Chemical Structure, Property and Potential Applications of Biosurfactants Produced by Bacillus subtilis in Petroleum Recovery and Spill Mitigation
Abstract
:1. Introduction
2. Structure and Property
2.1. Structure Characterization
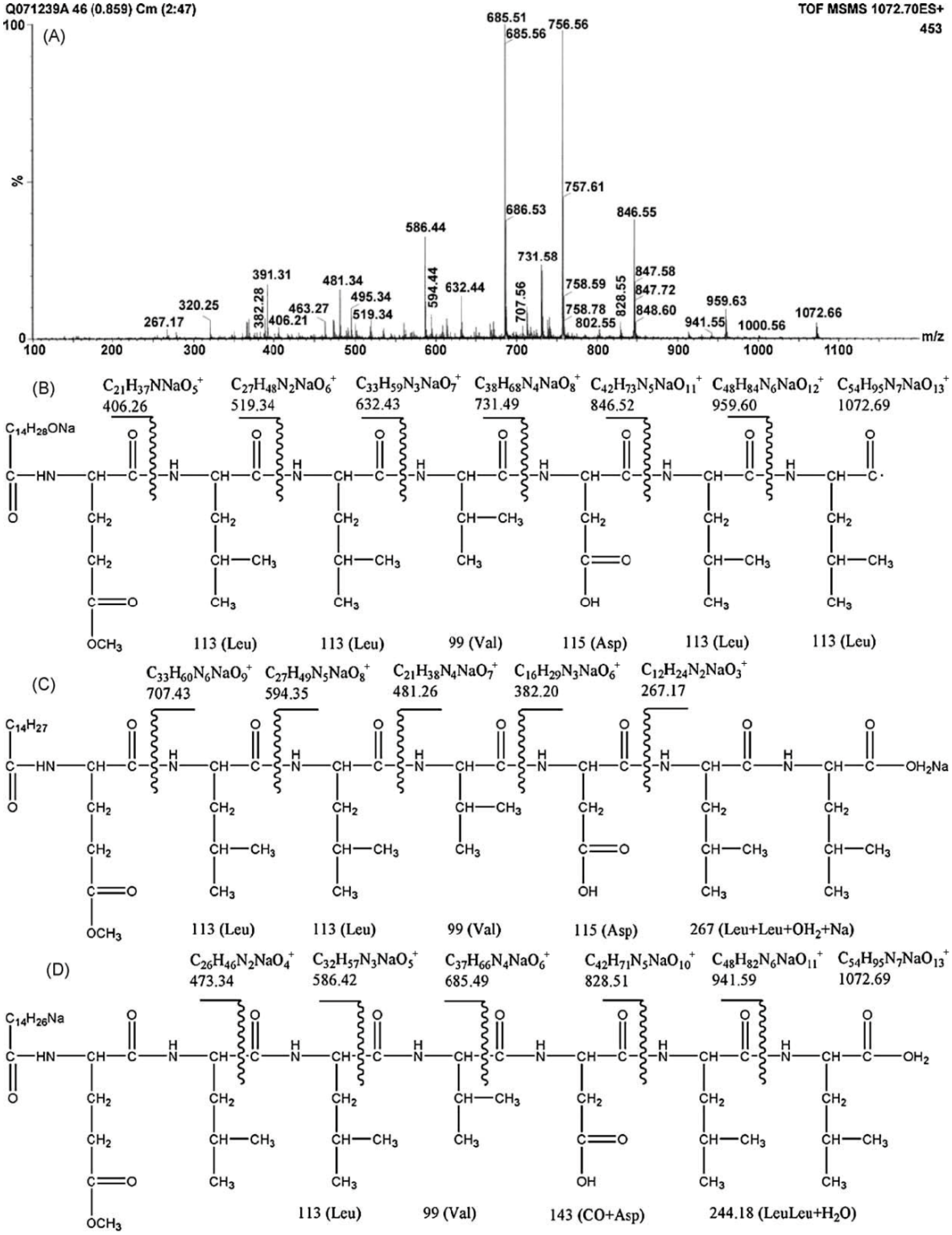
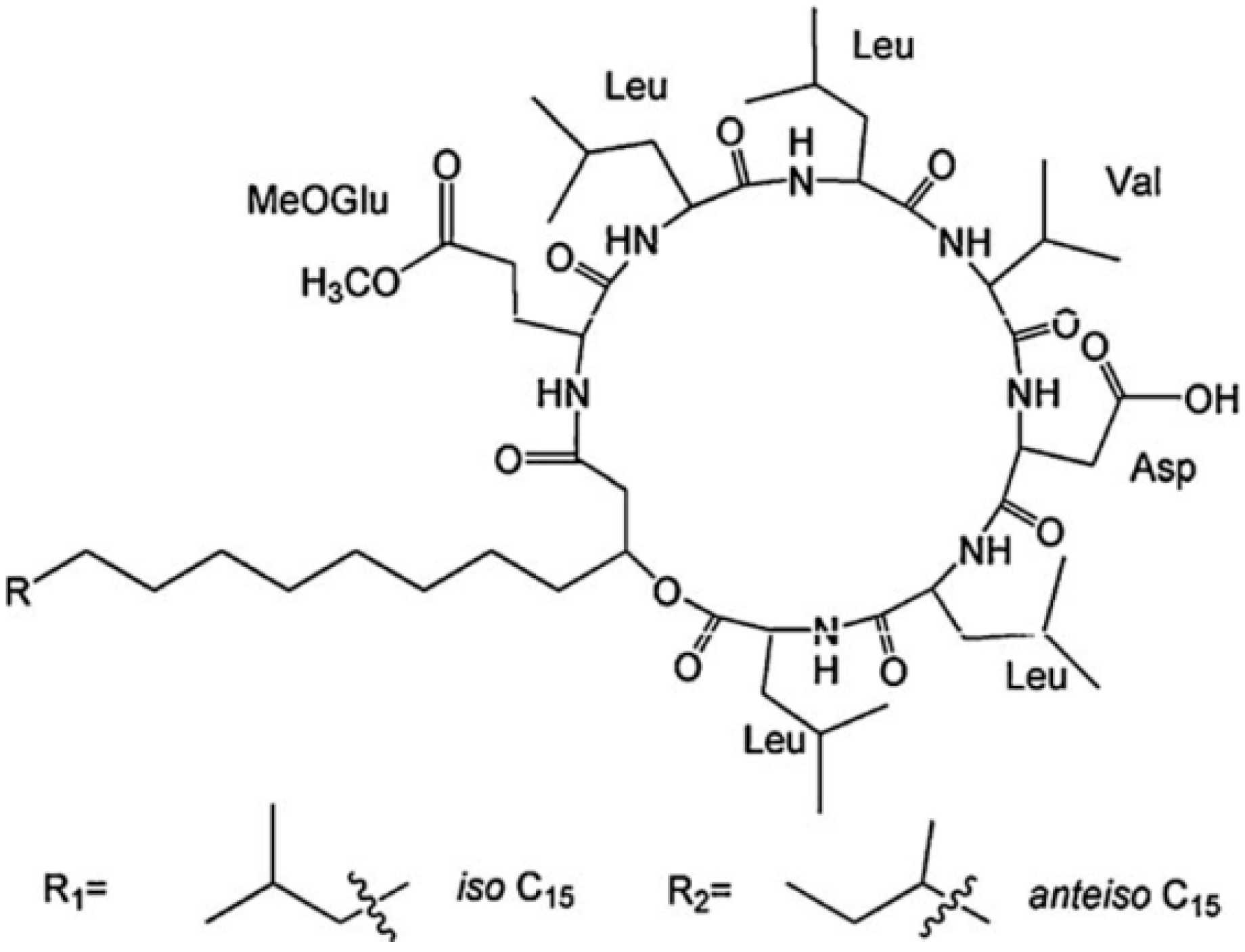
2.2. Molecular Behavior at Interface and in Solution
2.2.1. Interfacial Adsorption
2.2.2. Monolayer at Interface


2.2.3. Molecular Behavior in Solution


2.3. The Structure–Property Relationship
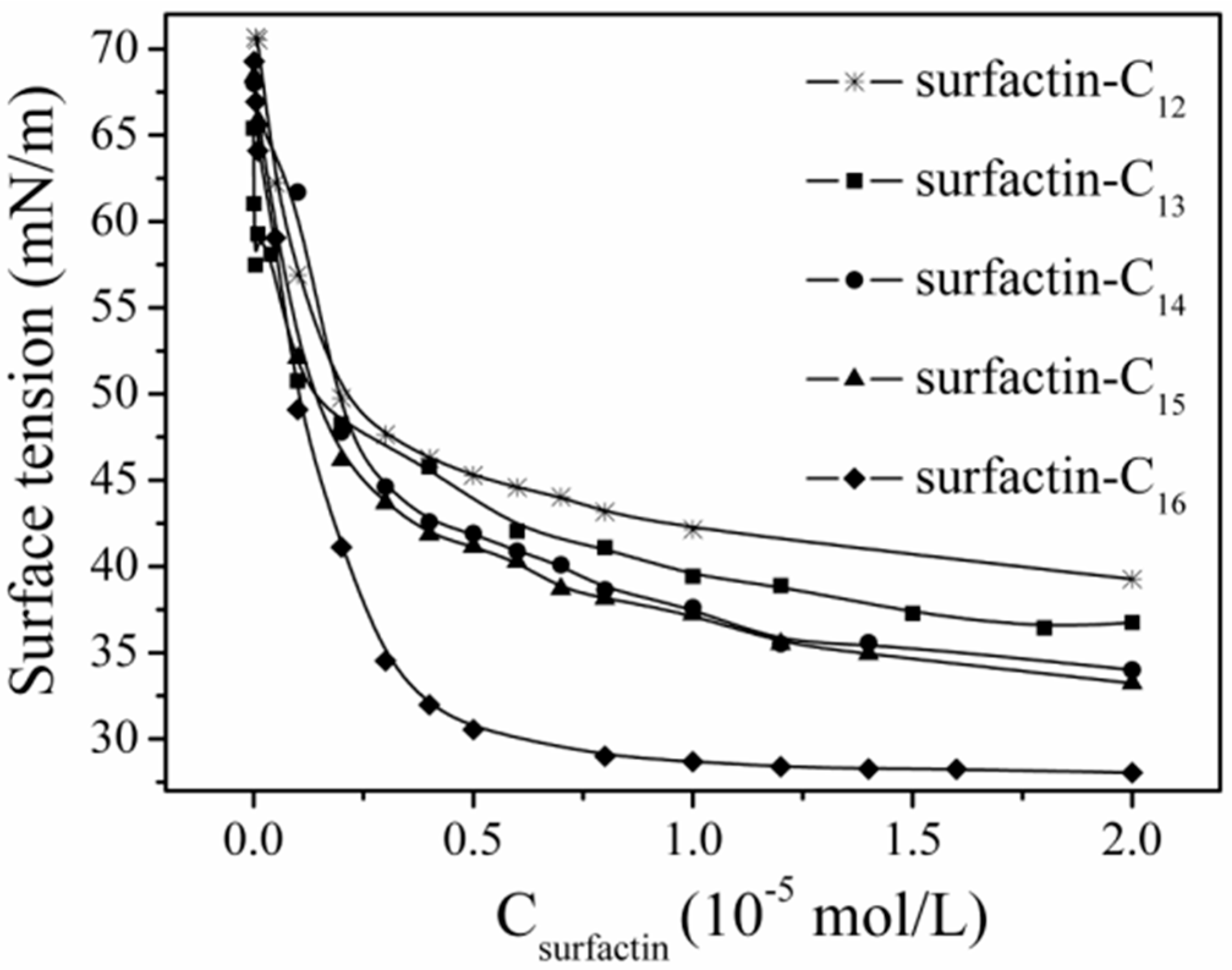
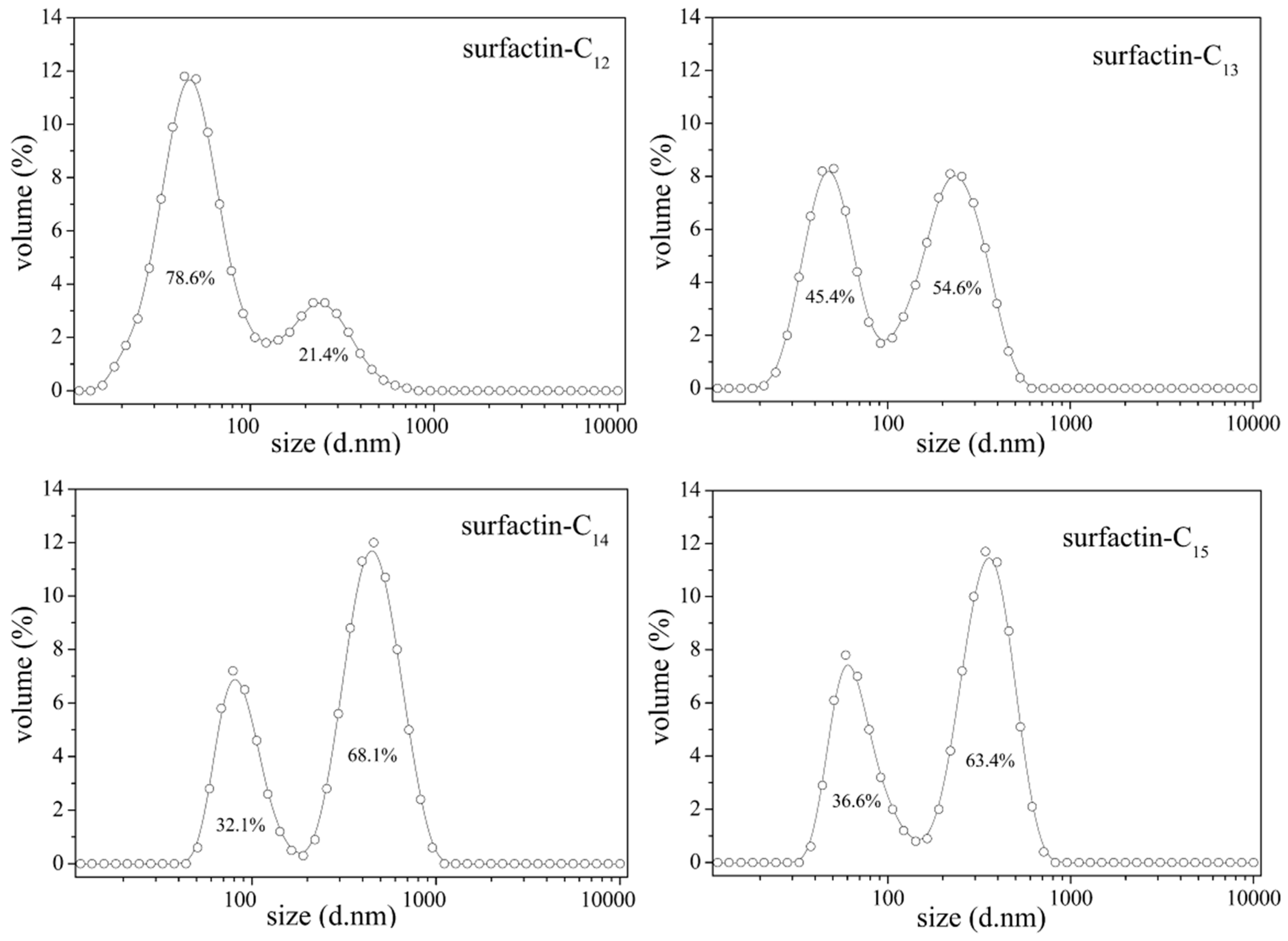
3. Potential Applications
3.1. Microbial Enhanced Oil Recovery (MEOR)
3.2. Oil Spill Contamination and Remediation
3.3. Other Applications
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Desai, J.D.; Banat, I.M. Microbial production of surfactants and their commercial potential. Microbiol. Mol. Biol. Rev. 1997, 61, 47–64. [Google Scholar] [PubMed]
- Silva, R.D.C.F.S.; Almeida, D.G.; Rufino, R.D.; Luna, J.M.; Santos, V.A.; Sarubbo, L.A. Applications of biosurfactants in the petroleum industry and the remediation of oil spills. Int. J. Mol. Sci. 2014, 15, 12523–12542. [Google Scholar] [CrossRef] [PubMed]
- Deleu, M.; Razafindralambo, H.; Popineau, Y.; Jacques, P.; Thonart, P.; Paquot, M. Interfacial and emulsifying properties of lipopeptides from Bacillus subtilis. Colloids Surf. Physicochem. Eng. Asp. 1999, 152, 3–10. [Google Scholar] [CrossRef]
- Peypoux, F.; Bonmatin, J.M.; Wallach, J. Recent trends in the biochemistry of surfactin. Appl. Microbiol. Biotechnol. 1999, 51, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Grgurina, I. Lipopeptide secondary metabolites from the phytopathogenic bacterium Pseudomonas syringae. In Advances in Microbial Toxin Research and Its Biotechnological Exploitation; Upadhyay, R., Ed.; Springer US: New York, NY, USA, 2002; pp. 105–140. [Google Scholar]
- Baltz, R.H.; Miao, V.; Wrigley, S.K. Natural products to drugs: Daptomycin and related lipopeptide antibiotics. Nat. Prod. Rep. 2005, 22, 717–741. [Google Scholar] [CrossRef] [PubMed]
- Hino, M.; Fujie, A.; Iwamoto, T.; Hori, Y.; Hashimoto, M.; Tsurumi, Y.; Sakamoto, K.; Takase, S.; Hashimoto, S. Chemical diversity in lipopeptide antifungal antibiotics. J. Ind. Microbiol. Biotechnol. 2001, 27, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Yang, S.Z.; Mu, B.Z. Molecular structures of microbial lipopeptides. Biotechnol. Bull. 2005, 4, 18–26. [Google Scholar]
