Clinical Evaluation of Extracellular ADMA Concentrations in Human Blood and Adipose Tissue
Abstract
:1. Introduction
2. Results and Discussion
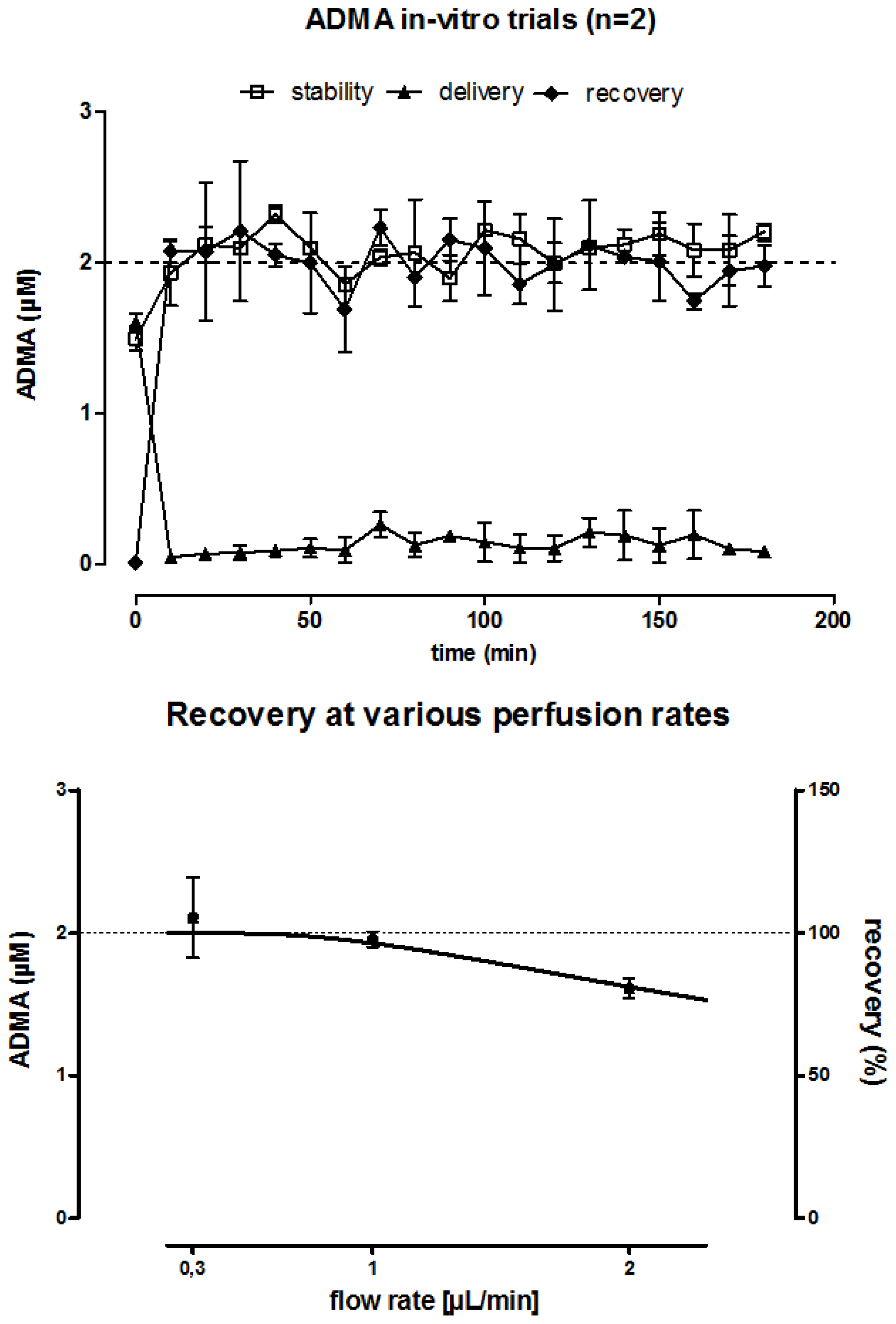
2.1. In Vitro Microdialysis Validation Experiments
2.2. Clinical Microdialysis Experiments
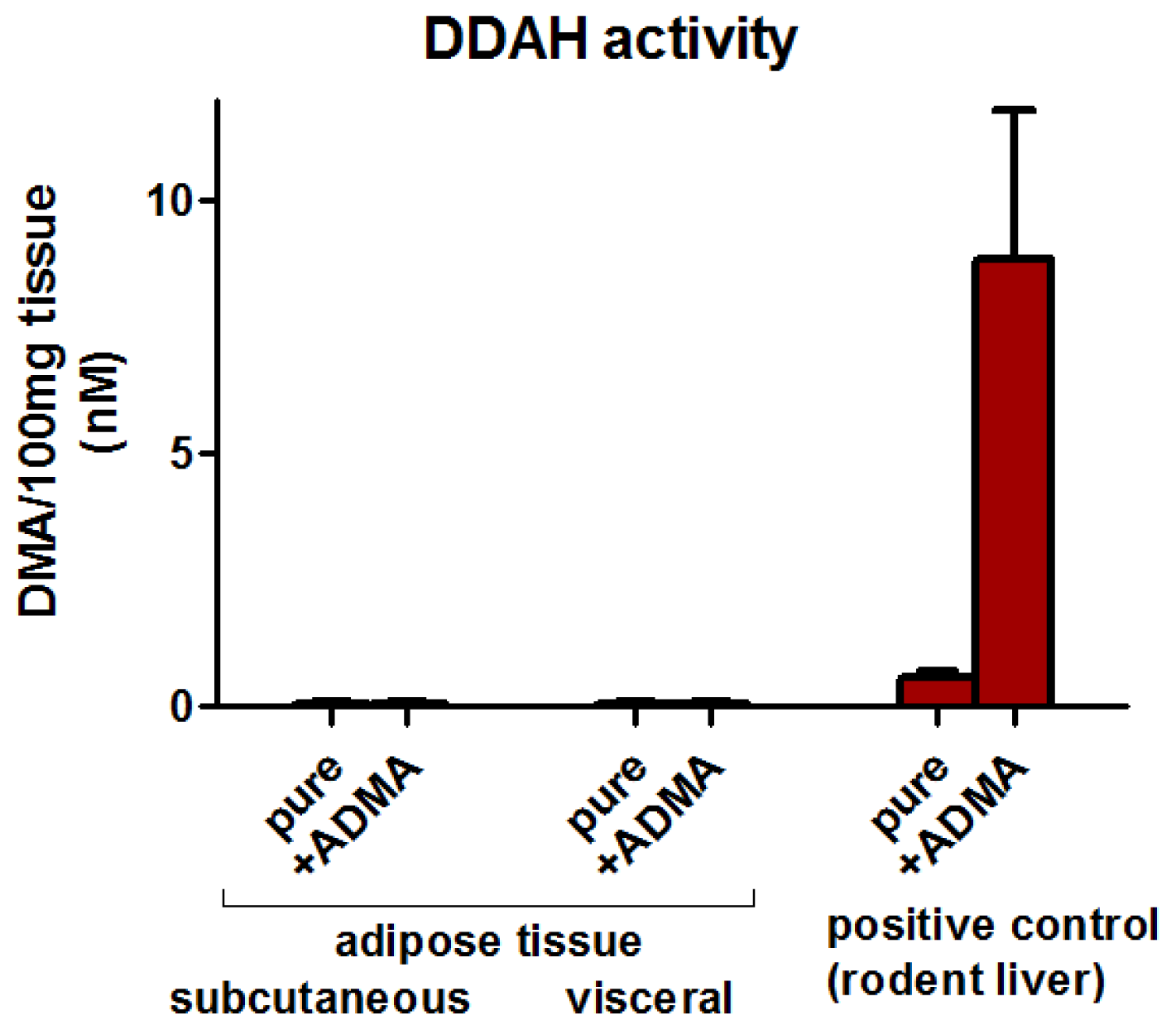
2.3. Adipose Tissue DDAH Activity
2.4. Discussion
3. Experimental Section
3.1. Study Subjects
3.2. Microdialysis
3.3. Adipose Tissue Collection
3.4. Analytical Procedures
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Ziccardi, P.; Nappo, F.; Giugliano, G.; Esposito, K.; Marfella, R.; Cioffi, M.; D’Andrea, F.; Molinari, A.M.; Giugliano, D. Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 2002, 105, 804–809. [Google Scholar]
- Hamdy, O.; Ledbury, S.; Mullooly, C.; Jarema, C.; Porter, S.; Ovalle, K.; Moussa, A.; Caselli, A.; Caballero, A.E.; Economides, P.A.; et al. Lifestyle modification improves endothelial function in obese subjects with the insulin resistance syndrome. Diabetes Care 2003, 26, 2119–2125. [Google Scholar]
- Pi-Sunyer, F.X. The obesity epidemic: Pathophysiology and consequences of obesity. Obes. Res 2002, 10, 97S–104S. [Google Scholar]
- Caballero, A.E. Endothelial dysfunction in obesity and insulin resistance: a road to diabetes and heart disease. Obes. Res 2003, 11, 1278–1289. [Google Scholar]
- Mohamed-Ali, V.; Pinkney, J.H.; Coppack, S.W. Adipose tissue as an endocrine and paracrine organ. Int. J. Obes. Relat. Metab. Disord 1998, 22, 1145–1158. [Google Scholar]
- Bergman, R.N.; Mittelman, S.D. Central role of the adipocyte in insulin resistance. J. Basic Clin. Physiol. Pharmacol 1998, 9, 205–221. [Google Scholar]
- Yudkin, J.S.; Eringa, E.; Stehouwer, C.D.A. “Vasocrine” signalling from perivascular fat: A mechanism linking insulin resistance to vascular disease. Lancet 2005, 365, 1817–1820. [Google Scholar]
- Stuhlinger, M.C.; Abbasi, F.; Chu, J.W.; Lamendola, C.; McLaughlin, T.L.; Cooke, J.P.; Reaven, G.M.; Tsao, P.S. Relationship between insulin resistance and an endogenous nitric oxide synthase inhibitor. JAMA 2002, 287, 1420–1426. [Google Scholar]
- Cooke, J.P. Does ADMA cause endothelial dysfunction? Arterioscler. Thromb. Vasc. Biol 2000, 20, 2032–2037. [Google Scholar]
