Environmental Stimuli Shape Biofilm Formation and the Virulence of Periodontal Pathogens
Abstract
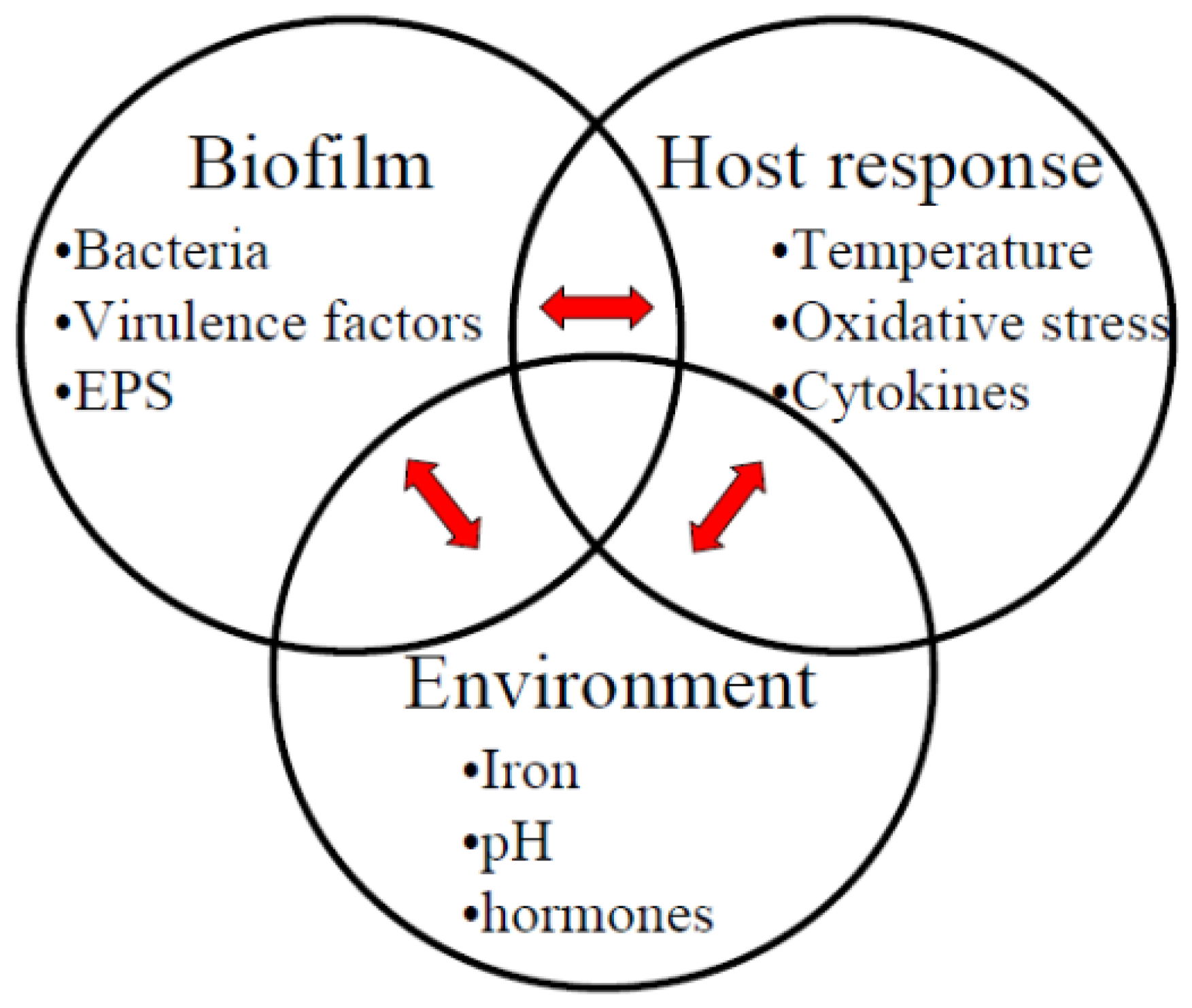
:1. Introduction
2. Host Inflammatory Reaction-Related Stimuli
2.1. Temperature
2.2. Oxidative Stress
2.3. Inflammatory Cytokines
3. Periodontitis-Associated Environmental Factors
3.1. Alkaline pH
3.2. Iron and Hemin
3.3. Hormones
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bartold, P.M.; van Dyke, T.E. Periodontitis: A host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontol 2000 2013, 62, 203–217. [Google Scholar]
- Griffen, A.L.; Beall, C.J.; Campbell, J.H.; Firestone, N.D.; Kumar, P.S.; Yang, Z.K.; Podar, M.; Leys, E.J. Distinct and complex bacterial profiles in human periodontitis and health revealed by 16S pyrosequencing. ISME J 2012, 6, 1176–1185. [Google Scholar]
- Rescala, B.; Rosalem, W., Jr; Teles, R.P.; Fischer, R.G.; Haffajee, A.D.; Socransky, S.S.; Gustafsson, A.; Figueredo, C.M. Immunologic and microbiologic profiles of chronic and aggressive periodontitis subjects. J. Periodontol. 2010, 81, 1308–1316. [Google Scholar]
- Fritschi, B.Z.; Albert-Kiszely, A.; Persson, G.R. Staphylococcus aureus and other bacteria in untreated periodontitis. J. Dent. Res 2008, 87, 589–593. [Google Scholar]
- Saygun, I.; Kubar, A.; Sahin, S.; Sener, K.; Slots, J. Quantitative analysis of association between herpesviruses and bacterial pathogens in periodontitis. J. Periodontal. Res 2008, 43, 352–359. [Google Scholar]
- Imbronito, A.V.; Okuda, O.S.; Maria de Freitas, N.; Moreira Lotufo, R.F.; Nunes, F.D. Detection of herpesviruses and periodontal pathogens in subgingival plaque of patients with chronic periodontitis, generalized aggressive periodontitis, or gingivitis. J. Periodontol 2008, 79, 2313–2321. [Google Scholar]
- Canabarro, A.; Valle, C.; Farias, M.R.; Santos, F.B.; Lazera, M.; Wanke, B. Association of subgingival colonization of Candida albicans and other yeasts with severity of chronic periodontitis. J. Periodontal. Res 2013, 48, 428–432. [Google Scholar]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol 2010, 8, 623–633. [Google Scholar]
- Lewis, J.P. Metal Uptake in host-pathogen interactions: Role of iron in Porphyromonas gingivalis interactions with host organisms. Periodontol. 2000 2010, 52, 94–116. [Google Scholar]
- McDougald, D.; Rice, S.A.; Barraud, N.; Steinberg, P.D.; Kjelleberg, S. Should we stay or should we go: Mechanisms and ecological consequences for biofilm dispersal. Nat. Rev. Microbiol 2012, 10, 39–50. [Google Scholar]
- Tonetti, M.S.; Imboden, M.A.; Lang, N.P. Neutrophil Migration into the gingival sulcus is associated with transepithelial gradients of interleukin-8 and ICAM-1. J. Periodontol 1998, 69, 1139–1147. [Google Scholar]
- Shapiro, R.S.; Cowen, L.E. Thermal control of microbial development and virulence: Molecular mechanisms of microbial temperature sensing. MBio 2012, 3. [Google Scholar] [CrossRef]
