Zinc as Allosteric Ion Channel Modulator: Ionotropic Receptors as Metalloproteins
Abstract
:1. Introduction
1.1. Zinc Background and Relevant Chemical Principles
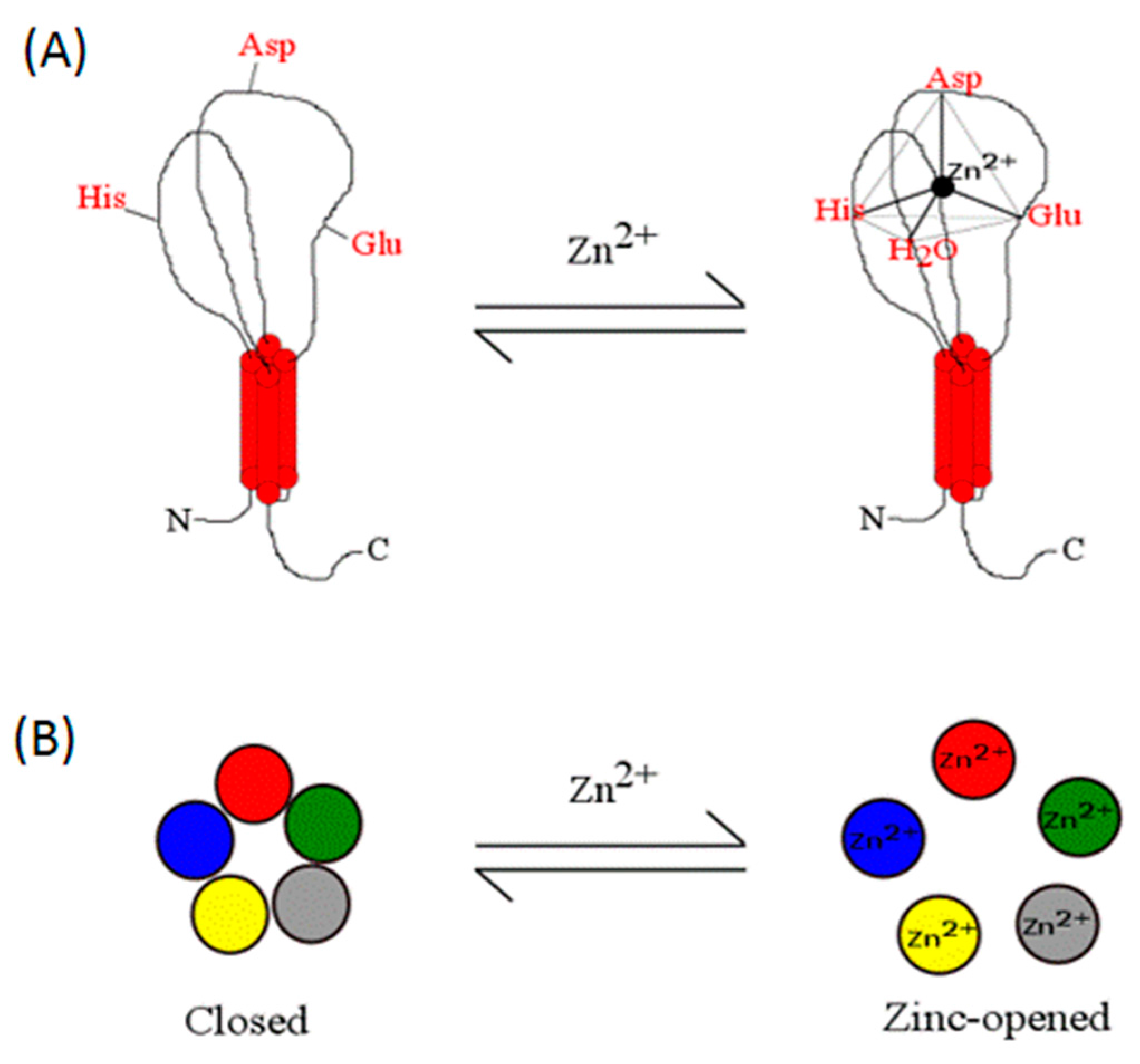
1.2. Zinc Coordination in Proteins
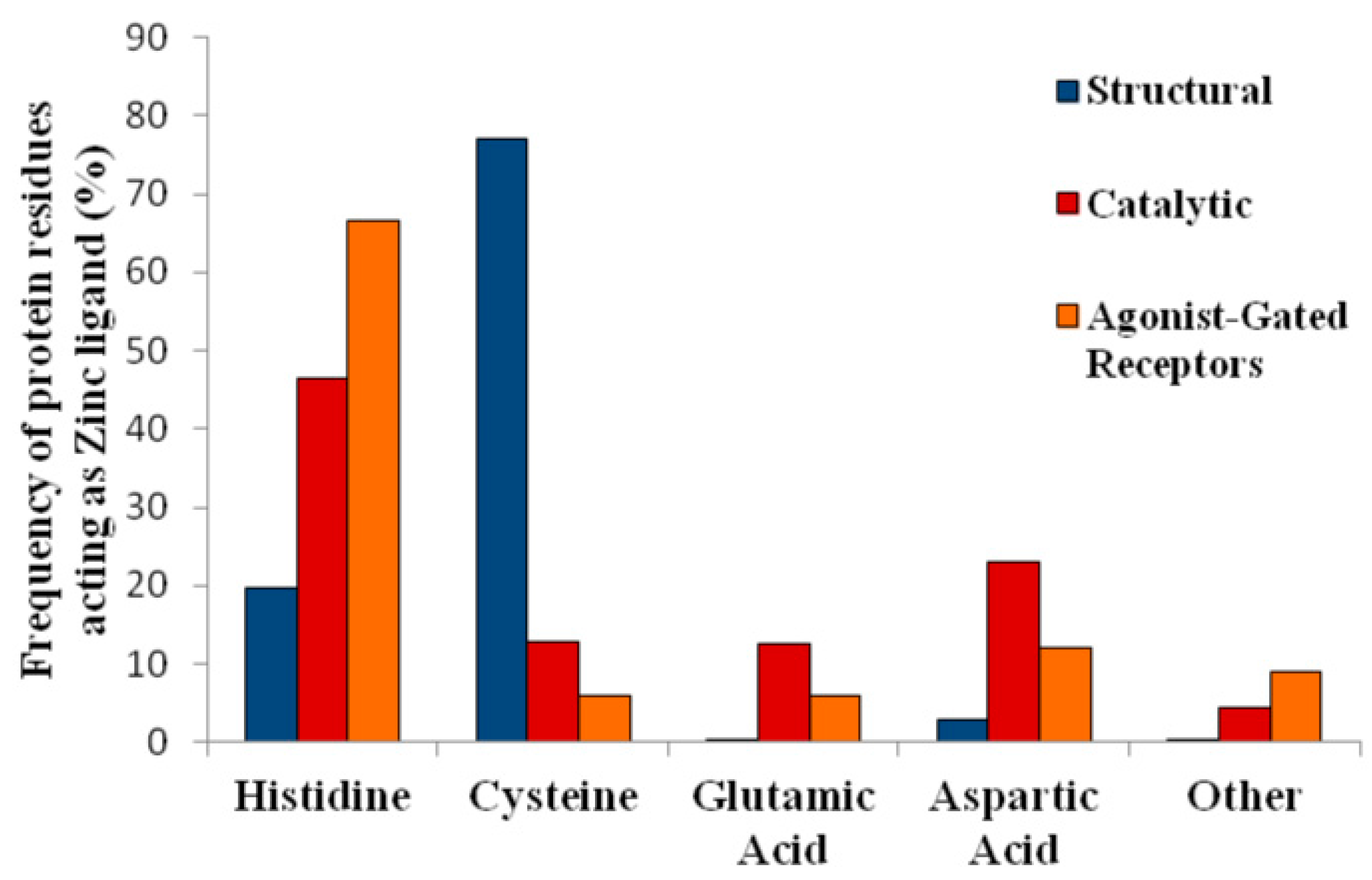
1.3. Zinc Coordination in Proteins: Residues Involved in Structural versus Catalytic Sites
1.4. Supporting Physicochemical Principles for the Zinc Coordination Sphere
2. Significance of Zinc Homeostasis; Role of Metallothioneines in Human Metal Determinations
2.1. Zinc Storage Mediated by Multiple Metal Transporters
2.1.1. The ZnT Family of Carriers
2.1.2. The Zinc ZIP Transporter Family
3. Allosteric Modulation: A Regulatory Mode in Proteins and Receptor Channels
4. Ion Channels
4.1. Zinc-Induced Modulation of Voltage-Gated Channels
4.1.1. K+ Channels
4.1.2. Na+ Channels
4.1.3. Ca2+ Channels
4.1.4. H+ Channels
4.1.5. Cl− Channels
4.2. Zinc-Induced Modulation of Agonist-Gated Receptors
4.2.1. GABA-A Receptors
4.2.2. Glycine Receptors
4.2.3. Nicotinic Ach Receptors
4.2.4. 5-HT3 Receptors
4.2.5. NMDA and Related Glutamate Receptors
4.2.6. H+ and ENAC, Na+ Receptors, Belonging to the Trimer Receptor Family
Protons Activate the Acid-Sensitive Cationic Channels (ASICs), Widespread in the Body
The ENaC Receptor
P2X Purinoceptors
Transient Receptor Potential Channels (TRPs)
