Toxicological Assessment of Inhaled Nanoparticles: Role of in Vivo, ex Vivo, in Vitro, and in Silico Studies
Abstract
:1. Introduction
2. In Vitro Systems
2.1. Cellular Models for NP Exposure
2.2. Commercial in Vitro Co-Cultures
2.3. ALI-Based Exposure Systems
3. Ex Vivo Systems
4. In Vivo Systems
4.1. Inhalation Exposure Models
4.2. Whole-Body Exposure
4.3. Nose/Head Only Exposure
4.4. Lung-Only Exposure
4.5. Limitations of in Vivo Systems
4.5.1. Interspecies Differences in Lung Physiology
4.5.2. Species-Specific Reaction to Particulates
5. In Silico Modeling
5.1. Mechanisms of Deposition
5.2. Deposition Models
5.2.1. Single Path Models
5.2.2. Multiple Path Models
6. Conclusions
Conflicts of Interest
References
- Royal Academy of Engineering Nanoscience and Nanotechnologies: Opportunities and Uncertainties; Royal Academy of Engineering: London, UK, 2004.
- Frampton, M.W. Systemic and cardiovascular effects of airway injury and inflammation: Ultrafine particle exposure in humans. Environ. Health Perspect 2001, 109, 529–532. [Google Scholar]
- Kan, H.; Heiss, G.; Rose, K.M.; Whitsel, E.A.; Lurmann, F.; London, S.J. Prospective analysis of traffic exposure as a risk factor for incident coronary heart disease: The Atherosclerosis Risk in Communities (ARIC) study. Environ. Health Perspect 2008, 116, 1463–1468. [Google Scholar]
- Nawrot, T.S.; Alfaro-Moreno, E.; Nemery, B. Update in occupational and environmental respiratory disease 2007. Am. J. Respir. Crit. Care Med 2008, 177, 696–700. [Google Scholar]
- Pope, C.A., 3rd; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer cardiopulmonary mortality and long-term exposure to fine particulate air pollution. JAMA 2002, 287, 1132–1141. [Google Scholar]
- Pope, C.A., 3rd; Burnett, R.T.; Thurston, G.D.; Thun, M.J.; Calle, E.E.; Krewski, D.; Godleski, J.J. Cardiovascular mortality and long-term exposure to particulate air pollution: Epidemiological evidence of general pathophysiological pathways of disease. Circulation 2004, 109, 71–77. [Google Scholar]
- Ruckerl, R.; Schneider, A.; Breitner, S.; Cyrys, J.; Peters, A. Health effects of particulate air pollution: A review of epidemiological evidence. Inhal. Toxicol 2011, 23, 555–592. [Google Scholar]
- Song, Y.; Li, X.; Du, X. Exposure to nanoparticles is related to pleural effusion pulmonary fibrosis and granuloma. Eur. Respir. J 2009, 34, 559–567. [Google Scholar]
- Sager, T.M.; Kommineni, C.; Castranova, V. Pulmonary response to intratracheal instillation of ultrafine vs fine titanium dioxide: Role of particle surface area. Part. Fibre Toxicol 2008, 5, 17. [Google Scholar]
- Kaluza, S.; Kleine Balderhaar, J.; Orthen, B.; Honnert, B.; Jankowska, E.; Pietrowski, P.; Rosell, M.G.; Tanarro, C.; Tejedor, J. Workplace Exposure to Nanoparticles; European Agency for Safety and Health at Work: Bilbao, Spain, 2009. [Google Scholar]
- Kuhlbusch, T.A.; Asbach, C.; Fissan, H.; Gohler, D.; Stintz, M. Nanoparticle exposure at nanotechnology workplaces: A review. Part. Fibre Toxicol 2011, 8, 22. [Google Scholar]
- Elder, A.; Gelein, R.; Silva, V.; Feikert, T.; Opanashuk, L.; Carter, J.; Potter, R.; Maynard, A.; Ito, Y.; Finkelstein, J.; et al. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ. Health Perspect 2006, 114, 1172–1178. [Google Scholar]
- De Lorenzo, A.J. Electron microscopic observations of the olfactory mucosa and olfactory nerve. J. Biophys. Biochem. Cytol 1957, 3, 839–850. [Google Scholar]
- Oberdorster, G.; Elder, A.; Rinderknecht, A. Nanoparticles and the brain: Cause for concern? J. Nanosci. Nanotechnol 2009, 9, 4996–5007. [Google Scholar]
- Radostits, O.; Houston, D.; Mayhew, I. Veterinary Clinical Examination and Diagnosis; W.B. Saunders: Philadelphia, PA, USA, 2000. [Google Scholar]
- Nemmar, A.; Hoet, P.H.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of inhaled particles into the blood circulation in humans. Circulation 2002, 105, 411–414. [Google Scholar]
- Kreyling, W.G.; Semmler, M.; Erbe, F.; Mayer, P.; Takenaka, S.; Schulz, H.; Oberdorster, G.; Ziesenis, A. Translocation of ultrafine insoluble iridium particles from lung epithelium to extrapulmonary organs is size dependent but very low. J. Toxicol. Environ. Health A 2002, 65, 1513–1530. [Google Scholar]
- Mills, N.L.; Amin, N.; Robinson, S.D.; Anand, A.; Davies, J.; Patel, D.; de la Fuente, J.M.; Cassee, F.R.; Boon, N.A.; Macnee, W.; et al. Do inhaled carbon nanoparticles translocate directly into the circulation in humans? Am. J. Respir. Crit. Care Med 2006, 173, 426–431. [Google Scholar]
- Wiebert, P.; Sanchez-Crespo, A.; Seitz, J.; Falk, R.; Philipson, K.; Kreyling, W.G.; Moller, W.; Sommerer, K.; Larsson, S.; Svartengren, M. Negligible clearance of ultrafine particles retained in healthy and affected human lungs. Eur. Respir. J 2006, 28, 286–290. [Google Scholar]
- Nemmar, A.; Vanbilloen, H.; Hoylaerts, M.F.; Hoet, P.H.; Verbruggen, A.; Nemery, B. Passage of intratracheally instilled ultrafine particles from the lung into the systemic circulation in hamster. Am. J. Respir. Crit. Care Med 2001, 164, 1665–1668. [Google Scholar]
- Takenaka, S.; Karg, E.; Roth, C.; Schulz, H.; Ziesenis, A.; Heinzmann, U.; Schramel, P.; Heyder, J. Pulmonary and systemic distribution of inhaled ultrafine silver particles in rats. Environ. Health Perspect 2001, 109, 547–551. [Google Scholar]
- Muhlfeld, C.; Gehr, P.; Rothen-Rutishauser, B. Translocation and cellular entering mechanisms of nanoparticles in the respiratory tract. Swiss Med. Wkly. 2008, 138, 387–391. [Google Scholar]
- Sauer, U.G.; Vogel, S.; Hess, A.; Kolle, S.N.; Ma-Hock, L.; van Ravenzwaay, B.; Landsiedel, R. In vivo-in vitro comparison of acute respiratory tract toxicity using human 3D airway epithelial models and human A549 and murine 3T3 monolayer cell systems. Toxicol. in Vitro 2013, 27, 174–190. [Google Scholar]
