Differential Proteomic Analysis of the Pancreas of Diabetic db/db Mice Reveals the Proteins Involved in the Development of Complications of Diabetes Mellitus
Abstract
:1. Introduction
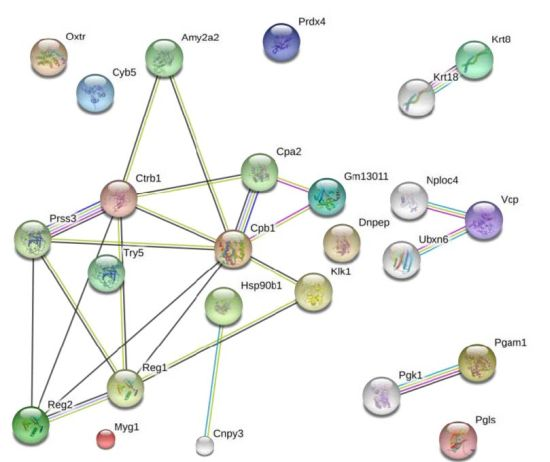
2. Results and Discussion

| Spot number | Protein | Gene name | UniProt accession number | Molecular mass (kD) | Peptide matched | Mascot score | Sequence coverage (%) | Fold change |
|---|---|---|---|---|---|---|---|---|
| 111 | Cytochrome b5 | Cyb5a | P56395 | 15.232 | 6 | 84 | 49 | +2.39 |
| 106 | Lithostathine 1 | Reg1 | P43137 | 18.906 | 9 | 132 | 55 | +2.38 |
| 110 | Lithostathine-2 | Reg2 | Q08731 | 19.793 | 7 | 109 | 52 | +2.51 |
| 610 | Prss3 protein | Prss3 | Q4G0C2 | 26.643 | 7 | 74 | 24 | +2.8 |
| 618 | Chymotrypsinogen B | Ctrb1 | Q9CR35 | 28.585 | 8 | 89 | 42 | a |
| 619 | Trypsinogen 11 | Trypsinogen | Q792Z0 | 268.88 | 7 | 80 | 31 | a |
| 158 | Chymotrypsinogen B precursor | Ctrb1 | Q9CR35 | 28.585 | 5 | 78 | 31 | +1.55 |
| 143 | Trypsin 5 precursor | Try5 | Q9QUK9 | 27.112 | 9 | 80 | 31 | +2.41 |
| 142 | Trypsin 5 precursor | Try5 | Q9QUK9 | 27.112 | 10 | 111 | 31 | +1.71 |
| 146 | 6-Phosphogluconolactonase | Pgls | Q9CQ60 | 27.522 | 8 | 112 | 43 | +2.07 |
| 141 | Peroxiredoxin-4 precursor | Prdx4 | O08807 | 31.317 | 9 | 131 | 43 | +1.6 |
| 145 | Phosphoglycerate mutase 1 | Pgam1 | Q9DBJ1 | 28.956 | 11 | 131 | 55 | +2.2 |
| 166 | Ela3 protein, partial | Ela3 | A2A9U8 | 28.321 | 7 | 83 | 29 | +2.25 |
| 616 | Ela3 protein, partial | Ela3 | A2A9U8 | 28.321 | 7 | 102 | 47 | a |
| 170 | Ela3 protein, partial | Ela3 | A2A9U8 | 28.321 | 10 | 129 | 68 | +1.63 |
| 3 | Kallikrein | Klk1 | P15947 | 29.495 | 8 | 97 | 37 | +1.68 |
| 258 | Protein MYG1, mitochondrial | Myg1 | Q9JK81 | 43.095 | 7 | 74 | 24 | +1.81 |
| 284 | Pancreatic Carboxypeptidase B1 precursor | Cpb1 | B2RS76 | 48.041 | 15 | 204 | 48 | +2.25 |
| 305 | Carboxypeptidase A2, pancreatic, isoform CRA_b | Cpa2 | Q504N0 | 37.185 | 10 | 99 | 26 | +1.98 |
| 620 | Aspartyl aminopeptidase | Dnpep | Q9Z2W0 | 52.744 | 9 | 69 | 17 | b |
| 621 | Endoplasmin | Hsp90b1 | P08113 | 92.703 | 13 | 112 | 13 | b |