- Arima, K.; Kakinuma, A.; Tamura, G. Surfactin, a crystalline peptide-lipid surfactant produced by Bacillus subtilis, isolation, characterization and its inhibition of fibrin clot formation. Biochem. Biophys. Res. Commun. 1968, 31, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Yakimov, M.M.; Abraham, W.-R.; Meyer, H.; Laura, G.; Golyshin, P.N. Structural characterization of lichenysin a components by fast atom bombardment tandem mass spectrometry. Biochim. Biophys. Acta 1999, 1438, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Peypoux, F.; Guinand, M.; Michel, G.; Delcambe, L.; Das, B.C.; Lederer, E. Structure of iturin A, a peptidolipid antibiotic from Bacillus subtilis. Biochemistry 1978, 17, 3992–3996. [Google Scholar] [CrossRef] [PubMed]
- Vanittanakom, N.; Loeffler, W.; Koch, U.; Jung, G. Fengycin—A novel antifungal lipopeptide antibiotic produced by Bacillus subtilis F-29-3. J. Antibiot. 1986, 39, 888–901. [Google Scholar] [CrossRef] [PubMed]
- Lang, S. Biological amphiphiles (microbial biosurfactants). Curr. Opin. Colloid Interface Sci. 2002, 7, 12–20. [Google Scholar] [CrossRef]
- Rosenberg, E.; Ron, E.Z. High- and low-molecular-mass microbial surfactants. Appl. Microbiol. Biotechnol. 1999, 52, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, Y.; Osman, M.; Nakahara, H.; Sano, Y.; Ishiguro, R.; Matsumoto, M. Significace of β-sheet formation for micellization and surface adsorption of surfactin. Colloids Surf. B 1995, 4, 341–348. [Google Scholar] [CrossRef]
- Kakinuma, A.; Hori, M.; Isono, M.; Tamura, G. Determination of amino acid sequence in surfaction, a crystalline peptide lipid surfactant produced by Bacillus subtilis. Agric. Biol. Chem. 1969, 33, 971–972. [Google Scholar] [CrossRef]
- Bonmatin, J.-M.; Laprevote, O.; Peypoux, F. Diversity among microbial cyclic lipopeptides: Iturins and surfactins: Activity-structure relationships to design new bioactive agents. Comb. Chem. High Throughput Screen 2003, 6, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, F.; Kluge, B.; Ullrich, C.; Vater, J.; Ziessow, D. Identification of amino acid substitutions in the lipopeptide surfactin using 2D NMR spectroscopy. Biochem. Biophys. Res. Commun. 1991, 177, 998–1005. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Z.; Wei, D.Z.; Mu, B.Z. Determination of the amino acid sequence in a cyclic lipopeptide using MS with DHT mechanism. J. Biochem. Biophys. Methods 2006, 68, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Z.; Wei, D.Z.; Mu, B.Z. Determination of the structure of the fatty acid chain in a cyclic lipopeptide using GC-MS. J. Biochem. Biophys. Methods 2007, 70, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Haddad, N.I.A.; Yang, S.Z.; Mu, B.Z. Structural characterization of eight cyclic lipopeptides produced by Bacillus subtilis HSO121. Protein Pept. Lett. 2007, 14, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Yang, S.Z.; Mu, B.Z. Isolation and characterization of a C12-lipopeptide produced by Bacillus subtilis HSO 121. J. Pept. Sci. 2008, 14, 864–875. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Yang, S.Z.; Mu, B.Z. Production and characterization of a C15-surfactin-O-methyl ester by a lipopeptide producing strain Bacillus subtilis HSO121. Process Biochem. 2009, 44, 1144–1151. [Google Scholar] [CrossRef]
- Kowall, M.; Vater, J.; Kluge, B.; Stein, T.; Franke, P.; Ziessow, D. Separation and characterization of surfactin isoforms produced by Bacillus subtilis okb 105. J. Colloid Interface Sci. 1998, 204, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jacques, P.; Hbid, C.; Destain, J.; Razafindralambo, H.; Paquot, M.; de Pauw, E.; Thonart, P. Optimization of biosurfactant lipopeptide production from Bacillus subtilis S499 by plackett-burman design. Appl. Biochem. Biotechnol. 1999, 77, 223–233. [Google Scholar] [CrossRef]
- Kim, P.I.; Ryu, J.; Kim, Y.H.; Chl, Y.-T. Production of biosurfactant lipopeptides iturin A, fengycin, and surfactin A from Bacillus subtilis CMB32 for control of colletotrichum gloeosporioides. J. Microbiol. Biotechnol. 2010, 20, 138–145. [Google Scholar] [PubMed]
- Athukorala, S.N.P.; Fernando, W.G.D.; Rashid, K.Y. Identification of antifungal antibiotics of Bacillus species isolated from different microhabitats using polymerase chain reaction and MALDI-TOF mass spectrometry. Can. J. Microbiol. 2009, 55, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.R.; Moses, V. Synthesis and surface activity properties of hydrophobic/hydrophilic peptides. Int. J. Pept. Protein Res. 1994, 43, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, E.T.; Kezdy, F.J. Amphiphilic secondary structure: Design of peptide hormones. Science (New York, NY) 1984, 223, 249–255. [Google Scholar] [CrossRef]
- Gallet, X.; Deleu, M.; Razafindralambo, H.; Jacques, P.; Thonart, P.; Paquot, M.; Brasseur, R. Computer simulation of surfactin conformation at a hydrophobic/hydrophilic interface. Langmuir 1999, 15, 2409–2413. [Google Scholar] [CrossRef]
- Maget-Dana, R.; Ptak, M. Interfacial properties of surfactin. J. Colloid Interface Sci. 1992, 153, 285–291. [Google Scholar] [CrossRef]
- Razafindralambo, H.; Thonart, P.; Paquot, M. Dynamic and equilibrium surface tensions of surfactin aqueous solutions. J. Surfactants Deterg. 2004, 7, 41–46. [Google Scholar] [CrossRef]