The Special Case of Intercellular or Hemichannels
5. Deciphering the Zinc Binding Sites in Receptor-Gated Channels
5.1. Identification of Strategic Amino Acid Residues of the Metal Coordination Sphere
5.2. Structural Evidence in Agonist-Gated Ionotropic Receptors
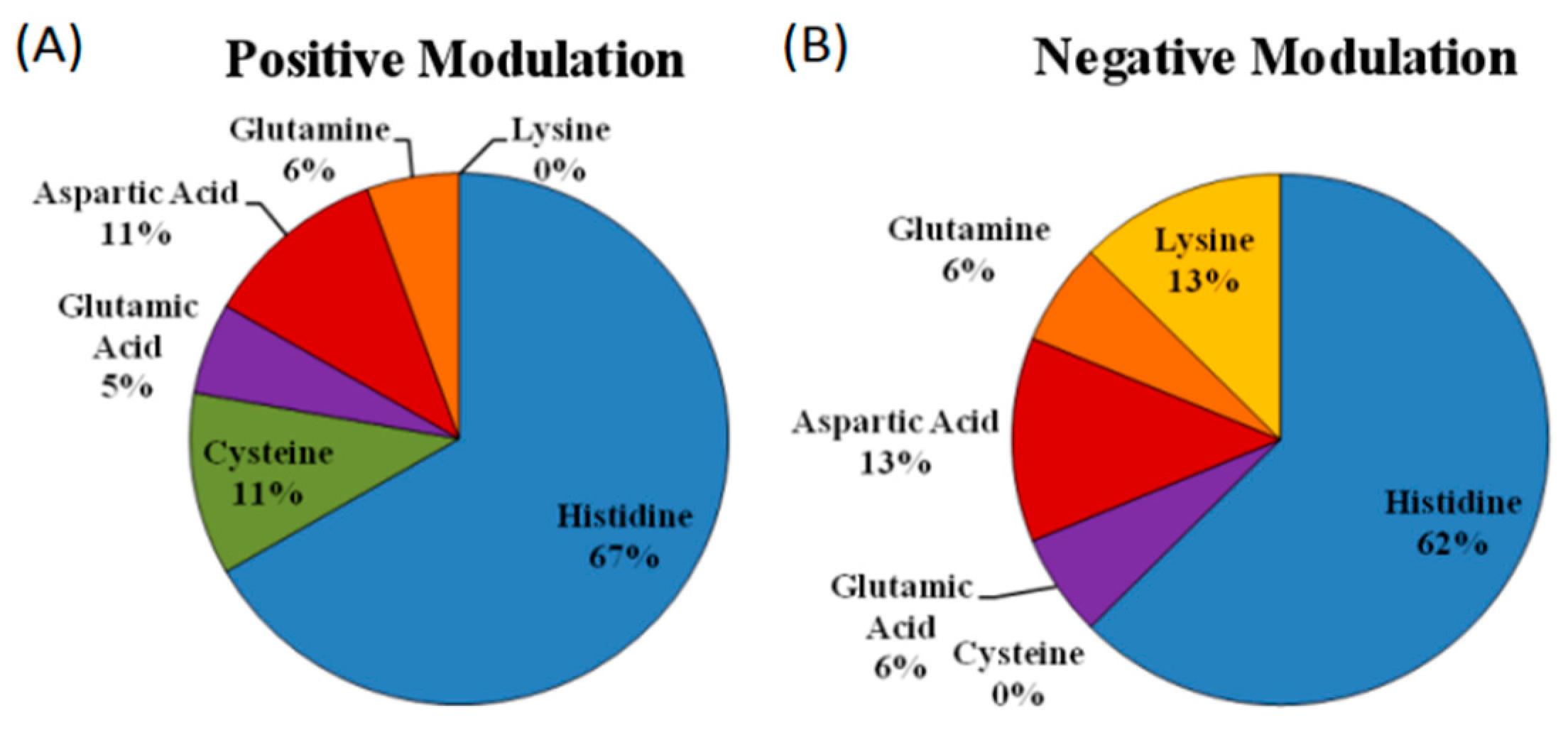
5.3. Is There a Difference in the Zinc Coordination Sphere When Comparing Positive and Negative Allosteric Modulators?
6. Zinc as a Ligand of Ionotropic Receptor Channels
7. Zinc-Associated Pathologies: A Link between Channel Receptors and Zinc Modulation
8. Biological Implications and Future Perspectives
9. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maret, W.; Li, Y. Coordination dynamics of zinc in proteins. Chem. Rev. 2009, 109, 4682–4707. [Google Scholar] [CrossRef] [PubMed]
- Coddou, C.; Yan, Z.; Obsil, T.; Huidobro-Toro, J.P.; Stojilkovic, S.S. Activation and regulation of purinergic P2X receptor channels. Pharmacol. Rev. 2011, 63, 641–683. [Google Scholar] [CrossRef] [PubMed]
- Elinder, F.; Rhem, P. Metal ion effects on ion channel gating. Q. Rev. Biophys. 2003, 36, 373–427. [Google Scholar] [CrossRef] [PubMed]
- Coddou, C.; Stojilkovic, S.S.; Huidobro-Toro, J.P. Allosteric modulation of ATP-gated P2X receptor channels. Rev. Neurosci. 2011, 22, 335–354. [Google Scholar] [CrossRef] [PubMed]
- Andreini, C.; Bertini, I.; Cavallaro, G. Minimal functional sites allow a classification of zinc sites in proteins. PLoS ONE 2011, 6, e26325. [Google Scholar] [CrossRef] [PubMed]
- Auld, D.S. Zinc Biochemistry, Physiology, and Homeostasis. In Zinc Coordination Sphere in Biochemical Zinc Sites; Springer Netherlands: New Youk, NY, USA, 2001; pp. 85–127. [Google Scholar]
- Kambe, T.; Tsuji, T.; Hashimoto, A.; Itsumura, N. The physiological, biochemical, and molecular roles of zinc transporters in zinc homeostasis and metabolism. Physiol. Rev. 2015, 95, 749–784. [Google Scholar] [CrossRef] [PubMed]
- Laitaoja, M.; Valjakka, J.; Jänis, J. Zinc coordination spheres in protein structures. Inorg. Chem. 2013, 52, 10983–10991. [Google Scholar] [CrossRef] [PubMed]
- Sousa, S.F.; Lopes, A.B.; Fernandes, P.A.; Ramos, M.J. The Zinc proteome: A tale of stability and functionality. Dalton Trans. 2009, 38, 7946–7956. [Google Scholar] [CrossRef] [PubMed]
- Colvin, R.A.; Holmes, W.R.; Fontaine, C.P.; Maret, W. Cytosolic zinc buffering and muffling: Their role in intracellular zinc homeostasis. Metallomics 2010, 2, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Baran, E.J. Química Bioinorgánica, 1st ed.; McGraw-Hill: Madrid, Spain, 1995; pp. 3–10. [Google Scholar]
- Herrmann, A.G.; Blanchard, D.P.; Haskin, L.A.; Jacobs, J.W.; Knake, D.; Korotev, R.L.; Brannon, J.C. Major, minor, and trace element compositions of peridotitic and basaltic komatiites from the precambrian crust of Southern Africa. Contrib. Mineral. Petrol. 1976, 59, 1–12. [Google Scholar] [CrossRef]
- Ekpo, B.O.; Ibok, U.J. Seasonal variation and partition of trace metals (Fe, Zn, Cu, Mn, Cr, Cd and Pb) in surface sediments: relationship with physico-chemical variables of water from the Calabar River, South Eastern Nigeria. Environ. Geochem. Health 1998, 20, 113–121. [Google Scholar] [CrossRef]
- Fisher, S.Z.; Kovalevsky, A.Y.; Domsic, J.F.; Mustyakimov, M.; McKenna, R.; Silverman, D.N.; Langan, P.A. Neutron structure of human carbonic anhydrase II: Implications for proton transfer. Biochemistry 2010, 49, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Omichinski, J.G.; Clore, G.M.; Robien, M.; Sakaguchi, K.; Appella, E.; Gronenborn, A.M. High-resolution solution structure of the double Cys2His2 zinc finger from the human enhancer binding protein MBP-1. Biochemistry 1992, 31, 3907–3917. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.D.; Pangborn, W.A.; Blessing, R.H. The structure of T 6 human insulin at 1.0 Å resolution. Acta Crystallogr. Sect. D 2003, 59, 474–482. [Google Scholar] [CrossRef]
- Hallman, P.S.; Perrin, D.D.; Watt, A.E. The computed distribution of copper(II) and zinc(II) ions among seventeen amino acids present in human blood plasma. Biochem. J. 1971, 121, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Alberts, I.L.; Nadassy, K.; Wodak, S.J. Analysis of zinc binding sites in protein crystal structures. Protein Sci. 1998, 7, 1700–1716. [Google Scholar] [CrossRef] [PubMed]
- Tamames, B.; Sousa, S.F.; Tamames, J.; Fernandes, P.A.; Ramos, M.J. Analysis of zinc-ligand bond lengths in metalloproteins: Trends and patterns. Proteins Struct. Funct. Genet. 2007, 69, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Veran, J.; Kumar, J.; Pinheiro, P.S.; Athané, A.; Mayer, M.L.; Perrais, D.; Mulle, C. Zinc potentiates GluK3 glutamate receptor function by stabilizing the ligand binding domain dimer interface. Neuron 2012, 76, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, B.; Mihalak, K.B.; Repicky, S.E.; Everhart, D.; Mederos, A.H.; Malhotra, A.; Luetje, C.W. Determinants of zinc potentiation on the alpha4 subunit of neuronal nicotinic receptors. Mol. Pharmacol. 2006, 69, 27–36. [Google Scholar] [PubMed]