| 555 | Endoplasmin | Hsp90b1 | P08113 | 92.998 | 20 | 174 | 25 | −2.04 |
| 532 | Transitional endoplasmic reticulum ATPase | Vcp | Q01853 | 89.950 | 15 | 113 | 16 | −1.5 |
| 368 | Keratin, type II cytoskeletal 8 | Krt8 | P11679 | 54.531 | 24 | 205 | 47 | −1.99 |
| 16 | Pancreatic alpha-amylase | Amy2a | P00688 | 57.966 | 8 | 89 | 21 | −1.83 |
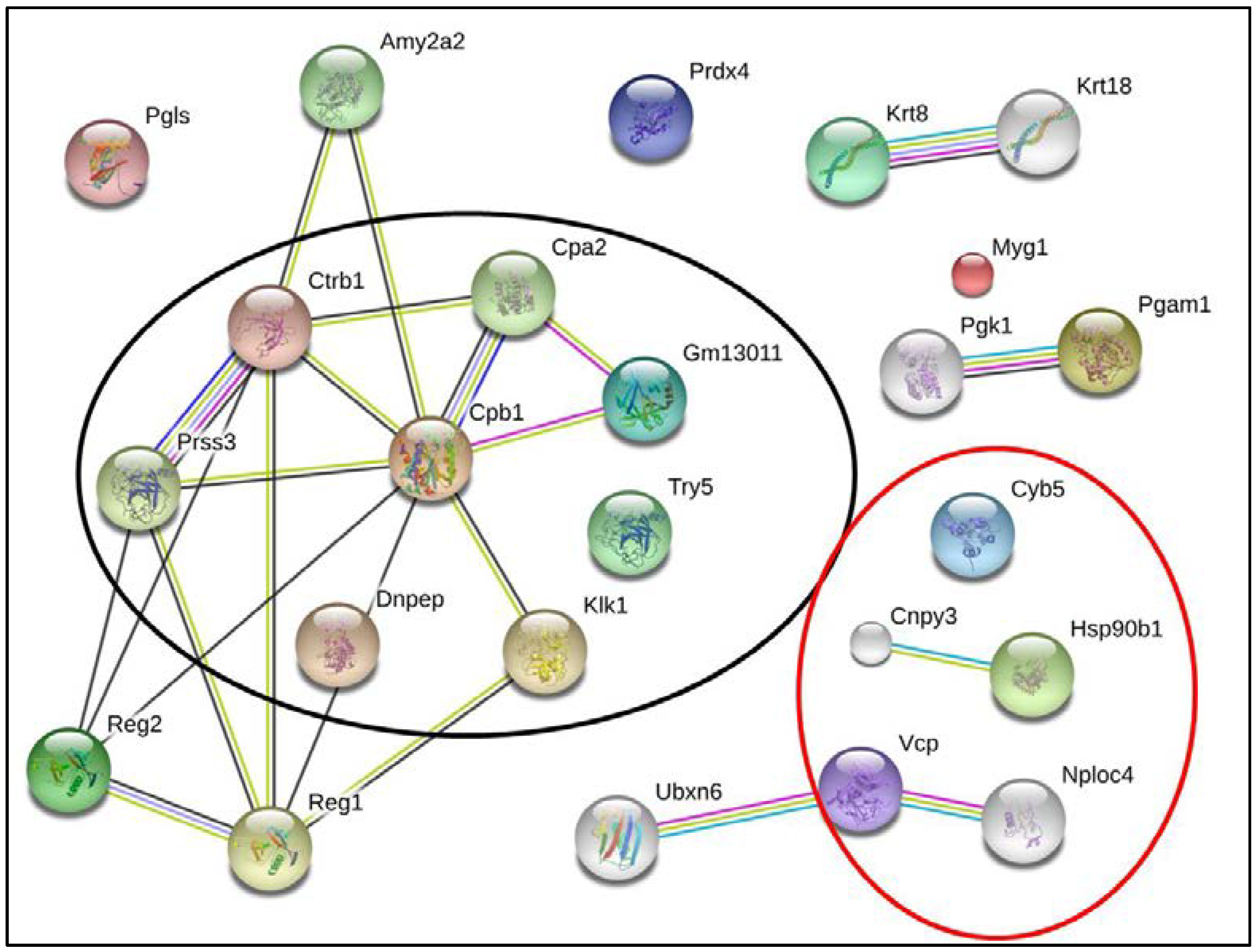
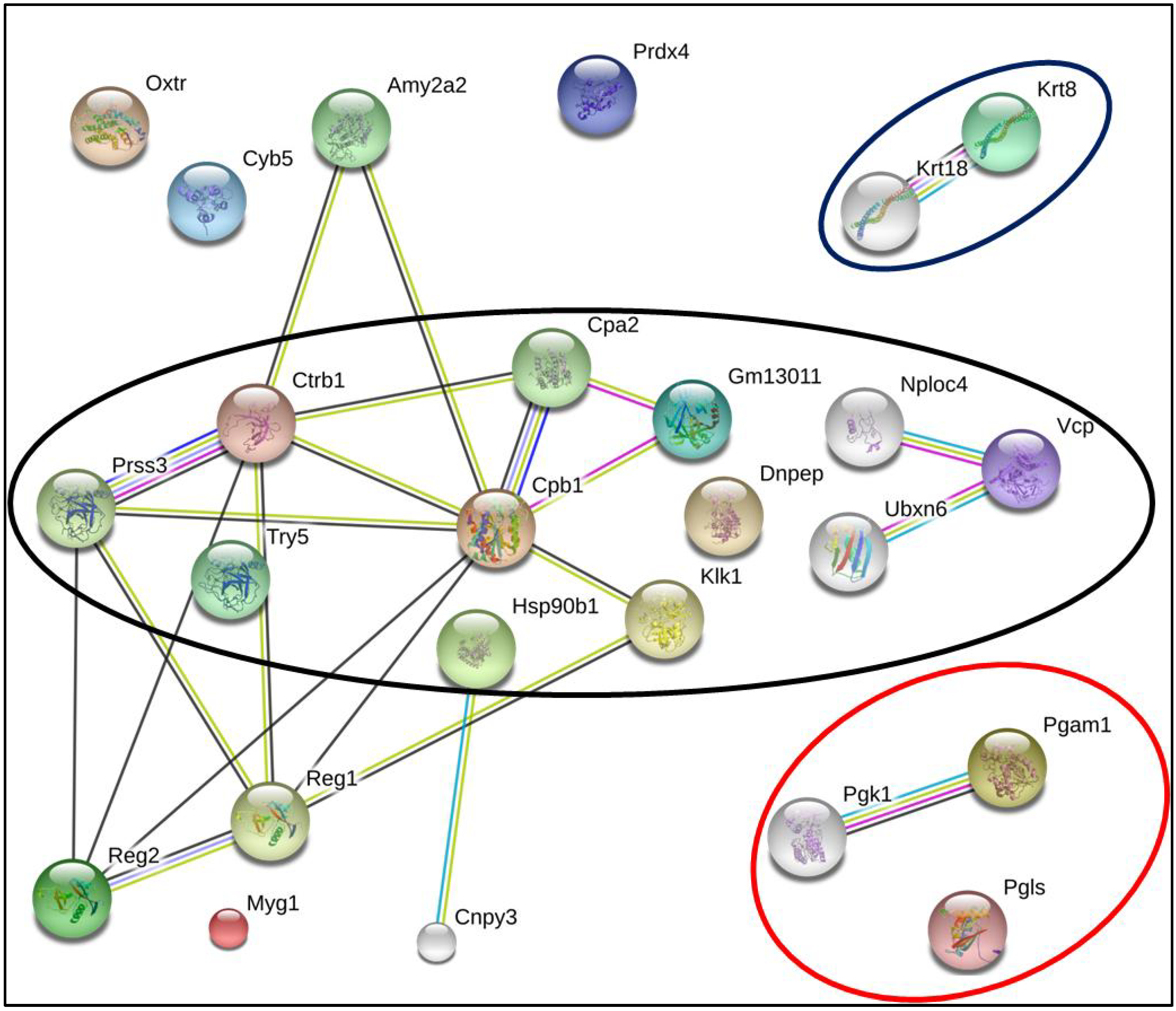
3. Experimental Section
3.1. Animals
3.2. Sample Preparation and 2D-Polyacrylamide Gel Electrophoresis
3.3. Mass Spectrometry
3.4. Bioinformatics Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alonso-Magdalena, P.; Quesada, I.; Nadal, A. Endocrine disruptors in the etiology of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2011, 7, 346–353. [Google Scholar] [CrossRef]
- Nolan, C.J.; Damm, P.; Prentki, M. Type 2 diabetes across generations: From pathophysiology to prevention and management. Lancet 2011, 378, 169–181. [Google Scholar]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef]
- Lee, A.W.; Cox, R.D. Use of mouse models in studying type 2 diabetes mellitus. Expert. Rev. Mol. Med. 2011, 13. [Google Scholar] [CrossRef]
- Lapolla, A.; Porcu, S.; Traldi, P. Some views on proteomics in diabetes. Clin. Chem. Lab. Med. 2011, 49, 943–957. [Google Scholar]
- Sanchez, J.C.; Chiappe, D.; Converset, V.; Hoogland, C.; Binz, P.A.; Paesano, S.; Appel, R.D.; Wang, S.; Sennitt, M.; Nolan, A.; et al. The mouse swiss-2d page database: A tool for proteomics study of diabetes and obesity. Proteomics 2001, 1, 136–163. [Google Scholar] [CrossRef]
- Qiu, L.; List, E.O.; Kopchick, J.J. Differentially expressed proteins in the pancreas of diet-induced diabetic mice. Mol. Cell Proteomics 2005, 4, 1311–1318. [Google Scholar] [CrossRef]
- Wang, M.Y.; Grayburn, P.; Chen, S.; Ravazzola, M.; Orci, L.; Unger, R.H. Adipogenic capacity and the susceptibility to type 2 diabetes and metabolic syndrome. Proc. Natl. Acad. Sci. USA 2008, 105, 6139–6144. [Google Scholar] [CrossRef]
