Trans-Cinnamic Acid Increases Adiponectin and the Phosphorylation of AMP-Activated Protein Kinase through G-Protein-Coupled Receptor Signaling in 3T3-L1 Adipocytes
Abstract
:1. Introduction
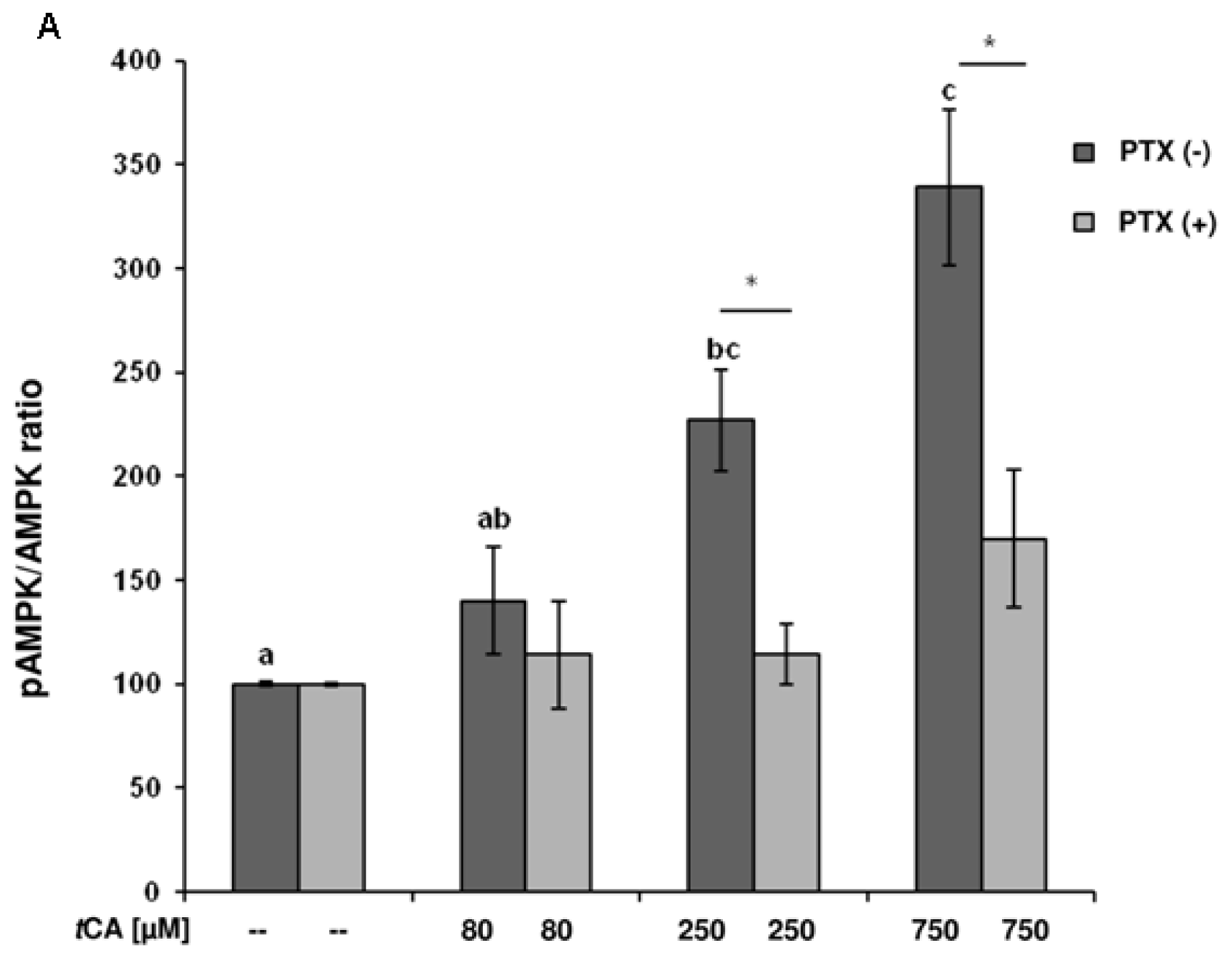
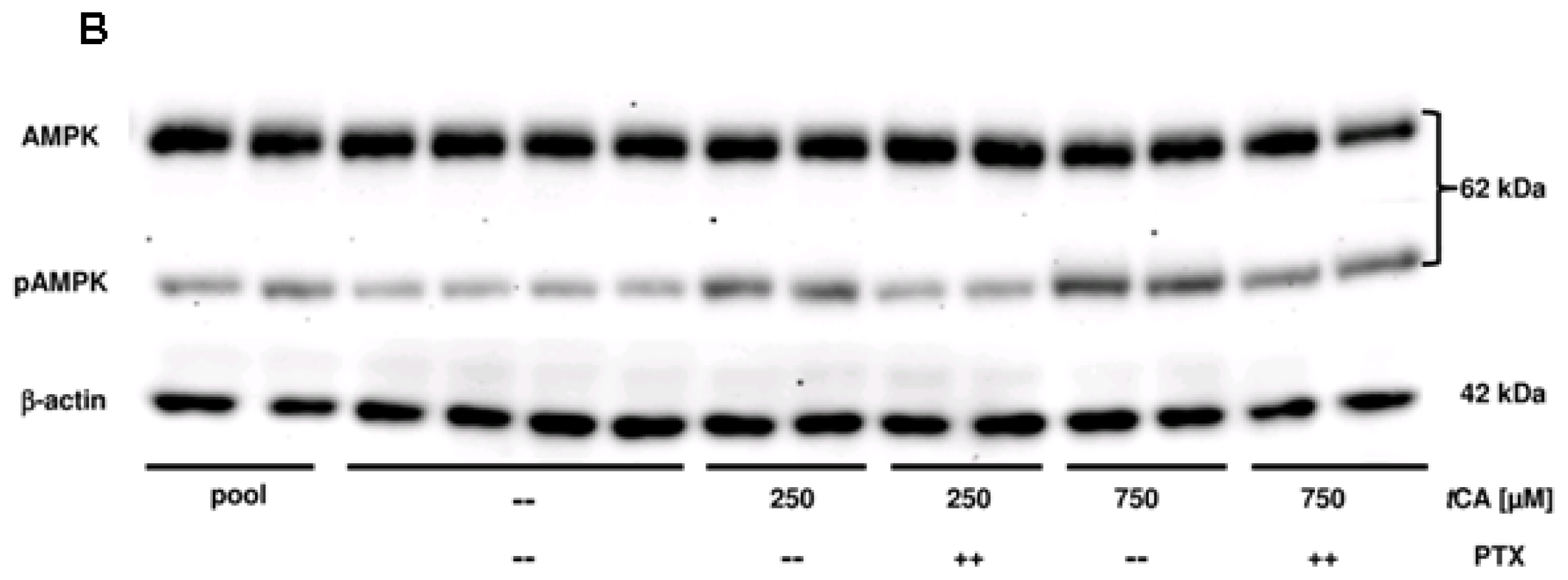
2. Results
3. Discussion
4. Experimental Section
4.1. Cell Culture
4.2. Treatment of Cells
4.3. Western Blot
4.4. Measurement of AdipoQ Secreted from 3T3-L1 Adipocytes
4.5. Statistical Analyses
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jarvill-Taylor, K.J.; Anderson, R.A.; Graves, D.J. A Hydroxychalcone derived from cinnamon functions as a mimetic for insulin in 3T3-L1 adipocytes. J. Am. Coll. Nutr 2001, 20, 27–36. [Google Scholar]
- Khan, A.; Safdar, M.; Khan, M.; Khattak, K.N.; Anderson, R.A. Cinnamon improves glucose and lipids of people with type 2 diabetes. Diabetes Care 2003, 26, 3215–3218. [Google Scholar]
- Kim, S.H.; Choung, S.Y. Antihyperglycemic and antihyperlipidemic action of Cinnamomi cassiae (cinnamon bark) extract in C57BL/Ks db/db mice. Arch. Pharm. Res 2010, 33, 325–333. [Google Scholar]
- Scherer, P.E.; Williams, S.; Fogliano, M.; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q exclusively in adipocytes. J. Biol. Chem 1995, 270, 26746–26749. [Google Scholar]
- Kadowaki, T.; Yamauchi, T. Adiponectin and adiponectin receptors. Endocr Rev 2005, 26, 439–451. [Google Scholar]
- Thornton, C.; Snowden, M.A.; Carling, D. Identification of a novel AMP-activated protein kinase b subunit isoform that is highly expressed in skeletal muscle. J. Biol. Chem 1998, 273, 12443–12450. [Google Scholar]
- Hardie, D.G.; Sakamoto, K. AMPK: A key sensor of fuel and energy status in skeletal muscle. Physiology 2006, 21, 48–60. [Google Scholar]
- Daval, M.; Foufelle, F.; Ferré, P. Functions of AMP-activated protein kinase in adipose tissue. J. Physiol 2006, 574, 55–62. [Google Scholar]
