Role of VEGF-A and Its Receptors in Sporadic and MEN2-Associated Pheochromocytoma
Abstract
:1. Introduction
2. Results
2.1. Patients
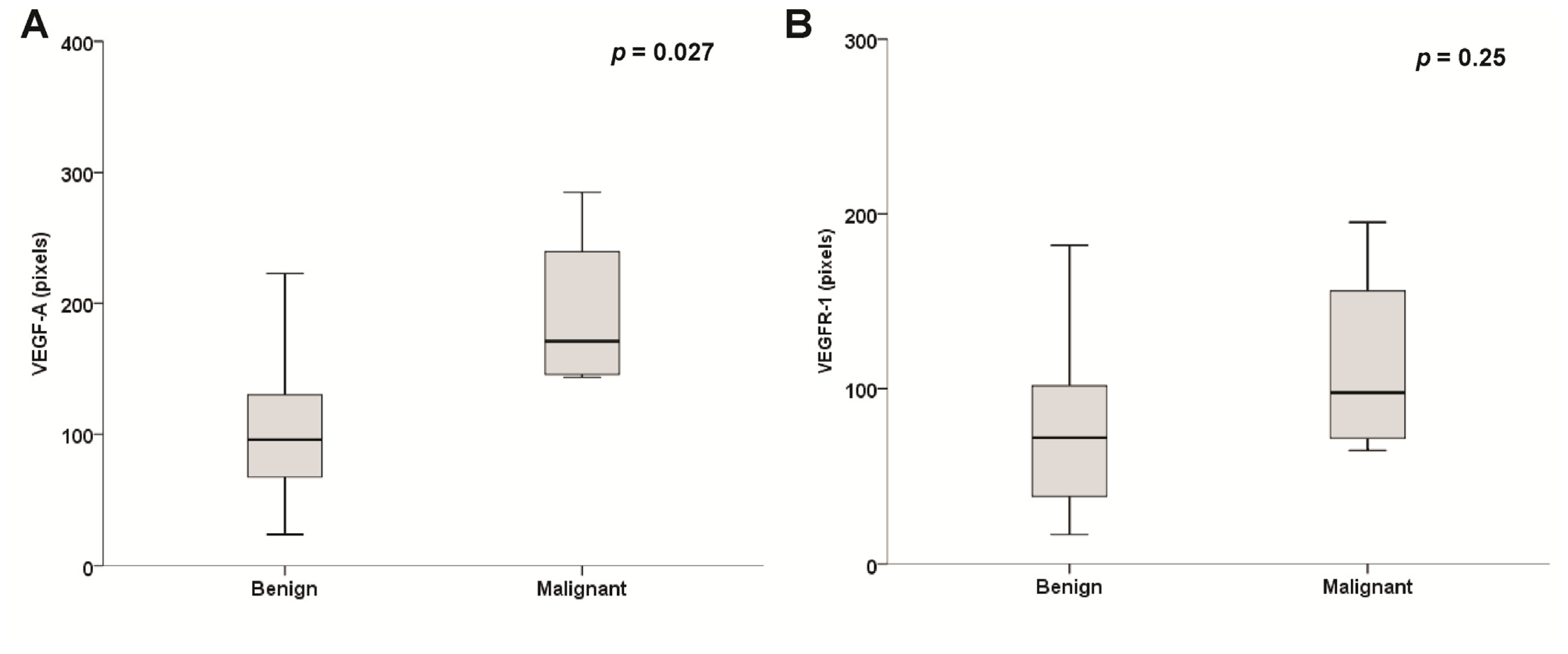
2.2. Expression of Vascular Endothelial Growth Factor (VEGF)-A, Vascular Endothelial Growth Factor Receptor (VEGFR)-1 and VEGFR-2 in Pheochromocytomas
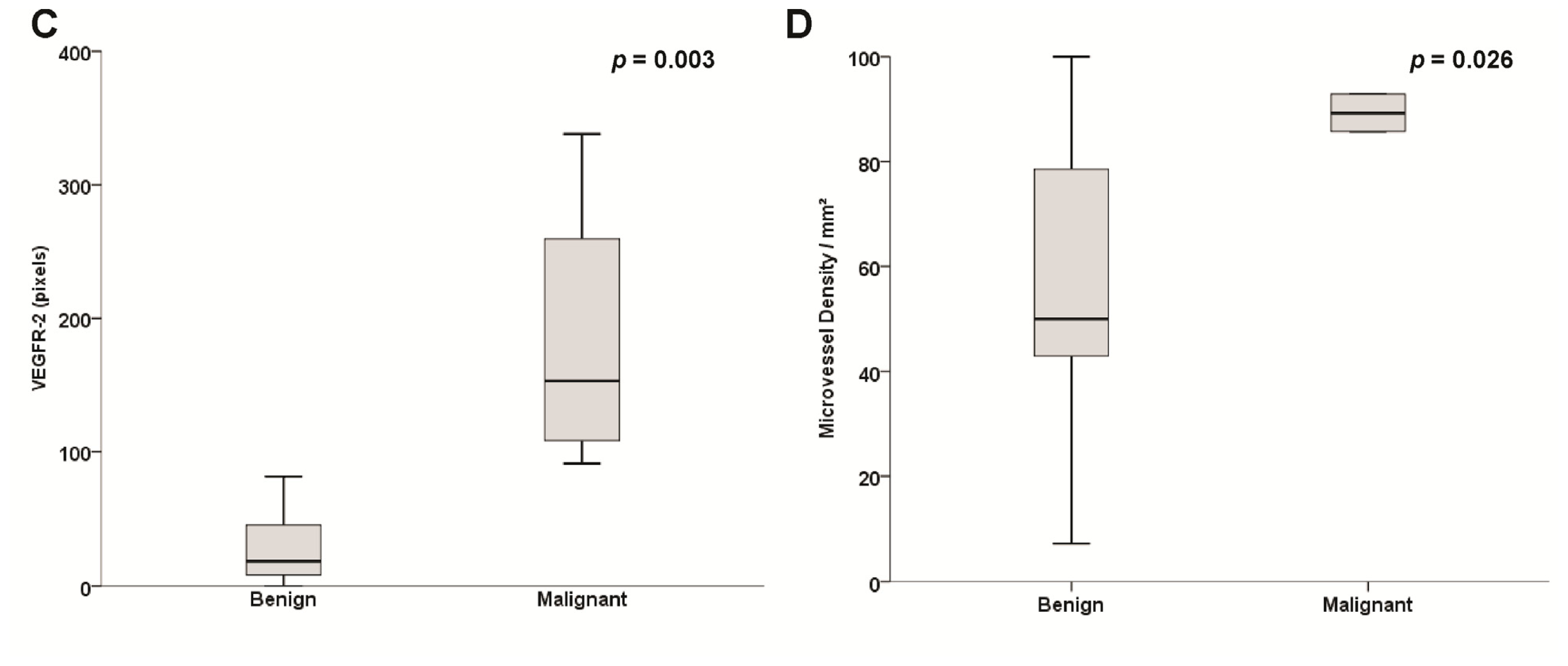
2.3. Microvessel Density (MVD) Assessment
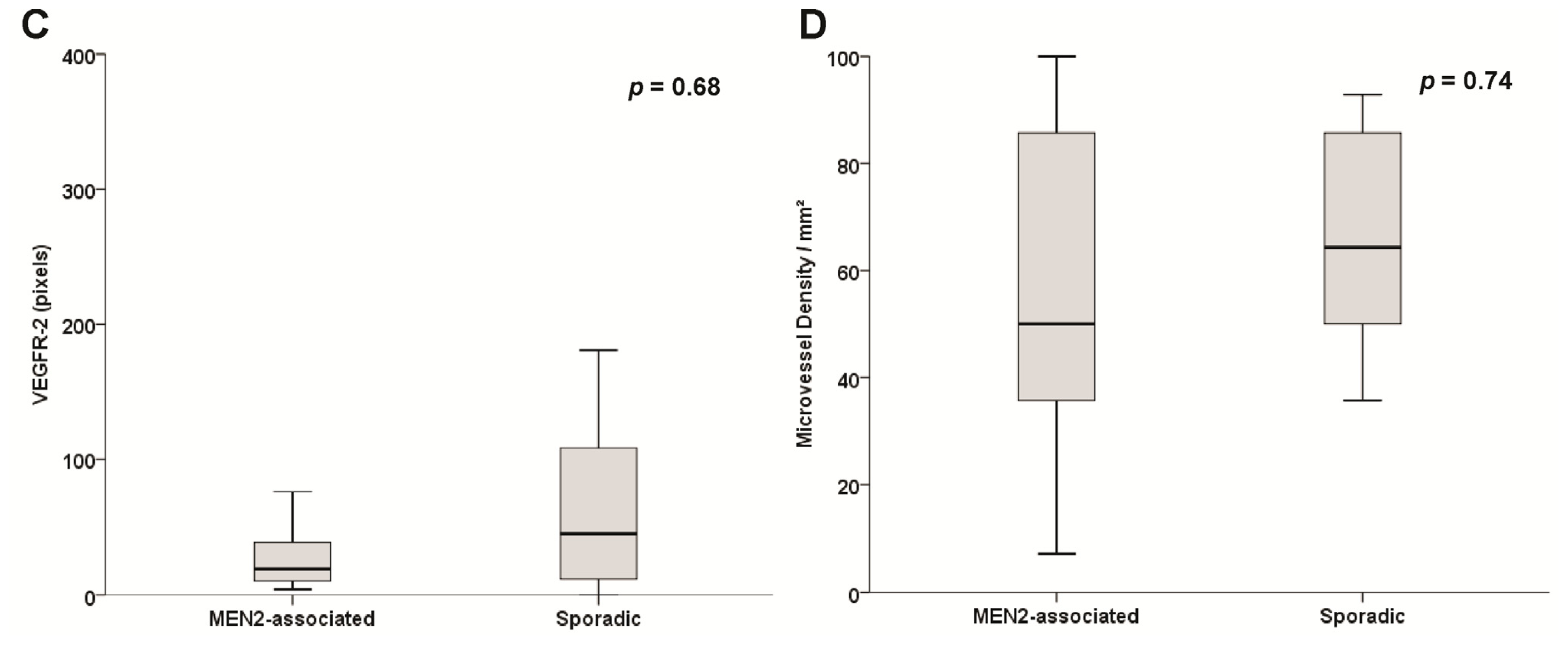
2.4. Sporadic and Hereditary Pheochromocytoma (PHEO) Tumors
3. Discussion
4. Material and Methods
4.1. Patients
4.2. Immunohistochemistry Analysis (IHC)
4.3. Semi-Quantitative Analysis for the Intensity of Positive Staining in Tissues
4.4. Microvessel Density (MVD) Assessment
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsConception and design: C.V.F., D.R.S., A.L.M. Provision of study materials or patients: A.L.M. Collection and assembly of data: C.V.F., D.R.S., M.R., L.C., A.L.M. Data analysis and interpretation: All authors. Manuscript writing: C.V.F., D.R.S., A.L.M. Final approval of manuscript: All authors.
References
- Lenders, J.W.; Eisenhofer, G.; Mannelli, M.; Pacak, K. Phaeochromocytoma. Lancet 2005, 366, 665–675. [Google Scholar]
- Waguespack, S.G.; Rich, T.; Grubbs, E.; Ying, A.K.; Perrier, N.D.; Ayala-Ramirez, M.; Jimenez, C. A current review of the etiology, diagnosis, and treatment of pediatric pheochromocytoma and paraganglioma. J. Clin. Endocrinol. Metab 2010, 95, 2023–2037. [Google Scholar]
- Neumann, H.P.; Bausch, B.; McWhinney, S.R.; Bender, B.U.; Gimm, O.; Franke, G.; Schipper, J.; Klisch, J.; Altehoefer, C.; Zerres, K. Germ-line mutations in nonsyndromic pheochromocytoma. N. Engl. J. Med. 2002, 346, 1459–1466. [Google Scholar]