- Ahmed, M.; Bergsten, P. Glucose-induced changes of multiple mouse islet proteins analyzed by two-dimensional gel electrophoresis and mass spectrometry. Diabetologia 2005, 48, 477–485. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, X.P.; Yu, Z.W.; Wang, L.S.; Zhu, Y.; Yu, X.F.; Wu, K.; Zeng, Y.; Xu, M.Y. Hyperlipidemic versus healthy pancreases: A proteomic analysis using an animal model. IUBMB Life 2010, 62, 781–789. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, Y.; Zeng, Y.; Yu, X.; Yao, J.; Zhao, S.; Bao, Z.; Chen, J.; Wang, X. Hyperlipidemic versus normal-lipid acute necrotic pancreatitis: Proteomic analysis using an animal model. Pancreas 2012, 41, 317–322. [Google Scholar] [CrossRef]
- Mathew, J.; Loranger, A.; Gilbert, S.; Faure, R.; Marceau, N. Keratin 8/18 regulation of glucose metabolism in normal versus cancerous hepatic cells through differential modulation of hexokinase status and insulin signaling. Exp. Cell Res. 2013, 319, 474–486. [Google Scholar] [CrossRef]
- Hinder, L.M.; Vivekanandan-Giri, A.; McLean, L.L.; Pennathur, S.; Feldman, E.L. Decreased glycolytic and tricarboxylic acid cycle intermediates coincide with peripheral nervous system oxidative stress in a murine model of type 2 diabetes. J. Endocrinol. 2013, 216, 1–11. [Google Scholar] [CrossRef]
- Giebelstein, J.; Poschmann, G.; Hojlund, K.; Schechinger, W.; Dietrich, J.W.; Levin, K.; Beck-Nielsen, H.; Podwojski, K.; Stuhler, K.; Meyer, H.E.; et al. The proteomic signature of insulin-resistant human skeletal muscle reveals increased glycolytic and decreased mitochondrial enzymes. Diabetologia 2012, 55, 1114–1127. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, C.; Fransson, U.; Hallgard, E.; Spegel, P.; Holm, C.; Krogh, M.; Warell, K.; James, P.; Mulder, H. Metabolomic and proteomic analysis of a clonal insulin-producing β-cell line (INS-1 832/13). J. Proteome Res. 2008, 7, 400–411. [Google Scholar] [CrossRef]
- Folli, F.; Guzzi, V.; Perego, L.; Coletta, D.K.; Finzi, G.; Placidi, C.; La Rosa, S.; Capella, C.; Socci, C.; Lauro, D.; et al. Proteomics reveals novel oxidative and glycolytic mechanisms in type 1 diabetic patients’ skin which are normalized by kidney-pancreas transplantation. PLoS One 2010, 5. [Google Scholar] [CrossRef]
