The Constituents of Roots and Stems of Illigera luzonensis and Their Anti-Platelet Aggregation Effects
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification and Characterization
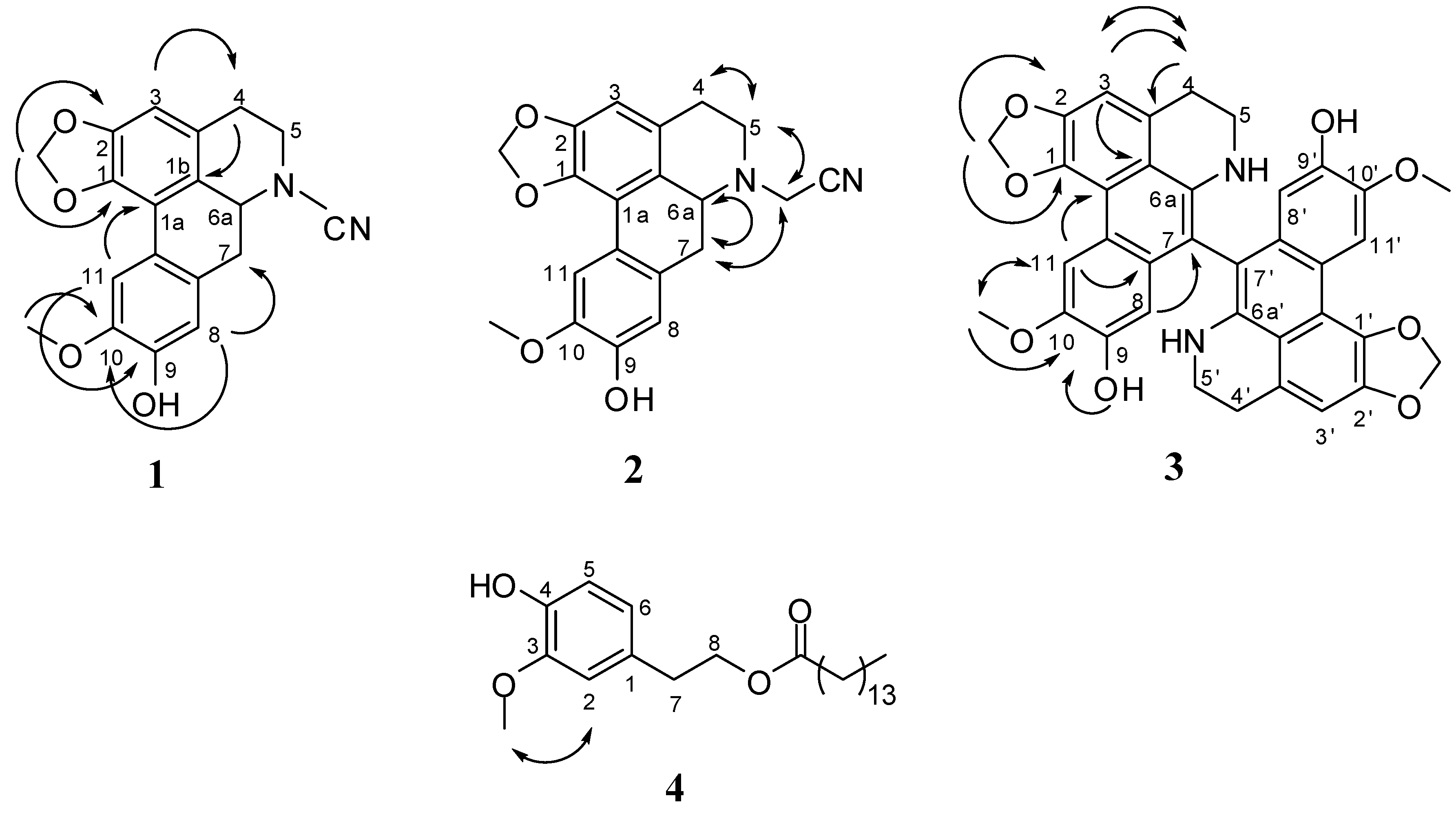
2.2. Structural Elucidation of Compounds 1–4
| Position | 1 | 2 | |
|---|---|---|---|
| dH (J, Hz) | dC | dH (J, Hz) | |
| 1 | 142.2 | ||
| 1a | 116.7 | ||
| 1b | 125.2 | ||
| 2 | 147.4 | ||
| 3 | 6.53, s | 107.0 | 6.52, s |
| 3a | 123.2 | ||
| 4 | 3.10, m | 28.1 | 2.91, m |
| 2.74, dt (16.2, 3.8) | 2.68, br d (15.6) | ||
| 5 | 3.43, td (12.6, 3.8) | 47.2 | 3.12, m |
| 3.67, m | 3.00, dd (11.2, 5.2) | ||
| 6a | 4.25, dd (14.0, 4.8) | 54.9 | 3.62, br d (13.8) |
| 7 | 2.9, t (14.0) | 33.7 | |
| 3.12, dd (14.0, 4.8) | 2.90, br d (13.8) | ||
| 7a | 127.0 | 2.59, t (13.8) | |
| 8 | 6.85, s | 114.5 | 6.82, s |
| 9 | 145.5 | ||
| 10 | 145.7 | ||
| 11 | 7.63, s | 110.0 | 7.63, s |
| 11a | 122.5 | ||
| OH-9 b | 5.71, br s | 5.71, br s | |
| OCH3-10 | 3.99, s | 56.2 | 4.04, s |
| –OCH2O– | 5.96, d (0.8) | 100.9 | 5.94, d (1.8) |
| 6.11, d (0.8) | 6.09, d (1.8) | ||
| –CH2CN | 4.09, d (17.6) | ||
| 3.73, d (17.6) | |||
| CN | 110.2 | ||
2.3. Anti-Platelet Aggregation Evaluation Bioassay
3. Experimental Section
3.1. General
3.2. Plant Materials
3.3. Extraction and Isolation
3.3.1. Illigeluzine A (1)
3.3.2. Illigeluzine B (2)
| Aggregation (%) | ||||||
|---|---|---|---|---|---|---|
| Inducer | Control | Conc. (μg/mL) | 1 | 5 | 6 | 7 |
| AA (100 μM) | 90.2 ± 0.8 | 100 | 0.0 ± 0.0 **** | 0.0 ± 0.0 **** | 0.0 ± 0.0 **** | 0.0 ± 0.0 **** |
| 50 | 4.7 ± 2.8 **** | – | 5.8 ± 5.2 **** | 0.0 ± 0.0 **** | ||
| 20 | 48.0 ± 13.7 *** | – | 24.9 ± 14.1 **** | 16.5 ± 11.1 **** | ||
| 10 | 78.7 ± 4.2 *** | – | 68.4 ± 8.6 *** | 49.9 ± 14.2 *** | ||
| 5 | 85.6 ± 3.2 * | – | 86.9 ± 1.2 *** | 83.8 ± 2.0 *** | ||
| 2 | – | 0.0 ± 0.0 **** | – | – | ||
| 1 | – | 17.8 ± 11.4 **** | – | – | ||
| 0.5 | – | 30.6 ± 13.5 **** | – | – | ||
| 0.2 | – | 83.6 ± 2.5 *** | – | – | ||
| Thr (0.1 U/mL) | 94.5 ± 1.0 | 100 | 90.9 ± 2.1 * | 89.5 ± 3.2 * | 92.4 ± 2.1 | 80.3 ± 2.6 *** |
| 50 | – | – | – | – | ||
| 20 | – | – | – | – | ||