- Opocher, G.; Schiavi, F.; Conton, P.; Scaroni, C.; Mantero, F. Clinical and genetic aspects of phaeochromocytoma. Horm. Res 2003, 59, 56–61. [Google Scholar]
- Plouin, P.F.; Duclos, J.M.; Soppelsa, F.; Boublil, G.; Chatellier, G. Factors associated with perioperative morbidity and mortality in patients with pheochromocytoma: Analysis of 165 operations at a single center. J. Clin. Endocrinol. Metab 2001, 86, 1480–1486. [Google Scholar]
- De Toma, G.; Letizia, C.; Cavallaro, G.; Giacchino, V.; Mosiello, G.; Cavallaro, A.; Basile, U. Malignant pheochromocytoma. Personal experience, review of the literature. Ann. Ital. Chir 2002, 73, 413–418, discussion 418–419. [Google Scholar]
- Harari, A.; Inabnet, W.B., III. Malignant pheochromocytoma: A review. Am. J. Surg 2011, 201, 700–708. [Google Scholar]
- Whalen, R.K.; Althausen, A.F.; Daniels, G.H. Extra-adrenal pheochromocytoma. J. Urol 1992, 147, 1–10. [Google Scholar]
- Papetti, M.; Herman, I.M. Mechanisms of normal and tumor-derived angiogenesis. Am. J. Physiol. Cell Physiol 2002, 282, C947–C970. [Google Scholar]
- Folkman, J. What is the evidence that tumors are angiogenesis dependent? J. Natl. Cancer Inst 1990, 82, 4–6. [Google Scholar]
- Bergers, G.; Benjamin, L.E. Tumorigenesis and the angiogenic switch. Nat. Rev. Cancer 2003, 3, 401–410. [Google Scholar]
- Shibuya, M.; Claesson-Welsh, L. Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp. Cell Res 2006, 312, 549–560. [Google Scholar]
- Petrova, T.V.; Makinen, T.; Alitalo, K. Signaling via vascular endothelial growth factor receptors. Exp. Cell Res 1999, 253, 117–130. [Google Scholar]
- Zeng, Y.; Zhao, G.F.; Qian, J.; Wang, Z.L.; She, M.P. Effect of oxLDL on the uptake and clearance rate of cholesterol in vascular smooth muscle cells originated from human apoAI transgenic mice. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2001, 23, 328–332. [Google Scholar]
