Serum Metabolomic Profiling Identifies Characterization of Non-Obstructive Azoospermic Men
Abstract
:1. Introduction
2. Results
2.1. Demographics of Study Participants
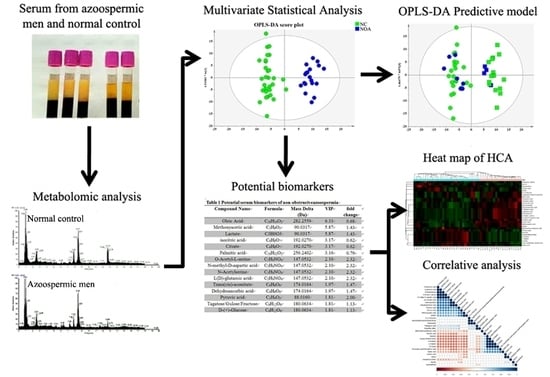
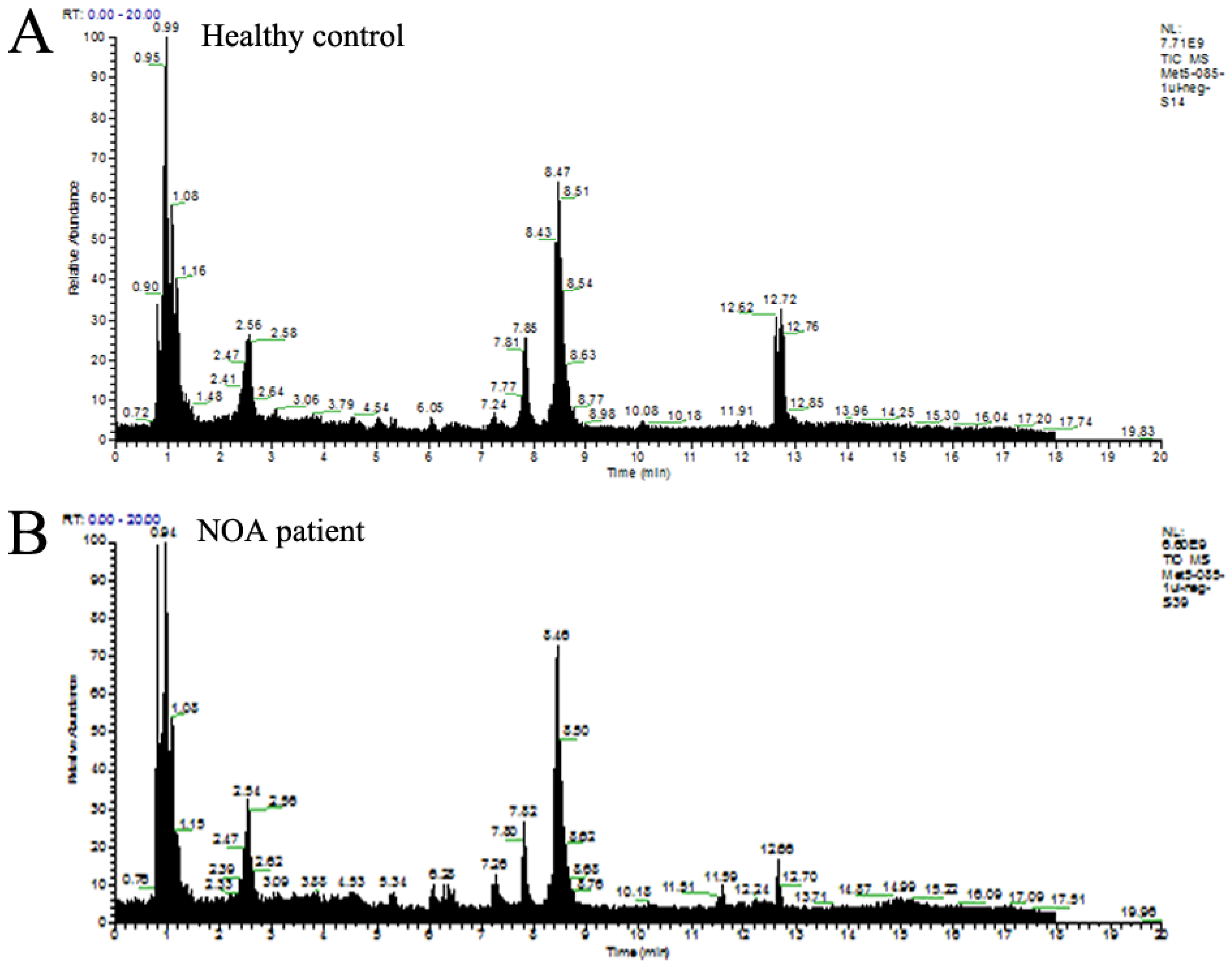
2.2. Multivariate Statistical Analysis of Metabolic Profiles
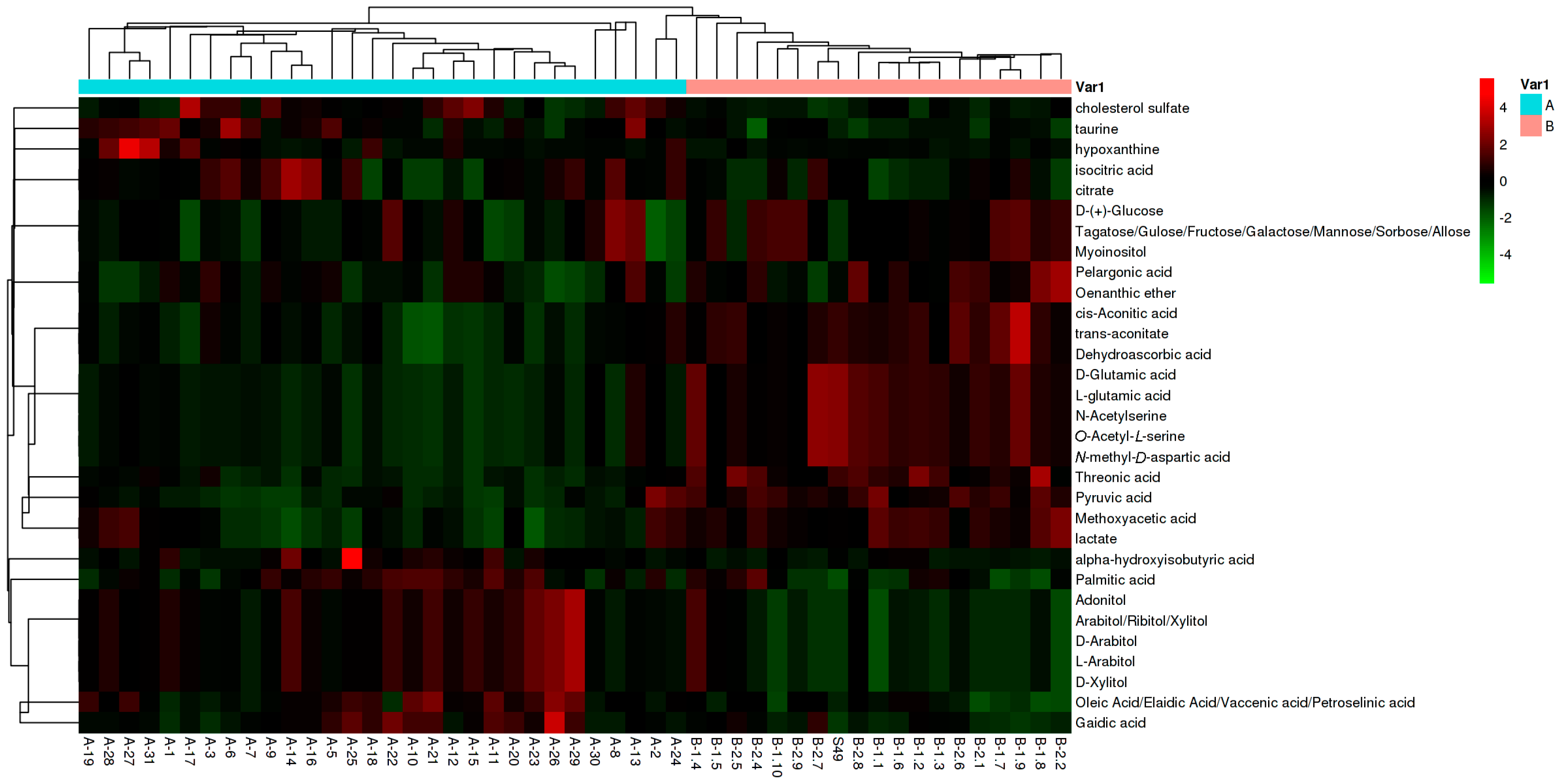
2.3. Identification of Potential Biomarkers in NOA
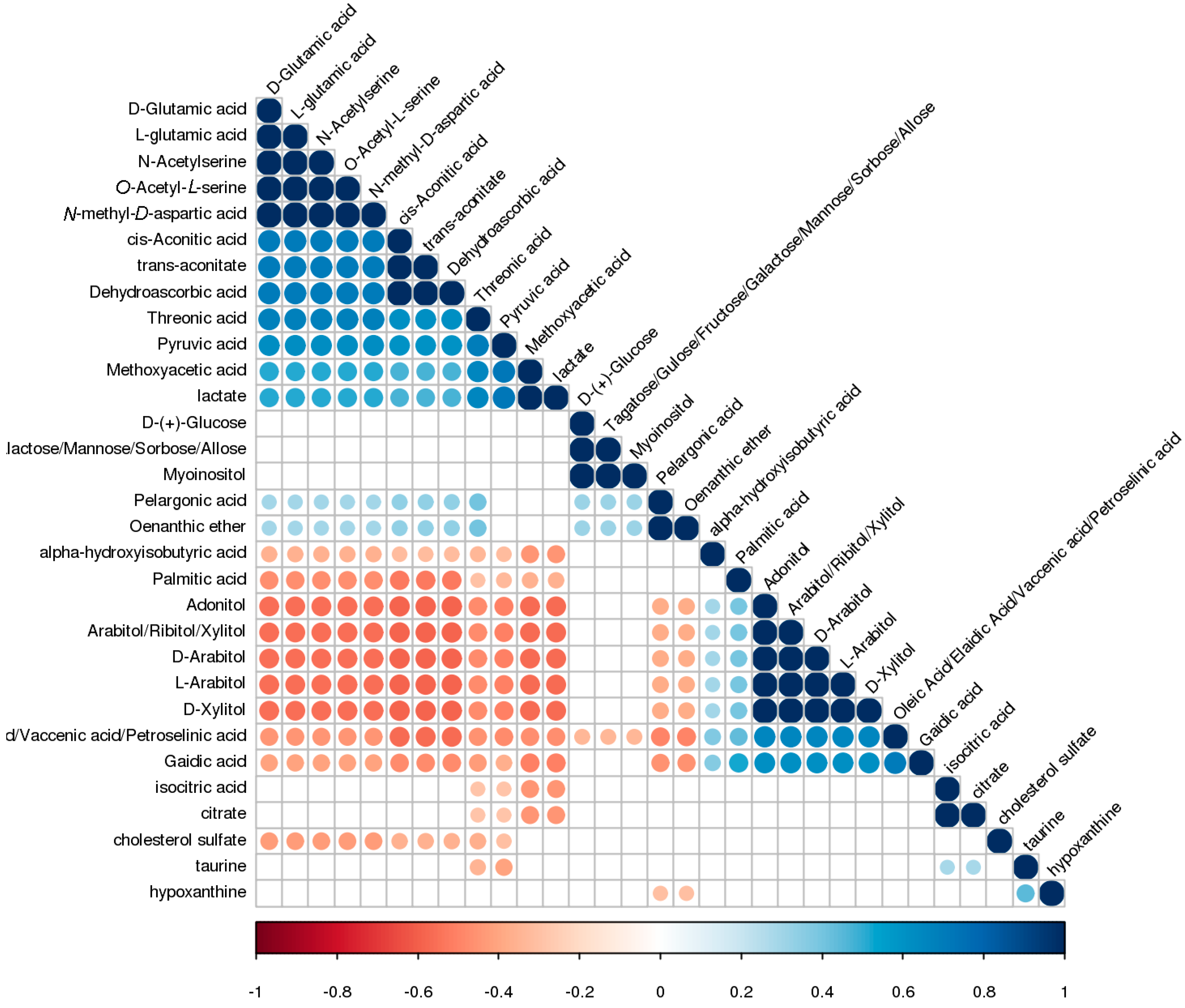
2.4. Metabolic Pathway and Linkages Analysis of Metabolites in NOA
3. Discussion
4. Materials and Methods
4.1. Study Design and Participant Recruitment
4.2. Serum Collection and Preparation
4.3. HPLC-MS/MS Analysis
4.4. Data Processing and Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ART | Assisted Reproductive Technology |
| FDR | False Discovery Rate |
| GPC | Glycerylphosphorylcholine |
| GPE | Glycerylphosphorylethanolamine |
| HCA | Hierarchical Clustering Analysis |
| HPLC-MS/MS | High-Performance Liquid Chromatography-tandem Mass Spectrometry |
| LH | Luteinizing Hormone |
| MA | Maturation Arrest |
| NOA | Non-obstructiveAzoospermia |
| OA | Obstructive Azoospermia |
| OPLS-DA | Orthogonal Partial Least Squares-Discriminant Analysis |
| PCA | Principal Component Analysis |
| PLS-DA | Partial Least Squares-Discriminant Analysis |
| ROS | Reactive Oxygen Species |
| VIP | Variable Importance in the Project |
References
- The Practice Committee of the American Society for Reproductive Medicine. Diagnostic evaluation of the infertile male: A committee opinion. Fertil. Steril. 2015, 103, e18–e25. [Google Scholar]
- Tournaye, H.; Krausz, C.; Oates, R.D. Concepts in diagnosis and therapy for male reproductive impairment. Lancet Diabetes Endocrinol. 2016. [Google Scholar] [CrossRef]