- Harvey, R.J.; Thomas, P.; James, C.H.; Wilderspin, A.; Smart, T.G. Identification of an inhibitory Zn2+ binding site on the human glycine receptor α1 subunit. J. Physiol. 1999, 520(Pt. 1), 53–64. [Google Scholar] [PubMed]
- Baron, A.; Schaefer, L.; Lingueglia, E.; Champigny, G.; Lazdunski, M. Zn2+ and H+ are Coactivators of Acid-sensing Ion Channels. J. Biol. Chem. 2001, 276, 35361–35367. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Winarski, K.L.; Myerburg, M.M.; Pitt, B.R.; Sheng, S. Probing the Structural Basis of Zn2+ Regulation of the Epithelial Na+ Channel. J. Biol. Chem. 2012, 287, 35589–35598. [Google Scholar] [CrossRef] [PubMed]
- Clyne, J.D.; LaPointe, L.D.; Hume, R.I. The role of histidine residues in modulation of the rat P2X2 purinoceptor by zinc and pH. J. Physiol. 2002, 539, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Tittle, R.K.; Saar, N.; Dellal, S.S.; Hume, R.I. An intersubunit zinc binding site in rat P2X2 receptors. J. Biol. Chem. 2005, 280, 25982–25993. [Google Scholar] [CrossRef] [PubMed]
- Coddou, C.; Acuña-Castillo, C.; Bull, P.; Huidobro-Toro, J.P. Dissecting the facilitator and inhibitor allosteric metal sites of the P2X4 receptor channel: Critical roles of CYS132 for zinc potentiation and ASP138 for copper inhibition. J. Biol. Chem. 2007, 282, 36879–36886. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Xiong, K.M.; Wu, Y.X.; Liu, Y.W.; Chen, L.; Stewart, R.R.; Peoples, R.W.; Yi, C.L. Conserved extracellular cysteines differentially regulate the potentiation produced by Zn2+ in rat P2X4 receptors. Eur. J. Pharmacol. 2013, 707, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Fayyazuddin, A.; Villarroel, A.; Le Goff, A.; Lerma, J.; Neyton, J. Four residues of the extracellular N-terminal domain of the NR2A subunit control high-affinity Zn2+ binding to NMDA receptors. Neuron 2000, 25, 683–694. [Google Scholar] [CrossRef]
- Zheng, F.; Erreger, K.; Low, C.M.; Banke, T.; Lee, C.J.; Conn, P.J.; Traynelis, S.F. Allosteric interaction between the amino terminal domain and the ligand binding domain of NR2A. Nat. Neurosci. 2001, 4, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-L.; Hackam, A.; Guggino, W.B.; Cutting, G.R. A Single histidine residue is essential for zinc inhibition of GABA. J. Neurosci. 1995, 15, 7684–7691. [Google Scholar] [PubMed]
- Chu, X.; Wemmie, J.A.; Wang, W.; Zhu, X.; Saugstad, J.A.; Price, M.P.; Simon, R.P.; Xiong, Z. Subunit-dependent high-affinity zinc inhibition of acid-sensing ion channels. J. Neurosci. 2004, 24, 8678–8689. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Surprenant, A.; Mao, H.-J.; Roger, S.; Xia, R.; Bradley, H.; Jiang, L.-H. Identification of key residues coordinating functional inhibition of P2X7 receptors by zinc and copper. Mol. Pharmacol. 2008, 73, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Acuña-Castillo, C.; Coddou, C.; Bull, P.; Brito, J.; Huidobro-Toro, J.P. Differential role of extracellular histidines in copper, zinc, magnesium and proton modulation of the P2X7 purinergic receptor. J. Neurochem. 2007, 101, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Amin, E.A.; Truhlar, D.G. Zn coordination chemistry: Development of benchmark suites for geometries, dipole moments, and bond dissociation energies and their use to test and validate density functionals and molecular orbital theory. J. Chem. Theory Comput. 2008, 4, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Trzaskowski, B.; Adamowicz, L.; Deymier, P.A. A theoretical study of zinc(II) interactions with amino acid models and peptide fragments. J. Biol. Inorg. Chem. 2008, 13, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Kochańczyk, T.; Drozd, A.; Krężel, A. Relationship between the architecture of zinc coordination and zinc binding affinity in proteins—Insights into zinc regulation. Metallomics 2015, 7, 244–257. [Google Scholar] [CrossRef] [PubMed]
- Burgess, J.; Prince, R.H. Zinc: Inorganic & Coordination Chemistry. In Encyclopedia of Inorganic and Bioinorganic Chemistry; John Wiley & Sons: Chichester, UK, 2011. [Google Scholar]
- Burdette, S.C.; Lippard, S.J. Meeting of the minds: Metalloneurochemistry. Proc. Natl. Acad. Sci. USA 2003, 100, 3605–3610. [Google Scholar] [CrossRef] [PubMed]
- Hambidge, K.M.; Casey, C.E.; Krebs, N.F. Zinc. In Trace Elements in Human and Animal Nutrition; Mertz, W., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 1986; pp. 1–137. [Google Scholar]
- Dawson, J.B.; Walker, B.E. Direct determination of zinc in whole blood, plasma and urine by atomic absorption spectroscopy. Clin. Chim. Acta 1969, 26, 465–475. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Koh, J.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Marger, L.; Schubert, C.R.; Bertrand, D. Zinc: An underappreciated modulatory factor of brain function. Biochem. Pharmacol. 2014, 91, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Maret, W.; Vallee, B.L. Differential fluorescence labeling of cysteinyl clusters uncovers high tissue levels of thionein. Proc. Natl. Acad. Sci. USA 2001, 98, 5556–5559. [Google Scholar] [CrossRef] [PubMed]
