Hip Osteoarthritis in Dogs: A Randomized Study Using Mesenchymal Stem Cells from Adipose Tissue and Plasma Rich in Growth Factors
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Animal Population
2.1.2. Pain Assessment
| Characteristic | aMSCs | PRGF | p-Value |
|---|---|---|---|
| Age (months) | 53 ± 43 (8–135) | 93 ± 35.5 (18–66) | 0.007 |
| Weight | 34.9 ± 12.8 (18.3–66.2) | 36.5 ± 10.6 (20–62.8) | 0.805 |
| Gender (male-female) | 10-8 | 14-3 | 0.201 |
| Radiographic Osteoarthritis degree Bioarth Score * | 15.47 ± 6.02 (13.37–17.57) | 17.36 ± 3.8 (16.07–18.65) | 0.259 |
| Functional limitation | 5.88 ± 2.83 (2–11) | 8.22 ± 3.39 (1–14) | 0.003 |
| Joint mobility | 4.18 ± 0.63 (3–5) | 3.92 ± 0.84 (2–6) | 0.131 |
| Muscle atrophy | 1.05 ± 0.55572 | 1.0526 ± 0.62126 | 0.890 |
| Owner Vas Score | 28.29 ± 14.49 (6.57–72.37) | 42.55 ± 20.52 (9.21–72.37) | 0.023 |
| Patients | 18 | 17 |
| Variable | Time | Group | N | Mean | Std. Deviation | 95% Confidence Interval for Mean | Minimum | Maximum | p-Value between Groups | |
|---|---|---|---|---|---|---|---|---|---|---|
| Lower Bound | Upper Bound | |||||||||
| Owner VAS | baseline | aMSCs | 34 | 28.6353 | 14.33904 | 23.6322 | 33.6384 | 6.57 | 53.94 | 0.001 |
| PRGF | 36 | 42.9756 | 20.61379 | 36.0009 | 49.9503 | 9.21 | 72.37 | |||
| Total | 70 | 36.0103 | 19.13089 | 31.4487 | 40.5719 | 6.57 | 72.37 | |||
| 1 month | aMSCs | 34 | 15.4312 | 11.03277 | 11.5817 | 19.2807 | 0.00 | 48.68 | 0.000 | |
| PRGF | 36 | 27.5978 | 13.61288 | 22.9918 | 32.2037 | 5.92 | 59.21 | |||
| Total | 70 | 21.6883 | 13.77408 | 18.4040 | 24.9726 | 0.00 | 59.21 | |||
| 3 months | aMSCs | 34 | 17.5500 | 15.87660 | 12.0104 | 23.0896 | 1.31 | 57.89 | 0.014 | |
| PRGF | 36 | 27.4081 | 16.72219 | 21.7501 | 33.0660 | 4.60 | 72.37 | |||
| Total | 70 | 22.6199 | 16.94175 | 18.5802 | 26.6595 | 1.31 | 72.37 | |||
| 6 months | aMSCs | 30 | 16.8620 | 13.55143 | 11.8018 | 21.9222 | 2.63 | 51.31 | 0.046 | |
| PRGF | 34 | 24.6088 | 16.43983 | 18.8727 | 30.3450 | 0.00 | 56.58 | |||
| Total | 64 | 20.9775 | 15.53332 | 17.0974 | 24.8576 | 0.00 | 56.58 | |||
| Veterinarian VAS | baseline | aMSCs | 34 | 23.8200 | 10.77498 | 20.0604 | 27.5796 | 6.57 | 50.66 | 0.000 |
| PRGF | 36 | 32.9692 | 9.88435 | 29.6248 | 36.3136 | 10.60 | 48.68 | |||
| Total | 70 | 28.5253 | 11.23820 | 25.8456 | 31.2049 | 6.57 | 50.66 | |||
| 1 month | aMSCs | 34 | 14.0894 | 6.38365 | 11.8621 | 16.3168 | 6.57 | 32.89 | 0.000 | |
| PRGF | 36 | 24.8964 | 7.74936 | 22.2744 | 27.5184 | 8.00 | 42.76 | |||
| Total | 70 | 19.6473 | 8.91895 | 17.5206 | 21.7739 | 6.57 | 42.76 | |||
| 3 months | aMSCs | 34 | 10.8635 | 5.89049 | 8.8082 | 12.9188 | 3.95 | 26.31 | 0.000 | |
| PRGF | 36 | 22.0383 | 7.50788 | 19.4980 | 24.5786 | 6.00 | 40.79 | |||