- Konukoglu, D.; Firtina, S.; Serin, O. The relationship between plasma asymmetrical dimethyl-L-arginine and inflammation and adhesion molecule levels in subjects with normal, impaired, and diabetic glucose tolerance. Metabolism 2008, 57, 110–115. [Google Scholar]
- Tran, C.T.; Leiper, J.M.; Vallance, P. The DDAH/ADMA/NOS pathway. Atheroscler. Suppl 2003, 4, 33–40. [Google Scholar]
- Lin, K.Y.; Ito, A.; Asagami, T.; Tsao, P.S.; Adimoolam, S.; Kimoto, M.; Tsuji, H.; Reaven, G.M.; Cooke, J.P. Impaired nitric oxide synthase pathway in diabetes mellitus: Role of asymmetric dimethylarginine and dimethylarginine dimethylaminohydrolase. Circulation 2002, 106, 987–992. [Google Scholar]
- Spoto, B.; Parlongo, R.M.; Parlongo, G.; Sgro’, E.; Zoccali, C. The enzymatic machinery for ADMA synthesis and degradation is fully expressed in human adipocytes. J. Nephrol 2007, 20, 554–559. [Google Scholar]
- Ekberg, N.R.; Wisniewski, N.; Brismar, K.; Ungerstedt, U. Measurement of glucose and metabolites in subcutaneous adipose tissue during hyperglycemia with microdialysis at various perfusion flow rates. Clin. Chim. Acta 2005, 359, 53–64. [Google Scholar]
- Strindberg, L.; Lonnroth, P. Validation of an endogenous reference technique for the calibration of microdialysis catheters. Scand. J. Clin. Lab. Invest 2000, 60, 205–211. [Google Scholar]
- Reinstrup, P.; Stahl, N.; Mellergard, P.; Uski, T.; Ungerstedt, U.; Nordstrom, C.H. Intracerebral microdialysis in clinical practice: Baseline values for chemical markers during wakefulness, anesthesia, and neurosurgery. Neurosurgery 2000, 47, 701–709, discussion 709–710. [Google Scholar]
- Engeli, S.; Tsikas, D.; Lehmann, A.C.; Böhnke, J.; Haas, V.; Strauß, A.; Janke, J.; Gorzelniak, K.; Luft, F.C.; Jordan, J. Influence of dietary fat ingestion on asymmetrical dimethylarginine in lean and obese human subjects. Nutr. Metab. Cardiovasc. Dis 2012, 22, 720–726. [Google Scholar]
- Wasserman, D.H. Four grams of glucose. Am. J. Physiol. Endocrinol. Metab 2009, 296, E11–E21. [Google Scholar]
- Boger, R.H.; Vallance, P.; Cooke, J.P. Asymmetric dimethylarginine (ADMA): A key regulator of nitric oxide synthase. Atheroscler. Suppl 2003, 4, 1–3. [Google Scholar]
