New Coumarins and Anti-Inflammatory Constituents from the Fruits of Cnidium monnieri
Abstract
:1. Introduction

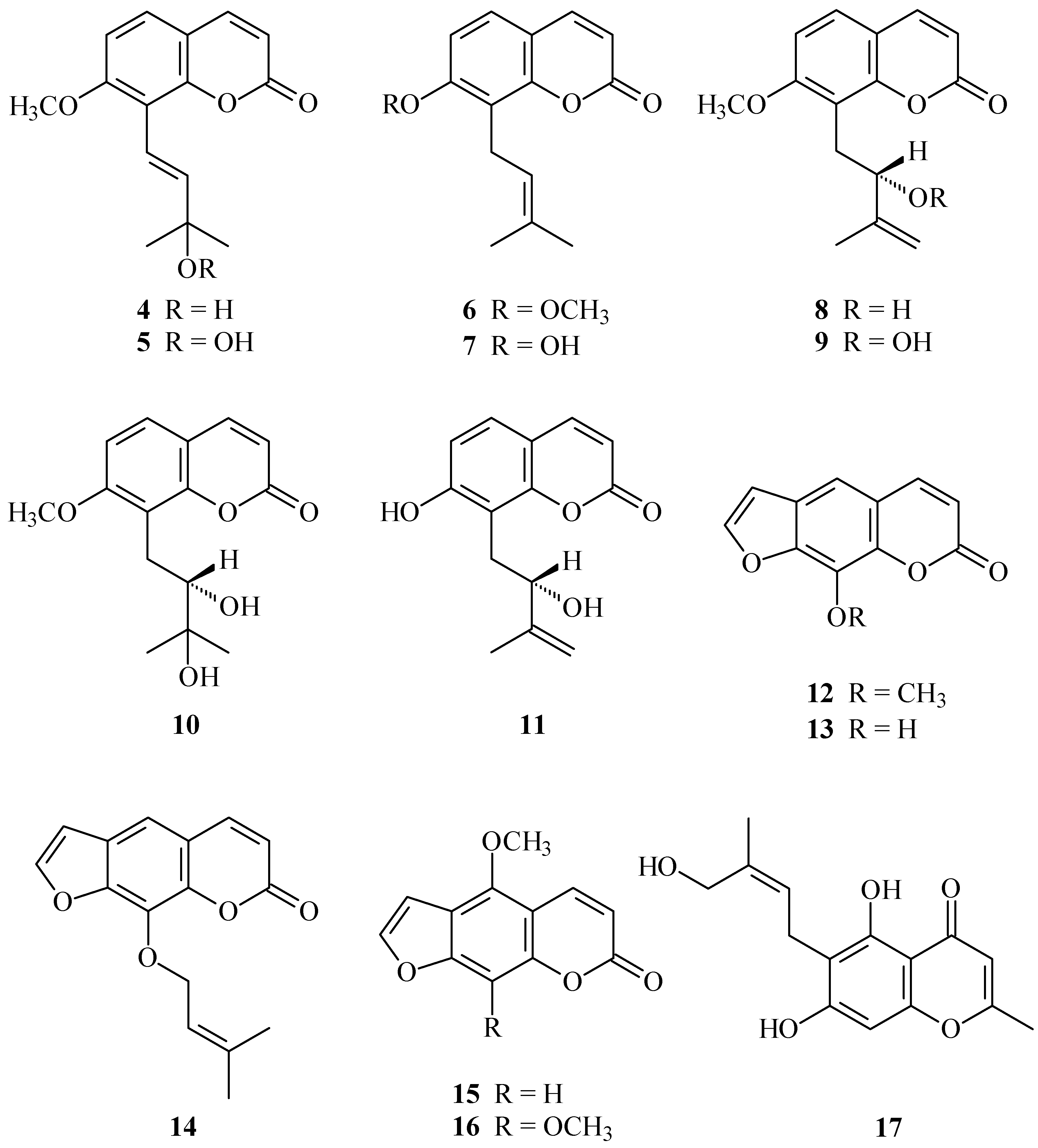
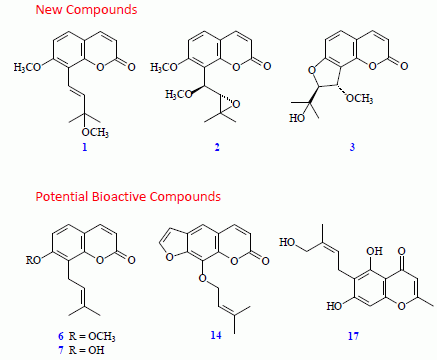
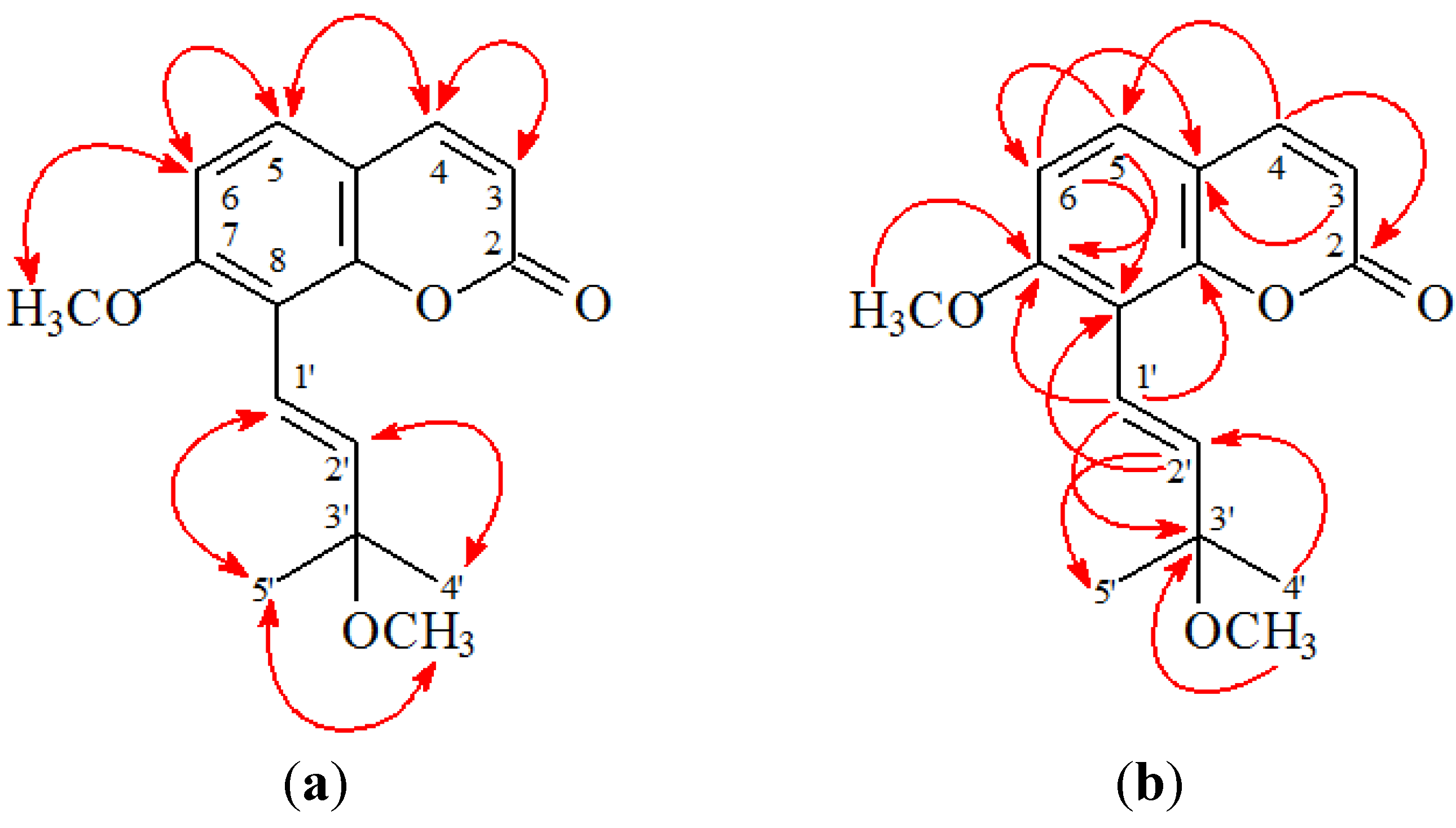
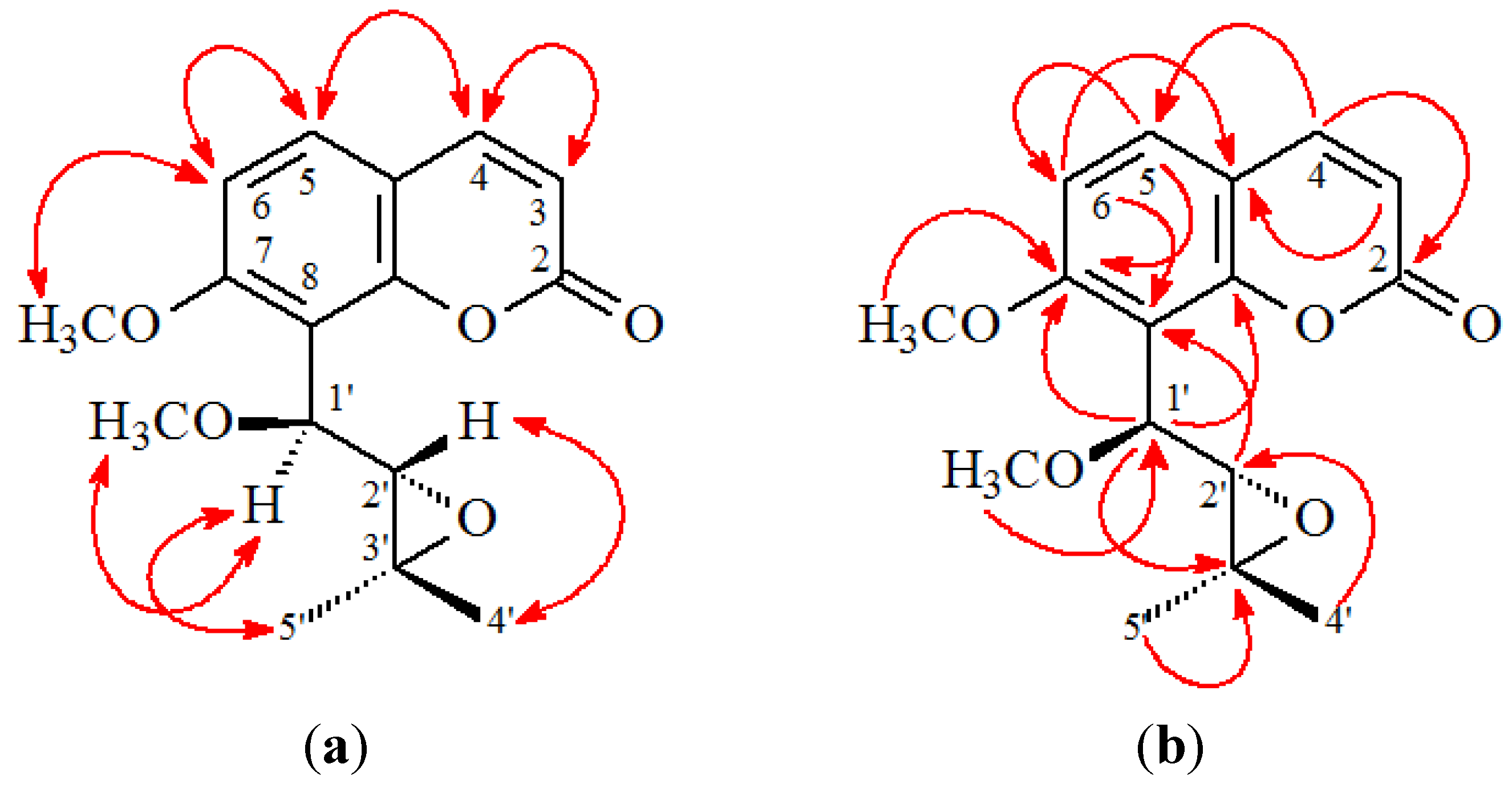
2. Results

| Compounds | Superoxide Anion | Elastase |
|---|---|---|
| IC50 [µg/mL] b or (Inh %) c | ||
| 3'-O-Methylmurraol (1) | 6.24 ± 0.50 g | (35.10 ± 5.71) f |
| 1'-O-Methylphlojodicarpin (2) | (15.04 ± 1.26) g | (6.02 ± 1.56) e |