- Sorokin, S.P. Properties of alveolar cells and tissues that strengthen alveolar defenses. Arch. Intern. Med 1970, 126, 450–463. [Google Scholar]
- Mondrinos, M.J.; Koutzaki, S.; Lelkes, P.I.; Finck, C.M. A tissue-engineered model of fetal distal lung tissue. Am. J. Physiol. Lung Cell Mol. Physiol 2007, 293, L639–L650. [Google Scholar]
- Klein, S.G.; Hennen, J.; Serchi, T.; Blomeke, B.; Gutleb, A.C. Potential of coculture in vitro models to study inflammatory and sensitizing effects of particles on the lung. Toxicol. in Vitro 2011, 25, 1516–1534. [Google Scholar]
- Chono, S.; Tauchi, Y.; Morimoto, K. Pharmacokinetic analysis of the uptake of liposomes by macrophages and foam cells in vitro and their distribution to atherosclerotic lesions in mice. Drug Metab. Pharmacokinet 2006, 21, 37–44. [Google Scholar]
- Hidalgo, I.J.; Raub, T.J.; Borchardt, R.T. Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology 1989, 96, 736–749. [Google Scholar]
- Kreda, S.M.; Okada, S.F.; van Heusden, C.A.; O’Neal, W.; Gabriel, S.; Abdullah, L.; Davis, C.W.; Boucher, R.C.; Lazarowski, E.R. Coordinated release of nucleotides and mucin from human airway epithelial Calu-3 cells. J. Physiol 2007, 584, 245–259. [Google Scholar]
- Ross, A.J.; Dailey, L.A.; Brighton, L.E.; Devlin, R.B. Transcriptional profiling of mucociliary differentiation in human airway epithelial cells. Am. J. Respir. Cell Mol. Biol 2007, 37, 169–185. [Google Scholar]
- Rothen-Rutishauser, B.; Blank, F.; Muhlfeld, C.; Gehr, P. In vitro models of the human epithelial airway barrier to study the toxic potential of particulate matter. Expert. Opin. Drug Metabol. Toxicol 2008, 4, 1075–1089. [Google Scholar]
- Gehr, P.; Green, F.H.; Geiser, M.; Im Hof, V.; Lee, M.M.; Schurch, S. Airway surfactant a primary defense barrier: Mechanical and immunological aspects. J. Aerosol Med 1996, 9, 163–181. [Google Scholar]
- Bakand, S.; Winder, C.; Khalil, C.; Hayes, A. A novel in vitro exposure technique for toxicity testing of selected volatile organic compounds. J. Environ. Monit 2006, 8, 100–105. [Google Scholar]
- Joeng, L.; Hayes, A.; Bakand, S. Validation of the dynamic direct exposure method for toxicity testing of diesel exhaust in vitro. ISRN Toxicol 2013, 2013, 139512. [Google Scholar]
- Coecke, S.; Blaauboer, B.; Elaut, G.; Freeman, S.; Freidig, A.; Gensmantel, N.; Hoet, P.H.; Kapoulas, V.; Ladstetter, B.; Langley, G.; et al. Final Document Subgroup of Experts on Toxicokinetics and Metabolism in the Context of the Follow-up of the 7th Amendment on the Cosmetics Directive; ECVAM: Ispra, Italy, 2004. [Google Scholar]
- Castell, J.V.; Donato, M.T.; Gomez-Lechon, M.J. Metabolism and bioactivation of toxicants in the lung The in vitro cellular approach. Exp. Toxicol. Pathol 2005, 57, 189–204. [Google Scholar]
- Zabner, J.; Karp, P.; Seiler, M.; Phillips, S.L.; Mitchell, C.J.; Saavedra, M.; Welsh, M.; Klingelhutz, A.J. Development of cystic fibrosis and noncystic fibrosis airway cell lines. Am. J. Physiol. Lung Cell Mol. Physiol 2003, 284, L844–L854. [Google Scholar]
- Sakagami, M. In vivo in vitro and ex vivo models to assess pulmonary absorption and disposition of inhaled therapeutics for systemic delivery. Adv. Drug Deliv. Rev 2006, 58, 1030–1060. [Google Scholar]
- Rothen-Rutishauser, B.M.; Kiama, S.G.; Gehr, P. A three-dimensional cellular model of the human respiratory tract to study the interaction with particles. Am. J. Respir. Cell Mol. Biol 2005, 32, 281–289. [Google Scholar]
- Alfaro-Moreno, E.; Nawrot, T.S.; Vanaudenaerde, B.M.; Hoylaerts, M.F.; Vanoirbeek, J.A.; Nemery, B.; Hoet, P.H. Co-cultures of multiple cell types mimic pulmonary cell communication in response to urban PM10. Eur. Respir. J 2008, 32, 1184–1194. [Google Scholar]
- Klein, S.G.; Serchi, T.; Hoffmann, L.; Blomeke, B.; Gutleb, A.C. An improved 3D tetraculture system mimicking the cellular organisation at the alveolar barrier to study the potential toxic effects of particles on the lung. Part. Fibre Toxicol 2013, 10, 31. [Google Scholar]
- Nguyen Hoang, A.T.; Chen, P.; Juarez, J.; Sachamitr, P.; Billing, B.; Bosnjak, L.; Dahlen, B.; Coles, M.; Svensson, M. Dendritic cell functional properties in a three-dimensional tissue model of human lung mucosa. Am. J. Physiol. Lung Cell Mol. Physiol 2012, 302, L226–L237. [Google Scholar]
- Huh, D.; Hamilton, G.A.; Ingber, D.E. From 3D cell culture to organs-on-chips. Trends Cell Biol 2011, 21, 745–754. [Google Scholar]
- Tomei, A.A.; Choe, M.M.; Swartz, M.A. Effects of dynamic compression on lentiviral transduction in an in vitro airway wall model. Am. J. Physiol. Lung Cell Mol. Physiol 2008, 294, L79–L86. [Google Scholar]
- Geys, J.; Coenegrachts, L.; Vercammen, J.; Engelborghs, Y.; Nemmar, A.; Nemery, B.; Hoet, P.H. In vitro study of the pulmonary translocation of nanoparticles: A preliminary study. Toxicol. Lett 2006, 160, 218–226. [Google Scholar]
- Schulze, C.; Schäfer, U.; Voetz, M.; Wohlleben, W.; Venzago, C.; Lehr, C. Transport of metal oxide nanoparticles across Calu-3 monolayers modelling the air-blood barrier. EURO-NanoTox-Letters 2011, 1, 0001–0011. [Google Scholar]
- Zhang, X.; Zheng, N.; Zou, P.; Zhu, H.; Hinestroza, J.P.; Rosania, G.R. Cells on pores: A simulation-driven analysis of transcellular small molecule transport. Mol. Pharm 2010, 7, 456–467. [Google Scholar]
- Berube, K.; Prytherch, Z.; Job, C.; Hughes, T. Human primary bronchial lung cell constructs: The new respiratory models. Toxicology 2010, 278, 311–318. [Google Scholar]
- Voisin, C.; Aerts, C.; Jakubczk, E.; Tonnel, A.B. La culture cellulaire en phase gazeuse Un nouveau modele experimental d’etude in vitro des activites des macrophages alveolaires. Bull. Eur. Physiopathol. Respir 1977, 13, 69–82. [Google Scholar]
