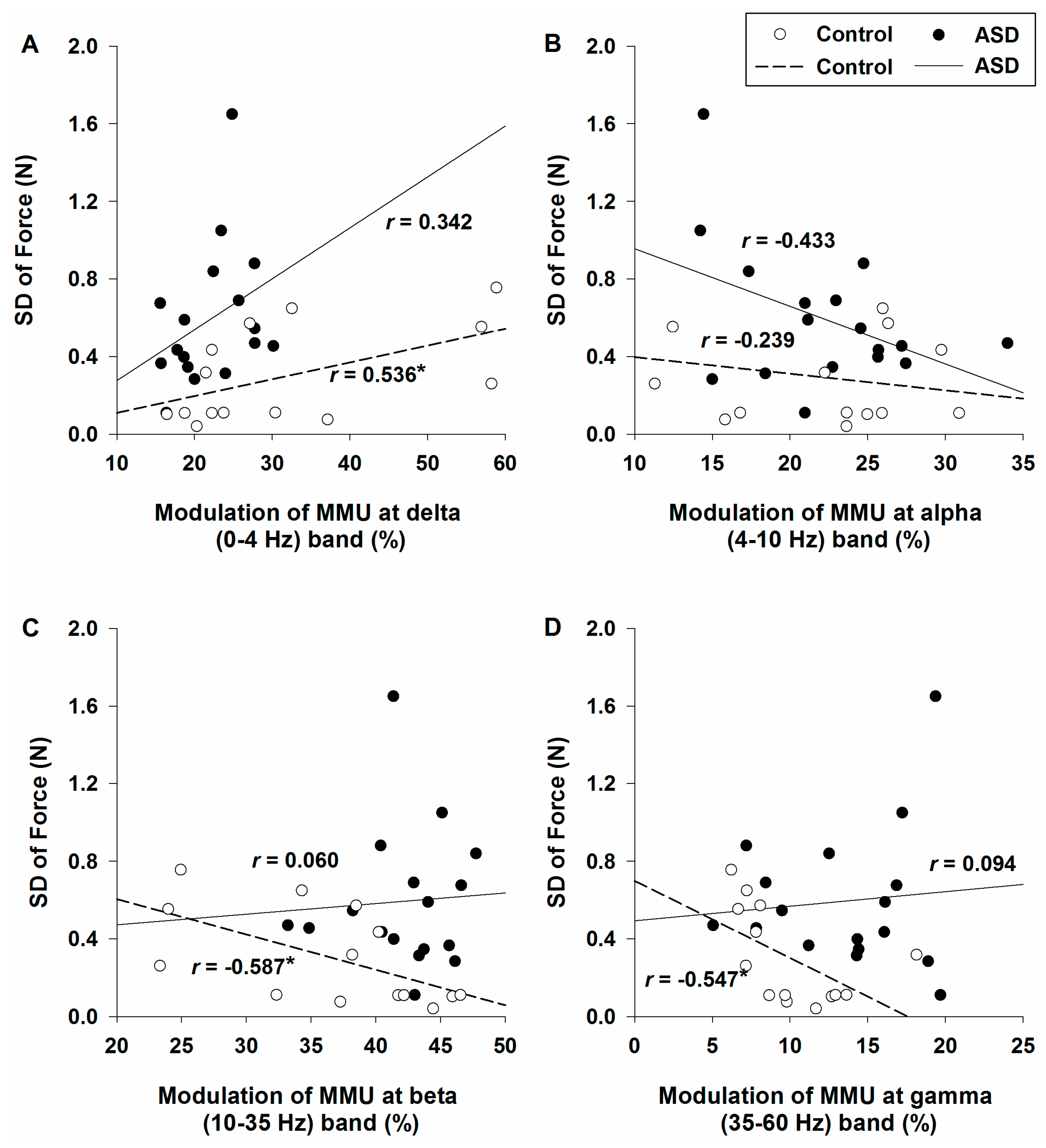
2.3. Relationship between Force Performance and Modulation of the FDI Muscle Motorneuron Pool Activity
2.4. Demographic and Clinical Correlations
3. Discussion
3.1. Altered Force Production in Autism Spectrum Disorder (ASD)
3.2. Altered Motorneuron Pool Activation during Force Control in ASD
3.3. Neuromotor Deficits, Demographic Characteristics and Clinical Symptoms in ASD
3.4. Study Limitation
4. Materials and Methods
4.1. Participants
4.2. Apparatus and Procedures
4.3. Data Processing and Analyses
4.3.1. Force Data
4.3.2. sEMG Data and Decomposition Procedures for Motor Units’ Activities
4.4. Clinical Measures
4.5. Statistical Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ASD | Autism spectrum disorder |
| ADOS-2 | Autism diagnostic observation schedule-2 |
| ADI-R | Autism diagnostic inventory-Revised |
| sEMG | Surface electromyography |
| dEMG | Decomposition-based quantitative electromyography |
| MVC | Maximum voluntary contraction |
| FDI muscle | First dorsal interosseus muscle |
| Motor unit | A single motorneuron and the muscle fiber that it innervates |
| Motorneuron pool | A motorneuron pool consists of all individual motorneurons that innervate a single muscle |
| Motor unit recruitment or Modulation of motor units | The CNS is responsible for the orderly recruitment of motorneurons through two distinct ways: spatial and temporal recruitment. Spatial recruitment activates more motor units to produce greater force. Temporal recruitment, or rate coding, deals with the frequency or activation rate of motor units firing |
| Size principle | Henneman’s size principle [22] explains spatial recruitment of motor units, in which motor units are recruited from smallest to largest based on the amount of force production. For smaller force, slow twitch, low-force, fatigue-resistant muscle fibers are activated prior to the recruitment of the fast twitch, high-force, less fatigue-resistant muscle fibers |
| Motor unit discharge rate | Motor unit discharge rate describes temporal recruitment of motor units represented by spike firing frequency or rate of action potentials |
Appendix A. Motor Unit Selection

References
- Fournier, K.A.; Hass, C.J.; Naik, S.K.; Lodha, N.; Cauraugh, J.H. Motor Coordination in Autism Spectrum Disorders: A Synthesis and Meta-Analysis. J. Autism Dev. Disord. 2010, 40, 1227–1240. [Google Scholar] [CrossRef] [PubMed]
- Ming, X.; Brimacombe, M.; Wagner, G.C. Prevalence of motor impairment in autism spectrum disorders. Brain Dev. 2007, 29, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Loh, A.; Soman, T.; Brian, J.; Bryson, S.E.; Roberts, W.; Szatmari, P.; Smith, I.M.; Zwaigenbaum, L. Stereotyped motor behaviors associated with autism in high-risk infants: A pilot videotape analysis of a sibling sample. J. Autism Dev. Disord. 2007, 37, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Landa, R.J. Diagnosis of autism spectrum disorders in the first 3 years of life. Nat. Clin. Pract. Neurol. 2008, 4, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Hannant, P.; Tavassoli, T.; Cassidy, S. The Role of Sensorimotor Difficulties in Autism Spectrum Conditions. Front. Neurol. 2016, 7, 124. [Google Scholar] [CrossRef] [PubMed]