- Eeman, M.; Berquand, A.; Dufrene, Y.; Paquot, M.; Dufour, S.; Deleu, M. Penetration of surfactin into phospholipid monolayers: Nanoscale interfacial organization. Langmuir 2006, 22, 11337–11345. [Google Scholar] [CrossRef] [PubMed]
- Gang, H.Z.; Liu, J.F.; Mu, B.Z. Interfacial behavior of surfactin at the decane/water interface: A molecular dynamics simulation. J. Phys. Chem. B 2010, 114, 14947–14954. [Google Scholar] [CrossRef] [PubMed]
- Song, C.S.; Ye, R.Q.; Mu, B.Z. Effect of compression speed on aggregation behavior of surfactin in langmuir monolayer. Acta Chim. Sin. 2009, 69, 2038–2042. [Google Scholar]
- Song, C.S.; Ye, R.Q.; Mu, B.Z. Aggregation behavior and surface morphology studies of surfactin in langmuir-blodgett films. Colloids Surf. Physicochem. Eng. Asp. 2008, 330, 49–54. [Google Scholar] [CrossRef]
- Song, C.S.; Ye, R.Q.; Mu, B.Z. Molecular behavior of a microbial lipopeptide monolayer at the air-water interface. Colloids Surf. Physicochem. Eng. Asp. 2007, 302, 82–87. [Google Scholar] [CrossRef]
- Gang, H.Z.; Liu, J.F.; Mu, B.Z. Molecular dynamics study of surfactin monolayer at the air/water interface. J. Phys. Chem. B 2011, 115, 12770–12777. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zou, A.H.; Mu, B.Z. Toluidine blue: Aggregation properties and distribution behavior in surfactin micelle solution. Colloids Surf. B. Biointerfaces 2010, 75, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Huang, X.; Cao, M.; Wang, Y. Micellization of surfactin and its effect on the aggregate conformation of amyloid β (1–40). J. Phys. Chem. B 2008, 112, 15195–15201. [Google Scholar] [CrossRef] [PubMed]
- Knoblich, A.; Matsumoto, M.; Ishiguro, R.; Murata, K.; Fujiyoshi, Y.; Ishigami, Y.; Osman, M. Electron cryo-microscopic studies on micellar shape and size of surfactin, an anionic lipopeptide. Colloids Surf. B. Biointerfaces 1995, 5, 43–48. [Google Scholar] [CrossRef]
- Li, Y.; Zou, A.H.; Ye, R.Q.; Mu, B.Z. Counterion-induced changes to the micellization of surfactin-C-16 aqueous solution. J. Phys. Chem. B 2009, 113, 15272–15277. [Google Scholar] [CrossRef] [PubMed]
- She, A.Q.; Gang, H.Z.; Mu, B.Z. Temperature influence on the structure and interfacial properties of surfactin micelle: A molecular dynamics simulation study. J. Phys. Chem. B 2012, 116, 12735–12743. [Google Scholar] [CrossRef] [PubMed]
- Osman, M.; Hoiland, H.; Holmsen, H. Micropolarity and microviscosity in the micelles of the heptapeptide biosurfactant “surfactin”. Colloids Surf. B. Biointerfaces 1998, 11, 167–175. [Google Scholar] [CrossRef]