- Salt, I.P.; Connell, J.M.; Gould, G.W. 5-aminoimidazole-4-carboxamide ribonucleoside (AICAR) inhibits insulin-stimulated glucose transport in 3T3-L1 adipocytes. Diabetes 2000, 49, 1649–1656. [Google Scholar]
- Huang, B.; Yuan, H.D.; Kim, D.Y.; Quan, H.Y.; Chung, S.H. Cinnamaldehyde prevents adipocyte differentiation and adipogenesis via regulation of peroxisome proliferator-activated receptor-γ (PPARγ) and AMP-activated protein kinase (AMPK) pathways. J. Agric. Food Chem 2011, 59, 3666–3673. [Google Scholar]
- Bijland, S.; Mancini, S.J.; Salt, I.P. Role of AMP-activated protein kinase in adipose tissue metabolism and inflammation. Clin. Sci 2013, 124, 491–507. [Google Scholar]
- Ren, N.; Kaplan, R.; Hernandez, M.; Cheng, K.; Jin, L.; Taggart, A.K.; Zhu, A.Y.; Gan, X.; Wright, S.D.; Cai, T.Q. Phenolic acids suppress adipocyte lipolysis via activation of the nicotinic acid receptor GPR109A (HM74a/PUMA-G). J. Lipid Res 2009, 50, 908–914. [Google Scholar]
- Offermanns, S.; Colletti, S.L.; Lovenberg, T.W.; Semple, G.; Wise, A.; Ijzerman, A.P. International union of basic and clinical pharmacology. LXXXII: Nomenclature and classification of hydroxy-carboxylic acid receptors (GPR81, GPR109A, and GPR109B). Pharmacol. Rev 2011, 63, 269–290. [Google Scholar]
- Taggart, A.K.; Kero, J.; Gan, X.; Cai, T.-Q.; Cheng, K.; Ippolito, M.; Ren, N.; Kaplan, R.; Wu, K.; Wu, T.-J.; et al. D-β-hydroxybutyrate inhibits adipocyte lipolysis via the nicotinic acid receptor PUMA-G. J. Biol. Chem 2005, 280, 26649–26652. [Google Scholar]
- Plaisance, E.P.; Lukasova, M.; Offermanns, S.; Zhang, Y.; Cao, G.; Judd, R.L. Niacin stimulates adiponectin secretion through the GPR109A receptor. Am. J. Physiol. Endocrinol. Metab 2009, 296, 549–558. [Google Scholar]
- Imparl-Radosevich, J.; Deas, S.; Polansky, M.M.; Baedke, D.A.; Ingebrutsen, T.S.; Anderson, R.A.; Graves, D.J. Regulation of phosphotyrosine phosphatase (PTP-1) and insulin receptor kinase by fractions from cinnamon: implications for cinnamon regulation of insulin signaling. Horm. Res 1998, 50, 177–182. [Google Scholar]