| Col (10 μM) | 85.6 ± 0.8 | 100 | 6.8 ± 2.1 **** | 0.0 ± 0.0 **** | 9.5 ± 1.0 **** | 9.5 ± 1.0 **** |
| 50 | – | – | – | – | ||
| 20 | – | – | – | – | ||
| PAF (2 ng/mL) | 84.9 ± 0.6 | 100 | 57.5 ± 5.1 **** | 0.0 ± 0.0 **** | 64.3 ± 3.1 **** | 0.0 ± 0.0 **** |
| 50 | – | 51.5 ± 0.7 **** | – | 54.1 ± 2.9 **** | ||
| 20 | – | 71.2 ± 2.1 **** | – | 76.4 ± 3.3 ** | ||
| 10 | – | 78.1 ± 3.0 ** | – | 81.5 ± 1.9 * | ||
| 5 | – | 81.6 ± 2.4 * | – | 84.3 ± 0.4 | ||
| Aggregation (%) | ||||||
|---|---|---|---|---|---|---|
| Inducer | Control | Conc. (μg/mL) | 8 | 11 | 12 | 13 |
| AA (100 μM) | 90.2 ± 0.8 | 100 | 0.0 ± 0.0 **** | – | 0.0 ± 0.0 **** | 0.0 ± 0.0 **** |
| 50 | – | 25.7 ± 12.4 **** | 0.0 ± 0.0 **** | – | ||
| 20 | – | 43.4 ± 17.2 *** | 11.9 ± 10.6 **** | – | ||
| 10 | – | 62.6 ± 15.2 ** | 54.3 ± 14.4 *** | – | ||
| 5 | 0.0 ± 0.0 **** | 70.5 ± 14.1 * | 85.1 ± 3.9 * | 0.0 ± 0.0 **** | ||
| 2 | 4.8 ± 2.6 **** | 88.0 ± 2.4 | 90.2 ± 0.8 | 43.0 ± 12.3 **** | ||
| 1 | 30.2 ± 13.5 **** | – | – | 77.3 ± 8.6 * | ||
| 0.5 | 43.8 ± 16.4 *** | – | – | – | ||
| 0.2 | 84.1 ± 2.4 *** | – | – | – | ||
| Thr (0.1 U/mL) | 94.5 ± 1.0 | 100 | 86.6 ± 0.8 **** | – | 12.5 ± 7.7 **** | 87.8 ± 2.1 ** |
| 50 | – | 90.5 ± 0.8 ** | – | – | ||
| 20 | – | – | – | – | ||
| Col (10 μM) | 85.6 ± 0.8 | 100 | 0.0 ± 0.0 **** | – | 7.1 ± 5.8 **** | 5.6 ± 1.1 **** |
| 50 | – | 71.3 ± 4.6 ** | – | – | ||
| 20 | – | – | – | – | ||
| PAF (2 ng/mL) | 84.9 ± 0.6 | 100 | 56.3 ± 6.4 *** | – | 0.0 ± 0.0 **** | 71.3 ± 1.1 **** |
| 50 | – | 67.0 ± 1.8 **** | 0.0 ± 0.0 **** | – | ||
| 20 | – | – | 58.2 ± 2.2 **** | – | ||
| 10 | – | – | 76.7 ± 1.2 **** | – | ||
| 5 | – | – | 82.2 ± 1.9 * | – | ||
3.3.3. Bisdehydroactinodaphine (3)
3.3.4. Illigeraol A (4)
3.4. Antiplatelet Aggregatory Bioassay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, H.I.; Liu, T.S. Hernandiaceae. Flora of Taiwan; Editorial Committee of the Flora of Taiwan: Taipei, Taiwan, 1996; Volume 2, pp. 500–503. [Google Scholar]
- Guinaudeau, H.; Leboeuf, M.; Cavé, A. Aporphinoid alkaloids, V. J. Nat. Prod. 1994, 57, 1033–1135. [Google Scholar] [CrossRef]