| Total | 70 | 16.6106 | 8.76544 | 14.5205 | 18.7006 | 3.95 | 40.79 | |||
| 6 months | aMSCs | 32 | 9.5306 | 5.77949 | 7.4469 | 11.6144 | 3.95 | 29.60 | 0.000 | |
| PRGF | 34 | 24.8297 | 8.75538 | 21.7748 | 27.8846 | 6.00 | 50.00 | |||
| Total | 66 | 17.4120 | 10.68689 | 14.7848 | 20.0391 | 3.95 | 50.00 | |||
2.1.3. Degree of Osteoarthritis Based on the Radiographic Findings
2.1.4. Bioarth Scale Assessment
| Variable | Time | Group | N | Mean | Std. Deviation | 95% Confidence Interval for Mean | Minimum | Maximum | p-Value between Groups | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower Bound | Upper Bound | ||||||||||||
| OA degree | baseline | aMSCs | 34 | 15.47 | 6.02 | 13.37 | 17.57 | 3.00 | 21.00 | 0.259 | |||
| PRGF | 36 | 17.36 | 3.80 | 16.07 | 18.65 | 10.00 | 21.00 | ||||||
| Total | 70 | 16.44 | 5.06 | 15.24 | 17.65 | 3.00 | 21.00 | ||||||
| 1 month | aMSCs | 34 | 15.62 | 6.11 | 13.49 | 17.75 | 3.00 | 21.00 | 0.339 | ||||
| PRGF | 36 | 17.36 | 3.80 | 16.07 | 18.65 | 10.00 | 21.00 | ||||||
| Total | 70 | 16.51 | 5.10 | 15.30 | 17.73 | 3.00 | 21.00 | ||||||
| 3 months | aMSCs | 34 | 15.74 | 6.16 | 13.59 | 17.88 | 3.00 | 21.00 | 0.402 | ||||
| PRGF | 36 | 17.39 | 3.81 | 16.10 | 18.68 | 10.00 | 21.00 | ||||||
| Total | 70 | 16.59 | 5.12 | 15.36 | 17.81 | 3.00 | 21.00 | ||||||
| 6 months | aMSCs | 32 | 15.75 | 6.44 | 13.43 | 18.07 | 3.00 | 21.00 | 0.714 | ||||
| PRGF | 34 | 17.18 | 3.82 | 15.84 | 18.51 | 10.00 | 21.00 | ||||||
| Total | 66 | 16.48 | 5.26 | 15.19 | 17.78 | 3.00 | 21.00 | ||||||
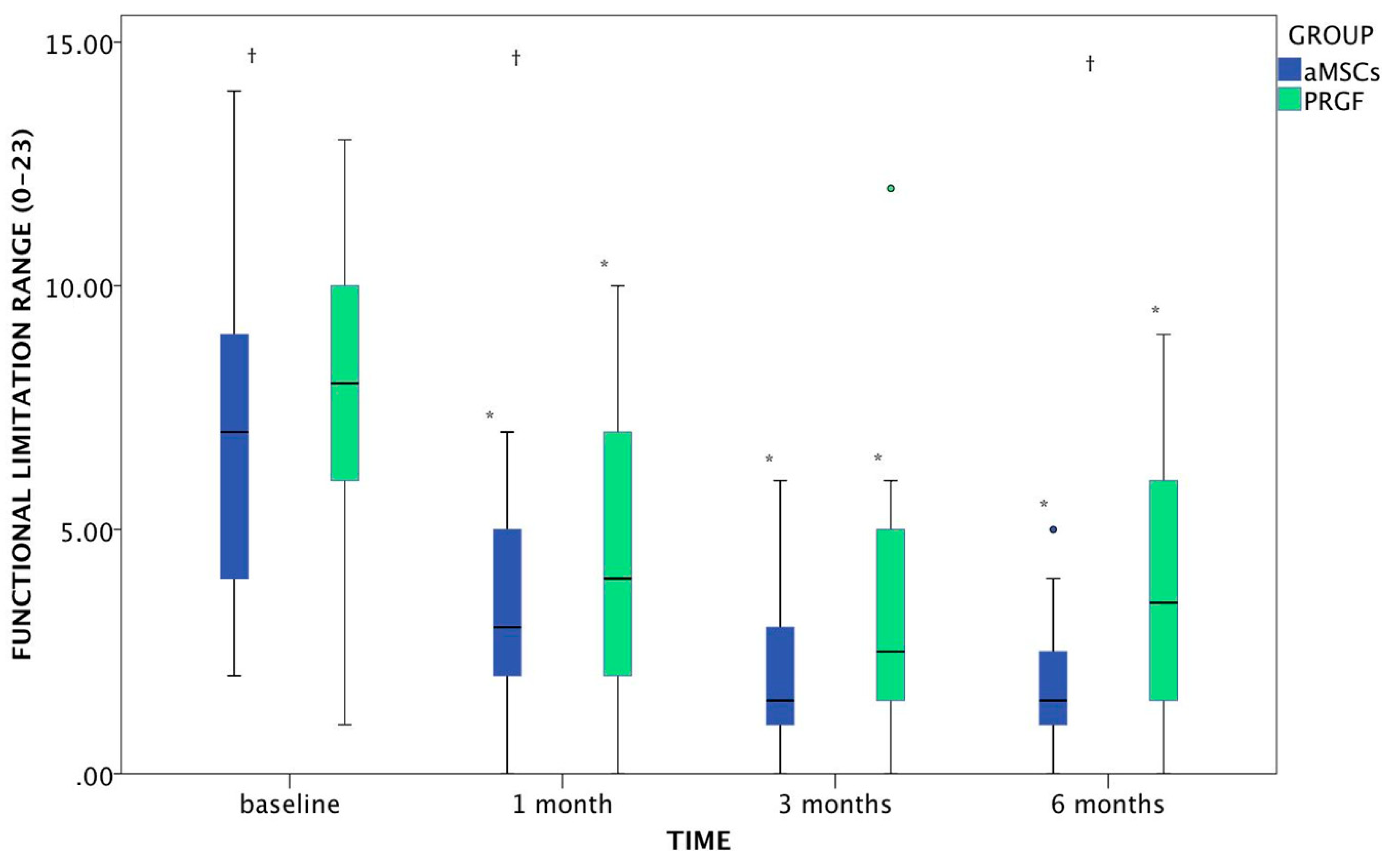
| Functional Limitation | baseline | aMSCs | 34 | 5.88 | 2.83 | 4.90 | 6.87 | 2.00 | 11.00 | 0.003 | |||
| PRGF | 36 | 8.22 | 3.39 | 7.08 | 9.37 | 1.00 | 14.00 | ||||||
| Total | 70 | 7.09 | 3.32 | 6.29 | 7.88 | 1.00 | 14.00 | ||||||
| 1 month | aMSCs | 34 | 2.85 * | 1.94 | 2.18 | 3.53 | 0.00 | 7.00 | 0.009 | ||||
| PRGF | 36 | 4.56 * | 2.75 | 3.62 | 5.49 | 0.00 | 10.00 | ||||||
| Total | 70 | 3.73 | 2.52 | 3.13 | 4.33 | 0.00 | 10.00 | ||||||
| 3 months | aMSCs | 34 | 2.26 * | 1.75 | 1.66 | 2.87 | 0.00 | 6.00 | 0.181 | ||||
| PRGF | 36 | 3.33 * | 2.95 | 2.34 | 4.33 | 0.00 | 12.00 | ||||||
| Total | 70 | 2.81 | 2.48 | 2.22 | 3.41 | 0.00 | 12.00 | ||||||
| 6 months | aMSCs | 30 | 1.80 * | 1.54 | 1.22 | 2.38 | 0.00 | 5.00 | 0.004 | ||||
| PRGF | 34 | 3.71 * | 2.76 | 2.74 | 4.67 | 0.00 | 9.00 | ||||||
| Total | 64 | 2.81 | 2.45 | 2.20 | 3.42 | 0.00 | 9.00 | ||||||
| Joint Mobility | baseline | aMSCs | 34 | 4.18 | 0.63 | 3.96 | 4.39 | 3.00 | 5.00 | 0.131 | |||
| PRGF | 36 | 3.92 | 0.84 | 3.63 | 4.20 | 2.00 | 6.00 | ||||||
| Total | 70 | 4.04 | 0.75 | 3.86 | 4.22 | 2.00 | 6.00 | ||||||
| 1 month | aMSCs | 34 | 2.4 * | 1.23 | 1.98 | 2.84 | 0.00 | 4.00 | 0.057 | ||||
| PRGF | 36 | 2.97 * | 0.88 | 2.68 | 3.27 | 1.00 | 4.00 | ||||||