- Ferrara, N. Molecular and biological properties of vascular endothelial growth factor. J. Mol. Med 1999, 77, 527–543. [Google Scholar]
- Gasparini, G.; Toi, M.; Gion, M.; Verderio, P.; Dittadi, R.; Hanatani, M.; Matsubara, I.; Vinante, O.; Bonoldi, E.; Boracchi, P.; et al. Prognostic significance of vascular endothelial growth factor protein in node-negative breast carcinoma. J. Natl. Cancer Inst 1997, 89, 139–147. [Google Scholar]
- Maeda, K.; Chung, Y.S.; Ogawa, Y.; Takatsuka, S.; Kang, S.M.; Ogawa, M.; Sawada, T.; Sowa, M. Prognostic value of vascular endothelial growth factor expression in gastric carcinoma. Cancer 1996, 77, 858–863. [Google Scholar]
- Salven, P.; Teerenhovi, L.; Joensuu, H. A high pretreatment serum vascular endothelial growth factor concentration is associated with poor outcome in non-Hodgkin’s lymphoma. Blood 1997, 90, 3167–3172. [Google Scholar]
- Takekoshi, K.; Isobe, K.; Yashiro, T.; Hara, H.; Ishii, K.; Kawakami, Y.; Nakai, T.; Okuda, Y. Expression of vascular endothelial growth factor (VEGF) and its cognate receptors in human pheochromocytomas. Life Sci 2004, 74, 863–871. [Google Scholar]
- Salmenkivi, K.; Heikkila, P.; Liu, J.; Haglund, C.; Arola, J. VEGF in 105 pheochromocytomas: Enhanced expression correlates with malignant outcome. APMIS 2003, 111, 458–464. [Google Scholar]
- Zielke, A.; Middeke, M.; Hoffmann, S.; Colombo-Benkmann, M.; Barth, P.; Hassan, I.; Wunderlich, A.; Hofbauer, L.C.; Duh, Q.Y. VEGF-mediated angiogenesis of human pheochromocytomas is associated to malignancy and inhibited by anti-VEGF antibodies in experimental tumors. Surgery 2002, 132, 1056–1063, discussion 1063. [Google Scholar]