- Guinaudeau, H.; Leboeuf, M.; Cavé, A. Aporphinoid alkaloids, IV. J. Nat. Prod. 1988, 51, 389–474. [Google Scholar] [CrossRef]
- Conserva, L.M.; Pereira, C.D.A.B.; Barbosa-Filho, J.M.; Cordell, G. Alkaloids of the Hernandiaceae: Occurrence and a Compilation of Their Biological Activities; Elsevier: Amsterdam, The Netherlands, 2005; Volume 54. [Google Scholar]
- Lakshmi, V.; Pandey, K.; Mishra, S.K.; Srivastava, S.; Mishra, M.; Agarwal, S.K. An overview of family Hernandiaceae. Rec. Nat. Prod. 2009, 3, 1–22. [Google Scholar]
- Wang, S.J.; Cheng, S.H. Alkaloids of Illigera luzonensis (Presl.) Merr. Yakugaku Zasshi 1968, 88, 1148–1150. [Google Scholar]
- Liu, S.L. Studies on the alkaloids of Illigera luzonensis (Presl.) Merr. J. Chin. Chem. Soc. 1977, 24, 209–211. [Google Scholar]
- Chen, C.Y.; Chang, F.R.; Wu, Y.C. The constituents from the stems of Annona cherimola. J. Chin. Chem. Soc. 1997, 44, 313–319. [Google Scholar]
- Chen, K.S.; Wu, Y.C.; Teng, C.M.; Ko, F.N. Bioactive alkaloids from Illigera luzonensis. J. Nat. Prod. 1997, 60, 645–647. [Google Scholar] [CrossRef]
- Voisin-Chiret, A.S.; Bazin, M.A.; Lancelot, J.C.; Rault, S. Synthesis of new l-ascorbic ferulic acid hybrids. Molecules 2007, 12, 2533–2545. [Google Scholar] [CrossRef]
- Yang, M.L.; Kuo, P.C.; Damu, A.G.; Chang, R.J.; Chiou, W.F.; Wu, T.S. A versatile route to the synthesis of 1-substituted β-carbolines by a single step Pictet–Spengler cyclization. Tetrahedron 2006, 62, 10900–10906. [Google Scholar] [CrossRef]
- Wu, Y.C.; Chao, Y.C.; Chang, F.R.; Chen, Y.Y. Alkaloids from Cassytha filiformis. Phytochemistry 1997, 46, 181–184. [Google Scholar] [CrossRef]
- El-Batta, A.; Jiang, C.; Zhao, W.; Anness, R.; Cooksy, A.L.; Bergdahl, M. Wittig reactions in water media employing stabilized ylides with aldehydes. Synthesis of α,β-unsaturated esters from mixing aldehydes, α-bromoesters, and Ph3P in aqueous NaHCO3. J. Org. Chem. 2007, 72, 5244–5259. [Google Scholar] [CrossRef]
- Acevedo, L.; Martinez, E.; Castañeda, P.; Franzblau, S.; Timmermann, B.N.; Linares, E.; Bye, R.; Mata, R. New phenylethanoids from Buddleja cordata subsp. cordata. Planta Med. 2000, 66, 257–261. [Google Scholar] [CrossRef]