- Tebani, A.; Abily-Donval, L.; Afonso, C.; Marret, S.; Bekri, S. Clinical Metabolomics: The New Metabolic Window for Inborn Errors of Metabolism Investigations in the Post-Genomic Era. Int. J. Mol. Sci. 2016, 17, 1167. [Google Scholar] [CrossRef] [PubMed]
- Robles, A.I.; Harris, C.C. Integration of multiple “OMIC” biomarkers: A precision medicine strategy for lung cancer. Lung Cancer 2016. [Google Scholar] [CrossRef] [PubMed]
- Courant, F.; Antignac, J.P.; Monteau, F.; Le Bizec, B. Metabolomics as a potential new approach for investigating human reproductive disorders. J. Proteome Res. 2013, 12, 2914–2920. [Google Scholar] [CrossRef] [PubMed]
- Kovac, J.R.; Pastuszak, A.W.; Lamb, D.J. The use of genomics, proteomics, and metabolomics in identifying biomarkers of male infertility. Fertil. Steril. 2013, 99, 998–1007. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, Z.; Chen, M.; Xia, Y.; Martin, F.L.; Hang, W.; Shen, H. Urinary metabolome identifies signatures of oligozoospermic infertile men. Fertil. Steril. 2014. [Google Scholar] [CrossRef] [PubMed]
- Aaronson, D.S.; Iman, R.; Walsh, T.J.; Kurhanewicz, J.; Turek, P.J. A novel application of 1H magnetic resonance spectroscopy: Non-invasive identification of spermatogenesis in men with non-obstructive azoospermia. Hum. Reprod. 2010, 25, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, K.; Yagi, N.; Mizushima, K.; Higashimura, Y.; Hirai, Y.; Okayama, T.; Yoshida, N.; Katada, K.; Kamada, K.; Handa, O.; et al. Serum metabolomics analysis for early detection of colorectal cancer. J. Gastroenterol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Shackleton, C. Clinical steroid mass spectrometry: A 45-year history culminating in HPLC-MS/MS becoming an essential tool for patient diagnosis. J. Steroid Biochem. Mol. Biol. 2010, 121, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Boussouar, F.; Benahmed, M. Lactate and energy metabolism in male germ cells. Trends Endocrinol. Metab. 