- Bonmatin, J.-M.; Genest, M.; Labbé, H.; Ptak, M. Solution three-dimensional structure of surfactin: A cyclic lipopeptide studied by 1H-NMR, distance geometry, and molecular dynamics. Biopolymers 1994, 34, 975–986. [Google Scholar] [CrossRef] [PubMed]
- Tsan, P.; Volpon, L.; Besson, F.; Lancelin, J.-M. Structure and dynamics of surfactin studied by NMR in micellar media. J. Am. Chem. Soc. 2007, 129, 1968–1977. [Google Scholar] [CrossRef] [PubMed]
- Vass, E.; Besson, F.; Majer, Z.; Volpon, L.; Hollosi, M. Ca2+-induced changes of surfactin conformation: A FTIR and circular dichroism study. Biochem. Biophys. Res. Commun. 2001, 282, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.-H.; Thomas, R.K.; Chen, C.-Y.; Darton, R.C.; Baker, S.C.; Penfold, J. Aggregation of the naturally occurring lipopeptide, surfactin, at interfaces and in solution: An unusual type of surfactant? Langmuir 2009, 25, 4211–4218. [Google Scholar] [CrossRef] [PubMed]
- Grangemard, I.; Wallach, J.; Maget-Dana, R.; Peypoux, F. Lichenysin: A more efficient cation chelator than surfactin. Appl. Biochem. Biotechnol. 2001, 90, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Razafindralambo, H.; Paquot, M.; Baniel, A.; Popineau, Y.; Hbid, C.; Jacques, P.; Thonart, P. Foaming properties of a natural cyclic lipoheptapeptide belonging to a special class of amphiphilic molecules. Food Hydrocolloids 1997, 11, 59–62. [Google Scholar] [CrossRef]
- Li, Y.; Zou, A.H.; Ye, R.Q.; Mu, B.Z. Effects of molecular structure on surfactin micellization activity. Acta Phys.-Chim. Sin. 2011, 27, 1128–1134. [Google Scholar]
- Yakimov, M.M.; Fredrickson, H.L.; Timmis, K.N. Effect of heterogeneity of hydrophobic moieties on surface activity of lichenysin A, a lipopeptide biosurfactant from Bacillus licheniformis BAS50. Biotechnol. Appl. Biochem. 1996, 23, 13–18. [Google Scholar] [PubMed]
- Dufour, S.; Deleu, M.; Nott, K.; Wathelet, B.; Thonart, P.; Paquot, M. Hemolytic activity of new linear surfactin analogs in relation to their physico-chemical properties. Biochim. Biophys. Acta 2005, 1726, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, M.; Hirata, Y.; Imanaka, T. A study on the structure-function relationship of lipopeptide biosurfactants. Biochim. Biophys. Acta 2000, 1488, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Thimon, L.; Peypoux, F.; Das, B.C.; Wallach, J.; Michel, G. Selective esterification of surfactin: Preparation and properties of surfactin methyl esters. Biotechnol. Appl. Biochem. 1994, 20, 415–423. [Google Scholar]