- Tessari, P.; Cecchet, D.; Cosma, A.; Puricelli, L.; Millioni, R.; Vedovato, M.; Tiengo, A. Insulin resistance of amino acid and protein metabolism in type 2 diabetes. Clin. Nutr. 2011, 30, 267–272. [Google Scholar] [CrossRef]
- Gougeon, R. Insulin resistance of protein metabolism in type 2 diabetes and impact on dietary needs: A review. Can. J. Diabetes 2013, 37, 115–120. [Google Scholar] [CrossRef]
- T Hart, L.M.; Fritsche, A.; Nijpels, G.; van Leeuwen, N.; Donnelly, L.A.; Dekker, J.M.; Alssema, M.; Fadista, J.; Carlotti, F.; Gjesing, A.P.; et al. The CTRB1/2 locus affects diabetes susceptibility and treatment via the incretin pathway. Diabetes 2013, 62, 3275–3281. [Google Scholar] [CrossRef]
- Habara, Y.; Uehara, A.; Takasugi, Y.; Namiki, M.; Kanno, T. Characterization of secretory responses in exocrine pancreas of genetically obese zucker rats. Int. J. Pancreatol. 1991, 10, 237–245. [Google Scholar]
- Knecht, W.; Cottrell, G.S.; Amadesi, S.; Mohlin, J.; Skaregarde, A.; Gedda, K.; Peterson, A.; Chapman, K.; Hollenberg, M.D.; Vergnolle, N.; et al. Trypsin IV or mesotrypsin and p23 cleave protease-activated receptors 1 and 2 to induce inflammation and hyperalgesia. J. Biol. Chem. 2007, 282, 26089–26100. [Google Scholar] [CrossRef]
- Hu, X.; Friedman, D.; Hill, S.; Caprioli, R.; Nicholson, W.; Powers, A.C.; Hunter, L.; Limbird, L.E. Proteomic exploration of pancreatic islets in mice null for the α2a adrenergic receptor. J. Mol. Endocrinol. 2005, 35, 73–88. [Google Scholar] [CrossRef]
- Shimada, S.; Yamaguchi, K.; Takahashi, M.; Ogawa, M. Pancreatic elastase IIIa and its variants are expressed in pancreatic carcinoma cells. Int. J. Mol. Med. 2002, 10, 599–603. [Google Scholar]
- Rathmann, W.; Haastert, B.; Icks, A.; Giani, G.; Hennings, S.; Mitchell, J.; Curran, S.; Wareham, N.J. Low faecal elastase 1 concentrations in type 2 diabetes mellitus. Scand. J. Gastroenterol. 2001, 36, 1056–1061. [Google Scholar] [CrossRef]
- Yuan, G.; Deng, J.; Wang, T.; Zhao, C.; Xu, X.; Wang, P.; Voltz, J.W.; Edin, M.L.; Xiao, X.; Chao, L.; et al. Tissue kallikrein reverses insulin resistance and attenuates nephropathy in diabetic rats by activation of phosphatidylinositol 3-kinase/protein kinase B and adenosine 5'-monophosphate-activated protein kinase signaling pathways. Endocrinology 2007, 148, 2016–2026. [Google Scholar] [CrossRef]