- Colvin, R.A.; Lai, B.; Holmes, W.R.; Lee, D. Understanding metal homeostasis in primary cultured neurons. Studies using single neuron subcellular and quantitative metallomics. Metallomics 2015, 7, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Aedo, F.; Delgado, R.; Wolff, D.; Vergara, C. Copper and zinc as modulators of neuronal excitability in a physiologically significant concentration range. Neurochem. Int. 2007, 50, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Koeberl, C.; Bayer, P.M.; Hobarth, K. Determination of Rare earth and other trace element abundances in human kidney stones and brain tissue. J. Radioanal. Nucl. Chem. Artic. 1993, 169, 269–276. [Google Scholar] [CrossRef]
- Vanhoe, H.; Vandecasteele, C.; Versieck, J.; Dams, R. Determination of iron, cobalt, copper, zinc, rubidium, molybdenum, and cesium in human serum by inductively coupled plasma mass spectrometry. Anal. Chem. 1989, 61, 1851–1857. [Google Scholar] [CrossRef] [PubMed]
- Bouron, A.; Kiselyov, K.; Oberwinkler, J. Permeation, regulation and control of expression of TRP channels by trace metal ions. Pflügers Arch. 2015, 467, 1143–1164. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Kambe, T. The Functions of metallothionein and ZIP and ZnT transporters: An overview and perspective. Int. J. Mol. Sci. 2016, 17, 336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, E.D. Cellular transporters for zinc. Nutr. Rev. 2002, 60, 121–124. [Google Scholar] [PubMed]
- Palmiter, R.D.; Findley, S.D. Cloning and functional characterization of a mammalian zinc transporter that confers resistance to zinc. EMBO J. 1995, 14, 639–649. [Google Scholar] [PubMed]
- Palmiter, R.D.; Cole, T.B.; Findley, S.D. ZnT-2, a mammalian protein that confers resistance to zinc by facilitating vesicular sequestration. EMBO J. 1996, 15, 1784–1791. [Google Scholar] [PubMed]
- Palmiter, R.D.; Cole, T.B.; Quaife, C.J.; Findley, S.D. ZnT-3, a putative transporter of zinc into synaptic vesicles. Proc. Natl. Acad. Sci. USA 1996, 93, 14934–14939. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Gitschier, J. A novel gene involved in zinc transport is deficient in the lethal milk mouse. Nat. Genet. 1997, 17, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Kambe, T.; Narita, H.; Yamaguchi-Iwai, Y.; Hirose, J.; Amano, T.; Sugiura, N.; Sasaki, R.; Mori, K.; Iwanaga, T.; Nagao, M. Cloning and characterization of a novel mammalian zinc transporter, zinc transporter 5, abundantly expressed in pancreatic β cells. J. Biol. Chem. 2002, 277, 19049–19055. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Kirschke, C.P.; Gitschier, J. Functional characterization of a novel mammalian zinc transporter, ZnT6. J. Biol. Chem. 2002, 277, 26389–26395. [Google Scholar] [CrossRef] [PubMed]
- Kirschke, C.P.; Huang, L. ZnT7, a novel mammalian zinc transporter, accumulates zinc in the Golgi apparatus. J. Biol. Chem. 2003, 278, 4096–4102. [Google Scholar] [CrossRef] [PubMed]
- Chimienti, F.; Devergnas, S.; Favier, A.; Seve, M. Identification and cloning of a cell-specific zinc transporter, ZnT-8, localized into insulin secretory granules. Diabetes 2004, 53, 2330–2337. [Google Scholar] [CrossRef] [PubMed]
- Bosomworth, H.J.; Thornton, J.K.; Coneyworth, L.J.; Ford, D.; Valentine, R.A. Efflux function, tissue-specific expression and intracellular trafficking of the Zn transporter ZnT10 indicate roles in adult Zn homeostasis. Metallomics 2012, 4, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Grotz, N.; Fox, T.; Connolly, E.; Park, W.; Guerinot, M.L.; Eide, D. Identification of a family of zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc. Natl. Acad. Sci. USA 1998, 95, 7220–7224. [Google Scholar] [CrossRef] [PubMed]
- Eng, B.H.; Guerinot, M.L.; Eide, D.; Saier, M.H. Sequence analyses and phylogenetic characterization of the ZIP family of metal ion transport proteins. J. Membr. Biol. 1998, 166, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kambe, T.; Yamaguchi-Iwai, Y.; Sasaki, R.; Nagao, M. Overview of mammalian zinc transporters. Cell. Mol. Life Sci. 2004, 61, 49–68. [Google Scholar] [CrossRef] [PubMed]
- Gerhart, J.C.; Pardee, A.B. The enzymology of control by feedback inhibition. J. Biol. Chem. 1962, 237, 891–896. [Google Scholar] [PubMed]
- Monod, J.; Changeux, J.P.; Jacob, F. Allosteric proteins and cellular control systems. J. Mol. Biol. 1963, 6, 306–329. [Google Scholar] [CrossRef]
- Sigel, E.; Baur, I. Allosteric modulation by benzodiazepine receptor ligands GABAA RECEPTOR channel expressed in Xenopus oocytes. J. Neurosci. 1988, 8, 289–295. [Google Scholar] [PubMed]
- Monod, J.; Wyman, J.; Changeux, J.P. On the nature of allosteric transitions: A plausible model. J. Mol. Biol. 1965, 12, 88–118. [Google Scholar] [CrossRef]