- Travers, B.G.; Bigler, E.D.; Duffield, T.C.; Prigge, M.D.; Froehlich, A.L.; Lange, N.; Alexander, A.L.; Lainhart, J.E. Longitudinal development of manual motor ability in autism spectrum disorder from childhood to mid-adulthood relates to adaptive daily living skills. Dev. Sci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Takarae, Y. Oculomotor abnormalities parallel cerebellar histopathology in autism. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1359–1361. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, L.M.; Cook, E.H.; Sweeney, J.A.; Mosconi, M.W. Saccadic eye movement abnormalities in autism spectrum disorder indicate dysfunctions in both cerebellum and brainstem. Mol. Autism 2014, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Minshew, N.J.; Sung, K.; Jones, B.L.; Furman, J.M. Underdevelopment of the postural control system in autism. Neurology 2007, 63, 2056–2061. [Google Scholar] [CrossRef]
- Wang, Z.; Hallac, R.R.; Conroy, K.C.; White, S.P.; Kane, A.A.; Collinsworth, A.L.; Sweeney, J.A.; Mosconi, M.W. Postural orientation and equilibrium processes associated with increased postural sway in autism spectrum disorder (ASD). J. Neurodev. Disord. 2016, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Hallett, M.; Lebiedowska, M.K.; Thomas, S.L.; Stanhope, S.J.; Benckla, M.B.; Rumsey, J. Locomotion of autistic adults. Arch. Neurol. 1993, 50, 1304–1308. [Google Scholar] [CrossRef] [PubMed]
- Kindregan, D.; Gallagher, L.; Gormley, J. Gait deviations in children with autism spectrum disorders: A review. Autism Res. Treat. 2015, 2015, 741480. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, C.T.; Mostofsky, S.H.; Bastian, A.J. Children with autism show specific handwriting impairments. Neurology 2009, 73, 1532–1537. [Google Scholar] [CrossRef] [PubMed]
- Hardan, A.Y.; Kilpatrick, M.; Keshavan, M.S.; Minshew, N.J. Motor Performance and Anatomic Magnetic Resonance Imaging (MRI) of the Basal Ganglia in Autism. J. Child Neurol. 2003, 18, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Kern, J.K.; Geier, D.A.; Adams, J.B.; Troutman, M.R.; Davis, G.; King, P.G.; Young, J.L.; Geier, M.R. Autism severity and muscle strength: A correlation analysis. Res. Autism Spectr. Disord. 2011, 5, 1011–1015. [Google Scholar] [CrossRef]
- Mosconi, M.W.; Mohanty, S.; Greene, R.K.; Cook, E.H.; Vaillancourt, D.E.; Sweeney, J.A. Feedforward and feedback motor control abnormalities implicate cerebellar dysfunctions in autism spectrum disorder. J. Neurosci. 2015, 35, 2015–2025. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Magnon, G.C.; White, S.P.; Greene, R.K.; Vaillancourt, D.E.; Mosconi, M.W. Individuals with autism spectrum disorder show abnormalities during initial and subsequent phases of precision gripping. J. Neurophysiol. 2015, 113, 1989–2001. [Google Scholar] [CrossRef] [PubMed]
- Grosse, P.; Cassidy, M.J.; Brown, P. EEG-EMG, MEG-EMG and EMG-EMG frequency analysis: Physiological principles and clinical applications. Clin. Neurophysiol. 2002, 113, 1523–1531. [Google Scholar] [CrossRef]
- De Luca, C.J.; LeFever, R.S.; McCue, M.P.; Xenakis, A.P. Behaviour of human motor units in different muscles during linearly varying contractions. J. Physiol. 1982, 329, 113–128. [Google Scholar] [CrossRef] [PubMed]