- Voisin, C.; Wallaert, B. Occupational dust exposure and chronic obstructive bronchopulmonary disease Etiopathogenic approach to the problem of compensation in the mining environment. Bull. Acad. Natl. Med 1992, 176, 243–250; discussion 250–242. [Google Scholar]
- Bitterle, E.; Karg, E.; Schroeppel, A.; Kreyling, W.G.; Tippe, A.; Ferron, G.A.; Schmid, O.; Heyder, J.; Maier, K.L.; Hofer, T. Dose-controlled exposure of A549 epithelial cells at the air-liquid interface to airborne ultrafine carbonaceous particles. Chemosphere 2006, 65, 1784–1790. [Google Scholar]
- Tippe, A.; Heinzmann, U.; Roth, C. Deposition of fine and ultrafine aerosol particles during exposure at the air/cell interface. J. Aerosol Sci 2002, 33, 207–218. [Google Scholar]
- Gaschen, A.; Lang, D.; Kalberer, M.; Savi, M.; Geiser, T.; Gazdhar, A.; Lehr, C.M.; Bur, M.; Dommen, J.; Baltensperger, U.; et al. Cellular responses after exposure of lung cell cultures to secondary organic aerosol particles. Environ. Sci. Technol 2010, 44, 1424–1430. [Google Scholar]
- Savi, M.; Kalberer, M.; Lang, D.; Ryser, M.; Fierz, M.; Gaschen, A.; Ricka, J.; Geiser, M. A novel exposure system for the efficient and controlled deposition of aerosol particles onto cell cultures. Environ. Sci. Technol 2008, 42, 5667–5674. [Google Scholar]
- Phillips, J.; Kluss, B.; Richter, A.; Massey, E. Exposure of bronchial epithelial cells to whole cigarette smoke: Assessment of cellular responses. Altern. Lab. Anim 2005, 33, 239–248. [Google Scholar]
- Brandenberger, C.; Muhlfeld, C.; Ali, Z.; Lenz, A.G.; Schmid, O.; Parak, W.J.; Gehr, P.; Rothen-Rutishauser, B. Quantitative evaluation of cellular uptake and trafficking of plain and polyethylene glycol-coated gold nanoparticles. Small 2010, 6, 1669–1678. [Google Scholar]
- Brandenberger, C.; Rothen-Rutishauser, B.; Muhlfeld, C.; Schmid, O.; Ferron, G.A.; Maier, K.L.; Gehr, P.; Lenz, A.G. Effects and uptake of gold nanoparticles deposited at the air-liquid interface of a human epithelial airway model. Toxicol. Appl. Pharmacol 2010, 242, 56–65. [Google Scholar]
- Lenz, A.G.; Karg, E.; Lentner, B.; Dittrich, V.; Brandenberger, C.; Rothen-Rutishauser, B.; Schulz, H.; Ferron, G.A.; Schmid, O. A dose-controlled system for air-liquid interface cell exposure and application to zinc oxide nanoparticles. Part. Fibre Toxicol 2009, 6, 32. [Google Scholar]
- De Bruijne, K.; Ebersviller, S.; Sexton, K.G.; Lake, S.; Leith, D.; Goodman, R.; Jetters, J.; Walters, G.W.; Doyle-Eisele, M.; Woodside, R.; et al. Design and testing of Electrostatic Aerosol in Vitro Exposure System (EAVES): An alternative exposure system for particles. Inhal. Toxicol 2009, 21, 91–101. [Google Scholar]
- Volckens, J.; Dailey, L.; Walters, G.; Devlin, R.B. Direct particle-to-cell deposition of coarse ambient particulate matter increases the production of inflammatory mediators from cultured human airway epithelial cells. Environ. Sci. Technol 2009, 43, 4595–4599. [Google Scholar]
- Aufderheide, M.; Mohr, U. CULTEX–An alternative technique for cultivation and exposure of cells of the respiratory tract to airborne pollutants at the air/liquid interface. Exp. Toxicol. Pathol 2000, 52, 265–270. [Google Scholar]
- Ritter, D.; Knebel, J.W.; Aufderheide, M. Exposure of human lung cells to inhalable substances: A novel test strategy involving clean air exposure periods using whole diluted cigarette mainstream smoke. Inhal. Toxicol 2003, 15, 67–84. [Google Scholar]
- Aufderheide, M.; Mohr, U. A modified CULTEX system for the direct exposure of bacteria to inhalable substances. Exp. Toxicol. Pathol 2004, 55, 451–454. [Google Scholar]
- Gasser, M.; Riediker, M.; Mueller, L.; Perrenoud, A.; Blank, F.; Gehr, P.; Rothen-Rutishauser, B. Toxic effects of brake wear particles on epithelial lung cells in vitro. Part. Fibre Toxicol 2009, 6, 30. [Google Scholar]
- Paur, H.R.; Mülhopt, S.; Weiss, C.; Diabaté, S. In Vitro exposure systems and bioassays for the assessment of toxicity of nanoparticles to the human lung. J. Verbr. Lebensm 2008, 3, 3. [Google Scholar]
- Rothen-Rutishauser, B.; Grass, R.N.; Blank, F.; Limbach, L.K.; Muhlfeld, C.; Brandenberger, C.; Raemy, D.O.; Gehr, P.; Stark, W.J. Direct combination of nanoparticle fabrication and exposure to lung cell cultures in a closed setup as a method to simulate accidental nanoparticle exposure of humans. Environ. Sci. Technol 2009, 43, 2634–2640. [Google Scholar]
- Blank, F.; Rothen-Rutishauser, B.M.; Schurch, S.; Gehr, P. An optimized in vitro model of the respiratory tract wall to study particle cell interactions. J. Aerosol. Med 2006, 19, 392–405. [Google Scholar]
- Fröhlich, E.; Bonstingl, G.; Hofler, A.; Meindl, C.; Leitinger, G.; Pieber, T.R.; Roblegg, E. Comparison of two in vitro systems to assess cellular effects of nanoparticles-containing aerosols. Toxicol. in Vitro 2013, 27, 409–417. [Google Scholar]
- Aufderheide, M.; Scheffler, S.; Mohle, N.; Halter, B.; Hochrainer, D. Analytical in vitro approach for studying cyto-and genotoxic effects of particulate airborne material. Anal. Bioanal. Chem 2011, 401, 3213–3220. [Google Scholar]
- Byron, P.R.; Patton, J.S. Drug delivery via the respiratory tract. J. Aerosol. Med 1994, 7, 49–75. [Google Scholar]
- Meiring, J.J.; Borm, P.J.; Bagate, K.; Semmler, M.; Seitz, J.; Takenaka, S.; Kreyling, W.G. The influence of hydrogen peroxide and histamine on lung permeability and translocation of iridium nanoparticles in the isolated perfused rat lung. Part. Fibre Toxicol 2005, 2, 3. [Google Scholar]
- Oberdorster, G.; Sharp, Z.; Atudorei, V.; Elder, A.; Gelein, R.; Lunts, A.; Kreyling, W.; Cox, C. Extrapulmonary translocation of ultrafine carbon particles following whole-body inhalation exposure of rats. J. Toxicol. Environ. Health A 2002, 65, 1531–1543. [Google Scholar]
- Ressmeyer, A.R.; Larsson, A.K.; Vollmer, E.; Dahlen, S.E.; Uhlig, S.; Martin, C. Characterisation of guinea pig precision-cut lung slices: Comparison with human tissues. Eur. Respir. J 2006, 28, 603–611. [Google Scholar]