2004, 15, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Rato, L.; Alves, M.G.; Socorro, S.; Duarte, A.I.; Cavaco, J.E.; Oliveira, P.F. Metabolic regulation is important for spermatogenesis. Nat. Rev. Urol. 2012, 9, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mu, X.; Xia, Y.; Martin, F.L.; Hang, W.; Liu, L.; Tian, M.; Huang, Q.; Shen, H. Metabolomic analysis reveals a unique urinary pattern in normozoospermic infertile men. J. Proteome Res. 2014, 13, 3088–3099. [Google Scholar] [CrossRef] [PubMed]
- Hamamah, S.; Seguin, F.; Barthelemy, C.; Akoka, S.; Le Pape, A.; Lansac, J.; Royere, D. 1H nuclear magnetic resonance studies of seminal plasma from fertile and infertile men. J. Reprod. Fertil. 1993, 97, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Bonache, S.; Martinez, J.; Fernandez, M.; Bassas, L.; Larriba, S. Single nucleotide polymorphisms in succinate dehydrogenase subunits and citrate synthase genes: Association results for impaired spermatogenesis. Int. J. Androl. 2007, 30, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Tavilani, H.; Doosti, M.; Abdi, K.; Vaisiraygani, A.; Joshaghani, H.R. Decreased polyunsaturated and increased saturated fatty acid concentration in spermatozoa from asthenozoospermic males as compared with normozoospermic males. Andrologia 2006, 38, 173–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersen, J.M.; Ronning, P.O.; Herning, H.; Bekken, S.D.; Haugen, T.B.; Witczak, O. Fatty acid composition of spermatozoa is associated with BMI and with semen quality. Andrology 2016, 4, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, G.; Trocchia, S.; Abdel-Gawad, F.K.; Ciarcia, G. Roles of reactive oxygen species in the spermatogenesis regulation. Front. Endocrinol. 2014, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Ko, E.Y.; Sabanegh, E.S., Jr.; Agarwal, A. Male infertility testing: Reactive oxygen species and antioxidant capacity. Fertil. Steril. 2014, 102, 1518–1527. [Google Scholar] [CrossRef] [PubMed]