- Cardounel, A.J.; Cui, H.; Samouilov, A.; Johnson, W.; Kearns, P.; Tsai, A.L.; Berka, V.; Zweier, J.L. Evidence for the pathophysiological role of endogenous methylarginines in regulation of endothelial NO production and vascular function. J. Biol. Chem 2007, 282, 879–887. [Google Scholar]
- Vallance, P.; Leone, A.; Calver, A.; Collier, J.; Moncada, S. Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 1992, 339, 572–575. [Google Scholar]
- Boger, R.H. The emerging role of asymmetric dimethylarginine as a novel cardiovascular risk factor. Cardiovasc. Res 2003, 59, 824–833. [Google Scholar]
- Miyazaki, H.; Matsuoka, H.; Cooke, J.P.; Usui, M.; Ueda, S.; Okuda, S.; Imaizumi, T. Endogenous nitric oxide synthase inhibitor: A novel marker of atherosclerosis. Circulation 1999, 99, 1141–1146. [Google Scholar]
- Asagami, T.; Abbasi, F.; Stuelinger, M.; Lamendola, C.; McLaughlin, T.; Cooke, J.P.; Reaven, G.M.; Tsao, P.S. Metformin treatment lowers asymmetric dimethylarginine concentrations in patients with type 2 diabetes. Metabolism 2002, 51, 843–846. [Google Scholar]
- Eid, H.M.; Arnesen, H.; Hjerkinn, E.M.; Lyberg, T.; Seljeflot, I. Relationship between obesity, smoking, and the endogenous nitric oxide synthase inhibitor, asymmetric dimethylarginine. Metabolism 2004, 53, 1574–1579. [Google Scholar]
- Raitakari, M.; Ilvonen, T.; Ahotupa, M.; Lehtimaki, T.; Harmoinen, A.; Suominen, P.; Elo, J.; Hartiala, J.; Raitakari, O.T. Weight reduction with very-low-caloric diet and endothelial function in overweight adults: role of plasma glucose. Arterioscler. Thromb. Vasc. Biol 2004, 24, 124–128. [Google Scholar]
- Krzyzanowska, K.; Mittermayer, F.; Kopp, H.P.; Wolzt, M.; Schernthaner, G. Weight loss reduces circulating asymmetrical dimethylarginine concentrations in morbidly obese women. J. Clin. Endocrinol. Metab 2004, 89, 6277–6281. [Google Scholar]
- McLaughlin, T.; Stuhlinger, M.; Lamendola, C.; Abbasi, F.; Bialek, J.; Reaven, G.M.; Reaven, G.M.; Tsao, P.S. Plasma asymmetric dimethylarginine concentrations are elevated in obese insulin-resistant women and fall with weight loss. J. Clin. Endocrinol. Metab 2006, 91, 1896–1900. [Google Scholar]
- Anderson, T.J.; Uehata, A.; Gerhard, M.D.; Meredith, I.T.; Knab, S.; Delagrange, D.; Lieberman, E.H.; Ganz, P.; Creager, M.A.; Yeung, A.C.; et al. Close relation of endothelial function in the human coronary and peripheral circulations. J. Am. Coll. Cardiol 1995, 26, 1235–1241. [Google Scholar]