- Fedi, P.F., Jr; Killoy, W.J. Temperature differences at periodontal sites in health and disease. J. Periodontol. 1992, 63, 24–27. [Google Scholar]
- Percival, R.S.; Marsh, P.D.; Devine, D.A.; Rangarajan, M.; Aduse-Opoku, J.; Shepherd, P.; Curtis, M.A. Effect of temperature on growth, hemagglutination, and protease activity of Porphyromonas gingivalis. Infect. Immun 1999, 67, 1917–1921. [Google Scholar]
- Amano, A.; Fujiwara, T.; Nagata, H.; Kuboniwa, M.; Sharma, A.; Sojar, H.T.; Genco, R.J.; Hamada, S.; Shizukuishi, S. Prophyromonas gingivalis fimbriae mediate coaggregation with Streptococcus oralis through specific domains. J. Dent. Res 1997, 76, 852–857. [Google Scholar]
- Murakami, Y.; Nagata, H.; Amano, A.; Takagaki, M.; Shizukuishi, S.; Tsunemitsu, A.; Aimoto, S. Inhibitory effects of human salivary histatins and lysozyme on coaggregation between Porphyromonas gingivalis and Streptococcus mitis. Infect. Immun 1991, 59, 3284–3286. [Google Scholar]
- Sato, T.; Nakazawa, F. Coaggregation between Prevotella oris and Porphyromonas gingivalis. J. Microbiol. Immunol. Infect. 2012. [Google Scholar] [CrossRef]
- Curtis, M.A.; Percival, R.S.; Devine, D.; Darveau, R.P.; Coats, S.R.; Rangarajan, M.; Tarelli, E.; Marsh, P.D. Temperature-dependent modulation of Porphyromonas gingivalis lipid A structure and interaction with the innate host defenses. Infect. Immun 2011, 79, 1187–1193. [Google Scholar]
- Haffajee, A.D.; Socransky, S.S.; Smith, C.; Dibart, S.; Goodson, J.M. Subgingival temperature (III). Relation to microbial counts. J. Clin. Periodontol 1992, 19, 417–422. [Google Scholar]
- Imlay, J.A. Cellular defenses against superoxide and hydrogen peroxide. Annu. Rev. Biochem 2008, 77, 755–776. [Google Scholar]
- Cabiscol, E.; Tamarit, J.; Ros, J. Oxidative stress in bacteria and protein damage by reactive oxygen species. Int. Microbiol 2000, 3, 3–8. [Google Scholar]
- Bradshaw, D.J.; Marsh, P.D.; Watson, G.K.; Allison, C. Role of Fusobacterium nucleatum and coaggregation in anaerobe survival in planktonic and biofilm oral microbial communities during aeration. Infect. Immun 1998, 66, 4729–4732. [Google Scholar]
- Silva, V.L.; Diniz, C.G.; Cara, D.C.; Santos, S.G.; Nicoli, J.R.; Carvalho, M.A.; Farias, L.M. Enhanced pathogenicity of Fusobacterium nucleatum adapted to oxidative stress. Microb. Pathog 2005, 39, 131–138. [Google Scholar]
- Gursoy, U.K.; Pöllänen, M.; Könönen, E.; Uitto, V.J. Biofilm formation enhances the oxygen tolerance and invasiveness of Fusobacterium nucleatum in an oral mucosa culture model. J. Periodontol 2010, 81, 1084–1091. [Google Scholar]
- Steeves, C.H.; Potrykus, J.; Barnett, D.A.; Bearne, S.L. Oxidative stress response in the opportunistic oral pathogen Fusobacterium nucleatum. Proteomics 2011, 11, 2027–2037. [Google Scholar]