- Talapatra, B.; Goswami, S.; Ghosh, A.; Talapatra, S.K. Alkaloids of Machilus glaucescens wight: Machigline, a new phenolic oxaporphine alkaloid. J. Indian Chem. Soc. 1982, 59, 1364–1368. [Google Scholar]
- Chen, K.S.; Chang, F.R.; Chia, Y.C.; Wu, T.S.; Wu, Y.C. Chemical constituents of Neolitsea parvigemma and Neolitsea konishii. J. Chin. Chem. Soc. 1998, 45, 103–110. [Google Scholar]
- Xiao, H.; Parkin, K. Isolation and identification of phase II enzyme-inducing agents from nonpolar extracts of green onion (Allium spp.). J. Agric. Food Chem. 2006, 54, 8417–8424. [Google Scholar] [CrossRef]
- Song, Y.; Cao, P.; Pan, D.S.; Han, S.; Yu, C.Y.; Zhao, Q.J.; Liang, Y. Alkaloids from Lycoris caldwellii and their particular cytotoxicities against the astrocytoma and glioma cell lines. Arch. Pharm. Res. 2013, 36, 927–932. [Google Scholar] [CrossRef]
- Rachmatiah, T.; Mukhtar, M.R.; Nafiah, M.A.; Hanafi, M.; Kosela, S.; Morita, H.; Litaudon, M.; Awang, K.; Omar, H.; Hadi, A.H. (+)-N-(2-Hydroxypropyl)lindcarpine: A new cytotoxic aporphine isolated from actinodaphne pruinosa nees. Molecules 2009, 14, 2850–2856. [Google Scholar] [CrossRef]
- Chang, Y.C.; Chen, C.Y.; Chang, F.R.; Wu, Y.C. Alkaloids from Lindera glauca. J. Chin. Chem. Soc. 2001, 48, 811–815. [Google Scholar]
- Khare, N.K.; Khare, M.P.; Khare, A. Two pregnane ester glycosides from Pergularia pallida. Phytochemistry 1984, 23, 2931–2936. [Google Scholar] [CrossRef]
- Chen, C.Y.; Chang, F.R.; Teng, C.M.; Wu, Y.C. Cheritamine, a new N-fatty acyl tryptamine and other constituents from the stems of Annona cherimola. J. Chin. Chem. Soc. 1999, 46, 77–86. [Google Scholar]
- Qi, H.L.; Chen, D.S.; Ye, J.S.; Huang, J.M. Electrochemical technique and copper-promoted transformations: Selective hydroxylation and amination of arylboronic acids. J. Org. Chem. 2013, 78, 7482–7487. [Google Scholar] [CrossRef]
- Jia, J.; Jiang, C.; Zhang, X.; Jiang, Y.; Ma, D. CuI-catalyzed hydroxylation of aryl bromides under the assistance of 5-bromo-2-(1H-imidazol-2-yl)pyridine and related ligands. Tetrahedron Lett. 2011, 52, 5593–5595. [Google Scholar] [CrossRef]