- Nassimi, M.; Schleh, C.; Lauenstein, H.D.; Hussein, R.; Lubbers, K.; Pohlmann, G.; Switalla, S.; Sewald, K.; Muller, M.; Krug, N.; et al. Low cytotoxicity of solid lipid nanoparticles in in vitro and ex vivo lung models. Inhal. Toxicol 2009, 21, 104–109. [Google Scholar]
- Horev-Azaria, L.; Baldi, G.; Beno, D.; Bonacchi, D.; Golla-Schindler, U.; Kirkpatrick, J.C.; Kolle, S.; Landsiedel, R.; Maimon, O.; Marche, P.N.; et al. Predictive toxicology of cobalt ferrite nanoparticles: Comparative in vitro study of different cellular models using methods of knowledge discovery from data. Part. Fibre Toxicol 2013, 10, 32. [Google Scholar]
- Choi, J.Y.; Ramachandran, G.; Kandlikar, M. The impact of toxicity testing costs on nanomaterial regulation. Environ. Sci. Technol 2009, 43, 3030–3034. [Google Scholar]
- Jud, C.; Clift, M.J.; Petri-Fink, A.; Rothen-Rutishauser, B. Nanomaterials and the human lung: What is known and what must be deciphered to realise their potential advantages? Swiss Med. Wkly. 2013, 143, w13758. [Google Scholar]
- Wheeler, C.W.; Park, S.S.; Guenthner, T.M. Immunochemical analysis of a cytochrome P-450IA1 homologue in human lung microsomes. Mol. Pharmacol 1990, 38, 634–643. [Google Scholar]
- Geiser, M.; Kreyling, W.G. Deposition and biokinetics of inhaled nanoparticles. Part. Fibre Toxicol 2010, 7, 2. [Google Scholar]
- Kreyling, W.G.; Semmler-Behnke, M.; Seitz, J.; Scymczak, W.; Wenk, A.; Mayer, P.; Takenaka, S.; Oberdorster, G. Size dependence of the translocation of inhaled iridium and carbon nanoparticle aggregates from the lung of rats to the blood and secondary target organs. Inhal. Toxicol 2009, 21, 55–60. [Google Scholar]
- Semmler-Behnke, M.; Takenaka, S.; Fertsch, S.; Wenk, A.; Seitz, J.; Mayer, P.; Oberdorster, G.; Kreyling, W.G. Efficient elimination of inhaled nanoparticles from the alveolar region: Evidence for interstitial uptake and subsequent reentrainment onto airways epithelium. Environ. Health Perspect 2007, 115, 728–733. [Google Scholar]
- Geiser, M.; Cruz-Orive, L.M.; Im Hof, V.; Gehr, P. Assessment of particle retention and clearance in the intrapulmonary conducting airways of hamster lungs with the fractionator. J. Microsci 1990, 160, 75–88. [Google Scholar]
- Geiser, M.; Baumann, M.; Cruz-Orive, L.M.; Im Hof, V.; Waber, U.; Gehr, P. The effect of particle inhalation on macrophage number and phagocytic activity in the intrapulmonary conducting airways of hamsters. Am. J. Respir. Cell Mol. Biol 1994, 10, 594–603. [Google Scholar]
- Phalen, R.F. Inhalation exposure of animals. Environ. Health Perspect 1976, 16, 17–24. [Google Scholar]
- Shvedova, A.A.; Kisin, E.; Murray, A.R.; Johnson, V.J.; Gorelik, O.; Arepalli, S.; Hubbs, A.F.; Mercer, R.R.; Keohavong, P.; Sussman, N.; et al. Inhalation vs aspiration of single-walled carbon nanotubes in C57BL/6 mice: Inflammation fibrosis oxidative stress and mutagenesis. Am. J. Physiol. Lung Cell Mol. Physiol 2008, 295, L552–L565. [Google Scholar]
- Osier, M.; Oberdorster, G. Intratracheal inhalation vs intratracheal instillation: Differences in particle effects. Fundam. Appl. Toxicol 1997, 40, 220–227. [Google Scholar]
- Warheit, D.B.; Brock, W.J.; Lee, K.P.; Webb, T.R.; Reed, K.L. Comparative pulmonary toxicity inhalation and instillation studies with different TiO2 particle formulations: Impact of surface treatments on particle toxicity. Toxicol. Sci 2005, 88, 514–524. [Google Scholar]
- Grassian, V.H.; O’Shaughnessy, P.T.; Adamcakova-Dodd, A.; Pettibone, J.M.; Thorne, P.S. Inhalation exposure study of titanium dioxide nanoparticles with a primary particle size of 2 to 5 nm. Environ. Health Perspect 2007, 115, 397–402. [Google Scholar]
- Yi, J.; Chen, B.T.; Schwegler-Berry, D.; Frazer, D.; Castranova, V.; McBride, C.; Knuckles, T.L.; Stapleton, P.A.; Minarchick, V.C.; Nurkiewicz, T.R. Whole-body nanoparticle aerosol inhalation exposures. J. Vis. Exp 2013, 75, e50263. [Google Scholar]
- Griffis, L.C.; Wolff, R.K.; Beethe, R.L.; Hobbs, C.H.; McClellan, R.O. Evaluation of a multitiered inhalation exposure chamber. Fundam. Appl. Toxicol 1981, 1, 8–12. [Google Scholar]
- Phalen, R. Methods in Inhalation Toxicology; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar]
- Thorne, P.S. Inhalation toxicology models of endotoxin-and bioaerosol-induced inflammation. Toxicology 2000, 152, 13–23. [Google Scholar]
- Farina, F.; Sancini, G.; Longhin, E.; Mantecca, P.; Camatini, M.; Palestini, P. Milan PM1 induces adverse effects on mice lungs and cardiovascular system. Biomed. Res. Int 2013, 2013, 583513. [Google Scholar]
- Miyata, R.; Bai, N.; Vincent, R.; Sin, D.D.; van Eeden, S.F. Statins reduce ambient particulate matter-induced lung inflammation by promoting the clearance of particulate matter <10 mum from lung tissues. Chest 2013, 143, 452–460. [Google Scholar]
- Tronde, A.; Baran, G.; Eirefelt, S.; Lennernas, H.; Bengtsson, U.H. Miniaturized nebulization catheters: A new approach for delivery of defined aerosol doses to the rat lung. J. Aerosol. Med 2002, 15, 283–296. [Google Scholar]
- Rao, G.V.; Tinkle, S.; Weissman, D.N.; Antonini, J.M.; Kashon, M.L.; Salmen, R.; Battelli, L.A.; Willard, P.A.; Hoover, M.D.; Hubbs, A.F. Efficacy of a technique for exposing the mouse lung to particles aspirated from the pharynx. J. Toxicol. Environ. Health A 2003, 66, 1441–1452. [Google Scholar]
- Renne, R.; Miller, R. Microscopic Anatomy of Toxicologically Important Regions of the Larynx of the Rat, Mouse, and Hamster; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- National Research Council (US) Committee on Animal Models for Testing Interventions Against Aerosolized Bioterrorism Agents, Selection or Development of an Animal Model; National Academies Press: Washington, DC, USA, 2006.