- Okano, S.; Shibata, Y.; Shiroza, T.; Abiko, Y. Proteomics-based analysis of a counter-oxidative stress system in Porphyromonas gingivalis. Proteomics 2006, 6, 251–258. [Google Scholar]
- Amano, A.; Sharma, A.; Sojar, H.T.; Kuramitsu, H.K.; Genco, R.J. Effects of temperature stress on expression of fimbriae and superoxide dismutase by Porphyromonas gingivalis. Infect. Immun 1994, 62, 4682–4685. [Google Scholar]
- McKenzie, R.M.; Johnson, N.A.; Aruni, W.; Dou, Y.; Masinde, G.; Fletcher, H.M. Differential response of Porphyromonas gingivalis to varying levels and duration of hydrogen peroxide-induced oxidative stress. Microbiology 2012, 158, 2465–2479. [Google Scholar]
- Gamonal, J.; Acevedo, A.; Bascones, A.; Jorge, O.; Silva, A. Levels of interleukin-1 beta, −8, and −10 and RANTES in gingival crevicular fluid and cell populations in adult periodontitis patients and the effect of periodontal treatment. J. Periodontol 2000, 71, 1535–1545. [Google Scholar]
- Silva, N.; Dutzan, N.; Hernandez, M.; Dezerega, A.; Rivera, O.; Aguillon, J.C.; Aravena, O.; Lastres, P.; Pozo, P.; Vernal, R.; et al. Characterization of progressive periodontal lesions in chronic periodontitis patients: Levels of chemokines, cytokines, matrix metalloproteinase-13, periodontal pathogens and inflammatory cells. J. Clin. Periodontol 2008, 35, 206–214. [Google Scholar]
- Graves, D.T.; Cochran, D. The contribution of interleukin-1 and tumor necrosis factor to periodontal tissue destruction. J. Periodontol 2003, 74, 391–401. [Google Scholar]
- Ishihara, Y.; Nishihara, T.; Maki, E.; Noguchi, T.; Koga, T. Role of interleukin-1 and prostaglandin in in vitro bone resorption induced by Actinobacillus actinomycetemcomitans lipopolysaccharide. J. Periodontal. Res 1991, 26, 155–160. [Google Scholar]
- Cochran, D.L. Inflammation and bone loss in periodontal disease. J. Periodontol 2008, 79, 1569–1576. [Google Scholar]
- Eskan, M.A.; Benakanakere, M.R.; Rose, B.G.; Zhang, P.; Zhao, J.; Stathopoulou, P.; Fujioka, D.; Kinane, D.F. Interleukin-1beta modulates proinflammatory cytokine production in human epithelial cells. Infect. Immun 2008, 76, 2080–2089. [Google Scholar]
- Tanabe, S.; Bodet, C.; Grenier, D. Treponema denticola lipooligosaccharide activates gingival fibroblasts and upregulates inflammatory mediator production. J. Cell. Physiol 2008, 216, 727–731. [Google Scholar]
- Kelk, P.; Claesson, R.; Chen, C.; Sjöstedt, A.; Johansson, A. IL-1beta secretion induced by Aggregatibacter (Actinobacillus) actinomycetemcomitans is mainly caused by the leukotoxin. Int. J. Med. Microbiol 2008, 298, 529–541. [Google Scholar]
- Tanabe, S.I.; Grenier, D. Macrophage tolerance response to Aggregatibacter actinomycetemcomitans lipopolysaccharide induces differential regulation of tumor necrosis factor-alpha, interleukin-1 beta and matrix metalloproteinase 9 secretion. J. Periodontal. Res 2008, 43, 372–377. [Google Scholar]
