Inhibition of Aflatoxin Synthesis in Aspergillus flavus by Three Structurally Modified Lentinans
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
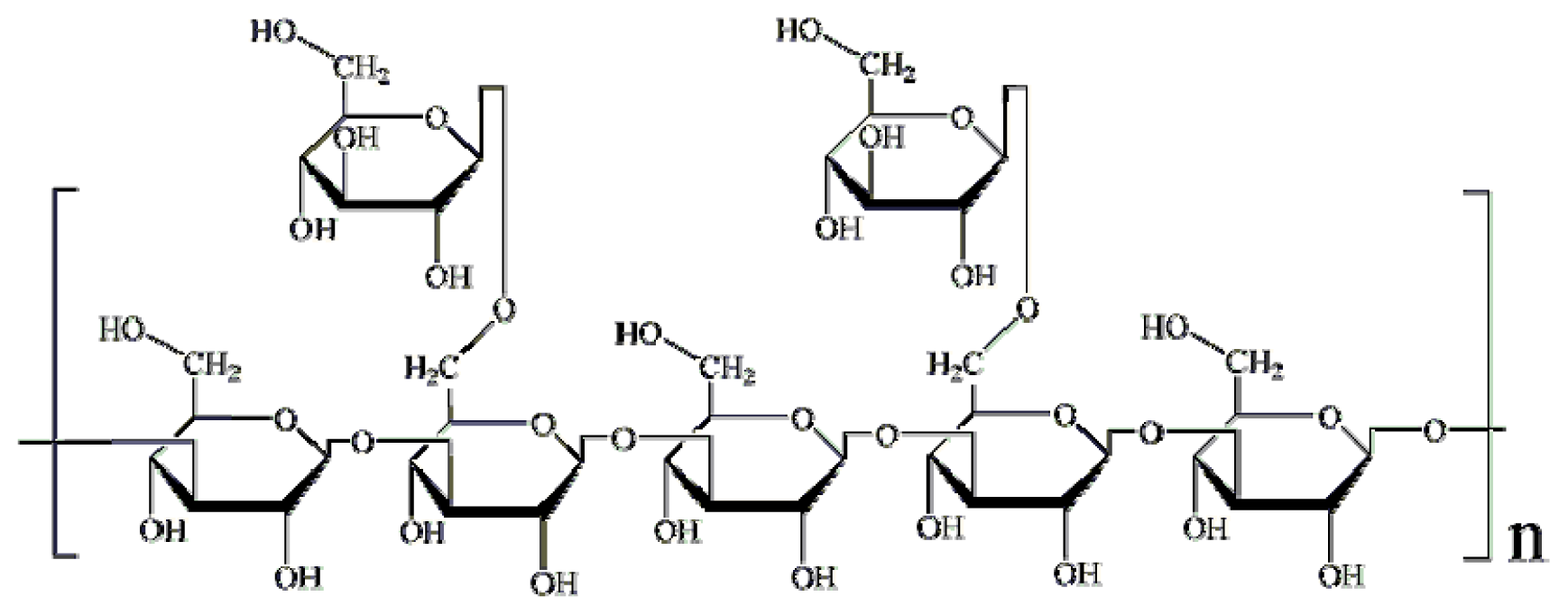
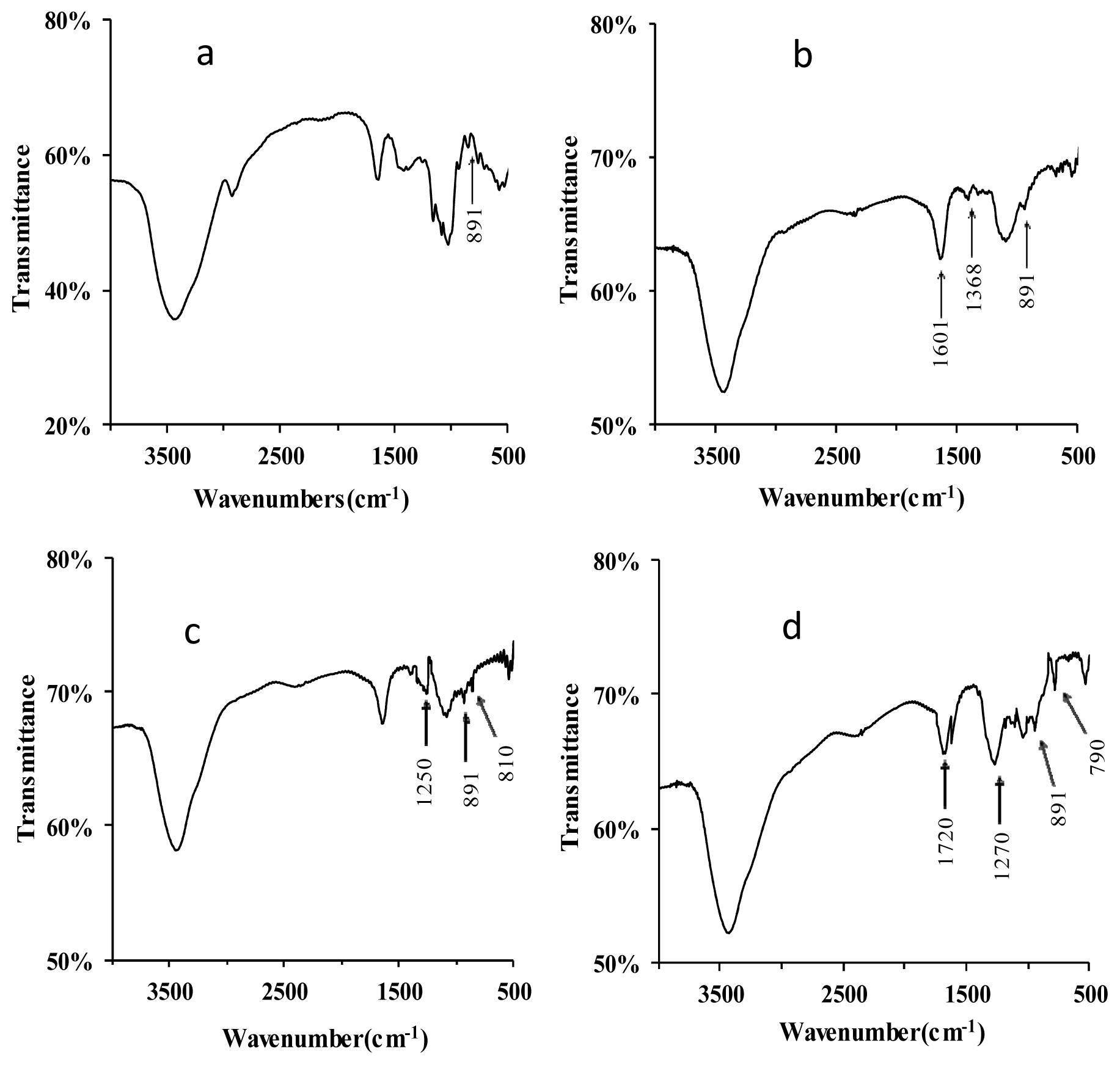
2.1.1. Structural Modification
2.1.2. Effects on Mycelia Growth and Aflatoxin Synthesis
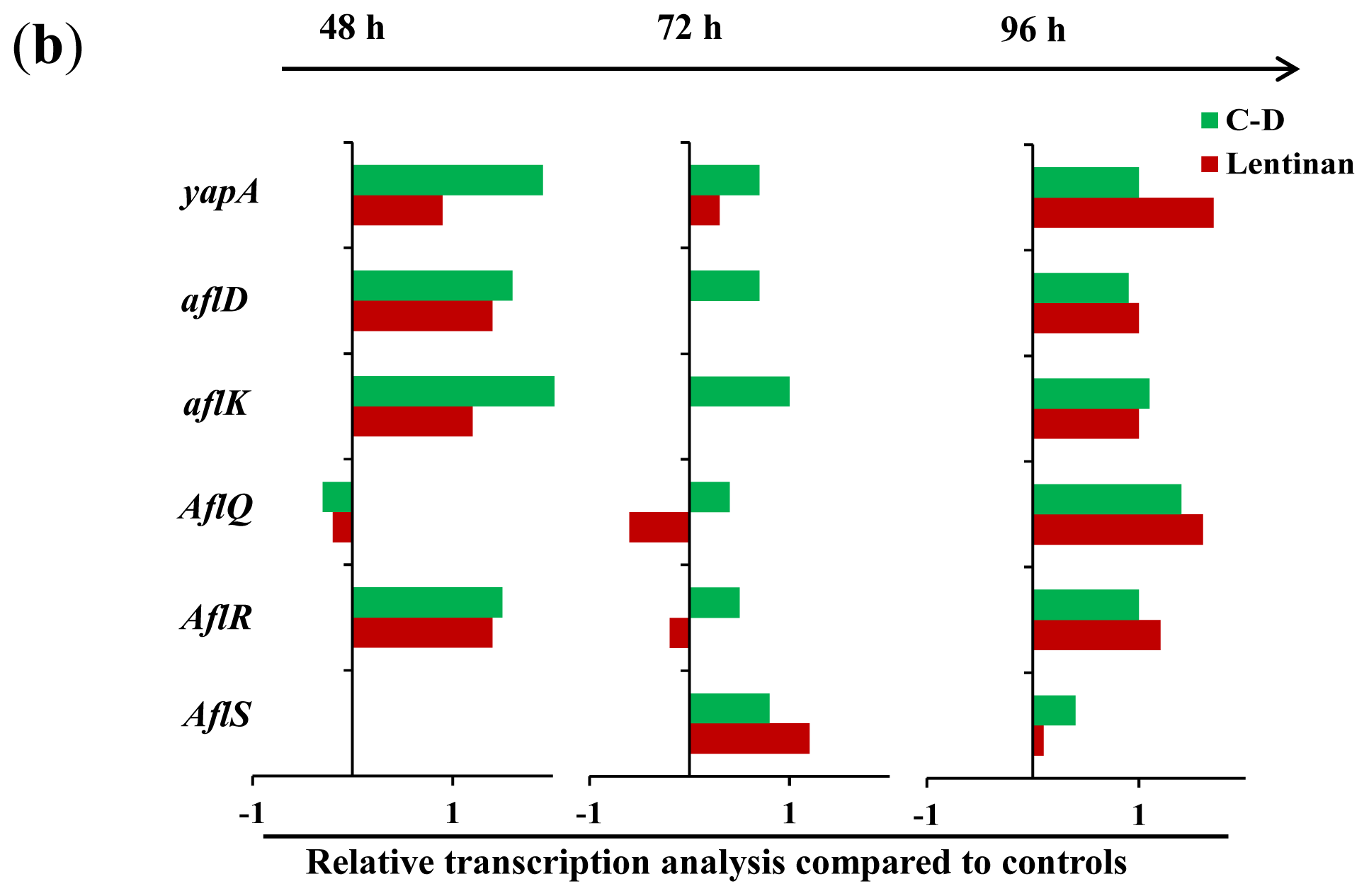
2.1.3. Gene Transcription Analysis
2.2. Discussion
3. Experimental Section
3.1. Culture of A. flavus
3.2. Aflatoxin Assay
3.3. Structural Modifications of Lentinan
3.3.1. Carboxymethylation
3.3.2. Sulfation
3.3.3. Phosphorylation
3.4. Fourier Transform Infrared (FT-IR) Spectra
3.5. Semi-Quantitative RT-PCR Analysis
3.6. Statistics
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Wagan, N.G. Aflatoxins as risk factors for hepatocellula carcinoma in huamans. Cancer Res. 1992, 52, 2114–2118. [Google Scholar]
- Yu, J.J.; Chang, P.K.; Ehrlich, K.C.; Cary, J.W.; Bhatnagar, D.; Cleveland, T.E.; Payne, G.A.; Linz, J.E.; Woloshuk, C.P.; Bennett, J.W. Clustered pathway genes in aflatoxin biosynthesis. Appl. Environ. Microbiol. 2004, 70, 1253–1262. [Google Scholar]
- Georgianna, D.R.; Payne, G.A. Genetic regulation of aflatoxin biosynthesis: From gene to genome. Fungal Genet. Biol 2009, 46, 113–125. [Google Scholar]
- Holmes, R.A.; Boston, R.S.; Payne, G.A. Diverse inhibitors of aflatoxin biosynthesis. Appl. Microbiol. Biotechnol 2008, 78, 559–572. [Google Scholar]
- Rasooli, I.; Fakoor, M.H.; Yadegarinia, D.; Gachkar, L.; Allameh, A.; Rezaei, M.B. Antimycotoxigenic characteristics of Rosmarinus officinalis and Trachyspermum copticum L essential oils. Int. J. Food Microbiol. 2008, 122, 135–139. [Google Scholar]