- Favier, J.; Plouin, P.F.; Corvol, P.; Gasc, J.M. Angiogenesis and vascular architecture in pheochromocytomas: Distinctive traits in malignant tumors. Am. J. Pathol 2002, 161, 1235–1246. [Google Scholar]
- Ohji, H.; Sasagawa, I.; Iciyanagi, O.; Suzuki, Y.; Nakada, T. Tumour angiogenesis and Ki-67 expression in phaeochromocytoma. BJU Int 2001, 87, 381–385. [Google Scholar]
- Ceolin, L.; Siqueira, D.R.; Romitti, M.; Ferreira, C.V.; Maia, A.L. Molecular basis of medullary thyroid carcinoma: The role of RET polymorphisms. Int. J. Mol. Sci 2012, 13, 221–239. [Google Scholar]
- Kouvaraki, M.A.; Shapiro, S.E.; Perrier, N.D.; Cote, G.J.; Gagel, R.F.; Hoff, A.O.; Sherman, S.I.; Lee, J.E.; Evans, D.B. RET proto-oncogene: A review and update of genotype-phenotype correlations in hereditary medullary thyroid cancer and associated endocrine tumors. Thyroid 2005, 15, 531–544. [Google Scholar]
- Capp, C.; Wajner, S.M.; Siqueira, D.R.; Brasil, B.A.; Meurer, L.; Maia, A.L. Increased expression of vascular endothelial growth factor and its receptors, VEGFR-1 and VEGFR-2, in medullary thyroid carcinoma. Thyroid 2010, 20, 863–871. [Google Scholar]
- Adler, J.T.; Meyer-Rochow, G.Y.; Chen, H.; Benn, D.E.; Robinson, B.G.; Sippel, R.S.; Sidhu, S.B. Pheochromocytoma: Current approaches and future directions. Oncologist 2008, 13, 779–793. [Google Scholar]
- Chen, H.; Sippel, R.S.; O’Dorisio, M.S.; Vinik, A.I.; Lloyd, R.V.; Pacak, K. The North American Neuroendocrine Tumor Society consensus guideline for the diagnosis and management of neuroendocrine tumors: Pheochromocytoma, paraganglioma, and medullary thyroid cancer. Pancreas 2010, 39, 775–783. [Google Scholar]
- Ayala-Ramirez, M.; Feng, L.; Habra, M.A.; Rich, T.; Dickson, P.V.; Perrier, N.; Phan, A.; Waguespack, S.; Patel, S.; Jimenez, C. Clinical benefits of systemic chemotherapy for patients with metastatic pheochromocytomas or sympathetic extra-adrenal paragangliomas: Insights from the largest single-institutional experience. Cancer 2011, 118, 2804–2812. [Google Scholar]