| Total | 70 | 2.70 | 1.09 | 2.44 | 2.96 | 0.00 | 4.00 | ||||||
| 3 months | aMSCs | 34 | 1.38 * | 1.52 | 0.85 | 1.91 | 0.00 | 4.00 | 0.000 | ||||
| PRGF | 36 | 2.75 * | 1.02 | 2.40 | 3.10 | 1.00 | 4.00 | ||||||
| Total | 70 | 2.09 | 1.45 | 1.74 | 2.43 | 0.00 | 4.00 | ||||||
| 6 months | aMSCs | 32 | 1.09 * | 1.40 | 0.59 | 1.60 | 0.00 | 4.00 | 0.000 | ||||
| PRGF | 34 | 2.82 * | 1.00 | 2.47 | 3.17 | 1.00 | 4.00 | ||||||
| Total | 66 | 1.98 | 1.48 | 1.62 | 2.35 | 0.00 | 4.00 | ||||||
| Muscle Perimeter (cm) | baseline | aMSCs | 34 | 32.56 | 5.37 | 30.69 | 34.44 | 25.00 | 47.00 | 0.609 | |||
| PRGF | 36 | 32.42 | 3.07 | 31.38 | 33.46 | 27.50 | 41.00 | ||||||
| Total | 70 | 32.49 | 4.31 | 31.46 | 33.52 | 25.00 | 47.00 | ||||||
| 1 month | aMSCs | 34 | 33.51 * | 4.97 | 31.78 | 35.25 | 27.00 | 45.00 | 0.005 | ||||
| PRGF | 36 | 32.56 | 3.24 | 31.46 | 33.65 | 26.00 | 40.00 | ||||||
| Total | 70 | 33.02 | 4.17 | 32.03 | 34.01 | 26.00 | 45.00 | ||||||
| 3 months | aMSCs | 34 | 33.26 * | 4.40 | 31.73 | 34.80 | 28.00 | 43.00 | 0.984 | ||||
| PRGF | 36 | 32.34 | 3.33 | 31.21 | 33.46 | 25.00 | 41.00 | ||||||
| Total | 70 | 32.79 | 3.89 | 31.86 | 33.71 | 25.00 | 43.00 | ||||||
| 6 months | aMSCs | 32 | 33.98 * | 4.51 | 32.36 | 35.61 | 27.00 | 43.00 | 0.000 | ||||
| PRGF | 34 | 32.50 | 3.35 | 31.33 | 33.67 | 26.00 | 40.00 | ||||||
| Total | 66 | 33.22 | 4.00 | 32.24 | 34.20 | 26.00 | 43.00 | ||||||
| Range of Motion | baseline | aMSCs | 34 | 64.12 | 12.06 | 59.91 | 68.33 | 40.00 | 90.00 | 0.000 | |||
| PRGF | 36 | 75.61 | 12.22 | 71.48 | 79.75 | 54.00 | 95.00 | ||||||
| Total | 70 | 70.03 | 13.37 | 66.84 | 73.22 | 40.00 | 95.00 | ||||||
| 1 month | aMSCs | 34 | 96.65 * | 13.21 | 92.04 | 101.26 | 60.00 | 125.00 | 0.002 | ||||
| PRGF | 36 | 87.69 * | 11.71 | 83.73 | 91.66 | 55.00 | 105.00 | ||||||
| Total | 70 | 92.04 | 13.16 | 88.90 | 95.18 | 55.00 | 125.00 | ||||||
| 3 months | aMSCs | 34 | 103.71 * | 12.26 | 99.43 | 107.98 | 60.00 | 125.00 | 0.000 | ||||
| PRGF | 36 | 92.67 * | 11.35 | 88.83 | 96.51 | 68.00 | 115.00 | ||||||
| Total | 70 | 98.03 | 12.96 | 94.94 | 101.12 | 60.00 | 125.00 | ||||||
| 6 months | aMSCs | 32 | 107.09 * | 8.56 | 104.01 | 110.18 | 90.00 | 122.00 | 0.000 | ||||
| PRGF | 34 | 90.50 * | 11.24 | 86.58 | 94.42 | 60.00 | 105.00 | ||||||
| Total | 66 | 98.55 | 12.99 | 95.35 | 101.74 | 60.00 | 122.00 | ||||||
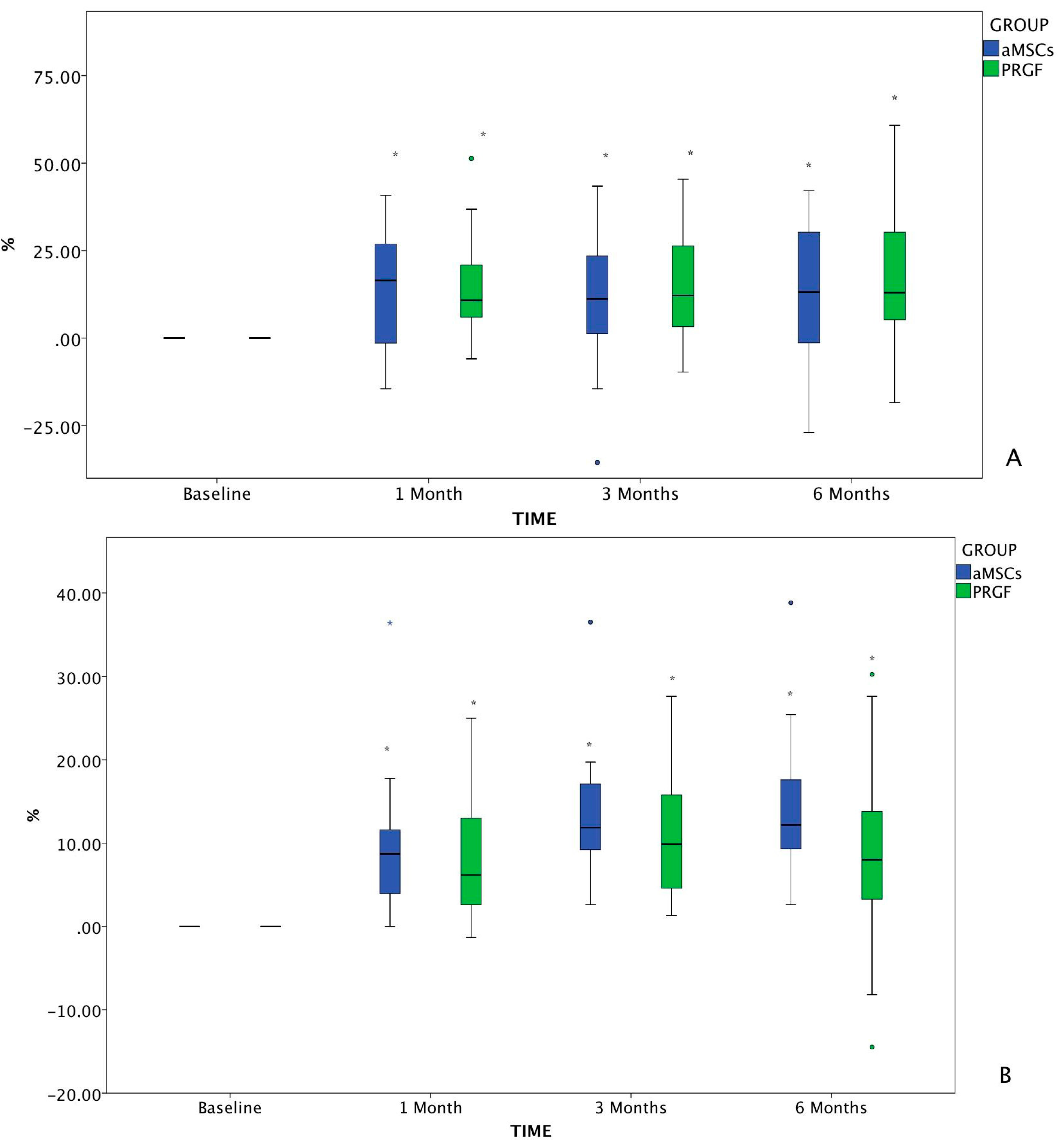
Functional Limitation
Joint Mobility
Muscle Atrophy
2.1.5. Overall Satisfaction with Treatment

2.1.6. Safety Assessment
2.1.7. Requirement of Rescue Analgesia
2.2. Discussion
3. Experimental Section
3.1. Animal Population and Randomization
- ○
- aMSCs: single intra-articular injection of autologous aMSCs (DogStem®, Fat-Stem, Aalst, Belgium) (2 mL containing 30 million aMSCs) (n = 18).