- De Luca, C.J.; LeFever, R.S.; McCue, M.P.; Xenakis, A.P. Control scheme governing concurrently active human motor units during voluntary contractions. J. Physiol. 1982, 329, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Moritz, C.T.; Barry, B.K.; Pascoe, M.A.; Enoka, R.M. Discharge rate variability influences the variation in force fluctuations across the working range of a hand muscle. J. Neurophysiol. 2005, 93, 2449–2459. [Google Scholar] [CrossRef] [PubMed]
- Henneman, E. Organization of the motorneuron pool: The size principle. In Medical Physiology, 14th ed.; Mountcastle, V.B., Ed.; Mosby: St. Louis, MO, USA, 1980; pp. 718–741. [Google Scholar]
- Seki, K.; Marusawa, M. Firing rate modulation of human motor units in different muscles during isometric contraction with various forces. Brain Res. 1996, 719, 1–7. [Google Scholar] [CrossRef]
- De Luca, C.J.; Erim, Z. Common drive of motor units in regulation of muscle force. Trends Neurosci. 1994, 17, 299–305. [Google Scholar] [CrossRef]
- De Luca, C.J.; Erim, Z. Common drive in motor units of a synergistic muscle pair. J. Neurophysiol. 2002, 87, 2200–2204. [Google Scholar] [CrossRef] [PubMed]
- Vallbo, A.B.; Wessberg, J. Organisation of motor output in slow finger movements in man. J. Physiol. 1993, 469, 673–691. [Google Scholar] [CrossRef] [PubMed]
- Wessberg, J.; Vallbo, A.B. Coding of pulsatile motor output by human muscle afferents during slow finger movements. J. Physiol. 1995, 485, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Marsden, C.D. Origins of normal and pathological tremor. In Movement Disorders: Tremor; Findley, L.J., Capideo, R., Eds.; MacMillan Press: London, UK, 1984; pp. 37–84. [Google Scholar]
- Conway, B.A.; Halliday, D.M.; Farmer, S.F.; Shahani, U.; Maas, P.; Weir, A.I.; Rosenberg, J.R. Synchronization between motor cortex and spinal motorneuronal pool during the performance of a maintained motor task in man. J. Physiol. 1995, 489, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Salenius, S.; Portin, K.; Kajola, M.; Salmelin, R.; Hari, R. Cortical control of human motorneuron firing during isometric contraction. J. Neurophysiol. 1997, 77, 3401–3405. [Google Scholar] [PubMed]
- Chakarov, V.; Naranjo, J.R.; Schulte-Monting, J.; Omlor, W.; Huethe, F.; Kristeva, R. β-Range EEG-EMG Coherence With Isometric Compensation for Increasing Modulated Low-Level Forces. J. Neurophysiol. 2009, 102, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Brown, P. Cortical drives to human muscle: The Piper and related rhythms Prog. Neurobiol. 2000, 60, 97–108. [Google Scholar] [CrossRef]
- Engel, A.K.; Fries, P. Beta-band oscillations-signalling the status quo? Curr. Opin. Neurobiol. 2010, 20, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Witte, M.; Patino, L.; Andrykiewicz, A.; Hepp-Reymond, M.C.; Kristeva, R. Modulation of human corticomuscular β-range coherence with low-level static forces. Eur. J. Neurosci. 2007, 26, 3564–3570. [Google Scholar] [CrossRef] [PubMed]