- Chrisoulidou, A.; Kaltsas, G.; Ilias, I.; Grossman, A.B. The diagnosis and management of malignant phaeochromocytoma and paraganglioma. Endocr. Relat. Cancer 2007, 14, 569–585. [Google Scholar]
- Nomura, K.; Kimura, H.; Shimizu, S.; Kodama, H.; Okamoto, T.; Obara, T.; Takano, K. Survival of patients with metastatic malignant pheochromocytoma and efficacy of combined cyclophosphamide, vincristine, and dacarbazine chemotherapy. J. Clin. Endocrinol. Metab 2009, 94, 2850–2856. [Google Scholar]
- Hahn, N.M.; Reckova, M.; Cheng, L.; Baldridge, L.A.; Cummings, O.W.; Sweeney, C.J. Patient with malignant paraganglioma responding to the multikinase inhibitor sunitinib malate. J. Clin. Oncol 2009, 27, 460–463. [Google Scholar]
- Joshua, A.M.; Ezzat, S.; Asa, S.L.; Evans, A.; Broom, R.; Freeman, M.; Knox, J.J. Rationale and evidence for sunitinib in the treatment of malignant paraganglioma/pheochromocytoma. J. Clin. Endocrinol. Metab 2009, 94, 5–9. [Google Scholar]
- Saito, Y.; Tanaka, Y.; Aita, Y.; Ishii, K.A.; Ikeda, T.; Isobe, K.; Kawakami, Y.; Shimano, H.; Hara, H.; Takekoshi, K. Sunitinib induces apoptosis in pheochromocytoma tumor cells by inhibiting VEGFR2/Akt/mTOR/S6K1 pathways through modulation of Bcl-2 and BAD. Am. J. Physiol. Endocrinol. Metab 2011, 302, E615–E625. [Google Scholar]
- Gimenez-Roqueplo, A.P.; Favier, J.; Rustin, P.; Mourad, J.J.; Plouin, P.F.; Corvol, P.; Rotig, A.; Jeunemaitre, X. The R22X mutation of the SDHD gene in hereditaryparaganglioma abolishes the enzymatic activity of complex II in the mitochondrial respiratory chain and activates the hypoxia pathway. Am. J. Hum. Genet 2001, 69, 1186–1197. [Google Scholar]