- Matsuki, Y.; Yamamoto, T.; Hara, K. Interleukin-1 mRNA-expressing macrophages in human chronically inflamed gingival tissues. Am. J. Pathol 1991, 138, 1299–1305. [Google Scholar]
- Hsi, E.D.; Remick, D.G. Monocytes are the major producers of interleukin-1 beta in an ex vivo model of local cytokine production. J. Interferon Cytokine Res 1995, 15, 89–94. [Google Scholar]
- Takeichi, O.; Saito, I.; Tsurumachi, T.; Saito, T.; Moro, I. Human polymorphonuclear leukocytes derived from chronically inflamed tissue express inflammatory cytokines in vivo. Cell. Immunol 1994, 156, 296–309. [Google Scholar]
- Vardar-Sengul, S.; Arora, S.; Baylas, H.; Mercola, D. Expression profile of human gingival fibroblasts induced by interleukin-1beta reveals central role of nuclear factor-kappa b in stabilizing human gingival fibroblasts during inflammation. J. Periodontol 2009, 80, 833–849. [Google Scholar]
- Uchida, Y.; Shiba, H.; Komatsuzawa, H.; Takemoto, T.; Sakata, M.; Fujita, T.; Kawaguchi, H.; Sugai, M.; Kurihara, H. Expression of IL-1 beta and IL-8 by human gingival epithelial cells in response to Actinobacillus actinomycetemcomitans. Cytokine 2001, 14, 152–161. [Google Scholar]
- Dickinson, B.C.; Moffatt, C.E.; Hagerty, D.; Whitmore, S.E.; Brown, T.A.; Graves, D.T.; Lamont, R.J. Interaction of oral bacteria with gingival epithelial cell multilayers. Mol. Oral Microbiol 2011, 26, 210–220. [Google Scholar]
- Stathopoulou, P.G.; Benakanakere, M.R.; Galicia, J.C.; Kinane, D.F. Epithelial cell pro-inflammatory cytokine response differs across dental plaque bacterial species. J. Clin. Periodontol 2010, 37, 24–29. [Google Scholar]
- Umeda, J.E.; Demuth, D.R.; Ando, E.S.; Faveri, M.; Mayer, M.P. Signaling transduction analysis in gingival epithelial cells after infection with Aggregatibacter actinomycetemcomitans. Mol. Oral Microbiol 2012, 27, 23–33. [Google Scholar]
- Peyyala, R.; Kirakodu, S.S.; Novak, K.F.; Ebersole, J.L. Oral microbial biofilm stimulation of epithelial cell responses. Cytokine 2012, 58, 65–72. [Google Scholar]
- Guggenheim, B.; Gmur, R.; Galicia, J.C.; Stathopoulou, P.G.; Benakanakere, M.R.; Meier, A.; Thurnheer, T.; Kinane, D.F. In vitro modeling of host-parasite interactions: The ‘subgingival’ biofilm challenge of primary human epithelial cells. BMC Microbiol. 2009, 9. [Google Scholar] [CrossRef] [Green Version]
- Peyyala, R.; Ebersole, J.L. Multispecies biofilms and host responses: “Discriminating the trees from the forest”. Cytokine 2013, 61, 15–25. [Google Scholar]
- Porat, R.; Clark, B.D.; Wolff, S.M.; Dinarello, C.A. Enhancement of growth of virulent strains of Escherichia coli by interleukin-1. Science 1991, 254, 430–432. [Google Scholar]
- Meduri, G.U.; Kanangat, S.; Stefan, J.; Tolley, E.; Schaberg, D. Cytokines IL-1beta, IL-6, and TNF-alpha enhance in vitro growth of bacteria. Am. J. Respir. Crit. Care Med 1999, 160, 961–967. [Google Scholar]