- Zhu, C.; Wang, R.; Falck, J.R. Mild and rapid hydroxylation of aryl/heteroaryl boronic acids and boronate esters with N-oxides. Org. Lett. 2012, 14, 3494–3497. [Google Scholar] [CrossRef]
- Bao, K.; Fan, A.; Dai, Y.; Zhang, L.; Zhang, W.; Cheng, M.; Yao, X. Selective demethylation and debenzylation of aryl ethers by magnesium iodide under solvent-free conditions and its application to the total synthesis of natural products. Org. Biomol. Chem. 2009, 7, 5084–5090. [Google Scholar] [CrossRef]
- Ahmad, I.; Chaudhary, B.A.; Janbaz, K.H.; Uzair, M.; Ashraf, M. Urease inhibitors and antioxidants from Vernonia cinerascens. J. Chem. Soc. Pak. 2011, 33, 114–117. [Google Scholar]
- Jiang, Z.H.; Wen, X.Y.; Tanaka, T.; Wu, S.Y.; Liu, Z.; Iwata, H.; Hirose, Y.; Wu, S.; Kouno, I. Cytotoxic hydrolyzable tannins from Balanophora japonica. J. Nat. Prod. 2008, 71, 719–723. [Google Scholar] [CrossRef]
- Spencer, P.A.; Tanaka, A.; Towers, G.H.N. An Agrobacterium signal compound from Grapevine cultivars. Phytochemistry 1990, 29, 3785–3788. [Google Scholar] [CrossRef]
- Taguchi, H.; Yosioka, I.; Yamasaki, K.; Kim, I. Studies on the constituents of Gastrodia elata Blume. Chem. Pharm. Bull. 1981, 29, 55–62. [Google Scholar] [CrossRef]
- Wu, T.S.; Chang, F.C.; Wu, P.L.; Kuoh, C.S.; Chen, I.S. Constituents of Leaves of Tetradium glabrifolium. J. Chin. Chem. Soc. 1995, 42, 929–934. [Google Scholar]
- Tao, R.; Wang, C.Z.; Kong, Z.W. Antibacterial/antifungal activity and synergistic interactions between polyprenols and other lipids isolated from Ginkgo Biloba L. leaves. Molecules 2013, 18, 2166–2182. [Google Scholar] [CrossRef]
- Wu, T.S.; Leu, Y.L.; Chan, Y.Y. Aristolochic acids as a defensive substance for the aristolochiaceous plant-feeding swallowtail butterfly, pachliopta aristolochiae interpositus. J. Chin. Chem. Soc. 2000, 47, 221–226. [Google Scholar]
- Shamma, M. The Isoquinoline Alkaloids Chemistry and Pharmacology; Academic Press, Inc.: London, UK, 1972; Volume 25. [Google Scholar]
- Borthakur, N.; Rastogi, R.C. Laetanine, a new noraporphine alkaloid from Litsea laeta. Phytochemistry 1979, 18, 910–911. [Google Scholar] [CrossRef]