- Favier, J.; Igaz, P.; Burnichon, N.; Amar, L.; Libe, R.; Badoual, C.; Tissier, F.; Bertherat, J.; Plouin, P.F.; Jeunemaitre, X.; et al. Rationale for anti-angiogenic therapy in pheochromocytoma and paraganglioma. Endocr. Pathol 2012, 23, 34–42. [Google Scholar]
- Weidner, N.; Carroll, P.R.; Flax, J.; Blumenfeld, W.; Folkman, J. Tumor angiogenesis correlates with metastasis in invasive prostate carcinoma. Am. J. Pathol 1993, 143, 401–409. [Google Scholar]
- Weidner, N.; Semple, J.P.; Welch, W.R.; Folkman, J. Tumor angiogenesis and metastasis— Correlation in invasive breast carcinoma. N. Engl. J. Med 1991, 324, 1–8. [Google Scholar]
- Macchiarini, P.; Fontanini, G.; Dulmet, E.; de Montpreville, V.; Chapelier, A.R.; Cerrina, J.; Ladurie, F.L.; Dartevelle, P.G. Angiogenesis: An indicator of metastasis in non-small cell lung cancer invading the thoracic inlet. Ann. Thorac. Surg 1994, 57, 1534–1539. [Google Scholar]
- Liu, Q.; Djuricin, G.; Staren, E.D.; Gattuso, P.; Gould, V.E.; Shen, J.; Saclarides, T.; Rubin, D.B.; Prinz, R.A. Tumor angiogenesis in pheochromocytomas and paragangliomas. Surgery 1996, 120, 938–942, discussion 942–943. [Google Scholar]
- Rooijens, P.P.; de Krijger, R.R.; Bonjer, H.J.; van der Ham, F.; Nigg, A.L.; Bruining, H.A.; Lamberts, S.W.; van der Harst, E. The significance of angiogenesis in malignant pheochromocytomas. Endocr. Pathol 2004, 15, 39–45. [Google Scholar]
- Punales, M.K.; Graf, H.; Gross, J.L.; Maia, A.L. RET codon 634 mutations in multiple endocrine neoplasia type 2: Variable clinical features and clinical outcome. J. Clin. Endocrinol. Metab 2003, 88, 2644–2649. [Google Scholar]