| 1'-O-Methylvaginol (3) | (11.99 ± 2.14) f | (6.42 ± 2.07) e |
| Murraol (4) | 2.83 ± 0.46 f | (18.16 ± 5.62) e |
| Peroxymurraol (5) | 2.87 ± 0.11 g | (5.78 ± 2.98) |
| Osthol (6) | 0.005 ± 0.0002 f | (31.96 ± 5.72) f |
| Osthenol (7) | 0.09 ± 0.01 g | 3.28 ± 0.90 g |
| Auraptenol (8) | 0.77 ± 0.11 e | (22.46 ± 2.65) f |
| Peroxyauraptenol (9) | 0.41 ± 0.06 e | 7.83 ± 1.17 e |
| Meranzin hydrate (10) | 7.31 ±1.62 e | 4.21 ± 1.41 e |
| Demethylauraptenol (11) | 0.54 ± 0.05 g | 4.36 ± 1.67 f |
| Xanthotoxin (12) | 0.32 ± 0.13 e | (14.81 ± 6.03) |
| Xanthotoxol (13) | (24.54 ± 2.47) g | (42.94 ± 4.29) g |
| Imperatorin (14) | 0.07 ± 0.02 g | (22.08 ± 2.93) f |
| Bergapten (15) | 0.36 ± 0.09 e | 4.62 ± 1.36 e |
| Isopimpinellin (16) | 2.75 ± 0.26 | (8.97 ± 2.28) e |
| Cnidimol A (17) | 3.65 ± 0.41 g | 3.20 ± 0.16 g |
| Diphenyleneiodonium d | 0.55 ± 0.20 g | - |
| Phenylmethylsulfonyl fluoride d | - | 34.4 ± 5.5 g |
3. Discussion
4. Experimental Section
4.1. Ethics Statement
4.2. General Experimental Procedures
4.3. Plant Material
4.4. Extraction and Isolation
4.4.1. 3'-O-Methylmurraol (1)
4.4.2. rel-(1'S,2'S)-1'-O-Methylphlojodicarpin (2)
4.4.3. (1'S,2'S)-1'-O-Methylvaginol (3)
4.4.4. Supplementary Files
4.5. Biological Assay
4.5.1. Preparation of Human Neutrophils
4.5.2. Measurement of Superoxide Anion Generation
4.5.3. Measurement of Elastase Release
4.5.4. Statistical Analysis
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2010; volume 1, pp. 295–296.