- Mayorga-Torres, B.J.; Camargo, M.; Cadavid, A.P.; du Plessis, S.S.; Cardona Maya, W.D. Are oxidative stress markers associated with unexplained male infertility? Andrologia 2016. [Google Scholar] [CrossRef] [PubMed]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sharma, R.K.; Sharma, R.; Assidi, M.; Abuzenadah, A.M.; Alshahrani, S.; Durairajanayagam, D.; Sabanegh, E. Characterizing semen parameters and their association with reactive oxygen species in infertile men. Reprod. Biol. Endocrinol. 2014, 12, 33. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zong, X.; Wu, G.; Lin, S.; Feng, Y.; Hu, J. Taurine increases testicular function in aged rats by inhibiting oxidative stress and apoptosis. Amino Acids 2015, 47, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wu, G.; Feng, Y.; Lv, Q.; Lin, S.; Hu, J. Effects of taurine on male reproduction in rats of different ages. J. Biomed. Sci. 2010, 17, S9. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, M.; Miura, C.; Iwai, T.; Miura, T. Trypsin regulates meiotic initiation in the Japanese eel (Anguilla japonica) by promoting the uptake of taurine into germ cells during spermatogenesis. Biol. Reprod. 2013, 89, 58. [Google Scholar] [CrossRef] [PubMed]
- Jindo, T.; Wine, R.N.; Li, L.H.; Chapin, R.E. Protein kinase activity is central to rat germ cell apoptosis induced by methoxyacetic acid. Toxicol. Pathol. 2001, 29, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Wade, M.G.; Kawata, A.; Williams, A.; Yauk, C. Methoxyacetic acid-induced spermatocyte death is associated with histone hyperacetylation in rats. Biol. Reprod. 