- Zhang, T.; Shi, Z.Q.; Hu, L.B.; Cheng, L.G.; Wang, F. Antifungal compounds from Bacillus subtilis B-FS06 inhibiting the growth of Aspergillus flavus. World J. Microbiol. Biotechnol. 2007, 24, 783–788. [Google Scholar]
- Reverberi, M.; Fabbri, A.A.; Zjalic, S.; Ricelli, A.; Punelli, F.; Fanelli, C. Antioxidant enzymes stimulation in Aspergillus parasiticus by Lentinus edodes inhibits aflatoxin production. Appl. Microbiol. Biotechnol. 2005, 69, 207–215. [Google Scholar]
- Yogeswari, R.; Murugesan, S.; Jagadeeswaran, A. Hepatoprotective effect of oyster mushroom (Pleurotus Sajor Caju) in broilers fed aflatoxin. Int. J. Vet. Sci. 2012, 1, 104–107. [Google Scholar]
- Akramiene, D.; Kondrotas, A.; Didziapetriene, J.; Kevelaitis, E. Effects of beta-glucans on the immune system. Medicina 2007, 43, 597–606. [Google Scholar]
- Li, G.; He, D.; Qian, Y.; Guan, B.; Gao, S.; Cui, Y.; Yokoyama, K.; Wang, L. Fungus-mediated green synthesis of silver nanoparticles using Aspergillus terreus. Int. J. Mol. Sci. 2011, 13, 466–476. [Google Scholar]
- Chen, X.; Zhang, L.; Cheung, P.C.K. Immunopotentiation and anti-tumor activity of carboxymethylated-sulfated β-(1→3)-d-glucan from Poria cocos. Int. Immunopharmacol. 2010, 10, 398–405. [Google Scholar]
- Chang, Y.J.; Lee, S.; Yoo, M.A.; Lee, H.G. Structural and biological characterization of sulfated-derivatized oat β-glucan. J. Agric. Food Chem. 2006, 54, 3815–3818. [Google Scholar]
- Huang, R.; Du, Y.; Yang, J.; Fan, L. Influence of functional groups on the in vitro anticoagulant activity of chitosan sulfate. Carbohydr. Res. 2003, 338, 483–489. [Google Scholar]