- Acuña-Castillo, C.; Morales, B.; Huidobro-Toro, J.P. Zinc and copper modulate differentially the P2X4 receptor. J. Neurochem. 2000, 74, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Coddou, C.; Morales, B.; González, J.; Grauso, M.; Gordillo, F.; Bull, P.; Rassendren, F.; Huidobro-Toro, J.P. Histidine 140 plays a key role in the inhibitory modulation of the P2X4 nucleotide receptor by copper but not zinc. J. Biol. Chem. 2003, 278, 36777–36785. [Google Scholar] [CrossRef] [PubMed]
- Huidobro-Toro, J.P.; Lorca, R.A.; Coddou, C. Trace metals in the brain: Allosteric modulators of ligand-gated receptor channels, the case of ATP-gated P2X receptors. Eur. Biophys. J. 2008, 37, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Zhu, R.; Zhu, L.; Qiu, T.; Cao, Z.; Kang, T. Potassium channels: Structures, diseases, and modulators. Chem. Biol. Drug Des. 2014, 83, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Jensen, H.S.; Callø, K.; Jespersen, T.; Jensen, B.S.; Olesen, S.P. The KCNQ5 potassium channel from mouse: A broadly expressed M-current like potassium channel modulated by zinc, pH, and volume changes. Mol. Brain Res. 2005, 139, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Park, J.; Kang, H.; Lee, E.-J.; Bang, H.; Lee, J. Zinc activates TREK-2 potassium channel activity. J. Pharmacol. Exp. Ther. 2005, 314, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Spires, S.; Begenisich, T. Modulation of potassium channel gating by external divalent cations. J. Gen. Physiol. 1994, 104, 675–692. [Google Scholar] [CrossRef] [PubMed]
- Harrison, N.L.; Radke, H.K.; Tamkun, M.M.; Lovinger, D.M. Modulation of gating of cloned rat and human K+ channels by micromolar Zn2+. Mol. Pharmacol. 1993, 43, 482–486. [Google Scholar] [PubMed]
- Zhang, S.; Kehl, S.J.; Fedida, D. Modulation of Kv1.5 potassium channel gating by extracellular zinc. Biophys. J. 2001, 81, 125–136. [Google Scholar] [CrossRef]
- Gruss, M.; Mathie, A.; Lieb, W.R.; Franks, N.P. The two-pore-domain K+ channels TREK-1 and TASK-3 are differentially modulated by copper and zinc. Mol. Pharmacol. 2004, 66, 530–537. [Google Scholar] [PubMed]
- Hou, S.; Vigeland, L.E.; Zhang, G.; Xu, R.; Li, M.; Heinemann, S.H.; Hoshi, T. Zn2+ activates large conductance Ca2+-activated K+ channel via an intracellular domain. J. Biol. Chem. 2010, 285, 6434–6442. [Google Scholar] [CrossRef] [PubMed]
- Gilly, W.F.; Armstrong, C.M. Slowing of sodium channel opening kinetics in squid axon by extracellular zinc. J. Gen. Physiol. 1982, 79, 935–964. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-C.; Chen, W.-Y.; Yang, Y.-C. Block of tetrodotoxin-resistant Na+ channel pore by multivalent cations: Gating modification and Na+ flow dependence. J. Gen. Physiol. 2004, 124, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Simms, B.A.; Zamponi, G.W. Neuronal voltage-gated calcium channels: Structure, function, and dysfunction. Neuron 2014, 82, 24–45. [Google Scholar] [CrossRef] [PubMed]
- Traboulsie, A.; Chemin, J.; Chevalier, M.; Quignard, J.-F.; Nargeot, J.; Lory, P. Subunit-specific modulation of T-type calcium channels by zinc. J. Physiol. 2007, 578, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.-S.; Hui, K.; Lee, D.W.K.; Feng, Z.-P. Zn2+ sensitivity of high- and low-voltage activated calcium channels. Biophys. J. 2007, 93, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Min, S.; Kang, H.; Lee, J. Differential zinc permeation and blockade of L-type Ca2+ channel isoforms Cav1.2 and Cav1.3. Biochim. Biophys. Acta 2015, 1848, 2092–2100. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Gao, C.; Chen, Y.; Jia, M.; Geng, J.; Zhang, H.; Zhan, Y.; Boland, L.M.; An, H. Divalent cations modulate TMEM16A calcium-activated chloride channels by a common mechanism. J. Membr. Biol. 2013, 246, 893–902. [Google Scholar] [CrossRef] [PubMed]
- De Coursey, T.E. Voltage-gated proton channels: Molecular biology, physiology, and pathophysiology of the H(V) family. Physiol. Rev. 2013, 93, 599–652. [Google Scholar] [CrossRef] [PubMed]
- Mahaut-Smith, M.P. The effect of zinc on calcium and hydrogen ion currents in intact snail neurones. J. Exp. Biol. 1989, 145, 455–464. [Google Scholar] [PubMed]
- Ramsey, I.S.; Moran, M.M.; Chong, J.A.; Clapham, D.E. A voltage-gated proton-selective channel lacking the pore domain. Nature 2006, 440, 1213–1216. [Google Scholar] [CrossRef] [PubMed]
- Musset, B.; Smith, S.M.E.; Rajan, S.; Cherny, V.V.; Sujai, S.; Morgan, D.; De Coursey, T.E. Zinc inhibition of monomeric and dimeric proton channels suggests cooperative gating. J. Physiol. 2010, 588, 1435–1449. [Google Scholar] [CrossRef] [PubMed]
- Verkman, A.S.; Galietta, L.J.V. Chloride channels as drug targets. Nat. Rev. Drug Discov. 2009, 8, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Stölting, G.; Fischer, M.; Fahlke, C. CLC channel function and dysfunction in health and disease. Front. Physiol. 2014, 5, 1–18. [Google Scholar]