- ○
- PRGF: single intra-articular injection of autologous PRGF (PRGF-Endoret®, BTI Biotechnology Institute, Vitoria-Gasteiz, Araba, Spain) (2 mL) (n = 19).
3.2. Treatments Applied
3.2.1. aMSCs
- Morphology: cells are plastic adherent, lack osmotic shrinkage, normal cell surface and fibroblastic shape.
- Ease of trypsinization: a single trypsinization.
- Color of the cultivation medium: Phenol red indicator is used to evaluate pH changes, which might affect growth of the stem cells.
- Proliferation time (doubling time): The population-doubling time (PDT) is defined as the time required for a colony area to expand twofold. Population doubling time calculates growth proliferation. PDT results are graphically visualized over passage time.
- Viability: Viability of the cells were quantified using trypan blue and cell counting methods were used.
- Cells were expanded and 30 million cells were used for clinical purpose.
3.2.2. PRGF
3.3. Outcome Variables
3.3.1. Pain Assessment


3.3.2. Bioarth Assessment Scale [82]
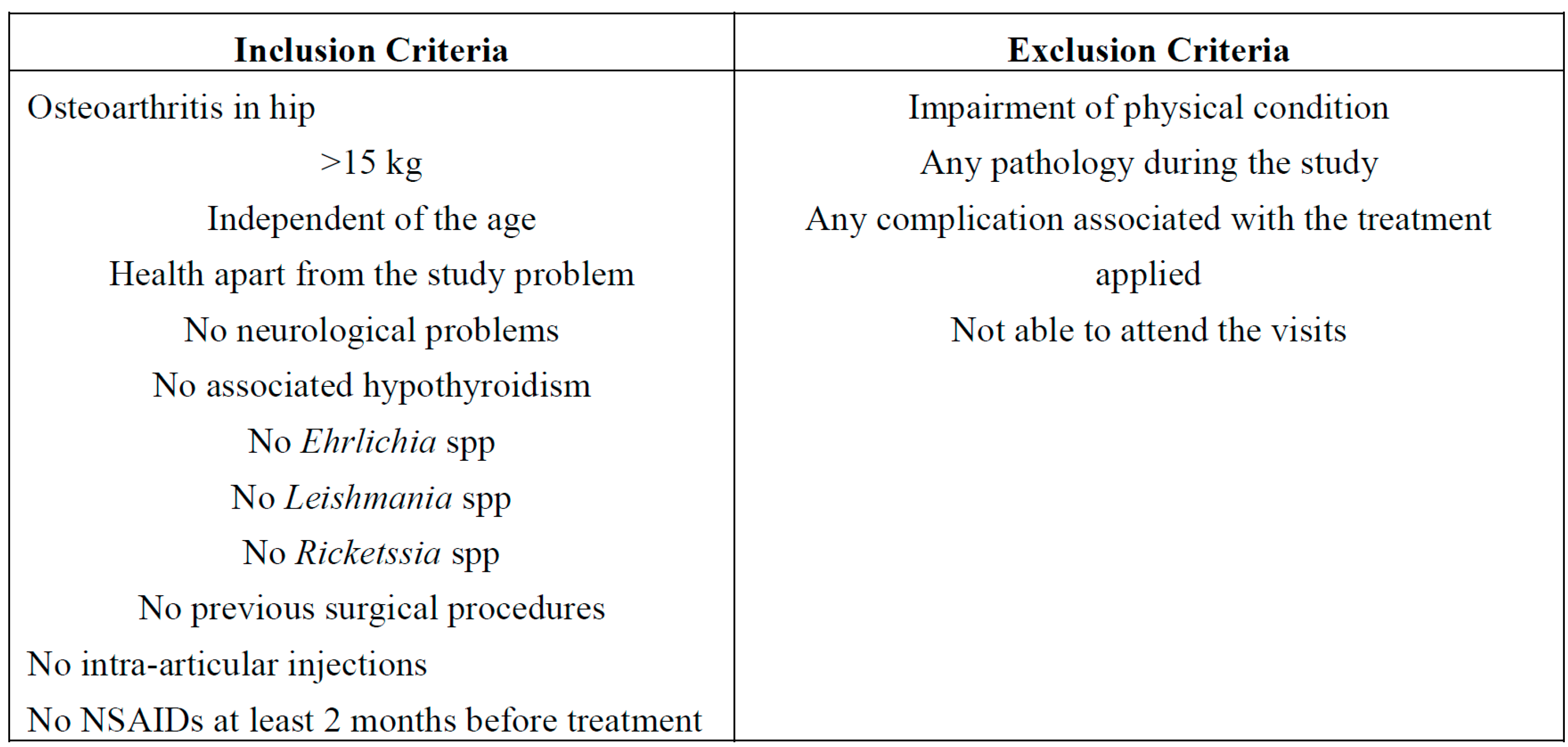
- Radiological assessment quantifies radiographic signs of osteoarthritis in canine elbows, hips and knees. Classifies the degrees of osteoarthritis into 4 categories 1: no signs of OA (0–2 points), 2: mild OA (3–8 points), 3: moderate OA (9–14 points), 4: severe OA (more than 14 points), based on a numerical assessment (0–3) of 7 anatomical points in the joint (limits of total score 0–21).