- Wei, D.; Cheng, W.; Wei, Y.; Zhang, L. Phosphorylated modification and in vitro antioxidant activity of Radix Hedysari polysaccharide. Glycoconj. J. 2012, 29, 167–172. [Google Scholar]
- Liu, J.; Sun, Y.; Yu, H.; Zhang, C.; Yue, L.; Yang, X.; Wang, L.; Liu, J. Purification and identification of one glucan from golden oyster mushroom (Pleurotus citrinopileatus (Fr) Singer). Carbohydr. Polym. 2012, 87, 348–352. [Google Scholar]
- Reverberi, M.; Zjalic, S.; Ricelli, A.; Punelli, F.; Camera, E.; Fabbri, C.; Picardo, M.; Fanelli, C.; Fabbri, A.A. Modulation of antioxidant defense in Aspergillus parasiticus is involved in aflatoxin biosynthesis: A role for the ApyapA gene. Eukaryot. Cell 2008, 7, 988–1000. [Google Scholar]
- Price, M.S.; Yu, J.; Nierman, W.C.; Kim, H.S.; Pritchard, B.; Jacobus, C.A.; Bhatnagar, D.; Cleveland, T.E.; Payne, G.A. The aflatoxin pathway regulator AflR induces gene transcription inside and outside of the aflatoxin biosynthetic cluster. FEMS Microbiol. Lett. 2006, 255, 275–279. [Google Scholar]
- Chang, P.K. The Aspergillus parasiticus protein AFLJ interacts with the aflatoxin pathway-specific regulator AFLR. Mol. Genet. Genomics 2003, 268, 711–719. [Google Scholar]
- Reverberi, M.; Zjalic, S.; Ricelli, A.; Fabbri, A.A.; Fanelli, C. Oxidant/antioxidant balance in Aspergillus parasiticus affects aflatoxin biosynthesis. Mycotoxin Res. 2006, 22, 39–47. [Google Scholar]
- Nelson, E.D.; Ramberg, J.E.; Best, T.; Sinnott, R.A. Neurologic effects of exogenous saccharides: A review of controlled human animal and in vitro studies. Nutr. Neurosci. 2012, 15, 149–162. [Google Scholar]