- Brondani, L.A.; de Souza, B.M.; Duarte, G.C.; Kliemann, L.M.; Esteves, J.F.; Marcon, A.S.; Gross, J.L.; Canani, L.H.; Crispim, D. The UCP1-3826A/G polymorphism is associated with diabetic retinopathy and increased UCP1 and MnSOD2 gene expression in human retina. Investig. Ophthalmol. Vis. Sci 2012, 53, 7449–7457. [Google Scholar]
- Vermeulen, P.B.; Gasparini, G.; Fox, S.B.; Colpaert, C.; Marson, L.P.; Gion, M.; Belien, J.A.; de Waal, R.M.; van Marck, E.; Magnani, E.; et al. Second international consensus on the methodology and criteria of evaluation of angiogenesis quantification in solid human tumours. Eur. J. Cancer 2002, 38, 1564–1579. [Google Scholar]
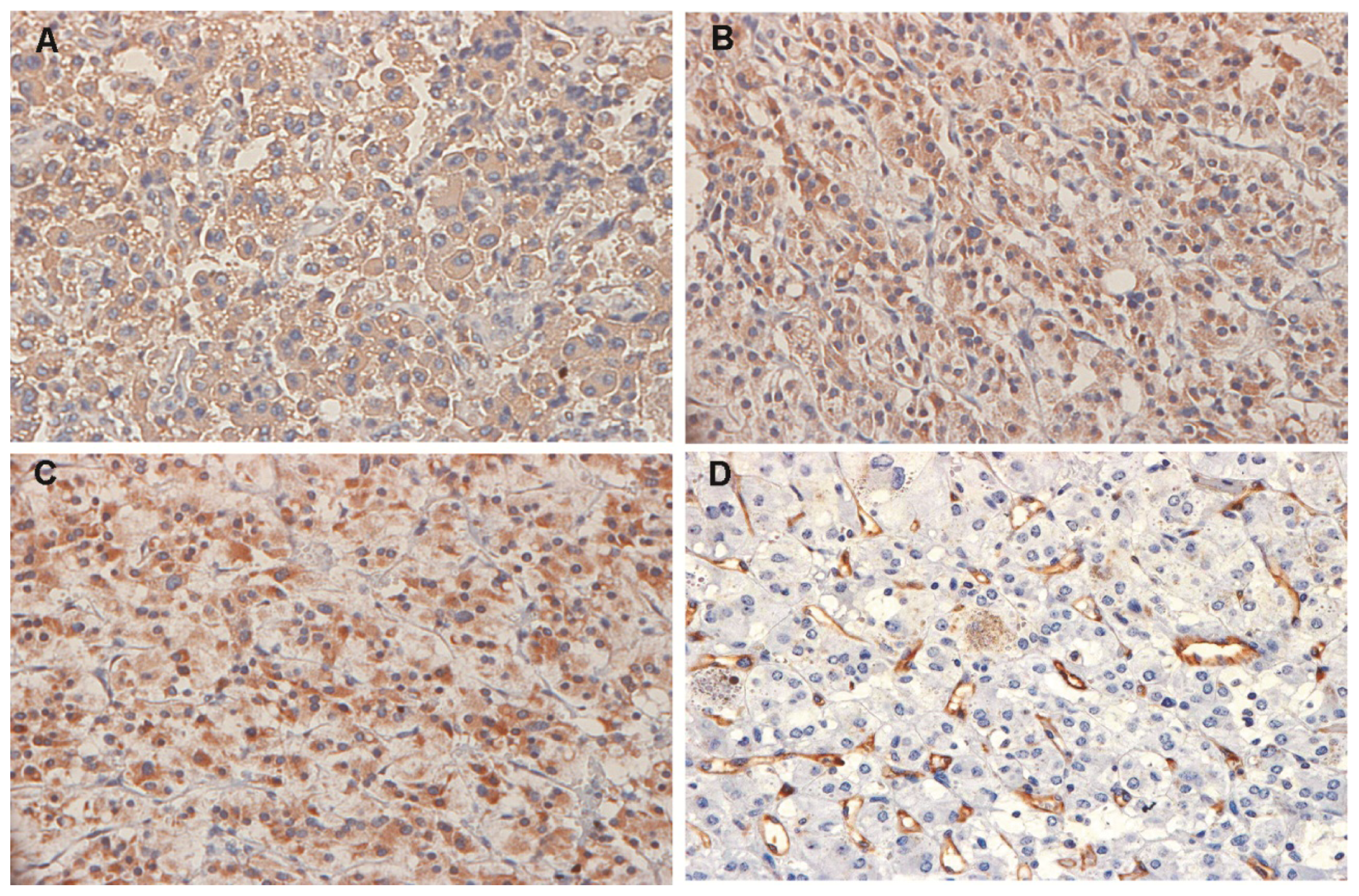

| Case | Phenotype | RET germline mutation | Age (years) 1/sex | Tumor size 2 | VEGF-A 3 | VEGFR-1 3 | VEGFR-2 3 | MVD 4 |
|---|---|---|---|---|---|---|---|---|
| 1 | MEN 2 A | C618R | 38/F | 7.50 | 118.6 | 102.8 | 16 | 78.6 |
| 2 | MEN 2 A | C634W | 36/F | 4.0 | 23.6 | 54 | 6.5 | 100 |
| 3 | MEN 2 A | C634R | 37/F | 6.40 | 319 | 27.6 | 4.2 | 35.7 |
| 4 | MEN 2 A | C634R | 21/F | 1.70 | 63.2 | 38.3 | 19.2 | 14.3 |
| 5 | MEN 2 A | C634R | 34/F | 5.50 | 78.5 | 182 | 29.6 | 92.9 |
| 6 | MEN 2 B | M918T | 49/F | N/A | 95.9 | 75.9 | N/A | 85.7 |
| 7 | MEN 2 B | M918T | 33/F | 2.5 | 67.3 | 47.6 | 67.6 | 50 |
| 8 | MEN 2 A | C634Y | 61/M | N/A | 199.1 | 35.5 | 38.7 | 50 |
| 9 | MEN 2 A | C634Y | 44/F | 1.00 | 97.2 | 81.1 | 17.3 | 7.14 |
| 10 | MEN 2 A | C634Y | 49/M | 1.20 | 222.9 | 101.6 | 75.9 | 7.14 |
| 11 | MEN 2 A | C634Y | 55/M | N/A | 102.6 | 163 | 52.2 | 57.1 |
| 12 | MEN 2 A | C634Y | 62/F | 3.50 | 220.1 | 35.8 | 4.5 | 42.9 |
| 13 | MEN 2 A | C634Y | 45/M | 2.5 | 65.8 | 62.7 | 10.3 | 92.8 |
| 14 | MEN 2 A | C634Y | 23/M | 1.4 | 112.5 | 253.1 | 33.2 | 50 |
| 15 | Sporadic 5 | 14/F | 6.50 | 143.5 | 64.6 | 338.1 | 92.8 | |
| 16 | Sporadic 5 | 30/F | 8.20 | 284.8 | 116.7 | 180.8 | 92.9 | |