- Functional assessment evaluates the 3 basic functional parameters: functional limitation, joint mobility and muscle atrophy.
- ○
- Functional limitation quantifies the weight-bearing or support of each extremity, changes in posture when standing still, signs of lameness when cold and when walking, endurance during walking and playing, endurance when going up stairs and strength limitations in small jumps (measurement scale: 0 to 23 points).
- ○
- Joint mobility score is a summation of joint motion limitation, of the degree of flexion and extension of the joint studied (ROM: extension minus flexion) and pain when performing this assessment, with the goniometer (measurement scale: 0 to 7 points).
- ○
- Muscle atrophy measured in centimeters, circumference of musculature at standard anatomical references. A measure ribbon was used taking three measurements and an average calculated. (Measurement scale: 0: no atrophy, 1: mild atrophy, 2: severe atrophy).
3.3.3. Owner Satisfaction with the Treatment
- ○
- very poor
- ○
- poor
- ○
- fair
- ○
- good
- ○
- excellent
3.3.4. Safety Assessments
3.3.5. Requirement of Rescue Analgesia
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Juni, P.; Reichenbach, S.; Dieppe, P. Osteoarthritis: Rational approach to treating the individual. Best Pract. Res. Clin. Rheumatol. 2006, 20, 721–740. [Google Scholar] [CrossRef]
- Malek, S.; Sample, S.J.; Schwartz, Z.; Nemke, B.; Jacobson, P.B.; Cozzi, E.M.; Schaefer, S.L.; Bleedorn, J.A.; Holzman, G.; Muir, P. Effect of analgesic therapy on clinical outcome measures in a randomized controlled trial using client-owned dogs with hip osteoarthritis. BMC Vet. Res. 2012, 8, 185. [Google Scholar] [CrossRef]
- Rychel, J.K. Diagnosis and treatment of osteoarthritis. Top. Companion Anim. Med. 2010, 25, 20–25. [Google Scholar] [CrossRef]
- Egloff, C.; Hugle, T.; Valderrabano, V. Biomechanics and pathomechanisms of osteoarthritis. Swiss Med. Wkly. 2012, 142, w13583. [Google Scholar] [CrossRef]
- Vaquerizo, V.; Plasencia, M.A.; Arribas, I.; Seijas, R.; Padilla, S.; Orive, G.; Anitua, E. Comparison of intra-articular injections of plasma rich in growth factors (prgf-endoret) versus durolane hyaluronic acid in the treatment of patients with symptomatic osteoarthritis: A randomized controlled trial. Arthroscopy 2013, 29, 1635–1643. [Google Scholar] [CrossRef]
- Singh, J.A. Stem cells and other innovative intra-articular therapies for osteoarthritis: What does the future hold? BMC Med. 2012, 10, 44. [Google Scholar] [CrossRef]
- Wu, L.; Cai, X.; Zhang, S.; Karperien, M.; Lin, Y. Regeneration of articular cartilage by adipose tissue derived mesenchymal stem cells: Perspectives from stem cell biology and molecular medicine. J. Cell. Physiol. 2013, 228, 938–944. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Adams, C.; Dhupa, S.; Sams, A.E.; Taylor, R.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of intraarticular injection of autologous adipose-derived mesenchymal stem and regenerative cells on clinical signs of chronic osteoarthritis of the elbow joint in dogs. Vet. Ther. 2008, 9, 192–200. [Google Scholar]
- Wu, W.; Chen, F.; Liu, Y.; Ma, Q.; Mao, T. Autologous injectable tissue-engineered cartilage by using platelet-rich plasma: Experimental study in a rabbit model. J. Oral Maxillofac. Surg. 2007, 65, 1951–1957. [Google Scholar] [CrossRef]
- Wang-Saegusa, A.; Cugat, R.; Ares, O.; Seijas, R.; Cusco, X.; Garcia-Balletbo, M. Infiltration of plasma rich in growth factors for osteoarthritis of the knee short-term effects on function and quality of life. Arch. Orthop. Trauma Surg. 2011, 131, 311–317. [Google Scholar] [CrossRef]
- Raghunath, J.; Salacinski, H.J.; Sales, K.M.; Butler, P.E.; Seifalian, A.M. Advancing cartilage tissue engineering: The application of stem cell technology. Curr. Opin. Biotechnol. 2005, 16, 503–509. [Google Scholar] [CrossRef]
- Oldershaw, R.A. Cell sources for the regeneration of articular cartilage: The past, the horizon and the future. Int. J. Exp. Pathol. 2012, 93, 389–400. [Google Scholar]
- Mizuno, H.; Tobita, M.; Uysal, A.C. Concise review: Adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells 2012, 30, 804–810. [Google Scholar] [CrossRef]
- Timmins, N.E.; Kiel, M.; Gunther, M.; Heazlewood, C.; Doran, M.R.; Brooke, G.; Atkinson, K. Closed system isolation and scalable expansion of human placental mesenchymal stem cells. Biotechnol. Bioeng. 2012, 109, 1817–1826. [Google Scholar] [CrossRef]
- Yang, S.; Huang, S.; Feng, C.; Fu, X. Umbilical cord-derived mesenchymal stem cells: Strategies, challenges, and potential for cutaneous regeneration. Front. Med. 2012, 6, 41–47. [Google Scholar] [CrossRef]
- Jackson, W.M.; Lozito, T.P.; Djouad, F.; Kuhn, N.Z.; Nesti, L.J.; Tuan, R.S. Differentiation and regeneration potential of mesenchymal progenitor cells derived from traumatized muscle tissue. J. Cell. Mol. Med. 2011, 15, 2377–2388. [Google Scholar] [CrossRef]
- Jones, B.A.; Pei, M. Synovium-derived stem cells: A tissue-specific stem cell for cartilage engineering and regeneration. Tissue Eng. Part B Rev. 2012, 18, 301–311. [Google Scholar] [CrossRef]
- Jones, E.A.; Crawford, A.; English, A.; Henshaw, K.; Mundy, J.; Corscadden, D.; Chapman, T.; Emery, P.; Hatton, P.; McGonagle, D. Synovial fluid mesenchymal stem cells in health and early osteoarthritis: Detection and functional evaluation at the single-cell level. Arthritis Rheumatol. 2008, 58, 1731–1740. [Google Scholar] [CrossRef]