- Kristeva, R.; Patino, L.; Omlor, W. β-Range cortical motor spectral power and corticomuscular coherence as a mechanism for effective corticospinal interaction during steady-state motor output. Neuroimage 2007, 36, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Brown, P. Muscle sounds in Parkinson’s disease. Lancet 1997, 349, 533–535. [Google Scholar] [CrossRef]
- Tracy, B.L.; Maluf, K.S.; Stephenson, J.L.; Hunter, S.K.; Enoka, R.M. Variability of motor unit discharge and force fluctuations across a range of muscle forces in older adults. Muscle Nerve 2005, 32, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kwon, M.; Solis, D.; Lodha, N.; Christou, E.A. Motor control differs for increasing and releasing force. J. Neurophysiol. 2016, 115, 2924–2930. [Google Scholar] [CrossRef] [PubMed]
- Neely, K.A.; Mohanty, S.; Schmitt, L.M.; Wang, Z.; Sweeney, J.A.; Mosconi, M.W. Motor Memory Deficits Contribute to Motor Impairments in Autism Spectrum Disorder. J. Autism Dev. Disord. 2016. [Google Scholar] [CrossRef] [PubMed]
- David, F.J.; Baranek, G.T.; Giuliani, C.A.; Mercer, V.S.; Poe, M.D.; Thorpe, D.E. A Pilot Study: Coordination of Precision Grip in Children and Adolescents with High Functioning Autism. Pediatr. Phys. Ther. 2009, 21, 205–211. [Google Scholar] [CrossRef] [PubMed]
- David, F.J.; Baranek, G.T.; Wiesen, C.; Miao, A.F.; Thorpe, D.E. Coordination of precision grip in 2–6 years-old children with autism spectrum disorders compared to children developing typically and children with developmental disabilities. Front. Integr. Neurosci. 2012, 6, 122. [Google Scholar] [PubMed]
- De Luca, C.J.; Adam, A.; Wotiz, R.; Gilmore, L.D.; Nawab, S.H. Decomposition of surface EMG signals. J. Neurophysiol. 2006, 96, 1646–1657. [Google Scholar] [CrossRef] [PubMed]
- Nawab, S.H.; Chang, S.S.; De Luca, C.J. High-yield decomposition of surface EMG signals. Clin. Neurophysiol. 2010, 121, 1602–1615. [Google Scholar] [CrossRef] [PubMed]
- Anzulewicz, A.; Sobota, K.; Delafield-Butt, J.T. Toward the Autism Motor Signature: Gesture patterns during smart tablet gameplay identify children with autism. Sci. Rep. 2016, 6, 31107. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.; McGill, K.C. Neuromuscular activation and motor-unit firing characteristics in cerebral palsy. Dev. Med. Child Neurol. 2005, 47, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Yoshitake, Y.; Shinohara, M. Low-frequency component of rectified EMG is temporally correlated with force and instantaneous rate of force fluctuations during steady contractions. Muscle Nerve 2013, 47, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Kim, C.; Kwon, M.; Chen, Y.T.; Onushko, T.; Lodha, N.; Christou, E.A. Force control is related to low-frequency oscillations in force and surface EMG. PLoS ONE 2014, 9, e109202. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.A.; Lundbye-Jensen, J.; Nielsen, J.B. Changes in corticospinal drive to spinal motorneurones following visuo-motor skill learning in humans. J. Physiol. 2006, 573, 843–855. [Google Scholar] [CrossRef] [PubMed]
- Onushko, T.; Baweja, H.S.; Christou, E.A. Practice improves motor control in older adults by increasing the motor unit modulation from 13 to 30 Hz. J. Neurophysiol. 2013, 110, 2393–2401. [Google Scholar] [CrossRef] [PubMed]
- Classen, J.; Gerloff, C.; Honda, M.; Hallett, M. Integrative visuomotor behavior is associated with interregionally coherent oscillations in the human brain. J. Neurophysiol. 1998, 79, 1567–1573. [Google Scholar] [PubMed]