- Williams, S.B.; Goldfine, A.B.; Timimi, F.K.; Ting, H.H.; Roddy, M.A.; Simonson, D.C.; Creager, M.A. Acute hyperglycemia attenuates endothelium-dependent vasodilation in humans in vivo. Circulation 1998, 97, 1695–1701. [Google Scholar]
- Jansson, P.A. Endothelial dysfunction in insulin resistance and type 2 diabetes. J. Intern. Med 2007, 262, 173–183. [Google Scholar]
- Plock, N.; Kloft, C. Microdialysis—Theoretical background and recent implementation in applied life-sciences. Eur. J. Pharm. Sci 2005, 25, 1–24. [Google Scholar]
- Wakino, S.; Hayashi, K.; Tatematsu, S.; Hasegawa, K.; Takamatsu, I.; Kanda, T.; Homma, K.; Yoshioka, K.; Sugano, N.; Saruta, T. Pioglitazone lowers systemic asymmetric dimethylarginine by inducing dimethylarginine dimethylaminohydrolase in rats. Hypertens. Res 2005, 28, 255–262. [Google Scholar]
- Chobanyan, K.; Thum, T.; Suchy, M.T.; Zhu, B.; Mitschke, A.; Gutzki, F.M.; Beckmann, B.; Stichtenoth, D.O.; Tsikas, D. GC-MS assay for hepatic DDAH activity in diabetic and non-diabetic rats by measuring dimethylamine (DMA) formed from asymmetric dimethylarginine (ADMA): Evaluation of the importance of S-nitrosothiols as inhibitors of DDAH activity in vitro and in vivo in humans. J. Chromatogr. B 2007, 858, 32–41. [Google Scholar]
- May, M.; Batkai, S.; Zoerner, A.A.; Tsikas, D.; Jordan, J.; Engeli, S. Enhanced human tissue microdialysis using hydroxypropyl-ss-cyclodextrin as molecular carrier. PLoS One 2013, 8, e60628. [Google Scholar]
- Tsikas, D.; Schubert, B.; Gutzki, F.M.; Sandmann, J.; Frolich, J.C. Quantitative determination of circulating and urinary asymmetric dimethylarginine (ADMA) in humans by gas chromatography-tandem mass spectrometry as methyl ester tri(N-pentafluoropropionyl) derivative. J. Chromatogr. B 2003, 798, 87–99. [Google Scholar]
- Tholance, Y.; Barcelos, G.; Quadrio, I.; Renaud, B.; Dailler, F.; Perret-Liaudet, A. Analytical validation of microdialysis analyzer for monitoring glucose, lactate and pyruvate in cerebral microdialysates. Clin. Chim. Acta 2010, 412, 647–654. [Google Scholar]
- Adams, F.; Jordan, J.; Schaller, K.; Luft, F.C.; Boschmann, M. Blood flow in subcutaneous adipose tissue depends on skin-fold thickness. Horm. Metab. Res 2005, 37, 68–73. [Google Scholar]