- Campbell, D.J.; Kladis, A.; Zhang, Y.; Jenkins, A.J.; Prior, D.L.; Yii, M.; Kenny, J.F.; Black, M.J.; Kelly, D.J. Increased tissue kallikrein levels in type 2 diabetes. Diabetologia 2010, 53, 779–785. [Google Scholar] [CrossRef]
- Nozaki, J.; Kubota, H.; Yoshida, H.; Naitoh, M.; Goji, J.; Yoshinaga, T.; Mori, K.; Koizumi, A.; Nagata, K. The endoplasmic reticulum stress response is stimulated through the continuous activation of transcription factors ATF6 and XBP1 in Ins2+/Akita pancreatic β cells. Genes Cells 2004, 9, 261–270. [Google Scholar] [CrossRef]
- Lee, J.W.; Kim, W.H.; Yeo, J.; Jung, M.H. ER stress is implicated in mitochondrial dysfunction-induced apoptosis of pancreatic β cells. Mol. Cells 2010, 30, 545–549. [Google Scholar] [CrossRef]
- Sims-Robinson, C.; Zhao, S.; Hur, J.; Feldman, E.L. Central nervous system endoplasmic reticulum stress in a murine model of type 2 diabetes. Diabetologia 2012, 55, 2276–2284. [Google Scholar] [CrossRef]
- Wojcik, C.; Rowicka, M.; Kudlicki, A.; Nowis, D.; McConnell, E.; Kujawa, M.; DeMartino, G.N. Valosin-containing protein (p97) is a regulator of endoplasmic reticulum stress and of the degradation of N-end rule and ubiquitin-fusion degradation pathway substrates in mammalian cells. Mol. Biol. Cell 2006, 17, 4606–4618. [Google Scholar] [CrossRef]
- Dreja, T.; Jovanovic, Z.; Rasche, A.; Kluge, R.; Herwig, R.; Tung, Y.C.; Joost, H.G.; Yeo, G.S.; Al-Hasani, H. Diet-induced gene expression of isolated pancreatic islets from a polygenic mouse model of the metabolic syndrome. Diabetologia 2010, 53, 309–320. [Google Scholar] [CrossRef]
- Jiang, Y.L.; Ning, Y.; Ma, X.L.; Liu, Y.Y.; Wang, Y.; Zhang, Z.; Shan, C.X.; Xu, Y.D.; Yin, L.M.; Yang, Y.Q. Alteration of the proteome profile of the pancreas in diabetic rats induced by streptozotocin. Int. J. Mol. Med. 2011, 28, 153–160. [Google Scholar]