| 17 | Sporadic 5 | 23/M | 13.0 | 194.1 | 195.2 | 91.3 | 85.7 | |
| 18 | Sporadic 5 | 52/M | 4.0 | 147.9 | 78.6 | 125.5 | 85.7 | |
| 19 | Sporadic | 20/F | 5.50 | 130.1 | 37.3 | 45.1 | 64.3 | |
| 20 | Sporadic | 18/M | 6.00 | 76 | 50 | 1.6 | 50 | |
| 21 | Sporadic | 34/F | 11.5 | 89.4 | 87.9 | 16 | 50 | |
| 22 | Sporadic | 45/F | 4.00 | 135.7 | 229.7 | 81.4 | 42.9 | |
| 23 | Sporadic | 39/F | 5.50 | 51.4 | 68.5 | 13.4 | 85.7 | |
| 24 | Sporadic | 23/F | 3.50 | 33.4 | 71.9 | 0 | 92.8 | |
| 25 | Sporadic | 23/F | 8.50 | 77.4 | 92.5 | 135 | 42.9 | |
| 26 | Sporadic | 35/F | 11.0 | 160.6 | 157.7 | 28.1 | 35.7 | |
| 27 | Sporadic | 38/F | 7.50 | 101.8 | 16.6 | 2.3 | 57.1 | |
| 28 | Sporadic | 61/F | 3.5 | 44 | 101.2 | 45.3 | 64.3 | |
| 29 | Sporadic | 58/M | 3.4 | 81.4 | 28.5 | 9.9 | 57.1 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ferreira, C.V.; Siqueira, D.R.; Romitti, M.; Ceolin, L.; Brasil, B.A.; Meurer, L.; Capp, C.; Maia, A.L. Role of VEGF-A and Its Receptors in Sporadic and MEN2-Associated Pheochromocytoma. Int. J. Mol. Sci. 2014, 15, 5323-5336. https://doi.org/10.3390/ijms15045323
Ferreira CV, Siqueira DR, Romitti M, Ceolin L, Brasil BA, Meurer L, Capp C, Maia AL. Role of VEGF-A and Its Receptors in Sporadic and MEN2-Associated Pheochromocytoma. International Journal of Molecular Sciences. 2014; 15(4):5323-5336. https://doi.org/10.3390/ijms15045323
Chicago/Turabian StyleFerreira, Carla Vaz, Débora Rodrigues Siqueira, Mírian Romitti, Lucieli Ceolin, Beatriz Assis Brasil, Luise Meurer, Clarissa Capp, and Ana Luiza Maia. 2014. "Role of VEGF-A and Its Receptors in Sporadic and MEN2-Associated Pheochromocytoma" International Journal of Molecular Sciences 15, no. 4: 5323-5336. https://doi.org/10.3390/ijms15045323