- Fellander, G.; Linde, B.; Bolinder, J. Evaluation of the microdialysis ethanol technique for monitoring of subcutaneous adipose tissue blood flow in humans. Int. J. Obes. Relat. Metab. Disord 1996, 20, 220–226. [Google Scholar]
- Farnebo, S.; Zettersten, E.K.; Samuelsson, A.; Tesselaar, E.; Sjoberg, F. Assessment of blood flow changes in human skin by microdialysis urea clearance. Microcirculation 2011, 18, 198–204. [Google Scholar]


| Healthy subjects (n = 4) | Patients with diabetes (n = 4) | Patients with morbid obesity before weight loss (n = 7) * | Patients after weight loss (n = 7) * | |
|---|---|---|---|---|
| Age (year) | 38 ± 4 | 54 ± 8 | 35 ± 3 | 36 ± 3 |
| Sex (m/f) | 4/0 | 1/3 | 2/5 | 2/5 |
| BMI (kg/m2) | 23.9 ± 1.8 | 30.0 ± 5.2 | 45.7 ± 1.5 | 34.4 ± 2.0 |
| Weight (kg) | 77 ± 2 | 80 ± 1 | 135 ± 4 | 102 ± 5 |
| HOMA | 1.2 ± 0.3 | 3.7 ± 0.3 | 5.6 ± 2.0 | 1.8 ± 0.7 |
| HbA1c (%) | <6.5 | 7.6 ± 1.3 | <6.5 | <6.5 |
| Hypertension (n) | 0 | 3 | 4 | 0 |
| Other comorbidities (n) | 0 | allergy (1) | migraine (2) | migraine (2) |
| Drug treatment (n) | none | ACE-inhibitors (2); beta blockers (1) thiazide diuretics (1) metformin (3) insulin (1) | AT1-blockers (1) ACE-inhibitors (1) diuretics (1) beta blockers (3) topiramate (1) diclofenac (1) omeprazole (3) | topiramate (1) diclofenac (1) omeprazole (1) |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
May, M.; Batkai, S.; Zörner, A.A.; Tsikas, D.; Jordan, J.; Engeli, S. Clinical Evaluation of Extracellular ADMA Concentrations in Human Blood and Adipose Tissue. Int. J. Mol. Sci. 2014, 15, 1189-1200. https://doi.org/10.3390/ijms15011189
May M, Batkai S, Zörner AA, Tsikas D, Jordan J, Engeli S. Clinical Evaluation of Extracellular ADMA Concentrations in Human Blood and Adipose Tissue. International Journal of Molecular Sciences. 2014; 15(1):1189-1200. https://doi.org/10.3390/ijms15011189
Chicago/Turabian StyleMay, Marcus, Sandor Batkai, Alexander A. Zörner, Dimitrios Tsikas, Jens Jordan, and Stefan Engeli. 2014. "Clinical Evaluation of Extracellular ADMA Concentrations in Human Blood and Adipose Tissue" International Journal of Molecular Sciences 15, no. 1: 1189-1200. https://doi.org/10.3390/ijms15011189