- Huang, X.Y.; Kong, X.F.; Wang, D.Y.; Hu, Y.L. Research progress on sulfating modification of polysaccharides and sulfated polysaccharides. Chin. Nat. Prod. Res. Dev. 2007, 4, 328–332. [Google Scholar]
- Suárez, E.R.; Kralovec, J.A.; Grindley, T.B. Isolation of phosphorylated polysaccharides from algae: The immunostimulatory principle of Chlorella pyrenoidosa. Carbohydr. Res. 2010, 345, 1190–1204. [Google Scholar]
- Yin, W.B.; Reinke, A.W.; Szilágyi, M.; Emri, T.; Chiang, Y.M.; Keating, A.E.; Pócsi, I.; Wang, C.C.C.; Keller, N.P. bZIP transcription factors affecting secondary metabolism sexual development and stress responses in Aspergillus nidulans. Microbiology 2013, 159, 77–88. [Google Scholar]
- Alberts, J.F.; Engelbrecht, Y.; Steyn, P.S.; Holzapfel, W.H.; van-Zyl, W.H. Biological degradation of aflatoxin B1 by Rhodococcus erythropolis cultures. Int. J. Food. Microbial. 2006, 109, 121–126. [Google Scholar]
- Somashekar, D.; Rati, E.R.; Chandrashekar, A. PCR-restriction fragment length analysis of aflR gene for differentiation and detection of Aspergillus flavus and Aspergillus parasiticusin maize. Int. J. Food Microbiol. 2004, 93, 101–107. [Google Scholar]


| Compounds | Mycelial weight (g) | |||||
|---|---|---|---|---|---|---|
| 0 (control) | 50 μg/mL | 100 μg/mL | 200 μg/mL | 400 μg/mL | 600 μg/mL | |
| lentinan | 0.176 ± 0.025 | 0.173 ± 0.013 | 0.145 ± 0.015 | 0.162 ± 0.030 | 0.187 ± 0.026 | 0.212 ± 0.024 |
| C-D | -- | 0.157 ± 0.022 | 0.197 ± 0.030 | 0.182 ± 0.019 | 0.208 ± 0.020 | 0.170 ± 0.006 |