- Stashenko, P.; Fujiyoshi, P.; Obernesser, M.S.; Prostak, L.; Haffajee, A.D.; Socransky, S.S. Levels of interleukin 1 beta in tissue from sites of active periodontal disease. J. Clin. Periodontol 1991, 18, 548–554. [Google Scholar]
- McLaughlin, R.A.; Hoogewerf, A.J. Interleukin-1beta-induced growth enhancement of Staphylococcus aureus occurs in biofilm but not planktonic cultures. Microb. Pathog 2006, 41, 67–79. [Google Scholar]
- Kanangat, S.; Bronze, M.S.; Meduri, G.U.; Postlethwaite, A.; Stentz, F.; Tolley, E.; Schaberg, D. Enhanced extracellular growth of Staphylococcus aureus in the presence of selected linear peptide fragments of human interleukin (IL)-1beta and IL-1 receptor antagonist. J. Infect. Dis 2001, 183, 65–69. [Google Scholar]
- Kanangat, S.; Postlethwaite, A.; Cholera, S.; Williams, L.; Schaberg, D. Modulation of virulence gene expression in Staphylococcus aureus by interleukin-1beta: Novel implications in bacterial pathogenesis. Microbes Infect 2007, 9, 408–415. [Google Scholar]
- Zav’yalov, V.P.; Chernovskaya, T.V.; Navolotskaya, E.V.; Karlyshev, A.V.; MacIntyre, S.; Vasiliev, A.M.; Abramov, V.M. Specific high affinity binding of human interleukin 1 beta by Caf1A usher protein of Yersinia pestis. FEBS Lett 1995, 371, 65–68. [Google Scholar]
- Zav’yalov, V.; Denesyuk, A.; Zav’yalova, G.; Korpela, T. Molecular modeling of the steric structure of the envelope F1 antigen of Yersinia pestis. Immunol. Lett 1995, 45, 19–22. [Google Scholar]
- Wu, L.; Estrada, O.; Zaborina, O.; Bains, M.; Shen, L.; Kohler, J.E.; Patel, N.; Musch, M.W.; Chang, E.B.; Fu, Y.X.; et al. Recognition of host immune activation by Pseudomonas aeruginosa. Science 2005, 309, 774–777. [Google Scholar]
- Diggle, S.P.; Stacey, R.E.; Dodd, C.; Camara, M.; Williams, P.; Winzer, K. The galactophilic lectin, LecA, contributes to biofilm development in Pseudomonas aeruginosa. Environ. Microbiol 2006, 8, 1095–1104. [Google Scholar]
- Cebo, C.; Dambrouck, T.; Maes, E.; Laden, C.; Strecker, G.; Michalski, J.C.; Zanetta, J.P. Recombinant human interleukins IL-1alpha, IL-1beta, IL-4, IL-6, and IL-7 show different and specific calcium-independent carbohydrate-binding properties. J. Biol. Chem 2001, 276, 5685–5691. [Google Scholar]
- Sherblom, A.P.; Decker, J.M.; Muchmore, A.V. The lectin-like interaction between recombinant tumor necrosis factor and uromodulin. J. Biol. Chem 1988, 263, 5418–5424. [Google Scholar]
- Cebo, C.; Vergoten, G.; Zanetta, J.P. Lectin activities of cytokines: Functions and putative carbohydrate-recognition domains. Biochim. Biophys. Acta 2002, 1572, 422–434. [Google Scholar]
- Vergoten, G.; Zanetta, J.P. Structural differences between the putative carbohydrate-recognition domains of human IL-1 alpha, IL-1 beta and IL-1 receptor antagonist obtained by in silico modeling. Glycoconj. J 2007, 24, 183–193. [Google Scholar]