- Koike, K.; Li, W.; Liu, L.; Hata, E.; Nikaido, T. New phenolic glycosides from the seeds of Cucurbita moschata. Chem. Pharm. Bull. 2005, 53, 225–228. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Topol, E.J. Scientific and therapeutic advances in antiplatelet therapy. Nat. Rev. Drug Discov. 2003, 2, 15–28. [Google Scholar] [CrossRef]
- Jackson, S.; Nesbitt, W.; Kulkarni, S. Signaling events underlying thrombus formation. J. Thromb. Haemost. 2003, 1, 1602–1612. [Google Scholar] [CrossRef]
- Wu, T.S.; Chern, H.J.; Damu, A.G.; Kuo, P.C.; Su, C.R.; Lee, E.J.; Teng, C.M. Flavonoids and ent-labdane diterpenoids from Andrographis paniculata and their antiplatelet aggregatory and vasorelaxing effects. J. Asian Nat. Prod. Res. 2008, 10, 17–24. [Google Scholar] [CrossRef]
- Teng, C.-M.; Chen, C.-C.; Ko, F.-N.; Lee, L.-G.; Huang, T.-F.; Chen, Y.-P.; Hsu, H.-Y. Two antiplatelet agents from Magnolia officinalis. Thromb. Res. 1988, 50, 757–765. [Google Scholar] [CrossRef]
- Born, G.; Cross, M. Effects of inorganic ions and of plasma proteins on the aggregation of blood platelets by adenosine diphosphate. J. Physiol. 1964, 170, 397–414. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, C.-H.; Chan, Y.-Y.; Kuo, P.-C.; Chen, Y.-F.; Chang, R.-J.; Chen, I.-S.; Wu, S.-J.; Wu, T.-S. The Constituents of Roots and Stems of Illigera luzonensis and Their Anti-Platelet Aggregation Effects. Int. J. Mol. Sci. 2014, 15, 13424-13436. https://doi.org/10.3390/ijms150813424
Huang C-H, Chan Y-Y, Kuo P-C, Chen Y-F, Chang R-J, Chen I-S, Wu S-J, Wu T-S. The Constituents of Roots and Stems of Illigera luzonensis and Their Anti-Platelet Aggregation Effects. International Journal of Molecular Sciences. 2014; 15(8):13424-13436. https://doi.org/10.3390/ijms150813424
Chicago/Turabian StyleHuang, Chieh-Hung, Yu-Yi Chan, Ping-Chung Kuo, Yu-Fon Chen, Ren-Jie Chang, Ih-Sheng Chen, Shwu-Jen Wu, and Tian-Shung Wu. 2014. "The Constituents of Roots and Stems of Illigera luzonensis and Their Anti-Platelet Aggregation Effects" International Journal of Molecular Sciences 15, no. 8: 13424-13436. https://doi.org/10.3390/ijms150813424