- Ding, Y.; Yamada, S.; Wang, K.Y.; Shimajiri, S.; Guo, X.; Tanimoto, A.; Murata, Y.; Kitajima, S.; Watanabe, T.; Izumi, H.; et al. Overexpression of peroxiredoxin 4 protects against high-dose streptozotocin-induced diabetes by suppressing oxidative stress and cytokines in transgenic mice. Antioxid. Redox Signal 2010, 13, 1477–1490. [Google Scholar] [CrossRef]
- Tavender, T.J.; Bulleid, N.J. Peroxiredoxin IV protects cells from oxidative stress by removing H2O2 produced during disulphide formation. J. Cell Sci. 2010, 123, 2672–2679. [Google Scholar] [CrossRef]
- Liu, Y.; Turdi, S.; Park, T.; Morris, N.J.; Deshaies, Y.; Xu, A.; Sweeney, G. Adiponectin corrects high-fat diet-induced disturbances in muscle metabolomic profile and whole-body glucose homeostasis. Diabetes 2013, 62, 743–752. [Google Scholar] [CrossRef]
- Goehring, I.; Sauter, N.S.; Catchpole, G.; Assmann, A.; Shu, L.; Zien, K.S.; Moehlig, M.; Pfeiffer, A.F.; Oberholzer, J.; Willmitzer, L.; et al. Identification of an intracellular metabolic signature impairing β cell function in the rat β cell line INS-1e and human islets. Diabetologia 2011, 54, 2584–2594. [Google Scholar] [CrossRef]
- Gupte, R.S.; Floyd, B.C.; Kozicky, M.; George, S.; Ungvari, Z.I.; Neito, V.; Wolin, M.S.; Gupte, S.A. Synergistic activation of glucose-6-phosphate dehydrogenase and NAD(P)H oxidase by Src kinase elevates superoxide in type 2 diabetic, Zucker fa/fa, rat liver. Free Radic. Biol. Med. 2009, 47, 219–228. [Google Scholar] [CrossRef]
- Schneider, A.M.; Rawat, D.; Weinstein, L.S.; Gupte, S.A.; Richards, W.O. Effects of laparoscopic Roux-en-Y gastric bypass on glucose-6 phosphate dehydrogenase activity in obese type 2 diabetics. Surg. Endosc. 2012, 26, 823–830. [Google Scholar] [CrossRef]
- Tripathi, Y.B.; Pandey, V. Obesity and endoplasmic reticulum (ER) stresses. Front. Immunol. 2012, 3, 240. [Google Scholar]
- Naidoo, N. The endoplasmic reticulum stress response and aging. Rev. Neurosci. 2009, 20, 23–37. [Google Scholar] [CrossRef]
- Gunasekaran, U.; Gannon, M. Type 2 diabetes and the aging pancreatic β cell. Aging 2011, 3, 565–575. [Google Scholar]