- Pass, D.; Freeth, G. The rat. Anzccart News 1993, 6, 1–4. [Google Scholar]
- Van Winkle, L.; Smiley-Jewel, S.; Pinkerton, K. Tracheobronchial airways. In Comprehensive Toxicology; Yost, G.E., Ed.; Springer: Oxford, UK, 2010. [Google Scholar]
- Wolff, R.; Dorato, M. Inhalation toxicology studies. In Comprehensive Toxicology; Yost, G.E., Ed.; Elsevier Ltd.: Oxford, UK, 2010. [Google Scholar]
- Hofmann, W.; Asgharian, B. Comparison of mucociliary clearance velocities in human and rat lungs for extrapolation modeling. Ann. Occup. Hyg 2002, 46, 323–325. [Google Scholar]
- Morgan, D. Part 3 Inhalation Toxicology; CRC Press LLC: Boca Raton, FL, USA, 1997. [Google Scholar]
- Irvin, C.G.; Bates, J.H. Measuring the lung function in the mouse: The challenge of size. Respir. Res 2003, 4, 4. [Google Scholar]
- Chapman, R.W. Canine models of asthma and COPD. Pulm. Pharmacol. Ther 2008, 21, 731–742. [Google Scholar]
- Kreyling, W.; Eder, K.; Erbe, F.; Ferron, G.; Haider, B.; Karg, E.; Ruprecht, L.; Schumann, G. Particle deposition in the canine respiratory tract. Ann. Occup. Hyg 1994, 38, 143–149. [Google Scholar]
- McClellan, R.O. Role of inhalation studies with animals in defining human health risks for vehicle and power plant emissions. Environ. Health Perspect 1983, 47, 283–292. [Google Scholar]
- Green, F.H.; Vallyathan, V.; Hahn, F.F. Comparative pathology of environmental lung disease: An overview. Toxicol. Pathol 2007, 35, 136–147. [Google Scholar]
- Nikula, K.J.; Vallyathan, V.; Green, F.H.; Hahn, F.F. Influence of exposure concentration or dose on the distribution of particulate material in rat and human lungs. Environ. Health Perspect 2001, 109, 311–318. [Google Scholar]
- Mauderly, J.L. Usefulness of animal models for predicting human responses to long-term inhalation of particles. Chest 1996, 109, 65S–68S. [Google Scholar]
- Moskal, A.; Sosnowski, T.; Gradon, L. Inhalation and Deposition of Nanoparticles. Fundamentals, Phenomenology and Practical Aspects; Springer: Dordrecht, The Netherland, 2010. [Google Scholar]
- Asgharian, B.; Price, O.T. Air flow distribution in the human lung and its influence on particle deposition. Inhal. Toxicol 2006, 18, 795–801. [Google Scholar]
- Hofmann, W. Modelling inhaled particle deposition in the human lung—A review. J. Aerosol. Sci 2011, 42, 693–724. [Google Scholar]
- Wang, C. A Brief History of Respiratory Depostion Modeling; RTI International: Research Triangle Park, NC, USA, 2011. [Google Scholar]
- United States Environmental Protection Agency, Air Quality Criteria for Particulate Matter, Volume II; Environmental Protection Agency: Washington, DC, USA, 2004.
- Mobley, C.; Hochhaus, G. Methods used to assess pulmonary deposition and absorption of drugs. Drug Discov. Today 2001, 6, 367–375. [Google Scholar]
- Asgharian, B.; Hofmann, W.; Bergmann, R. Particle deposition in a multiple-path model of the human lung. Aerosol Sci. Technol 2001, 34, 332–339. [Google Scholar]
- Broday, D.M.; Georgopoulous, G. Growth and deposition of hygroscopic particulate matter in the human lung. Aerosol Sci. Technol 2001, 34, 144–159. [Google Scholar]
- Hofmann, W.; Koblinger, L. Monte-Carlo modeling of aerosol deposition in human lungs 3 Comparison with experimental-data. J. Aerosol Med 1992, 23, 51–63. [Google Scholar]
- Koblinger, L.; Hofmann, W. Monte Carlo modeling of aerosol deposition in human lungs Part I: Simulation of particle transport in a stochastic lung structure. J. Aerosol. Sci 1990, 21, 661–674. [Google Scholar]
- Longest, P.W.; Holbrook, L.D. In silico models of aerosol delivery to the respiratory tract—Development and applications. Adv. Drug Deliv. Rev 2012, 64, 296–311. [Google Scholar]
- Longest, P.W.; Kleinstreuer, C.; Buchanan, J.R. Efficient computation of microparticle dynamics including wall effects. Comput. Fluids 2004, 33, 577–601. [Google Scholar]
- Mitsakou, C.; Helmis, C.; Housiadas, C. Eulerian modelling of lung deposition with sectional representation of aerosol dynamics. Aerosol. Sci. Technol 2005, 36, 75–94. [Google Scholar]
- Zhang, Z.; Kleinstreuer, C.; Kim, C.S. Comparison of analytical and CFD models with regard to micronparticle deposition in a human 16-generation tracheobronchial airway model. J. Aerosol. Sci 2009, 40, 16–28. [Google Scholar]
- Weibel, E. Morphometry of the Human Lung; Springer: Berlin, Germany, 1963. [Google Scholar]
- Yeh, H.C.; Schum, G.M. Models of the human lung airways and their application to inhaled particle deposition. Bull. Math. Biol 1980, 42, 461–480. [Google Scholar]
- Findeisen, W. Über das Absetzen kleiner in der Luft suspendierter Teilchen in der menschlichen Lunge bei der Atmung. Pflügers Arch. Gesamte Physiol. Menschen Tiere 1935, 236, 367–379. [Google Scholar]
- International commission on radiological protection Human respiratory tract model for radiological protection. Ann. ICRP 1994, 24, 1–482.