- Ohara, K.; Uchida, A.; Nagasaka, R.; Ushio, H.; Ohshima, T. The effects of hydroxycinnamic acid derivatives on adiponectin secretion. Phytomedicine 2009, 16, 130–137. [Google Scholar]
- Lafontan, M.; Viguerie, N. Role of adipokines in the control of energy metabolism: Focus on adiponectin. Curr. Opin. Pharmacol 2006, 6, 580–585. [Google Scholar]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med 2002, 8, 1288–1295. [Google Scholar]
- Westphal, S.; Borucki, K.; Taneva, E.; Makarova, R.; Luley, C. Extended-release niacin raises adiponectin and leptin. Atherosclerosis 2007, 193, 361–365. [Google Scholar]
- Iwaki, M.; Matsuda, M.; Maeda, N.; Funahashi, T.; Matsuzawa, Y.; Makishima, M.; Shimomura, I. Induction of adiponectin, a fat-derived antidiabetic and antiatherogenic factor, by nuclear receptors. Diabetes 2003, 52, 1655–1663. [Google Scholar]
- Chiarelli, F.; Di, M.D. Peroxisome proliferator-activated receptor-gamma agonists and diabetes: current evidence and future perspectives. Vasc. Health Risk Manag 2008, 4, 297–304. [Google Scholar]
- Zhang, Y.; Schmidt, R.J.; Foxworthy, P.; Emkey, R.; Oler, J.K.; Large, T.H.; Wang, H.; Su, E.W.; Mosior, M.K.; Eacho, P.I.; et al. Niacin mediates lipolysis in adipose tissue through its G-protein coupled receptor HM74A. Biochem. Biophys. Res. Commun 2005, 334, 729–732. [Google Scholar]
- Jeninga, E.H.; Bugge, A.; Nielsen, R.; Kersten, S.; Hamers, N.; Dani, C.; Wabitsch, M.; Berger, R.; Stunnenberg, H.G.; Mandrup, S.; et al. Peroxisome proliferator-activated receptor regulates expression of the anti-lipolytic G-protein-coupled receptor 81 (GPR81/Gpr81). J. Biol. Chem 2009, 25, 26385–26393. [Google Scholar]
- Digby, J.E.; McNeill, E.; Dyar, O.J.; Lam, V.; Greaves, D.R.; Choudhury, R.P. Anti-inflammatory effects of nicotinic acid in adipocytes demonstrated by suppression of fractalkine, RANTES, and MCP-1 and upregulation of adiponectin. Atherosclerosis 2010, 209, 89–95. [Google Scholar]