- Brovelli, A.; Ding, M.; Ledberg, A.; Chen, Y.T.; Nakamura, R.; Bressler, S.L. Beta oscillations in a large-scale sensorimotor cortical network: Directional influences revealed by Granger causality. Proc. Natl. Acad. Sci. USA 2004, 101, 9849–9854. [Google Scholar] [CrossRef] [PubMed]
- Takarae, Y.; Minshew, N.J.; Luna, B.; Sweeney, J.A. Atypical involvement of frontostriatal systems during sensorimotor control in autism. Psychiatry Res. 2007, 156, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.; Muller, R.A.; Courchesne, E. Cerebellar function in autism: Functional magnetic resonance image activation during a simple motor task. Biol. Psychiatry 2004, 56, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Suresh, A.K.; Li, X.; Rymer, W.Z.; Suresh, N.L. Impaired motor unit control in paretic muscle post stroke assessed using surface electromyography: A preliminary report. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2012, 2012, 4116–4119. [Google Scholar] [PubMed]
- De Luca, C.J.; Mambrito, B. Voluntary control of motor units in human antagonist muscles: Coactivation and reciprocal activation. J. Neurophysiol. 1987, 58, 525–542. [Google Scholar] [PubMed]
- Negro, F.; Holobar, A.; Farina, D. Fluctuations in isometric muscle force can be described by one linear projection of low-frequency components of motor unit discharge rates. J. Physiol. 2009, 587, 5925–5938. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.L.; Blakemore, S.J.; Press, C. Atypical basic movement kinematics in autism spectrum conditions. Brain 2013, 136, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Kalra, L.; Swift, C.G.; Jackson, H.D. Psychomotor performance and antihypertensive treatment. Br. J. Clin. Pharmacol. 1994, 37, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Arora, E.; Khajuria, V.; Tandon, V.R.; Sharma, A.; Choudhary, N. Comparative evaluation of aliskiren, ramipril, and losartan on psychomotor performance in healthy volunteers: A preliminary report. Perspect. Clin. Res. 2014, 5, 190–194. [Google Scholar] [PubMed]
- Dumont, G.J.; de Visser, S.J.; Cohen, A.F.; van Gerven, J.M.; Biomarker Working Group of the German Association for Applied Human, P. Biomarkers for the effects of selective serotonin reuptake inhibitors (SSRIs) in healthy subjects. Br. J. Clin. Pharmacol. 2005, 59, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Schrijvers, D.; Maas, Y.J.; Pier, M.P.; Madani, Y.; Hulstijn, W.; Sabbe, B.G. Psychomotor changes in major depressive disorder during sertraline treatment. Neuropsychobiology 2009, 59, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Reilly, J.L.; Lencer, R.; Bishop, J.R.; Keedy, S.; Sweeney, J.A. Pharmacological treatment effects on eye movement control. Brain Cogn. 2008, 68, 415–435. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, D. Wechsler Abbreviated Scale of Intelligence, 1st ed.; Psychological Corporation: New York, NY, USA, 1999. [Google Scholar]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Lord, C.; Rutter, M.; DiLavore, P.C.; Risi, S.; Gotham, K.; Bishop, S. Autism Disgnostic Observation Schedule, 2nd ed.; Western Psychological Services: Torrance, CA, USA, 2012. [Google Scholar]