2008, 78, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Wu, Z.; Hang, S.; Zhu, W.; Wu, G. Amino acid metabolism in intestinal bacteria and its potential implications for mammalian reproduction. Mol. Hum. Reprod. 2015, 21, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, S.; Volbracht, K.; Lundgaard, I.; Karadottir, R.T. Glutamate signalling: A multifaceted modulator of oligodendrocyte lineage cells in health and disease. Neuropharmacology 2016, 110, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Diao, R.; Zhu, X.; Li, Z.; Cai, Z. Metabolic characterization of asthenozoospermia using nontargeted seminal plasma metabolomics. Clin. Chim. Acta 2015, 450, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Strott, C.A.; Higashi, Y. Cholesterol sulfate in human physiology: What’s it all about? J. Lipid Res. 2003, 44, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Langlais, J.; Zollinger, M.; Plante, L.; Chapdelaine, A.; Bleau, G.; Roberts, K.D. Localization of cholesteryl sulfate in human spermatozoa in support of a hypothesis for the mechanism of capacitation. Proc. Natl. Acad. Sci. USA 1981, 78, 7266–7270. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.G.; Rato, L.; Carvalho, R.A.; Moreira, P.I.; Socorro, S.; Oliveira, P.F. Hormonal control of Sertoli cell metabolism regulates spermatogenesis. Cell. Mol. Life Sci. 2013, 70, 777–793. [Google Scholar] [CrossRef]
- Trumble, B.C.; Brindle, E.; Kupsik, M.; O’Connor, K.A. Responsiveness of the reproductive axis to a single missed evening meal in young adult males. Am. J. Hum. Biol. 2010, 22, 775–781. [Google Scholar] [CrossRef]
- Silva, F.R.; Leite, L.D.; Barreto, K.P.; D’Agostini, C.; Zamoner, A. Effect of 3,5,3′-triiodo-l-thyronine on amino acid accumulation and membrane potential in Sertoli cells of the rat testis. Life Sci. 2001, 69, 977–986. [Google Scholar] [CrossRef]