- Yakimov, M.M.; Timmis, K.N.; Wray, V.; Fredrickson, H.L. Characterization of a new lipopeptide surfactant produced by thermotolerant and halotolerant subsurface Bacillus licheniformis BAS50. Appl. Environ. Microbiol. 1995, 61, 1706–1713. [Google Scholar] [PubMed]
- Grangemard, I.; Bonmatin, J.-M.; Bernillon, J.; Das, B.C.; Peypoux, F. Lichenysins G, a novel family of lipopeptide biosurfactants from Bacillus licheniformis IM 1307: Production, isolation and structural evaluation by NMR and mass spectrometry. J. Antibiot. 1999, 52, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Bonmatin, J.-M.; Labbé, H.; Grangemard, I.; Peypoux, F.; Maget-Dana, R.; Ptak, M.; Michel, G. Production, isolation and characterization of [Leu4]- and [Ile4]surfactins from Bacillus subtilis. Lett. Pept. Sci. 1995, 2, 41–47. [Google Scholar] [CrossRef]
- Peypoux, F.; Bonmatin, J.M.; Labbe, H.; Grangemard, I.; Das, B.C.; Ptak, M.; Wallach, J.; Michel, G. [Ala4] surfactin, a novel isoform from Bacillus subtilis studied by mass and NMR spectroscopies. Eur. J. Biochem. 1994, 224, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Khire, J.M. Bacterial Biosurfactants, and Their Role in Microbial Enhanced Oil Recovery (MEOR). In Biosurfactants; Sen, R., Ed.; Springer New York: New York, NY, USA, 2010; Volume 672, pp. 146–157. [Google Scholar]
- Sarafzadeh, P.; Hezave, A.Z.; Ravanbakhsh, M.; Niazi, A.; Ayatollahi, S. Enterobacter cloacae as biosurfactant producing bacterium: Differentiating its effects on interfacial tension and wettability alteration mechanisms for oil recovery during MEOR process. Colloids Surf. B. Biointerfaces 2013, 105, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Al-Sulaimani, H.; Al-Wahaibi, Y.; Al-Bahry, S.; Elshafie, A.; Al-Bemani, A.; Joshi, S.; Ayatollahi, S. Residual-oil recovery through injection of biosurfactant chemical surfactant, and mixtures of both under reservoir temperatures: Induced-wettability and interfacial-tension effects. Spe Reserv. Eval. Eng. 2012, 15, 210–217. [Google Scholar] [CrossRef]
- Fiechter, A. Biosurfactants: Moving towards industrial application. Trends Biotechnol. 1992, 10, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Kowalewski, E.; Rueslatten, I.; Steen, K.H.; Bodtker, G.; Torsaeter, O. Microbial improved oil recovery—Bacterial induced wettability and interfacial tension effects on oil production. J. Pet. Sci. Eng. 2006, 52, 275–286. [Google Scholar] [CrossRef]
- Crescente, C.; Rasmussen, K.; Torsæter, O.; Strøm, A.; Kowalewski, E. An Experimental Study of Bacterial Improved Oil Recovery by using Rhodococcus sp.094. In Presented at International Symposium of the Society of Core Analysts, Toronto, Canada, 21–25 Auguest, 2005.