- National Council on Radiation Protection and Measurements, Deposition Retention and Dosimetry of Inhaled Radioactive Substances; The Council: Bethesda, MD, USA, 1997.
- Yeh, H.C.; Cuddihy, R.G.; Phalen, R.F.; Chang, I. Comparisons of calculated respiratory tract deposition of particles based on the proposed NCRP model and the new ICRP66 model. Aerosol. Sci. Technol 1996, 25, 134–140. [Google Scholar]
- Balashazy, I.; Hofmann, W.; Heistracher, T. Computation of local enhancement factors for the quantification of particle deposition patterns in airway bifurcations. J. Aerosol Sci 1999, 30, 185–203. [Google Scholar]
- Bell, K.A. Local Particle Deposition in Respiratory Airway Models; Wiley: New York, NY, USA, 1978. [Google Scholar]
- Gradon, L.; Orlicki, D. Deposition of inhaled aerosol particles in a generation of the tracheobronchial tree. J. Aerosol Sci 1990, 21, 3–19. [Google Scholar]
- Longest, P.W.; Xi, J. Effectiveness of direct Lagrangian tracking models for simulating nanoparticle deposition in the upper airways. Aerosol. Sci. Technol 2007, 41, 380–397. [Google Scholar]
- Zhang, Z.; Kleinstreuer, C. Airflow structures and nano-particle deposition in a human upper airway model. J. Comput. Phys 2004, 198, 178–210. [Google Scholar]
- Raabe, O.G.; Yeh, H.C.; Schum, G.M.; Phalen, R.F. Tracheobronchial Geometry: Human, Dog, Rat, Hamster; Lovelace Foundation: Albuquerque, NM, USA, 1976. [Google Scholar]
- Koblinger, L.; Hofmann, W. Analysis of human lung morphometric data for stochastic aerosol deposition calculations. Phys. Med. Biol 1985, 30, 541–556. [Google Scholar]
- Asgharian, B.; Price, O.T. Deposition of ultrafine (nano) particles in the human lung. Inhal. Toxicol 2007, 19, 1045–1054. [Google Scholar]
- Asgharian, B.; Price, O.T.; Hofmann, W. Prediction of particle deposition in the human lung using realistic models of lung ventilation. Aerosol. Sci. Technol 2006, 37, 1209–1221. [Google Scholar]
- Londahl, J.; Moller, W.; Pagels, J.H.; Kreyling, W.G.; Swietlicki, E.; Schmid, O. Measurement techniques for respiratory tract deposition of airborne nanoparticles: A critical review. J. Aerosol. Med. Pulm. Drug Deliv 2013. [Google Scholar] [CrossRef]
- Wu, S.; Salar-Behzadi, S.; Fröhlich, E. Role of in-silico modeling in drug development for inhalation treatment. J. Mol. Pharm. Org. Process 2013, 1, 106. [Google Scholar]
- Kondej, D.; Sosnowski, T.R. Alteration of biophysical activity of pulmonary surfactant by aluminosilicate nanoparticles. Inhal. Toxicol 2013, 25, 77–83. [Google Scholar]