- Zhu, Y.P. Chinese Material Medica. Chemistry, Pharmacology and Applications; Harwood Academic Publishers: Amsterdam, The Netherlands, 1998; pp. 624–625. [Google Scholar]
- Baba, K.; Kawanishi, H.; Taniguchi, M.; Kozawa, M. Chromones from Cnidium monnieri. Phytochemistry 1992, 31, 1367–1370. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, G.; Zhang, Q.; Wu, H.; Wu, Y. Fingerprint analysis of the fruits of Cnidium monnieri extract by high-performance liquid chromatography–diode array detection–electrospray ionization tandem mass spectrometry. J. Pharm. Biomed. Anal. 2007, 43, 926–936. [Google Scholar] [CrossRef]
- Basnet, P.; Yasuda, I.; Kumagai, N.; Tohda, C.; Nojima, H.; Kuraishi, Y.; Komatsu, K. Inhibition of itch-scratch response by fruits of Cnidium monnieri in mice. Biol. Pharm. Bull. 2001, 24, 1012–1015. [Google Scholar] [CrossRef]
- Cai, J.N.; Basnet, P.; Wang, Z.T.; Komatsu, K.; Xu, L.S.; Tani, T. Coumarins from the fruits of Cnidium monnieri. J. Nat. Prod. 2000, 63, 485–488. [Google Scholar] [CrossRef]
- Yang, L.L.; Wang, M.C.; Chen, L.G.; Wang, C.C. Cytotoxic activity of coumarins from the fruits of Cnidium monnieri on leukemia cell lines. Planta Med. 2003, 69, 1091–1095. [Google Scholar] [CrossRef]
- Dien, P.H.; Nhan, N.T.; Le Thuy, H.T.; Quang, D.N. Main constituents from the seeds of Vietnamese Cnidium monnieri and cytotoxic activity. Nat. Prod. Res. 2012, 26, 2107–2111. [Google Scholar]
- Honda, G.; Tabata, M.; Baba, K.; Kozawa, M. On the antidermatophytic constituents and the original plants of the traditional Chinese drug “She chuang zi”. Shoyakugaku Zasshi 1984, 38, 221–226. [Google Scholar]
- Yahara, S.; Sugimura, C.; Nohara, T.; Niiho, Y.; Nakajima, Y.; Ito, H. Studies on the constituents of Cnidii monnieri Fructus. Shoyakugaku Zasshi 1993, 47, 74–78. [Google Scholar]
- Kitajima, J.; Aoki, Y.; Ishikawa, T.; Tanaka, Y. Monoterpenoid glucosides of Cnidium monnieri fruit. Chem. Pharm. Bull. 1999, 47, 639–642. [Google Scholar] [CrossRef]
- Ito, C.; Furukawa, H. Constituents of Murraya exotica L. Structure elucidation of new coumarins. Chem. Pharm. Bull. 1987, 35, 4277–4285. [Google Scholar] [CrossRef]
- Ito, C.; Furukawa, H. Two new coumarins from Murraya plants. Chem. Pharm. Bull. 1989, 37, 819–820. [Google Scholar] [CrossRef]
- Gantimur, D.; Semenov, A.A. Coumarins from Phlojodicarpus sibiricus. Chem. Nat. Comp. 1981, 17, 41–43. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, M.; Liu, Y.; Zhang, G.; Luo, Y. Chromones and coumarins from the dried fructus of Cnidium monnieri. Fitoterapia 2011, 82, 767–771. [Google Scholar] [CrossRef]
- Zou, Y.; Lobera, M.; Snider, B.B. Synthesis of 2,3-dihydro-3-hydroxy-2-hydroxylalkylbenzofurans from epoxy aldehydes. One-step syntheses of brosimacutin G, vaginidiol, vaginol, smyrindiol, xanthoarnol, and avicenol A. Biomimetic syntheses of angelicin and psoralen. J. Org. Chem. 2005, 70, 1761–1770. [Google Scholar] [CrossRef]
- Razdan, T.K.; Qadri, B.; Harkar, S.; Waight, E.S. Chromones and coumarins from Skimmia laureola. Phytochemistry 1987, 26, 2063–2069. [Google Scholar] [CrossRef]