- Banat, I.M.; Franzetti, A.; Gandolfi, I.; Bestetti, G.; Martinotti, M.G.; Fracchia, L.; Smyth, T.J.; Marchant, R. Microbial biosurfactants production, applications and future potential. Appl. Microbiol. Biotechnol. 2010, 87, 427–444. [Google Scholar] [CrossRef] [PubMed]
- Al-Wahaibi, Y.; Joshi, S.; Al-Bahry, S.; Elshafie, A.; Al-Bemani, A.; Shibulal, B. Biosurfactant production by Bacillus subtilis B30 and its application in enhancing oil recovery. Colloids Surf. B Biointerfaces 2014, 114, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Schaller, K.D.; Fox, S.; Bruhn, D.; Noah, K.; Bala, G. Characterization of surfactin from Bacillus subtilis for application as an agent for enhanced oil recovery. Appl. Biochem. Biotechnol. 2004, 115, 827–836. [Google Scholar] [CrossRef]
- Makkar, R.S.; Cameotra, S.S. Structural characterization of a biosurfactant produced by Bacillus subtilis at 45 °C. J. Surfactants Deterg. 1999, 2, 367–372. [Google Scholar] [CrossRef]
- Youssef, N.; Simpson, D.R.; Duncan, K.E.; McInerney, M.J.; Folmsbee, M.; Fincher, T.; Knapp, R.M. In situ biosurfactant production by Bacillus strains injected into a limestone petroleum reservoir. Appl. Environ. Microbiol. 2007, 73, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Youssef, N.; Simpson, D.R.; McInerney, M.J.; Duncan, K.E. In-situ lipopeptide biosurfactant production by Bacillus strains correlates with improved oil recovery in two oil wells approaching their economic limit of production. Int. Biodeterior. Biodegrad. 2013, 81, 127–132. [Google Scholar] [CrossRef]
- Gao, C.H.; Zekri, A. Applications of microbial-enhanced oil recovery technology in the past decade. Energy Sources Part A—Recovery Util. Environ. Eff. 2011, 33, 972–989. [Google Scholar] [CrossRef]
- Maudgalya, S.; Knapp, R.; McInerney, M. Microbial Enhanced Oil-Recovery Technologies: A review of the Past, Present, and Future. In Presented at Production and Operations Symposium, Oklahoma City, OK, USA, 31 March–3 April, 2007.
- Weidong, W.; Junzhang, L.; Xueli, G.; Jing, W.; Ximing, L.; Yan, J.; Fengmin, Z. MEOR field test at block Luo801 of Shengli oil field in China. Pet. Sci. Technol. 2014, 32, 673–679. [Google Scholar] [CrossRef]
- Tuleva, B.K.; Ivanov, G.R.; Christova, N.E. Biosurfactant production by a new Pseudomonas putida strain. Z. Naturforsch. C 2002, 57, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Pruthi, V.; Cameotra, S.S. Short communication: Production and properties of a biosurfactant synthesized by Arthrobacter protophormiae: An antarctic strain. World J. Microbiol. Biotechnol. 1997, 13, 137–139. [Google Scholar] [CrossRef]
- Maier, R.M.; Soberon-Chavez, G. Pseudomonas aeruginosa rhamnolipids: Biosynthesis and potential applications. Appl. Microbiol. Biotechnol. 2000, 54, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Jenny, K.; Kappeli, O.; Fiechter, A. Biosurfactants from Bacillus licheniformis: Structural analysis and characterization. Appl. Microbiol. Biotechnol. 1991, 36, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante Barros, F.F.; de Quadrosi, C.P.; Pastore, G.M. Studies of emulsifying properties and stability of the biosurfactant produced by Bacillus subtilis in cassava wastewater. Cienc. Tecnol Aliment. 2008, 28, 979–985. [Google Scholar]
- Kim, J. Isolation and characterization of a biosurfactant-producing bacterium Bacillus pumilus IJ-1 from contaminated crude oil collected in Taean, Korea. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 5–14. [Google Scholar] [CrossRef]
- Saeki, H.; Sasaki, M.; Komatsu, K.; Miura, A.; Matsuda, H. Oil spill remediation by using the remediation agent JE1058BS that contains a biosurfactant produced by Gordonia sp strain JE-1058. Bioresour. Technol. 2009, 100, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Crescenzi, F.; Buffagni, M.; D'Angeli, E.; Porcelli, F. A new biosurfactant for use in the clean up of oil spills on sea water environment. In Oil and Hydrocarbon Spills III: Modelling, Analysis and Control; Brebbia, C.A., Ed.; Wit Press: Southampton, UK, 2002; Volume 11, pp. 245–251. [Google Scholar]
- Pavitran, S.; Jagtap, C.B.; Subramanian, S.B.; Titus, S.; Kumar, P.; Deb, P.C. Microbial bioremediation of fuel oil hydrocarbons in marine environment. Def. Sci. J. 2006, 56, 209–224. [Google Scholar] [CrossRef]
- Head, I.M.; Jones, D.M.; Roling, W.F.M. Marine microorganisms make a meal of oil. Nat. Rev. Micro 2006, 4, 173–182. [Google Scholar] [CrossRef]
- Harayama, S.; Kasai, Y.; Hara, A. Microbial communities in oil-contaminated seawater. Curr. Opin. Biotechnol. 2004, 15, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Yakimov, M.M.; Timmis, K.N.; Golyshin, P.N. Obligate oil-degrading marine bacteria. Curr. Opin. Biotechnol. 2007, 18, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Banat, I.M. Potentials for use of biosurfactants in oil spills cleanup and oil bioremediation. In Oil and Hydrocarbon Spills, Modelling, Analysis and Control II; Rodriguez, G.R., Brebbia, C.A., Eds.; Wit Press: Southampton, UK, 2000; Volume 8, pp. 177–185. [Google Scholar]
- Chhatre, S.; Purohit, H.; Shanker, R.; Khanna, P. Bacterial consortia for crude oil spill remediation. Water Sci. Technol. 1996, 34, 187–193. [Google Scholar] [CrossRef]