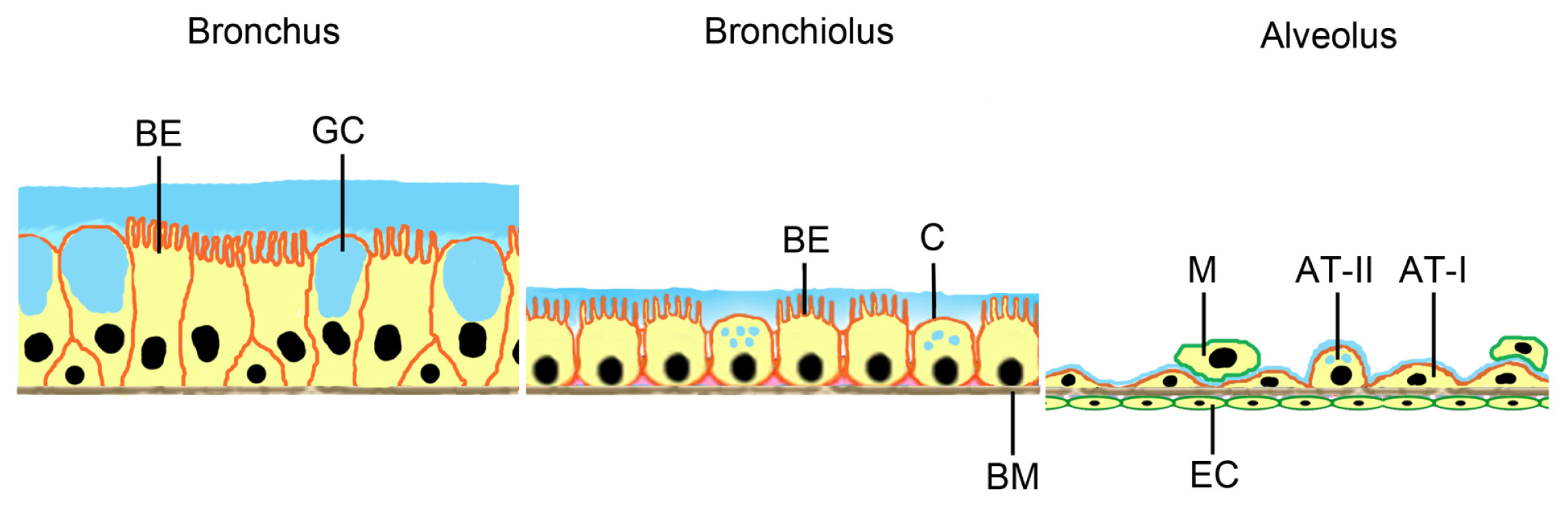
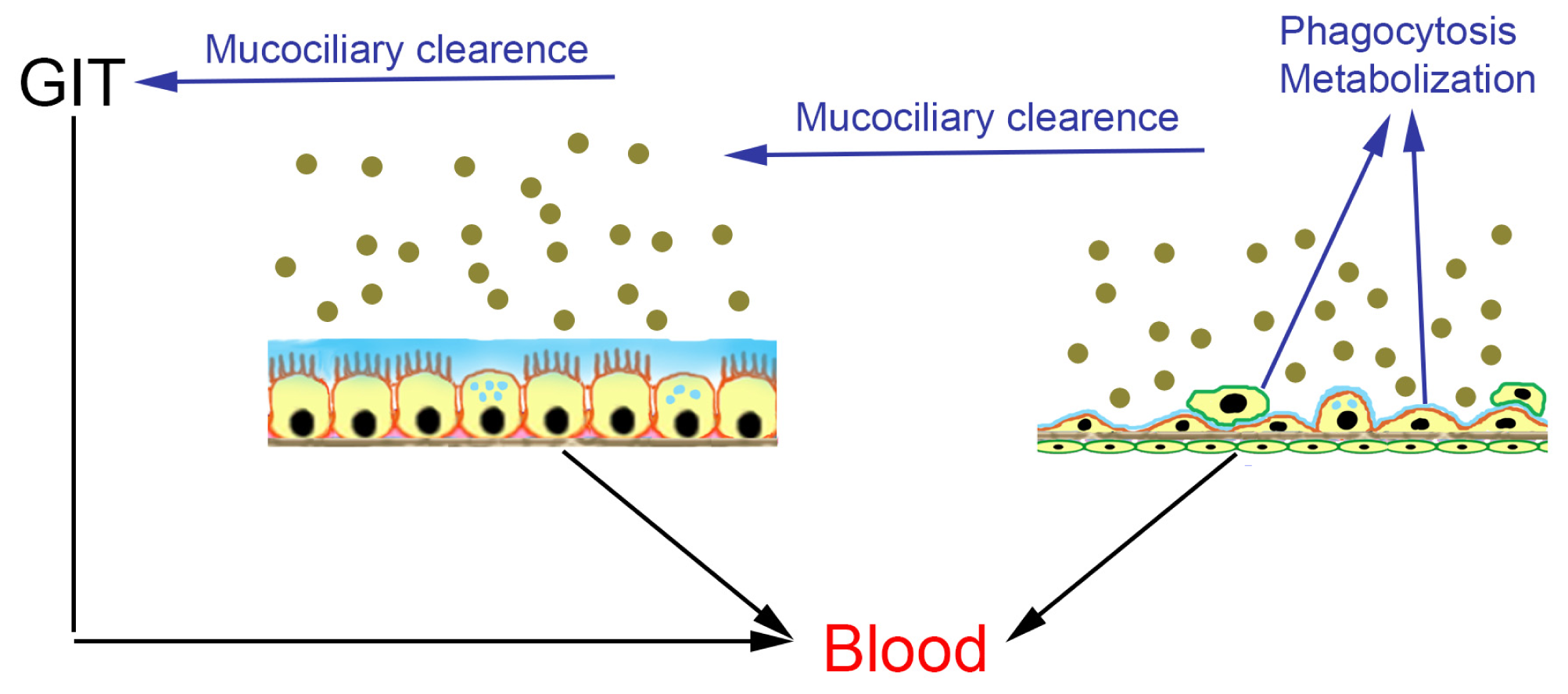
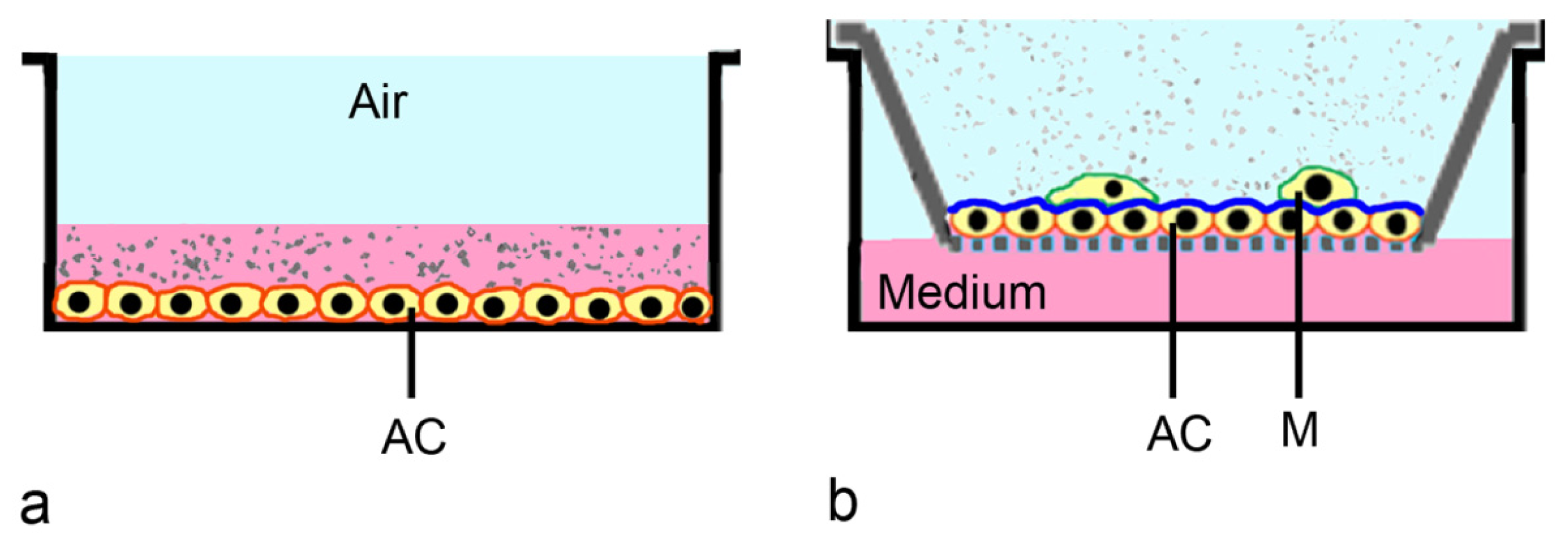
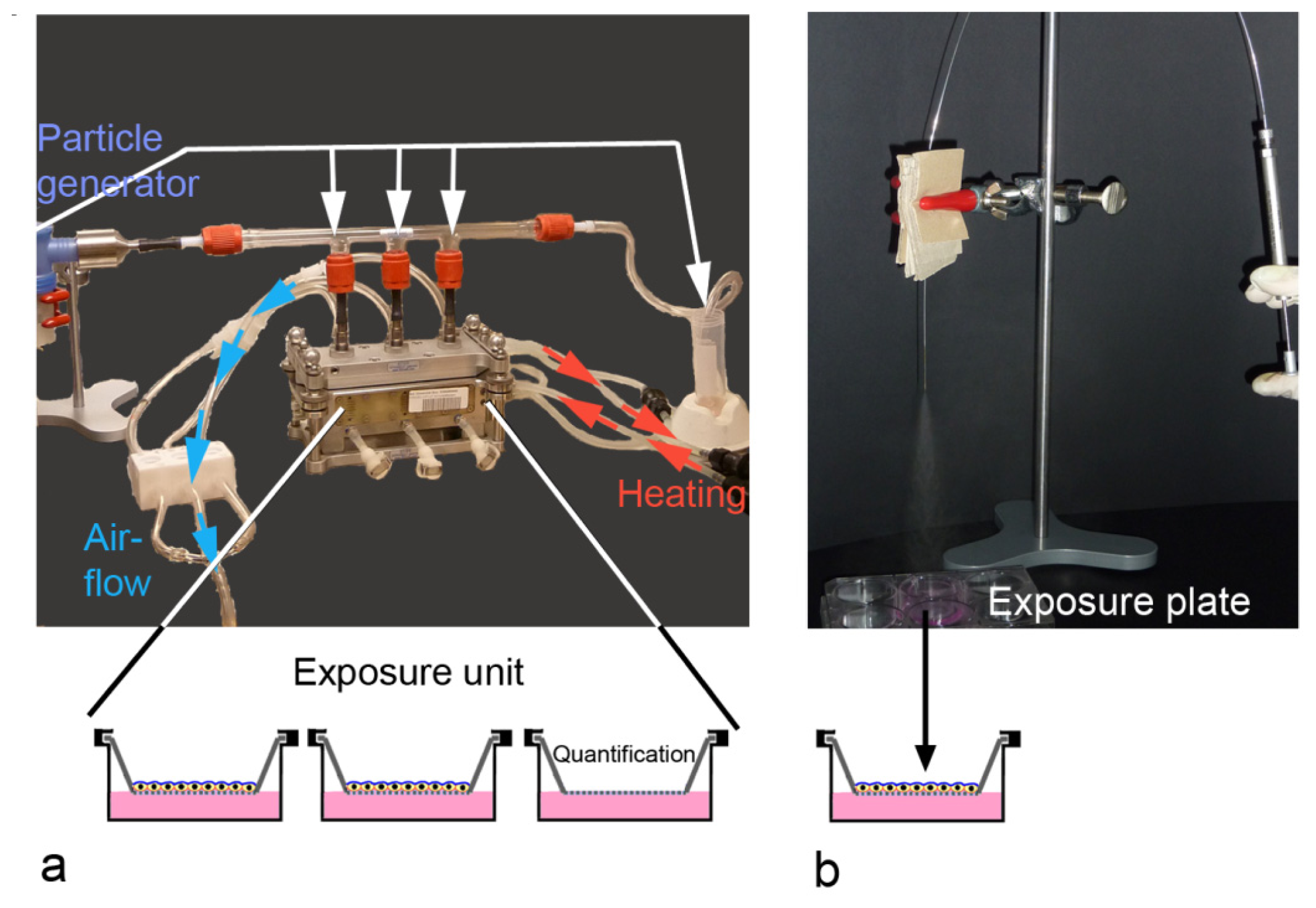

| Method | Application | Advantages | Limitations |
|---|---|---|---|
| In vitro techniques | |||
| Conventional exposure (submersed) | High-throughput testing | Controlled dosing | Exposure of non-differentiated cells |
| Initial screening for short-term effects | Easy to perform | Non-physiological exposure | |
| Efficient use of material | No information on permeation | ||
| No complex (multicellular) response | |||
| No long-term exposure | |||
| ALI (monoculture) + Suspension exposure | Mechanistic uptake and toxicity studies | Controlled dosing | Non-physiological exposure |
| Study of differentiated cells | No complex (multicellular) response | ||
| Efficient use of material | No long-term exposure | ||
| Advanced technology | |||
| ALI (monoculture) + Aerosol exposure chamber | Mechanistic uptake and toxicity studies | Relatively controlled dosing | No complex (multicellular) response |
| Permeation studies | Study of differentiated cells | No long-term exposure | |
| Efficient use of material | Complex exposure system | ||
| Aerosol loss in the exposure system | |||
| More complicated technology | |||
| ALI (mono/co-culture) + Aerosol spraying | Mechanistic uptake and toxicity studies | Controlled cellular dose | No long-term exposure |
| Permeation studies | Study of differentiated cells | Potential shear stress of the cells | |
| Efficient use of material | More complicated technology | ||
| ALI (co-culture) + Aerosol exposure chamber | Absorption studies | Controlled dosing | Technically demanding |
| Efficient use of material | No long-term exposure | ||
| Study on several cell types | Aerosol loss in the exposure system | ||
| Limited complex (multicellular) response | |||
| Ex-vivo techniques | |||
| Isolated perfused lung | Absorption studies | Relatively controlled dosing | Technically demanding |
| Complex (multicellular) response | Short observation time | ||
| Physiological exposure | |||
| Efficient use of material | |||
| Precision-cut lung slices | Toxicity studies | Controlled cellular dose | Non-physiological exposure |
| Complex (multicellular) response | Short observation time | ||
| Efficient use of material | |||
| In-vivo techniques | |||
| Whole-body exposure | ADME studies | Physiological way of exposure | Large amount of material needed |