- Yarak, S.; Okamoto, O.K. Human adipose-derived stem cells: Current challenges and clinical perspectives. An. Bras. Dermatol. 2010, 85, 647–656. [Google Scholar] [CrossRef]
- Rodríguez-Jiménez, F.J.; Valdes-Sánchez, T.; Carrillo, J.M.; Rubio, M.; Monleon-Prades, M.; García-Cruz, D.M.; García, M.; Cugat, R.; Moreno-Manzano, V. Platelet-rich plasma favors proliferation of canine adipose-derived mesenchymal stem cells in methacrylate-endcapped caprolactone porous scaffold niches. J. Funct. Biomater. 2012, 3, 556–568. [Google Scholar] [CrossRef]
- Agung, M.; Ochi, M.; Yanada, S.; Adachi, N.; Izuta, Y.; Yamasaki, T.; Toda, K. Mobilization of bone marrow-derived mesenchymal stem cells into the injured tissues after intraarticular injection and their contribution to tissue regeneration. Knee Surg. Sports Traumatol. Arthrosc. 2006, 14, 1307–1314. [Google Scholar] [CrossRef]
- Vilar, J.M.; Morales, M.; Santana, A.; Spinella, G.; Rubio, M.; Cuervo, B.; Cugat, R.; Carrillo, J.M. Controlled, blinded force platform analysis of the effect of intraarticular injection of autologous adipose-derived mesenchymal stem cells associated to prgf-endoret in osteoarthritic dogs. BMC Vet. Res. 2013, 9, 131. [Google Scholar] [CrossRef]
- Bubnov, R.; Yevseenko, V.; Semeniv, I. Ultrasound guided injections of platelets rich plasma for muscle injury in professional athletes. Comparative study. Med. Ultrason. 2013, 15, 101–105. [Google Scholar] [CrossRef]
- Grageda, E.; Lozada, J.L.; Boyne, P.J.; Caplanis, N.; McMillan, P.J. Bone formation in the maxillary sinus by using platelet-rich plasma: An experimental study in sheep. J. Oral Implantol. 2005, 31, 2–17. [Google Scholar] [CrossRef]
- Marx, R.E. Platelet-rich plasma: Evidence to support its use. J. Oral Maxillofac. Surg. 2004, 62, 489–496. [Google Scholar] [CrossRef]
- Pietrzak, W.S.; Eppley, B.L. Platelet rich plasma: Biology and new technology. J. Craniofac. Surg. 2005, 16, 1043–1054. [Google Scholar] [CrossRef]
- Anitua, E.; Andia, I.; Ardanza, B.; Nurden, P.; Nurden, A.T. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb. Haemost. 2004, 91, 4–15. [Google Scholar]
- Anitua, E.; Sanchez, M.; Orive, G.; Andia, I. The potential impact of the preparation rich in growth factors (prgf) in different medical fields. Biomaterials 2007, 28, 4551–4560. [Google Scholar] [CrossRef]
- Park, Y.G.; Han, S.B.; Song, S.J.; Kim, T.J.; Ha, C.W. Platelet-rich plasma therapy for knee joint problems: Review of the literature, current practice and legal perspectives in korea. Knee Surg. Relat. Res. 2012, 24, 70–78. [Google Scholar] [CrossRef]
- Velnar, T.; Bailey, T.; Smrkolj, V. The wound healing process: An overview of the cellular and molecular mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef]
- Filardo, G.; Kon, E.; Pereira Ruiz, M.T.; Vaccaro, F.; Guitaldi, R.; di Martino, A.; Cenacchi, A.; Fornasari, P.M.; Marcacci, M. Platelet-rich plasma intra-articular injections for cartilage degeneration and osteoarthritis: Single- versus double-spinning approach. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 2082–2091. [Google Scholar] [CrossRef]
- Fahie, M.A.; Ortolano, G.A.; Guercio, V.; Schaffer, J.A.; Johnston, G.; Au, J.; Hettlich, B.A.; Phillips, T.; Allen, M.J.; Bertone, A.L. A randomized controlled trial of the efficacy of autologous platelet therapy for the treatment of osteoarthritis in dogs. J. Am. Vet. Med. Assoc. 2013, 243, 1291–1297. [Google Scholar] [CrossRef]
- Tuan, R.S.; Boland, G.; Tuli, R. Adult mesenchymal stem cells and cell-based tissue engineering. Arthritis Res. Ther. 2003, 5, 32–45. [Google Scholar] [CrossRef] [Green Version]
- Mizuno, H. Adipose-derived stem cells for tissue repair and regeneration: Ten years of research and a literature review. J. Nippon Med. Sch. 2009, 76, 56–66. [Google Scholar] [CrossRef]
- Bigham-Sadegh, A.; Mirshokraei, P.; Karimi, I.; Oryan, A.; Aparviz, A.; Shafiei-Sarvestani, Z. Effects of adipose tissue stem cell concurrent with greater omentum on experimental long-bone healing in dog. Connect. Tissue Res. 2012, 53, 334–342. [Google Scholar] [CrossRef]
- Haghighat, A.; Akhavan, A.; Hashemi-Beni, B.; Deihimi, P.; Yadegari, A.; Heidari, F. Adipose derived stem cells for treatment of mandibular bone defects: An autologous study in dogs. Dent. Res. J. 2011, 8, S51–S57. [Google Scholar]
- Chung, C.S.; Fujita, N.; Kawahara, N.; Yui, S.; Nam, E.; Nishimura, R. A comparison of neurosphere differentiation potential of canine bone marrow-derived mesenchymal stem cells and adipose-derived mesenchymal stem cells. J. Vet. Med. Sci. 2013, 75, 879–886. [Google Scholar] [CrossRef]
- Martinello, T.; Bronzini, I.; Maccatrozzo, L.; Mollo, A.; Sampaolesi, M.; Mascarello, F.; Decaminada, M.; Patruno, M. Canine adipose-derived-mesenchymal stem cells do not lose stem features after a long-term cryopreservation. Res. Vet. Sci. 2011, 91, 18–24. [Google Scholar]
- Reich, C.M.; Raabe, O.; Wenisch, S.; Bridger, P.S.; Kramer, M.; Arnhold, S. Isolation, culture and chondrogenic differentiation of canine adipose tissue- and bone marrow-derived mesenchymal stem cells—A comparative study. Vet. Res. Commun. 2012, 36, 139–148. [Google Scholar] [CrossRef]
- Kang, B.J.; Ryu, H.H.; Park, S.S.; Koyama, Y.; Kikuchi, M.; Woo, H.M.; Kim, W.H.; Kweon, O.K. Comparing the osteogenic potential of canine mesenchymal stem cells derived from adipose tissues, bone marrow, umbilical cord blood, and wharton’s jelly for treating bone defects. J. Vet. Sci. 2012, 13, 299–310. [Google Scholar] [CrossRef]