- Chen, T.Y. Extracellular zinc ion inhibits ClC-0 chloride channels by facilitating slow gating. J. Gen. Physiol. 1998, 112, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.A.; Wang, W.; Hales, T.G.; Kirkness, E.F. A novel class of ligand-gated ion channel is activated by Zn2+. J. Biol. Chem. 2003, 278, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Bloc, A.; Cens, T.; Cruz, H.; Dunant, Y. Zinc-induced changes in ionic currents of clonal rat pancreatic -cells: Activation of ATP-sensitive K+ channels. J. Physiol. 2000, 529(Pt. 3), 723–734. [Google Scholar] [CrossRef] [PubMed]
- Prost, A.; Bloc, A.; Hussy, N.; Derand, R.; Vivaudou, M. Zinc is both an intracellular and extracellular regulator of KATP channel function. J. Physiol. 2004, 559, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Walker, M.C.; Fabian-Fine, R.; Kullmann, D.M. Endogenous zinc inhibits GABAA receptors in a hippocampal pathway. J. Neurophysiol. 2004, 91, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Feigenspan, A.; Weiler, R. Electrophysiological properties of mouse horizontal cell GABAA receptors. J. Neurophysiol. 2004, 92, 2789–2801. [Google Scholar] [CrossRef] [PubMed]
- Calvo, D.J.; Vazquez, A.E.; Miledi, R. Cationic modulation of rho 1-type γ-aminobutyrate receptors expressed in Xenopus oocytes. Proc. Natl. Acad. Sci. USA 1994, 91, 12725–12729. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Amin, J.; Weiss, D.S. Zinc is a mixed antagonist of homomeric rho 1 γ-aminobutyric acid-activated channels. Mol. Pharmacol. 1995, 47, 595–602. [Google Scholar] [PubMed]
- Hsiao, B.; Dweck, D.; Luetje, C.W. Subunit-dependent modulation of neuronal nicotinic receptors by zinc. J. Neurosci. 2001, 21, 1848–1856. [Google Scholar] [PubMed]
- Palma, E.; Maggi, L.; Miledi, R.; Eusebi, F. Effects of Zn2+ on wild and mutant neuronal α7 nicotinic receptors. Proc. Natl. Acad. Sci. USA 1998, 95, 10246–10250. [Google Scholar] [CrossRef] [PubMed]
- Ohno, Y.; Shimizu, S.; Tokudome, K. Pathophysiological roles of serotonergic system in regulating extrapyramidal motor functions. Biol. Pharm. Bull. 2013, 36, 1396–1400. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, P.C.; Lummis, S.C.R. Zn2+ enhancement of the recombinant 5-HT3 receptor is modulated by divalent cations. Eur. J. Pharmacol. 2000, 394, 189–197. [Google Scholar] [CrossRef]
- Chen, N.; Moshaver, A.; Raymond, L.A. Differential sensitivity of recombinant N-methyl-d-aspartate receptor subtypes to zinc inhibition. Mol. Pharmacol. 1997, 51, 1015–1023. [Google Scholar] [PubMed]
- Mott, D.D.; Benveniste, M.; Dingledine, R.J. pH-Dependent inhibition of kainate receptors by zinc. J. Neurosci. 2008, 28, 1659–1671. [Google Scholar] [CrossRef] [PubMed]
- Sheng, S.; Perry, C.J.; Kleyman, T.R. Extracellular Zn2+ activates epithelial Na+ channels by eliminating Na+ self-inhibition. J. Biol. Chem. 2004, 279, 31687–31696. [Google Scholar] [CrossRef] [PubMed]
- Kellenberger, S.; Gautschi, I.; Pfister, Y.; Schild, L. Intracellular thiol-mediated modulation of epithelial sodium channel activity. J. Biol. Chem. 2005, 280, 7739–7747. [Google Scholar] [CrossRef] [PubMed]
- Wildman, S.S.; King, B.F.; Burnstock, G. Modulatory activity of extracellular H+ and Zn2+ on ATP-responses at rP2X1 and rP2X3 receptors. Br. J. Pharmacol. 1999, 128, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, K.; Ohno, Y. Effects of neuroamines and divalent cations on cloned and mutated ATP-gated channels. Eur. J. Pharmacol. 1997, 325, 101–108. [Google Scholar] [CrossRef]
- Wildman, S.S.; King, B.F.; Burnstock, G. Zn2+ modulation of ATP-responses at recombinant P2X2 receptors and its dependence on extracellular pH. Br. J. Pharmacol. 1998, 123, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Xiong, K.; Peoples, R.W.; Montgomery, J.P.; Chiang, Y.; Stewart, R.R.; Weight, F.F.; Li, C. Differential modulation by copper and zinc of P2X2 and P2X4 receptor function. J. Neurophysiol. 1999, 81, 2088–2094. [Google Scholar] [PubMed]
- Garcia-Guzman, M.; Soto, F.; Gomez-Hernandez, J.M.; Lund, P.E.; Stühmer, W. Characterization of recombinant human P2X4 receptor reveals pharmacological differences to the rat homologue. Mol. Pharmacol. 1997, 51, 109–118. [Google Scholar] [PubMed]
- Wildman, S.S.; King, B.F.; Burnstock, G. Modulation of ATP-responses at recombinant rP2X4 receptors by extracellular pH and zinc. Br. J. Pharmacol. 1999, 126, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Wildman, S.S.; Brown, S.G.; Rahman, M.; Noel, C.A.; Churchill, L.; Burnstock, G.; Unwin, R.J.; King, B.F. Sensitization by extracellular Ca2+ of rat P2X5 receptor and its pharmacological properties compared with rat P2X1. Mol. Pharmacol. 2002, 62, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Virginio, C.; Church, D.; North, R.A.; Surprenant, A. Effects of divalent cations, protons and calmidazolium at the rat P2X7 receptor. Neuropharmacology 1997, 36, 1285–1294. [Google Scholar] [CrossRef]