- Ge, H.; Li, X.; Weiszmann, J.; Wang, P.; Baribault, H.; Chen, J.; Tian, H.; Li, Y. Activation of G protein-coupled receptor 43 in adipocytes leads to inhibition of lipolysis and suppression of plasma free fatty acids. Endocrinology 2008, 149, 4519–4526. [Google Scholar]
- Locher, L.F.; Meyer, N.; Weber, E.-M.; Rehage, J.; Meyer, U.; Dänicke, S.; Huber, K. Hormone-sensitive lipase protein expression and extent of phosphorylation in subcutaneous and retroperitoneal adipose tissues in the periparturient dairy cow. J. Dairy Sci 2011, 94, 4514–4523. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Mielenz, M.; Mielenz, B.; Singh, S.P.; Kopp, C.; Heinz, J.; Häussler, S.; Sauerwein, H. Development, validation, and pilot application of a semiquantitative Western blot analysis and an ELISA for bovine adiponectin. Domest. Anim. Endocrinol 2013, 44, 121–130. [Google Scholar]

© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kopp, C.; Singh, S.P.; Regenhard, P.; Müller, U.; Sauerwein, H.; Mielenz, M. Trans-Cinnamic Acid Increases Adiponectin and the Phosphorylation of AMP-Activated Protein Kinase through G-Protein-Coupled Receptor Signaling in 3T3-L1 Adipocytes. Int. J. Mol. Sci. 2014, 15, 2906-2915. https://doi.org/10.3390/ijms15022906
Kopp C, Singh SP, Regenhard P, Müller U, Sauerwein H, Mielenz M. Trans-Cinnamic Acid Increases Adiponectin and the Phosphorylation of AMP-Activated Protein Kinase through G-Protein-Coupled Receptor Signaling in 3T3-L1 Adipocytes. International Journal of Molecular Sciences. 2014; 15(2):2906-2915. https://doi.org/10.3390/ijms15022906
Chicago/Turabian StyleKopp, Christina, Shiva P. Singh, Petra Regenhard, Ute Müller, Helga Sauerwein, and Manfred Mielenz. 2014. "Trans-Cinnamic Acid Increases Adiponectin and the Phosphorylation of AMP-Activated Protein Kinase through G-Protein-Coupled Receptor Signaling in 3T3-L1 Adipocytes" International Journal of Molecular Sciences 15, no. 2: 2906-2915. https://doi.org/10.3390/ijms15022906