- Guercio, A.; di Marco, P.; Casella, S.; Cannella, V.; Russotto, L.; Purpari, G.; di Bella, S.; Piccione, G. Production of canine mesenchymal stem cells from adipose tissue and their application in dogs with chronic osteoarthritis of the humeroradial joints. Cell Biol. Int. 2012, 36, 189–194. [Google Scholar] [CrossRef]
- Oh, H.J.; Park, E.J.; Lee, S.Y.; Soh, J.W.; Kong, I.S.; Choi, S.W.; Ra, J.C.; Kang, S.K.; Lee, B.C. Comparison of cell proliferation and epigenetic modification of gene expression patterns in canine foetal fibroblasts and adipose tissue-derived mesenchymal stem cells. Cell Prolif. 2012, 45, 438–444. [Google Scholar] [CrossRef]
- Dhillon, M.; Patel, S.; Bali, K. Comment on: Platelet-rich plasma intra-articular knee injections for the treatment of degenerative cartilage lesions and osteoarthritis. Knee Surg. Sports Traumatol. Arthrosc. 2011, 19, 863–864, author reply 865–866. [Google Scholar] [CrossRef]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef]
- Wieland, H.A.; Michaelis, M.; Kirschbaum, B.J.; Rudolphi, K.A. Osteoarthritis—An untreatable disease? Nat. Rev. Drug Discov. 2005, 4, 331–344. [Google Scholar] [CrossRef]
- Lane, N.E.; Brandt, K.; Hawker, G.; Peeva, E.; Schreyer, E.; Tsuji, W.; Hochberg, M.C. Oarsi-fda initiative: Defining the disease state of osteoarthritis. Osteoarthr. Cartil. 2011, 19, 478–482. [Google Scholar] [CrossRef]
- Mifune, Y.; Matsumoto, T.; Takayama, K.; Ota, S.; Li, H.; Meszaros, L.B.; Usas, A.; Nagamune, K.; Gharaibeh, B.; Fu, F.H.; et al. The effect of platelet-rich plasma on the regenerative therapy of muscle derived stem cells for articular cartilage repair. Osteoarthr. Cartil. 2013, 21, 175–185. [Google Scholar] [CrossRef]
- Kon, E.; Buda, R.; Filardo, G.; di Martino, A.; Timoncini, A.; Cenacchi, A.; Fornasari, P.M.; Giannini, S.; Marcacci, M. Platelet-rich plasma: Intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surg. Sports Traumatol. Arthrosc. 2010, 18, 472–479. [Google Scholar] [CrossRef]
- Guilak, F.; Awad, H.A.; Fermor, B.; Leddy, H.A.; Gimble, J.M. Adipose-derived adult stem cells for cartilage tissue engineering. Biorheology 2004, 41, 389–399. [Google Scholar]
- Rada, T.; Reis, R.L.; Gomes, M.E. Distinct stem cells subpopulations isolated from human adipose tissue exhibit different chondrogenic and osteogenic differentiation potential. Stem Cell Rev. 2011, 7, 64–76. [Google Scholar] [CrossRef] [Green Version]
- Ringe, J.; Burmester, G.R.; Sittinger, M. Regenerative medicine in rheumatic disease-progress in tissue engineering. Nat. Rev. Rheumatol. 2012, 8, 493–498. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Gahring, D.; Adams, C.; Aron, D.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of adipose-derived mesenchymal stem and regenerative cells on lameness in dogs with chronic osteoarthritis of the coxofemoral joints: A randomized, double-blinded, multicenter, controlled trial. Vet. Ther. 2007, 8, 272–284. [Google Scholar]
- Emadedin, M.; Aghdami, N.; Taghiyar, L.; Fazeli, R.; Moghadasali, R.; Jahangir, S.; Farjad, R.; Baghaban Eslaminejad, M. Intra-articular injection of autologous mesenchymal stem cells in six patients with knee osteoarthritis. Arch. Iran. Med. 2012, 15, 422–428. [Google Scholar]
- Filardo, G.; Kon, E.; Buda, R.; Timoncini, A.; di Martino, A.; Cenacchi, A.; Fornasari, P.M.; Giannini, S.; Marcacci, M. Platelet-rich plasma intra-articular knee injections for the treatment of degenerative cartilage lesions and osteoarthritis. Knee Surg. Sports Traumatol. Arthrosc. 2011, 19, 528–535. [Google Scholar] [CrossRef]
- Liu, S.S.; Buvanendran, A.; Rathmell, J.P.; Sawhney, M.; Bae, J.J.; Moric, M.; Perros, S.; Pope, A.J.; Poultsides, L.; della Valle, C.J.; et al. A cross-sectional survey on prevalence and risk factors for persistent postsurgical pain 1 year after total hip and knee replacement. Reg. Anesth. Pain Med. 2012, 37, 415–422. [Google Scholar] [CrossRef]
- Hudson, J.T.; Slater, M.R.; Taylor, L.; Scott, H.M.; Kerwin, S.C. Assessing repeatability and validity of a visual analogue scale questionnaire for use in assessing pain and lameness in dogs. Am. J. Vet. Res. 2004, 65, 1634–1643. [Google Scholar] [CrossRef]
- Pak, J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: A case series. J. Med. Case Rep. 2011, 5, 296. [Google Scholar] [CrossRef]
- Van der Kraan, P.M. Osteoarthritis year 2012 in review: Biology. Osteoarthr. Cartil. 2012, 20, 1447–1450. [Google Scholar] [CrossRef]
- Braun, H.J.; Gold, G.E. Diagnosis of osteoarthritis: Imaging. Bone 2012, 51, 278–288. [Google Scholar] [CrossRef]
- Toghraie, F.; Razmkhah, M.; Gholipour, M.A.; Faghih, Z.; Chenari, N.; Torabi Nezhad, S.; Nazhvani Dehghani, S.; Ghaderi, A. Scaffold-free adipose-derived stem cells (ascs) improve experimentally induced osteoarthritis in rabbits. Arch. Iran. Med. 2012, 15, 495–499. [Google Scholar]
- Kon, E.; Filardo, G.; Matteo, B.D.; Marcacci, M. Prp for the treatment of cartilage pathology. Open Orthop. J. 2013, 7, 120–128. [Google Scholar] [CrossRef]
- Silva, R.F.; Carmona, J.U.; Rezende, C.M. Intra-articular injections of autologous platelet concentrates in dogs with surgical reparation of cranial cruciate ligament rupture. A pilot study. Vet. Comp. Orthop. Traumatol. 2013, 26, 285–290. [Google Scholar] [CrossRef]
- Roemer, F.W.; Crema, M.D.; Trattnig, S.; Guermazi, A. Advances in imaging of osteoarthritis and cartilage. Radiology 2011, 260, 332–354. [Google Scholar] [CrossRef]