- Flockerzi, V.; Nilius, B. Mammalian transient receptor potential (TRP) cation channels. In Handbook of Experimental Pharmacology; Nilius, B., Flockerzi, V., Eds.; Springer Berlin Heidelberg: Berlin, Germany, 2014; Volume 222. [Google Scholar]
- Yang, W.; Manna, P.T.; Zou, J.; Luo, J.; Beech, D.J.; Sivaprasadarao, A.; Jiang, L.H. Zinc inactivates melastatin transient receptor potential 2 channels via the outer pore. J. Biol. Chem. 2011, 286, 23789–23798. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K.; Tominaga, M. Extracellular zinc ion regulates transient receptor potential melastatin 5 (TRPM5) channel activation through its interaction with a pore loop domain. J. Biol. Chem. 2013, 288, 25950–25955. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhang, D.; McMahon, D.G. Zinc modulation of hemi-gap-junction channel currents in retinal horizontal cells. J. Neurophysiol. 2009, 101, 1774–1780. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Bandell, M.; Petrus, M.J.; Zhu, M.X.; Patapoutian, A. Zinc activates damage-sensing TRPA1 ion channels. Nat. Chem. Biol. 2009, 5, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Wang, Q.; Zhang, L.H.; Lee, H.C.; Zhang, L.; Yue, J. A cell permeable NPE caged ADP-Ribose for Studying TRPM2. PLoS ONE 2012, 7, e51028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inoue, K.; Branigan, D.; Xiong, Z.G. Zinc-induced neurotoxicity mediated by transient receptor potential melastatin 7 channels. J. Biol. Chem. 2010, 285, 7430–7439. [Google Scholar] [CrossRef] [PubMed]
- Shuart, N.G.; Haitin, Y.; Camp, S.S.; Black, K.D.; Zagotta, W.N. Molecular mechanism for 3:1 subunit stoichiometry of rod cyclic nucleotide-gated ion channels. Nat. Commun. 2011, 2, 457. [Google Scholar] [CrossRef] [PubMed]
- Bixby, K.A.; Nanao, M.H.; Shen, N.V.; Kreusch, A.; Bellamy, H.; Pfaffinger, P.J.; Choe, S. Zn2+-binding and molecular determinants of tetramerization in voltage-gated K+ channels. Nat. Struct. Biol. 1999, 6, 38–43. [Google Scholar] [PubMed]
- Rotter, I.; Kosik-Bogacka, D.; Dołęgowska, B.; Safranow, K.; Lubkowska, A.; Laszczyńska, M. Relationship between the concentrations of heavy metals and bioelements in aging men with metabolic syndrome. Int. J. Environ. Res. Public Health 2015, 12, 3944–3961. [Google Scholar] [CrossRef] [PubMed]
- Watt, N.T.; Whitehouse, I.J.; Hooper, N.M. The role of zinc in Alzheimer’s disease. Int. J. Alzheimers Dis. 2011, 2011, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.R.; Levenson, C.W. Ion channels and zinc: Mechanisms of neurotoxicity and neurodegeneration. J. Toxicol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-O.; Wang, J.-L.; Bjørklund, G.; Zhao, W.-N.; Yin, C.-H. Serum copper and zinc levels in individuals with autism spectrum disorders. Neuroreport 2014, 25, 1216–1220. [Google Scholar] [CrossRef] [PubMed]
- Macedoni-Lukšič, M.; Gosar, D.; Bjørklund, G.; Oražem, J.; Kodrič, J.; Lešnik-Musek, P.; Zupančič, M.; France-Štiglic, A.; Sešek-Briški, A.; Neubauer, D.; et al. Levels of metals in the blood and specific porphyrins in the urine in children with autism spectrum disorders. Biol. Trace Elem. Res. 2015, 163, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, L.R.; Godar, S.C.; Khoja, S.; Jakowec, M.W.; Alkana, R.L.; Bortolato, M.; Davies, D.L. Sociocommunicative and sensorimotor impairments in male P2X4-deficient mice. Neuropsychopharmacology 2013, 38, 1993–2002. [Google Scholar] [CrossRef] [PubMed]
- Nickols, H.H.; Conn, J.P. Development of allosteric modulators of GPCRs for treatment of CNS disorders. Neurobiol. Dis. 2014, 61, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Lorca, A.; Rozas, C.; Loyola, S.; Moreira-ramos, S.; Zeise, M.L.; Kirkwood, A.; Huidobro-Toro, J.P.; Morales, B. Zinc enhances long-term potentiation through P2X receptor modulation in the hippocampal CA1 region. Eur. J. Neurosci. 2011, 33, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Wang, Y.; Zhang, T.; Ren, G.; Yang, Z. Effects of nanoparticle zinc oxide on spatial cognition and synaptic plasticity in mice with depressive-like behaviors. J. Biomed. Sci. 2012, 19, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
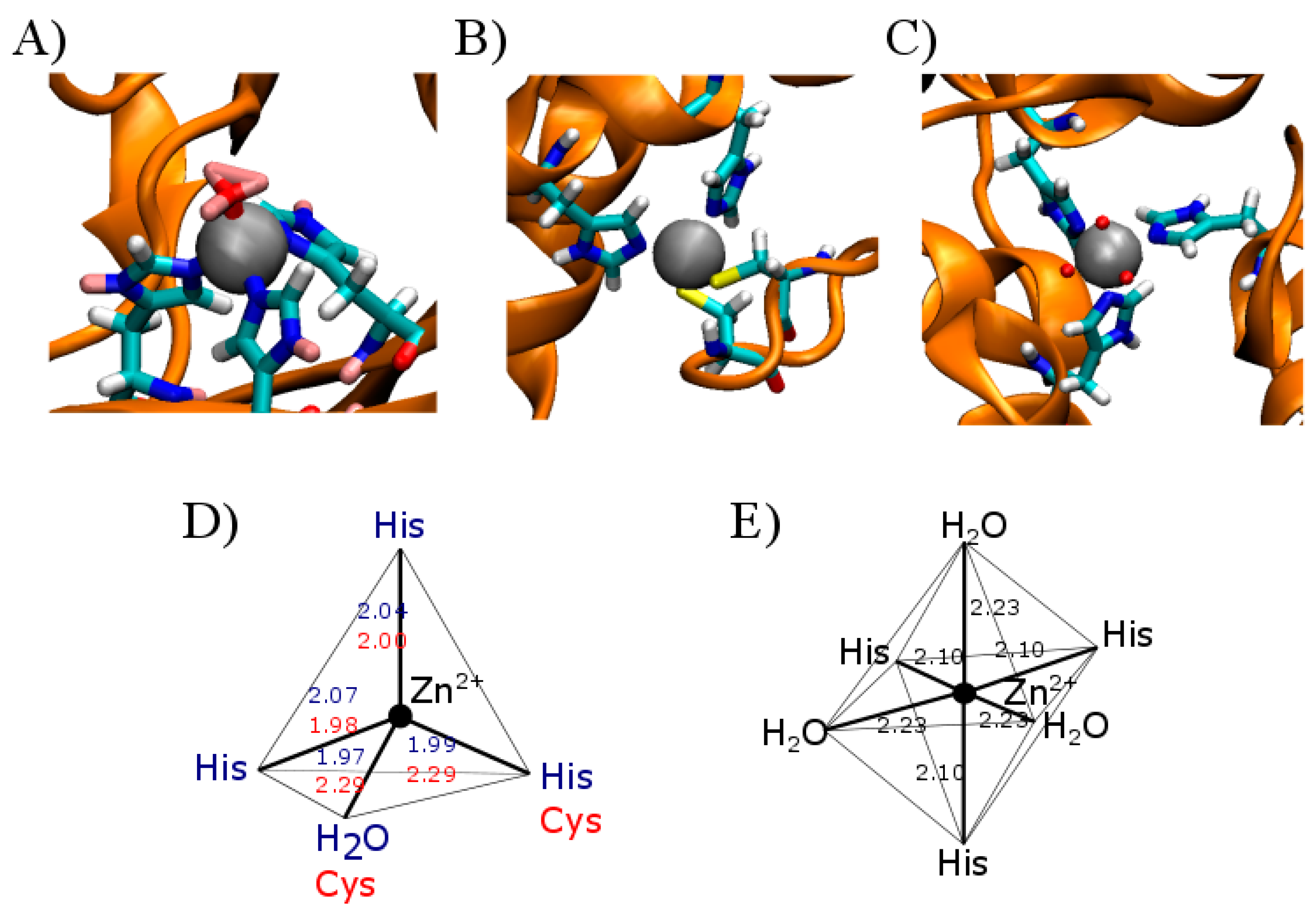

| Type of Zinc Modulation | Receptor | aa1 | aa2 | aa3 | aa4 |
|---|---|---|---|---|---|
| Positive Modulator | GluK3 | Q756 [20] | D759 [20] | H762 [20] | D730 [20] |
| NAChR α4β4 | α E59 [21] | α H61 [21] | α H162 [21] | β H469 [21] | |
| GlyRα1 | H107 [22] | ||||
| ASIC2a | H162 [23] | H339 [23] | |||
| ENaC γ | H193 [24] | H200 [24] | H202 [24] | ||
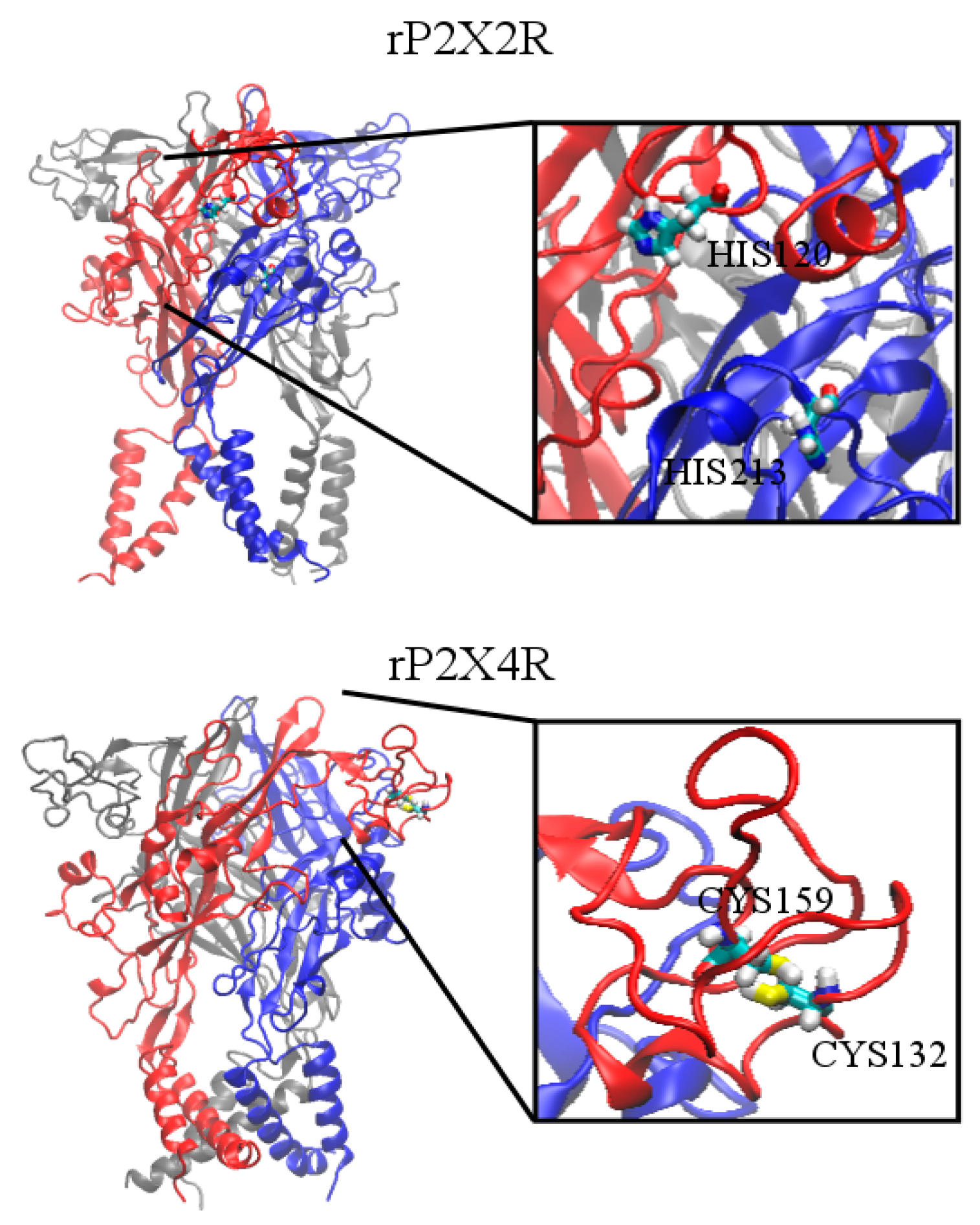
| P2X2 | H120 [25,26] | H213 [25,26] | |||
| P2X4 | C132 [27,28] | C159 [28] | |||
| Negative Modulator | NR1/NR2A | H44 [29,30] | H128 [29,30] | K233 [29] | E266 [29] |
| GABAρ1 | H156 [31] | ||||
| GlyRα1 | H107 [22] | H109 [22] | |||
| ASIC1a | K133 [32] | ||||
| ENaC γ | H88 [24] | ||||
| P2X4 | D136 [27] | H140 [27] | |||
| P2X7 | H62 [33] | D197 [33] | H219 [34] | H267 [34] |
| Amino Acid | Interaction | Energy (kJ/mol) * | Distance (Å) | Geometry of the Complex † |