| S-D | -- | 0.145 ± 0.022 | 0.152 ± 0.023 | 0.166 ± 0.018 | 0.202 ± 0.021 | 0.193 ± 0.005 |
| P-D | -- | 0.149 ± 0.026 | 0.176 ± 0.021 | 0.181 ± 0.021 | 0.190 ± 0.027 | 0.212 ± 0.087 |
| Gene | Primer sequence |
|---|---|
| yap | Forward 5′-TGCAACCTCTCTACAAGCCG-3′ Reverse 5′-CCGAAGTCTCGAGAAAGAGCC-3′ |
| aflK | Forward 5′-GAACTGCTTCAGTTGCCGTG-3′ Reverse 5′-ACGAGGGTTCGTTTCTGGAC-3′ |
| aflD | Forward 5′-TCCAGGCACACATGATGGTC-3′ Reverse 5′-TGTGGATAACGAAGTGCCCC-3′ |
| aflQ | Forward 5′-TTAAGGCAGCGGAATACAAG-3′ Reverse 5′-GACGCCCAAAGCCGAACACAAA-3′ |
| aflR | Forward 5′-GCACCCTGTCTTCCCTAACA-3′ Reverse 5′-ACGACCATGCTCAGCAAGTA-3′ |
| aflS | Forward 5′-GGAATGGGATGGAGATG-3′ Reverse 5′-GGAATATGGCTGTAGGAAG-3′ |
| 18S rDNA | Forward 5′-ATGGCCGTTCTTAGTTGGTG-3′ Reverse 5′-GTACAAAGGGCAGGGACGTA-3′ |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, J.; Mo, H.; Chen, Y.; Ding, D.; Hu, L. Inhibition of Aflatoxin Synthesis in Aspergillus flavus by Three Structurally Modified Lentinans. Int. J. Mol. Sci. 2014, 15, 3860-3870. https://doi.org/10.3390/ijms15033860
Ma J, Mo H, Chen Y, Ding D, Hu L. Inhibition of Aflatoxin Synthesis in Aspergillus flavus by Three Structurally Modified Lentinans. International Journal of Molecular Sciences. 2014; 15(3):3860-3870. https://doi.org/10.3390/ijms15033860
Chicago/Turabian StyleMa, Jinyou, Haizhen Mo, Ying Chen, Ding Ding, and Liangbin Hu. 2014. "Inhibition of Aflatoxin Synthesis in Aspergillus flavus by Three Structurally Modified Lentinans" International Journal of Molecular Sciences 15, no. 3: 3860-3870. https://doi.org/10.3390/ijms15033860