| Short-term/long-term, single exposure and multiple exposure | No anesthesia or discomfort for animals | Dose not well defined | |
| Complex (multicellular) response | |||
| Nose/head only exposure | ADME studies | Relatively physiological way of exposure | Slight discomfort for animals |
| Short-term/long-term, single exposure and multiple exposure | Not invasive, no anesthesia | Inexact dose control | |
| Complex (multicellular) response | |||
| Intratracheal instillation | ADME studies | Direct dosing to lungs | Non-physiological exposure |
| Short-term, single dose exposure | Complex (multicellular) response | Anesthesia needed | |
| No repeated dosing | |||
| Tissue injury | |||
| Labor intensive | |||
| Oropharyngeal instillation | ADME studies | Direct dosing to lungs | Non-physiological exposure |
| Short-term, single dose exposure | Intubation not required | No repeated dosing | |
| Complex (multicellular) response | Labor intensive | ||
| Oropharyngeal aspiration | ADME studies | Direct dosing to lungs | Non-physiological exposure |
| Short-term, single dose exposure | No intubation required | Potential aspiration of oral content into lungs | |
| Complex (multicellular) response | No repeated dosing | ||
| Labor intensive | |||
| Species | Breath rate (resting, per minute) | Tidal volume (mL) | Total lung capacity (mL) |
|---|---|---|---|
| Rat | 85 | 1 | 10 |
| Mouse | 163 | 0.15 | 1 |
| Hamster | 30 | 1 | 7 |
| Guinea pig | 84 | 1.7 | 23 |
| Human | 15 | 500 | 6000 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fröhlich, E.; Salar-Behzadi, S. Toxicological Assessment of Inhaled Nanoparticles: Role of in Vivo, ex Vivo, in Vitro, and in Silico Studies. Int. J. Mol. Sci. 2014, 15, 4795-4822. https://doi.org/10.3390/ijms15034795
Fröhlich E, Salar-Behzadi S. Toxicological Assessment of Inhaled Nanoparticles: Role of in Vivo, ex Vivo, in Vitro, and in Silico Studies. International Journal of Molecular Sciences. 2014; 15(3):4795-4822. https://doi.org/10.3390/ijms15034795
Chicago/Turabian StyleFröhlich, Eleonore, and Sharareh Salar-Behzadi. 2014. "Toxicological Assessment of Inhaled Nanoparticles: Role of in Vivo, ex Vivo, in Vitro, and in Silico Studies" International Journal of Molecular Sciences 15, no. 3: 4795-4822. https://doi.org/10.3390/ijms15034795
APA StyleFröhlich, E., & Salar-Behzadi, S. (2014). Toxicological Assessment of Inhaled Nanoparticles: Role of in Vivo, ex Vivo, in Vitro, and in Silico Studies. International Journal of Molecular Sciences, 15(3), 4795-4822. https://doi.org/10.3390/ijms15034795