|---|---|---|---|---|
| Cys | Zn–S (Cysteine) | −60.35 | 2.27 | Tetrahedral |
| His | Zn–N (Imidazol) | −34.26 | 2.07 | Octahedral |
| Asp, Glu | Zn–O (Carboxyl) | −4.49 | 2.18 | Octahedral |
| Channel Name | Subtype or Subunit | [Zn2+] (EC50 or IC50) |
|---|---|---|
| K+ Channels | Kv1.1 (NM) | N.D. [75] |
| Kv1.4 (NM) | N.D. [75] | |
| Kv1.5 (NM) | N.D. [75] | |
| TREK-1 (NM) | 659 µM [77] | |
| TREK-2 (PM) | 87.1 µM [73] | |
| TASK-3 (NM) | 12.7 µM [77]; 25.4 µM [73] | |
| Shaker (H4) K-channel (NM) | N.D. [74] | |
| Slo1 K (BK) channels (A) | 33.6 µM [78] | |
| KCNQ5 (PM) | 21.8 µM [72] | |
| ATP-sensitive K+ Channels | Kir6.2 (A) | 1.7 µM [94] |
| SUR1/Kir6.2 (A) | Extracellular N.D.; Intracellular 1.8 µM [95] | |
| SUR2A/Kir6.2 (NM, extracellular; A, intracellular) | Extracellular N.D.; Intracellular 60 µM [95] | |
| Na+ Channels | Nav (NM) | N.D. [79] |
| TTX-sensitive Na channels (NM) | N.D. [80] | |
| Ca2+ Channels | TMEM16A (NM) | 12.5 µM [85] |
| Cav1.2 (NM) | 10.9 µM [83]; 18.4 µM [84] | |
| Cav1.3 (NM) | 34.1 µM [84] | |
| Cav2.1 (NM) | 110 µM [83] | |
| Cav2.2 (NM) | 98.0 µM [83] | |
| Cav2.3 (NM) | 31.8 µM [83] | |
| Cav3.1 (NM) | 81.7 µM [82]; 196.1 µM [83] | |
| Cav3.2 (NM) | 0.78 µM [82]; 24.1 µM [83] | |
| Cav3.3 (NM) | 158.6 µM [82]; 15.2 µM [83] | |
| H+ Channels | HV1 (NM) | 2 µM [88]; 16 µM [87] |
| Cl− Channels | ClC-0 (NM) | 1–3 µM [92] |
| ZAC | ZAC (A) | 540 µM [93] |
| Receptor Type | Subtype or Subunit | Ligand | [Zn2+] (EC50 or IC50) |
|---|---|---|---|
| GABAergic | GABAρ1 (NM) | GABA 1 µM [98], GABA 3 µM [99] | 21.9 µM (1 min PI) [98], 20.4 µM [99] |
| GABAα1β2γ2 (NM) | GABA 3 µM [99] | 441.3 µM [99] | |
| GABAA (NM) | GABA (WC) [96], GABA 50 µM [97] | N.D. [96], 7.3 µM [97] | |
| Glycinergic | GlyRα1 (PM/NM) | Glycine 50 µM [22] | 80 nM (PM); 546 µM (NM) [22] |
| Cholinergic | α7 (NM) | Acetylcholine 3 µM [101] | 27 µM [101] |
| α2β2 (PM/NM) | Acetylcholine 3 µM [100] | 13 µM (PM); 52 µM (NM) [100] | |
| α2β4 (PM/NM) | Acetylcholine 3 µM [100] | 45 µM (PM); 590 µM (NM) [100] | |
| α3β2 (NM) | Acetylcholine 3 µM [100] | 97 µM [100] | |
| α3β4 (PM/NM) | Acetylcholine 3 µM [100] | 47 µM (PM); 3200 µM (NM) [100] | |
| α4β2 (PM/NM) | Acetylcholine 3 µM [100] | 16 µM (PM); 440 µM (NM) [100] | |
| α4β4 (PM/NM) | Acetylcholine 3 µM [100] | 22 µM (PM); 510 µM (NM) [100] | |
| Glutamatergic | NR1/NR2A (NM) | Glutamate 100 µM [104] | 5 nM (HAS); 79 µM (LAS) [104] |
| NR1/NR2B (NM) | Glutamate 100 µM [104] | 9.5 µM [104] | |
| GluR6R (NM) | AMPA 30–300 µM [105] | 67 µM (5 min PI) [105] | |
| GluR6R/KA1 (NM) | AMPA 30–300 µM [105] | 1.5 µM (5 min PI) [105] | |
| GluR6R/KA2 (NM) | AMPA 30–300 µM [105] | 2.1 µM (5 min PI) [105] | |
| GluK3 (PM/NM) | Glutamate 10 mM [20] | 46 µM (PM); 100 µM (NM) [20] | |
| Serotoninergic | 5-HT3A (PM) | 5-HT 1 µM [103] | N.D. [103] |
| H+ergic | ASIC2a (PM) | pH 5 [23] | 120 µM [23] |
| ASIC1a (NM) | pH 6.5 [32] | 7.0 nM [32] | |
| ASIC1a-ASIC2a (NM) | pH 6.0 [32] | 10.04 nM pH 6.5 [32] | |
| Na+ergic | ENaC αβγ (PM/NM) | Na+ 110 mM [24,106] | 1.74 µM [106], 2.1 µM [24] (PM); N.D. [107], 2.1 mM [24] (NM) |
| Purinergic | P2X1 (NM) | ATP 0.3 µM [108] | 9.34 µM, 0.82 µM (20 min PI), 1.1 µM (40 min PI) [108] |
| P2X2 (PM/NM) | ATP (WC) [109], 3 µM [110], 5 µM [25], 2 µM [26], 50 µM [111] | N.D. [25,109], 9.3 µM, 6.1 µM (5 min PI) [110], 7.9 µM [26], 19.6 µM [111] (PM); N.D. (>30 µM, 5 min PI) [110] (NM) | |
| P2X3 (PM/NM) | ATP 0.3 µM [108] | 10.9 µM (20 min PI), N.D. (<20 µM, 40 min PI) (PM); N.D. (>20 µM, 20 and 40 min PI) (NM) [108] | |
| P2X4 (PM/NM) | ATP 5 µM [111,112], 3 µM [113], 1 µM 25 µM [68] | N.D. (<10 µM) [112], 2.4 µM [111], 1.9 µM (0 and 15 min PI) [113], 2.4 µM, 4.9 µM (20 s PI) [68] (PM); N.D. (>100 µM) [112], N.D. (>30 µM, 0 and 15 min PI) [113] (NM) | |
| P2X5 (PM/NM) | ATP 300 nM [114] | 42.6 µM ( 0 and 15 min PI) (PM); N.D. (0 and 15 min PI) (NM) [114] | |
| P2X7 (NM) | BzATP 30 µM [33,115], ATP 600 µM [34] | 11.2 µM [115], 78 µM (10 s PI) [34], 4.6 µM [33] | |
| TRP | TRPM2 (NM) | ADPR [117] | N.D. [117] |
| TRPM5 (NM) | Intracellular Ca2+ 500 nM [118] | 4.3 µM [118] | |
| TRPA1 (A) | – | 2.3 µM [120] | |
| Hemichannels | Hemi-gap-junction channels (NM) | 0 Ca2+ [119] | 37 µM [119] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peralta, F.A.; Huidobro-Toro, J.P. Zinc as Allosteric Ion Channel Modulator: Ionotropic Receptors as Metalloproteins. Int. J. Mol. Sci. 2016, 17, 1059. https://doi.org/10.3390/ijms17071059
Peralta FA, Huidobro-Toro JP. Zinc as Allosteric Ion Channel Modulator: Ionotropic Receptors as Metalloproteins. International Journal of Molecular Sciences. 2016; 17(7):1059. https://doi.org/10.3390/ijms17071059
Chicago/Turabian StylePeralta, Francisco Andrés, and Juan Pablo Huidobro-Toro. 2016. "Zinc as Allosteric Ion Channel Modulator: Ionotropic Receptors as Metalloproteins" International Journal of Molecular Sciences 17, no. 7: 1059. https://doi.org/10.3390/ijms17071059