- Crown, A.; Clifton, D.K.; Steiner, R.A. Neuropeptide signaling in the integration of metabolism and reproduction. Neuroendocrinology 2007, 86, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Morgante, G.; Tosti, C.; Orvieto, R.; Musacchio, M.C.; Piomboni, P.; de Leo, V. Metformin improves semen characteristics of oligo-terato-asthenozoospermic men with metabolic syndrome. Fertil. Steril. 2011, 95, 2150–2152. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Wishart, D.S. MetPA: A web-based metabolomics tool for pathway analysis and visualization. Bioinformatics 2010, 26, 2342–2344. [Google Scholar] [CrossRef] [PubMed]


| Clinical Features | NC | NOA |
|---|---|---|
| Total number | 31 | 22 |
| Age (Years, Mean ± SD) | 28.1 ± 4.5 | 30.6 ± 4.3 |
| Testosterone < 14 nmol/L | NA | 2 |
| FSH > 11.1 mIU/mL | NA | 9 |
| Biopsy findings | NA | |
| Maturation arrest | 12 | |
| Sertoli-cell only | 10 | |
| Cytology findings | NA | |
| Spermatids or sperm | 5 |
| Compound Name | Formula | KEGG ID | Mass ∆ (Da) | VIP | Fold Change | Retention Time (min) | Mass Error (ppm) | Mass Library Score |
|---|---|---|---|---|---|---|---|---|
| Oleic acid | C18H34O2 | C00712 | 282.2559 | 6.33 | 0.68 | 0.9891 | 0 | 30 |
| Lactate/Methoxyacetic acid | C3H6O3 | C01432/NA | 90.0317 | 5.87 | 1.43 | 7.2385 | 6 | 83 |
| Citrate/isocitric acid | C6H8O7 | C00158/C00311 | 192.0270 | 3.17 | 0.62 | 12.9001 | 0 | 100 |
| Palmitic acid | C16H32O2 | C00249 | 256.2402 | 3.16 | 0.79 | 0.9891 | 0 | NA |
| O-Acetyl-l-serine/N-methyl-d-aspartic acid/N-Acetylserine/l(d)-glutamic acid | C5H9NO4 | C00979/C12269/NA/C00025 | 147.0532 | 2.10 | 2.32 | 11.1536 | 1 | 70 |
| Trans(cis)-aconitate/Dehydroascorbicacid | C6H6O6 | C02341/C05422 | 174.0164 | 1.97 | 1.47 | 11.7449 | 0 | 83 |