- Takahashi, H.; Oh-Ishi, M.; Shimizu, H.; Mori, M. Detection and identification of subcutaneous adipose tissue protein related to obesity in New Zealand obese mouse. Endocr. J. 2001, 48, 205–211. [Google Scholar] [CrossRef]
- Nikolajczyk, B.S.; Jagannathan-Bogdan, M.; Shin, H.; Gyurko, R. State of the union between metabolism and the immune system in type 2 diabetes. Genes Immun. 2011, 12, 239–250. [Google Scholar] [CrossRef]
- Guzman-Flores, J.M.; Lopez-Briones, S. Cells of innate and adaptive immunity in type 2 diabetes and obesity. Gac. Med. Mex. 2012, 148, 381–389. [Google Scholar]
- Keel, M.; Harter, L.; Reding, T.; Sun, L.K.; Hersberger, M.; Seifert, B.; Bimmler, D.; Graf, R. Pancreatic stone protein is highly increased during posttraumatic sepsis and activates neutrophil granulocytes. Crit. Care Med. 2009, 37, 1642–1648. [Google Scholar]
- Philips, M.A.; Vikesa, J.; Luuk, H.; Jonson, L.; Lillevali, K.; Rehfeld, J.F.; Vasar, E.; Koks, S.; Nielsen, F.C. Characterization of MYG1 gene and protein: Subcellular distribution and function. Biol. Cell. 2009, 101, 361–373. [Google Scholar] [CrossRef]
- Franceschini, A.; Szklarczyk, D.; Frankild, S.; Kuhn, M.; Simonovic, M.; Roth, A.; Lin, J.; Minguez, P.; Bork, P.; von Mering, C.; et al. String v. 9.1: Protein-protein interaction networks, with increased coverage and integration. Nucleic Acids Res. 2013, 41, D808–D815. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar]
- Lee, J.; Ozcan, U. Unfolded protein response signaling and metabolic diseases. J. Biol. Chem. 2014, 289, 1203–1211. [Google Scholar] [CrossRef]
- Back, S.H.; Kaufman, R.J. Endoplasmic reticulum stress and type 2 diabetes. Annu. Rev. Biochem. 2012, 81, 767–793. [Google Scholar] [CrossRef]
- Wieser, V.; Moschen, A.R.; Tilg, H. Inflammation, cytokines and insulin resistance: A clinical perspective. Arch. Immunol. Ther. Exp. 2013, 61, 119–125. [Google Scholar] [CrossRef]
- Eguchi, K.; Manabe, I. Macrophages and islet inflammation in type 2 diabetes. Diabetes Obes. Metab. 2013, 15, 152–158. [Google Scholar] [CrossRef]
- Horvat, S.; Bunger, L. Polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) assay for the mouse leptin receptor (Lepr(db)) mutation. Lab. Anim. 1999, 33, 380–384. [Google Scholar] [CrossRef]