- Sasaki, H.; Taguchi, H.; Endo, T.; Yosioka, I. The constituents of Glehnia littoralis Fr. Schmidt et Miq. Structure of a new coumarin glycoside, osthenol 7-O-β-gentiobioside. Chem. Pharm. Bull. 1980, 28, 1847–1852. [Google Scholar] [CrossRef]
- lrnai, F.; Kinoshita, T.; Sankawa, U. Constituents of the leaves of Murraya paniculata collected in Taiwan. Chem. Pharm. Bull. 1989, 37, 358–362. [Google Scholar] [CrossRef]
- Barik, B.R.; Dey, A.K.; Das, P.C.; Chatterjee, A.; Shoolery, J.N. Coumarins of Murraya exotica. Absolute configuration of auraptenol. Phytochemistry 1983, 22, 792–794. [Google Scholar] [CrossRef]
- Stevensona, P.C.; Simmondsa, M.S.J.; Yulea, M.A.; Veitcha, N.C.; Kitea, G.C.; Irwinb, D.; Leggb, M. Insect antifeedant furanocoumarins from Tetradium daniellii. Phytochemistry 2003, 63, 41–46. [Google Scholar] [CrossRef]
- Baba, K.; Hamasaki, F.; Tabata, Y.; Kozawa, M.; Honda, G.; Tabata, M. Chemical studies on Chinese crude drug “She Chuang Zi”. Shoyakugaku Zasshi 1985, 39, 282–290. [Google Scholar]
- Boyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J. Clin. Lab. Investig. 1968, 97, 77–89. [Google Scholar]
- Jauregui, H.O.; Hayner, N.T.; Driscoll, J.L.; Williams-Holland, R.; Lipsky, M.H.; Galletti, P.M. Trypan blue dye uptake and lactate dehydrogenase in adult rat hepatocytes-freshly isolated cells, cell suspensions, and primary monolayer cultures. In Vitro 1981, 17, 1100–1110. [Google Scholar] [CrossRef]
- Hwang, T.L.; Leu, Y.L.; Kao, S.H.; Tang, M.C.; Chang, H.L. Viscolin, a new chalcone from Viscum coloratum, inhibits human neutrophil superoxide anion and elastase release via a cAMP-dependent pathway. Free Radic. Biol. Med. 2006, 41, 1433–1441. [Google Scholar] [CrossRef]
- Chen, J.J.; Ting, C.W.; Wu, Y.C.; Hwang, T.L.; Cheng, M.J.; Sung, P.J.; Wang, T.C.; Chen, J.F. New labdane-type diterpenoids and anti-inflammatory constituents from Hedychium coronarium. Int. J. Mol. Sci. 2013, 14, 13063–13077. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, T.-H.; Chen, Y.-C.; Hwang, T.-L.; Shu, C.-W.; Sung, P.-J.; Lim, Y.-P.; Kuo, W.-L.; Chen, J.-J. New Coumarins and Anti-Inflammatory Constituents from the Fruits of Cnidium monnieri. Int. J. Mol. Sci. 2014, 15, 9566-9578. https://doi.org/10.3390/ijms15069566
Lee T-H, Chen Y-C, Hwang T-L, Shu C-W, Sung P-J, Lim Y-P, Kuo W-L, Chen J-J. New Coumarins and Anti-Inflammatory Constituents from the Fruits of Cnidium monnieri. International Journal of Molecular Sciences. 2014; 15(6):9566-9578. https://doi.org/10.3390/ijms15069566
Chicago/Turabian StyleLee, Tzong-Huei, Yuan-Chih Chen, Tsong-Long Hwang, Chih-Wen Shu, Ping-Jyun Sung, Yun-Ping Lim, Wen-Lung Kuo, and Jih-Jung Chen. 2014. "New Coumarins and Anti-Inflammatory Constituents from the Fruits of Cnidium monnieri" International Journal of Molecular Sciences 15, no. 6: 9566-9578. https://doi.org/10.3390/ijms15069566
APA StyleLee, T.-H., Chen, Y.-C., Hwang, T.-L., Shu, C.-W., Sung, P.-J., Lim, Y.-P., Kuo, W.-L., & Chen, J.-J. (2014). New Coumarins and Anti-Inflammatory Constituents from the Fruits of Cnidium monnieri. International Journal of Molecular Sciences, 15(6), 9566-9578. https://doi.org/10.3390/ijms15069566