| Pyruvic acid | C3H4O3 | C00022 | 88.0160 | 1.81 | 2.00 | 4.9982 | 6 | 67 |
| Tagatose/Gulose/Fructose/d-(+)-Glucose | C6H12O6 | C00795/C00267 | 180.0634 | 1.81 | 1.13 | 8.4792 | 0 | 97 |
| Threonic acid | C4H8O5 | NA | 136.0372 | 1.36 | 2.05 | 9.2863 | 0 | 66 |
| Gaidic acid | C16H30O2 | NA | 254.2246 | 1.35 | 0.61 | 1.21 | 0 | 44 |
| Taurine | C2H7NO3S | C00245 | 125.0147 | 1.33 | 0.65 | 8.3418 | 2 | NA |
| Pelargonic acid/Oenanthic ether | C9H18O2 | C01601/NA | 158.1307 | 1.26 | 1.34 | 1.4672 | 0 | 31 |
| Cholesterol sulfate | C27H46O4S | C18043 | 466.3117 | 1.24 | 0.57 | 0.8231 | 0 | 60 |
| Hypoxanthine | C5H4N4O | C00262 | 136.0385 | 1.09 | 0.34 | 4.8046 | 0 | 69 |
| Arabitol/Ribitol/Xylitol | C5H12O5 | C00532 | 152.0685 | 1.07 | 0.63 | 2.8157 | 0 | 22 |
| α-hydroxyisobutyric acid | C4H8O3 | C01188 | 104.0473 | 1.04 | 0.47 | 7.1775 | 4 | 78 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Zhang, Y.; Liu, C.; Zhao, M.; Yang, Y.; Wu, H.; Zhang, H.; Lin, H.; Zheng, L.; Jiang, H. Serum Metabolomic Profiling Identifies Characterization of Non-Obstructive Azoospermic Men. Int. J. Mol. Sci. 2017, 18, 238. https://doi.org/10.3390/ijms18020238
Zhang Z, Zhang Y, Liu C, Zhao M, Yang Y, Wu H, Zhang H, Lin H, Zheng L, Jiang H. Serum Metabolomic Profiling Identifies Characterization of Non-Obstructive Azoospermic Men. International Journal of Molecular Sciences. 2017; 18(2):238. https://doi.org/10.3390/ijms18020238
Chicago/Turabian StyleZhang, Zhe, Yingwei Zhang, Changjie Liu, Mingming Zhao, Yuzhuo Yang, Han Wu, Hongliang Zhang, Haocheng Lin, Lemin Zheng, and Hui Jiang. 2017. "Serum Metabolomic Profiling Identifies Characterization of Non-Obstructive Azoospermic Men" International Journal of Molecular Sciences 18, no. 2: 238. https://doi.org/10.3390/ijms18020238