- Salazar, E.; Diaz-Mejia, J.J.; Moreno-Hagelsieb, G.; Martinez-Batallar, G.; Mora, Y.; Mora, J.; Encarnacion, S. Characterization of the NifA-RpoN regulon in Rhizobium etli in free life and in symbiosis with Phaseolus vulgaris. Appl. Environ. Microbiol. 2010, 76, 4510–4520. [Google Scholar] [CrossRef]
- Higareda-Almaraz, J.C.; Enriquez-Gasca Mdel, R.; Hernandez-Ortiz, M.; Resendis-Antonio, O.; Encarnacion-Guevara, S. Proteomic patterns of cervical cancer cell lines, a network perspective. BMC Syst. Biol. 2011, 5, 96. [Google Scholar] [CrossRef]
- Perkins, D.N.; Pappin, D.J.; Creasy, D.M.; Cottrell, J.S. Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 1999, 20, 3551–3567. [Google Scholar] [CrossRef]
- UniProt Consortium. The universal protein resource (uniprot) 2009. Nucleic Acids Res. 2009, 37, D169–D174. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pérez-Vázquez, V.; Guzmán-Flores, J.M.; Mares-Álvarez, D.; Hernández-Ortiz, M.; Macías-Cervantes, M.H.; Ramírez-Emiliano, J.; Encarnación-Guevara, S. Differential Proteomic Analysis of the Pancreas of Diabetic db/db Mice Reveals the Proteins Involved in the Development of Complications of Diabetes Mellitus. Int. J. Mol. Sci. 2014, 15, 9579-9593. https://doi.org/10.3390/ijms15069579
Pérez-Vázquez V, Guzmán-Flores JM, Mares-Álvarez D, Hernández-Ortiz M, Macías-Cervantes MH, Ramírez-Emiliano J, Encarnación-Guevara S. Differential Proteomic Analysis of the Pancreas of Diabetic db/db Mice Reveals the Proteins Involved in the Development of Complications of Diabetes Mellitus. International Journal of Molecular Sciences. 2014; 15(6):9579-9593. https://doi.org/10.3390/ijms15069579
Chicago/Turabian StylePérez-Vázquez, Victoriano, Juan M. Guzmán-Flores, Daniela Mares-Álvarez, Magdalena Hernández-Ortiz, Maciste H. Macías-Cervantes, Joel Ramírez-Emiliano, and Sergio Encarnación-Guevara. 2014. "Differential Proteomic Analysis of the Pancreas of Diabetic db/db Mice Reveals the Proteins Involved in the Development of Complications of Diabetes Mellitus" International Journal of Molecular Sciences 15, no. 6: 9579-9593. https://doi.org/10.3390/ijms15069579