- Association, A.P. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Lord, C.; Rutter, M.; Le Couteur, A. Autism Diagnostic Interveiw-Revised: A revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J. Autism Dev. Disord. 1994, 24, 659–685. [Google Scholar] [CrossRef] [PubMed]
- Rutter, M.; Bailey, A.; Lord, C. The Social Communication Questionnaire: Manual, 1st ed.; Western Psychological Services: Torrance, CA, USA, 2003. [Google Scholar]
- Taylor, A.M.; Christou, E.A.; Enoka, R.M. Multiple features of motor unit activity influence force fluctuations during isometric contractions. J. Neurophysiol. 2003, 90, 1350–1361. [Google Scholar] [CrossRef] [PubMed]
- Kline, J.C.; De Luca, C.J. Error reduction in EMG signal decomposition. J. Neurophysiol. 2014, 112, 2718–2728. [Google Scholar] [CrossRef] [PubMed]
- Mottram, C.J.; Christou, E.A.; Meyer, F.G.; Enoka, R.M. Frequency modulation of motor unit discharge has task-dependent effects on fluctuations in motor output. J. Neurophysiol. 2005, 94, 2878–2887. [Google Scholar] [CrossRef] [PubMed]




| Control (n = 14) | Age | FSIQ | PIQ | VIQ | ||
| MVC | 0.399 | 0.590 * | 0.640 * | 0.477 | ||
| SD force | −0.576 * | 0.172 | 0.033 | 0.270 | ||
| Mean discharge rate | 0.462 | −0.175 | −0.067 | −0.235 | ||
| ASD (n = 17) | Age | FSIQ | PIQ | VIQ | ADOS.soc.com | ADOS.rrb |
| MVC | 0.787 ** | 0.296 | 0.310 | 0.230 | −0.709 ** | −0.058 |
| SD force | 0.143 | −0.300 | −0.108 | −0.453 | 0.026 | 0.329 |
| Mean discharge rate | −0.470 | −0.388 | −0.332 | −0.397 | 0.674 ** | 0.1474 |
| Demographic Characteristics | Control (n = 14) | ASD (n = 17) | t | p |
|---|---|---|---|---|
| Age (yr) | 19.57 ± 6.24 | 18.95 ± 7.14 | 0.067 | 0.798 |
| Range | 11–28 | 11–32 | ||
| % Male * | 85.7 (12/14) | 94.14 (16/17) | 0.576 | 0.425 |
| % Right-handed * | 92.9 (13/14) | 88.23 (15/17) | 0.653 | 0.422 |
| Verbal IQ | 112.62 ± 17.74 | 107.63 ± 17.14 | 0.589 | 0.449 |
| Range | 82–140 | 71–126 | ||
| Performance IQ | 112.69 ± 13.68 | 106.81 ± 17.68 | 0.965 | 0.335 |
| Range | 85–133 | 79–129 | ||
| Full-scale IQ | 114.77 ± 16.41 | 108.31 ± 18.34 | 0.975 | 0.449 |
| Range | 82–138 | 78–131 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Kwon, M.; Mohanty, S.; Schmitt, L.M.; White, S.P.; Christou, E.A.; Mosconi, M.W. Increased Force Variability Is Associated with Altered Modulation of the Motorneuron Pool Activity in Autism Spectrum Disorder (ASD). Int. J. Mol. Sci. 2017, 18, 698. https://doi.org/10.3390/ijms18040698
Wang Z, Kwon M, Mohanty S, Schmitt LM, White SP, Christou EA, Mosconi MW. Increased Force Variability Is Associated with Altered Modulation of the Motorneuron Pool Activity in Autism Spectrum Disorder (ASD). International Journal of Molecular Sciences. 2017; 18(4):698. https://doi.org/10.3390/ijms18040698
Chicago/Turabian StyleWang, Zheng, Minhyuk Kwon, Suman Mohanty, Lauren M. Schmitt, Stormi P. White, Evangelos A. Christou, and Matthew W. Mosconi. 2017. "Increased Force Variability Is Associated with Altered Modulation of the Motorneuron Pool Activity in Autism Spectrum Disorder (ASD)" International Journal of Molecular Sciences 18, no. 4: 698. https://doi.org/10.3390/ijms18040698