- Das, K.; Mukherjee, A.K. Crude petroleum-oil biodegradation efficiency of Bacillus subtilis and Pseudomonas aeruginosa strains isolated from a petroleum-oil contaminated soil from North-East India. Bioresour. Technol. 2007, 98, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, E.; Delille, D.; Delille, B. Crude oil bioremediation in sub-antarctic intertidal sediments: Chemistry and toxicity of oiled residues. Mar. Environ. Res. 2004, 57, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Harvey, S.; Elashvili, I.; Valdes, J.J.; Kamely, D.; Chakrabarty, A.M. Enhanced removal of exxon valdez spilled oil from alaskan gravel by a microbial surfactant. Nat. Biotechnol. 1990, 8, 228–230. [Google Scholar] [CrossRef]
- Swannell, R.P.; Lee, K.; McDonagh, M. Field evaluations of marine oil spill bioremediation. Microbiol. Rev. 1996, 60, 342–365. [Google Scholar] [PubMed]
- Marti, M.E.; Colonna, W.J.; Patra, P.; Zhang, H.; Green, C.; Reznik, G.; Pynn, M.; Jarrell, K.; Nyman, J.A.; Somasundaran, P.; et al. Production and characterization of microbial biosurfactants for potential use in oil-spill remediation. Enzym. Microb. Technol. 2014, 55, 31–39. [Google Scholar] [CrossRef]
- Asaka, O.; Shoda, M. Biocontrol of Rhizoctonia solani damping-off of tomato with Bacillus subtilis RB14. Appl. Environ. Microbiol. 1996, 62, 4081–4085. [Google Scholar] [PubMed]
- Joshi, S.; Bharucha, C.; Desai, A.J. Production of biosurfactant and antifungal compound by fermented food isolate Bacillus subtilis 20B. Bioresour. Technol. 2008, 99, 4603–4608. [Google Scholar] [CrossRef] [PubMed]
- Tendulkar, S.R.; Saikumari, Y.K.; Patel, V.; Raghotama, S.; Munshi, T.K.; Balaram, P.; Chattoo, B.B. Isolation, purification and characterization of an antifungal molecule produced by Bacillus licheniformis BC98, and its effect on phytopathogen magnaporthe grisea. J. Appl. Microbiol. 2007, 103, 2331–2339. [Google Scholar] [CrossRef] [PubMed]
- Bechard, J.; Eastwell, K.C.; Sholberg; Mazza, G.; Skura, B. Isolation and partial chemical characterization of an antimicrobial peptide produced by a strain of Bacillus subtilis. J. Agric. Food Chem. 1998, 46, 5355–5361. [Google Scholar] [CrossRef]
- Brötz, H.; Sahl, H.-G. New insights into the mechanism of action of lantibiotics-diverse biological effects by binding to the same molecular target. J. Antimicrob. Chemother. 2000, 46, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Heerklotz, H.; Seelig, J. Leakage and lysis of lipid membranes induced by the lipopeptide surfactin. Eur. Biophys. J. 2007, 36, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, C.; Teruel, J.A.; Aranda, F.J.; Ortiz, A. Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin. Biochim. Biophys. Acta 2003, 1611, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Lopez, S.; Kim, H.-S.; Choi, E.C.; Delgado, M.; Granja, J.R.; Khasanov, A.; Kraehenbuehl, K.; Long, G.; Weinberger, D.A.; Wilcoxen, K.M.; et al. Antibacterial agents based on the cyclic d,l-α-peptide architecture. Nature 2001, 412, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.J. Biosurfactants for cosmetic applications. Int. J. Cosmet. Sci. 1991, 13, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Nitschke, M.; Costa, S.G.V.A.O. Biosurfactants in food industry. Trends Food Sci. Technol. 2007, 18, 252–259. [Google Scholar] [CrossRef]
- Mulligan, C.N. Environmental applications for biosurfactants. Environ. Pollut. 2005, 133, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, C.N.; Yong, R.N.; Gibbs, B.F.; James, S.; Bennett, H.P.J. Metal removal from contaminated soil and sediments by the biosurfactant surfactin. Environ. Sci. Technol. 1999, 33, 3812–3820. [Google Scholar] [CrossRef]
- Zouboulis, A.I.; Matis, K.A.; Lazaridis, N.K.; Golyshin, P.N. The use of biosurfactants in flotation: Application for the removal of metal ions. Miner. Eng. 2003, 16, 1231–1236. [Google Scholar] [CrossRef]
- Iwamoto, T.; Fujie, A.; Sakamoto, K.; Tsurumi, Y.; Shigematsu, N.; Yamashita, M.; Hashimoto, S.; Okuhara, M.; Kohsaka, M. WF11899A, B and C, novel antifungal lipopeptides: I. Taxonomy, fermentation, isolation and physico-chemical properties. J. Antibiot. 1994, 47, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.H.; Thrane, C.; Christophersen, C.; Anthoni, U.; Sorensen, J. Structure, production characteristics and fungal antagonism of tensin—A new antifungal cyclic lipopeptide from Pseudomonas fluorescens strain 96.578. J. Appl. Microbiol. 2000, 89, 992–1001. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.I.; Bai, H.; Bai, D.; Chae, H.; Chung, S.; Kim, Y.; Park, R.; Chi, Y.T. Purification and characterization of a lipopeptide produced by Bacillus thuringiensis CMB26. J. Appl. Microbiol. 2004, 97, 942–949. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.-F.; Mbadinga, S.M.; Yang, S.-Z.; Gu, J.-D.; Mu, B.-Z. Chemical Structure, Property and Potential Applications of Biosurfactants Produced by Bacillus subtilis in Petroleum Recovery and Spill Mitigation. Int. J. Mol. Sci. 2015, 16, 4814-4837. https://doi.org/10.3390/ijms16034814
Liu J-F, Mbadinga SM, Yang S-Z, Gu J-D, Mu B-Z. Chemical Structure, Property and Potential Applications of Biosurfactants Produced by Bacillus subtilis in Petroleum Recovery and Spill Mitigation. International Journal of Molecular Sciences. 2015; 16(3):4814-4837. https://doi.org/10.3390/ijms16034814
Chicago/Turabian StyleLiu, Jin-Feng, Serge Maurice Mbadinga, Shi-Zhong Yang, Ji-Dong Gu, and Bo-Zhong Mu. 2015. "Chemical Structure, Property and Potential Applications of Biosurfactants Produced by Bacillus subtilis in Petroleum Recovery and Spill Mitigation" International Journal of Molecular Sciences 16, no. 3: 4814-4837. https://doi.org/10.3390/ijms16034814