- Xie, X.; Wang, Y.; Zhao, C.; Guo, S.; Liu, S.; Jia, W.; Tuan, R.S.; Zhang, C. Comparative evaluation of mscs from bone marrow and adipose tissue seeded in prp-derived scaffold for cartilage regeneration. Biomaterials 2012, 33, 7008–7018. [Google Scholar] [CrossRef]
- Molloy, T.; Wang, Y.; Murrell, G. The roles of growth factors in tendon and ligament healing. Sports Med. 2003, 33, 381–394. [Google Scholar] [CrossRef]
- Staudenmaier, R.; Froelich, K.; Birner, M.; Kindermann, J.; The Hoang, N.; Pueschel, R.C.; Mandlik, V. Optimization of platelet isolation and extraction of autogenous tgf-β in cartilage tissue engineering. Artif. Cells Blood Substit. Immobil. Biotechnol. 2009, 37, 265–272. [Google Scholar] [CrossRef]
- Akeda, K.; An, H.S.; Okuma, M.; Attawia, M.; Miyamoto, K.; Thonar, E.J.; Lenz, M.E.; Sah, R.L.; Masuda, K. Platelet-rich plasma stimulates porcine articular chondrocyte proliferation and matrix biosynthesis. Osteoarthr. Cartil. 2006, 14, 1272–1280. [Google Scholar] [CrossRef]
- Serra, C.I.; Soler, C.; Carillo, J.M.; Sopena, J.J.; Redondo, J.I.; Cugat, R. Effect of autologous platelet-rich plasma on the repair of full-thickness articular defects in rabbits. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 1730–1736. [Google Scholar]
- Sampson, S.; Reed, M.; Silvers, H.; Meng, M.; Mandelbaum, B. Injection of platelet-rich plasma in patients with primary and secondary knee osteoarthritis: A pilot study. Am. J. Phys. Med. Rehabil. 2010, 89, 961–969. [Google Scholar] [CrossRef]
- Hildner, F.; Albrecht, C.; Gabriel, C.; Redl, H.; van Griensven, M. State of the art and future perspectives of articular cartilage regeneration: A focus on adipose-derived stem cells and platelet-derived products. J. Tissue Eng. Regen. Med. 2011, 5, e36–e51. [Google Scholar] [CrossRef]
- Strem, B.M.; Hicok, K.C.; Zhu, M.; Wulur, I.; Alfonso, Z.; Schreiber, R.E.; Fraser, J.K.; Hedrick, M.H. Multipotential differentiation of adipose tissue-derived stem cells. Keio J. Med. 2005, 54, 132–141. [Google Scholar] [CrossRef]
- Pountos, I.; Jones, E.; Tzioupis, C.; McGonagle, D.; Giannoudis, P.V. Growing bone and cartilage. The role of mesenchymal stem cells. J. Bone Jt. Surg. 2006, 88, 421–426. [Google Scholar] [CrossRef]
- Roberts, S.; Genever, P.; McCaskie, A.; de Bari, C. Prospects of stem cell therapy in osteoarthritis. Regen. Med. 2011, 6, 351–366. [Google Scholar] [CrossRef]
- Singer, N.G.; Caplan, A.I. Mesenchymal stem cells: Mechanisms of inflammation. Annu. Rev. Pathol. 2011, 6, 457–478. [Google Scholar] [CrossRef]
- Khan, W.S.; Johnson, D.S.; Hardingham, T.E. The potential of stem cells in the treatment of knee cartilage defects. Knee 2010, 17, 369–374. [Google Scholar] [CrossRef]
- Fortier, L.A.; Barker, J.U.; Strauss, E.J.; McCarrel, T.M.; Cole, B.J. The role of growth factors in cartilage repair. Clin. Orthop. Relat. Res. 2011, 469, 2706–2715. [Google Scholar] [CrossRef]
- Andia, I.; Sanchez, M.; Maffulli, N. Joint pathology and platelet-rich plasma therapies. Expert Opin. Biol. Ther. 2012, 12, 7–22. [Google Scholar] [CrossRef]
- Cromer, M.S.; Bourne, R.M.; Fransen, M.; Fulton, R.; Wang, S.C. Responsiveness of quantitative cartilage measures over one year in knee osteoarthritis: Comparison of radiography and mri assessments. J. Magn. Reson. Imaging 2014, 39, 103–109. [Google Scholar] [CrossRef]
- Chen, L.; Dong, S.W.; Liu, J.P.; Tao, X.; Tang, K.L.; Xu, J.Z. Synergy of tendon stem cells and platelet-rich plasma in tendon healing. J. Orthop. Res. 2012, 30, 991–997. [Google Scholar] [CrossRef]
- Cho, H.H.; Jang, S.; Lee, S.C.; Jeong, H.S.; Park, J.S.; Han, J.Y.; Lee, K.H.; Cho, Y.B. Effect of neural-induced mesenchymal stem cells and platelet-rich plasma on facial nerve regeneration in an acute nerve injury model. Laryngoscope 2010, 120, 907–913. [Google Scholar]
- Patruno, M.; Martinello, T. Treatments of the injured tendon in veterinary medicine: From scaffolds to adult stem cells. Histol. Histopathol. 2014, 29, 417–422. [Google Scholar]
- Sanchez-Carmona, A.; Agut, A.; Chico, A.; Closa, J.M.; Rial, J.; Velasco, A. Desarrollo de una escala de valoracioón radioloógica del grado de osteoartrosis para las articulaciones de la rodilla y el codo en el perro—Escala “BIOARTH”. Revista oficial de la Asociación Veterinaria Española de Especialistas en Pequeños Animales, AVEPA 2006, 26, 269–275. (In Spanish) [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cuervo, B.; Rubio, M.; Sopena, J.; Dominguez, J.M.; Vilar, J.; Morales, M.; Cugat, R.; Carrillo, J.M. Hip Osteoarthritis in Dogs: A Randomized Study Using Mesenchymal Stem Cells from Adipose Tissue and Plasma Rich in Growth Factors. Int. J. Mol. Sci. 2014, 15, 13437-13460. https://doi.org/10.3390/ijms150813437
Cuervo B, Rubio M, Sopena J, Dominguez JM, Vilar J, Morales M, Cugat R, Carrillo JM. Hip Osteoarthritis in Dogs: A Randomized Study Using Mesenchymal Stem Cells from Adipose Tissue and Plasma Rich in Growth Factors. International Journal of Molecular Sciences. 2014; 15(8):13437-13460. https://doi.org/10.3390/ijms150813437
Chicago/Turabian StyleCuervo, Belen, Monica Rubio, Joaquin Sopena, Juan Manuel Dominguez, Jose Vilar, Manuel Morales, Ramón Cugat, and Jose Maria Carrillo. 2014. "Hip Osteoarthritis in Dogs: A Randomized Study Using Mesenchymal Stem Cells from Adipose Tissue and Plasma Rich in Growth Factors" International Journal of Molecular Sciences 15, no. 8: 13437-13460. https://doi.org/10.3390/ijms150813437