- Mandrell, R.E.; McLaughlin, R.; Aba Kwaik, Y.; Lesse, A.; Yamasaki, R.; Gibson, B.; Spinola, S.M.; Apicella, M.A. Lipooligosaccharides (LOS) of some Haemophilus species mimic human glycosphingolipids, and some LOS are sialylated. Infect. Immun 1992, 60, 1322–1328. [Google Scholar]
- Li, Y.; Sun, M.; Huang, S.; Yu, H.; Chokhawala, H.A.; Thon, V.; Chen, X. The hd0053 gene of Haemophilus ducreyi encodes an α2,3-sialyltransferase. Biochem. Biophys. Res. Commun 2007, 361, 555–560. [Google Scholar]
- Paino, A.; Tuominen, H.; Jääskeläinen, M.; Alanko, J.; Nuutila, J.; Asikainen, S.E.; Pelliniemi, L.J.; Pöllänen, M.T.; Chen, C.; Ihalin, R. Trimeric form of intracellular ATP synthase subunit beta of Aggregatibacter actinomycetemcomitans binds human interleukin-1beta. PLoS One 2011, 6, e18929. [Google Scholar]
- Paino, A.; Lohermaa, E.; Sormunen, R.; Tuominen, H.; Korhonen, J.; Pöllänen, M.T.; Ihalin, R. Interleukin-1β is internalised by viable Aggregatibacter actinomycetemcomitans biofilm and locates to the outer edges of nucleoids. Cytokine 2012, 60, 565–574. [Google Scholar]
- Paino, A.; Ahlstrand, T.; Nuutila, J.; Navickaite, I.; Lahti, M.; Tuominen, H.; Välimaa, H.; Lamminmäki, U.; Pöllänen, M.T.; Ihalin, R. Identification of a novel bacterial outer membrane interleukin-1beta-binding protein from Aggregatibacter actinomycetemcomitans. PLoS One 2013, 8, e70509. [Google Scholar]
- Alcami, A. Viral mimicry of cytokines, chemokines and their receptors. Nat. Rev. Immunol 2003, 3, 36–50. [Google Scholar]
- Treseler, C.B.; Maziarz, R.T.; Levitz, S.M. Biological activity of interleukin-2 bound to Candida albicans. Infect. Immun 1992, 60, 183–188. [Google Scholar]
- Bickel, M.; Munoz, J.L.; Giovannini, P. Acid-base properties of human gingival crevicular fluid. J. Dent. Res 1985, 64, 1218–1220. [Google Scholar]
- Bickel, M.; Cimasoni, G. The pH of human crevicular fluid measured by a new microanalytical technique. J. Periodontal. Res 1985, 20, 35–40. [Google Scholar]
- Eggert, F.M.; Drewell, L.; Bigelow, J.A.; Speck, J.E.; Goldner, M. The pH of gingival crevices and periodontal pockets in children, teenagers and adults. Arch. Oral Biol 1991, 36, 233–238. [Google Scholar]
- Takahashi, N. Acid-neutralizing activity during amino acid fermentation by Porphyromonas gingivalis, Prevotella intermedia and Fusobacterium nucleatum. Oral Microbiol. Immunol 2003, 18, 109–113. [Google Scholar]
- Zilm, P.S.; Rogers, A.H. Co-adhesion and biofilm formation by Fusobacterium nucleatum in response to growth pH. Anaerobe 2007, 13, 146–152. [Google Scholar]
- Zilm, P.S.; Bagley, C.J.; Rogers, A.H.; Milne, I.R.; Gully, N.J. The proteomic profile of Fusobacterium nucleatum is regulated by growth pH. Microbiology 2007, 153, 148–159. [Google Scholar]
- Kapatral, V.; Anderson, I.; Ivanova, N.; Reznik, G.; Los, T.; Lykidis, A.; Bhattacharyya, A.; Bartman, A.; Gardner, W.; Grechkin, G.; et al. Genome sequence and analysis of the oral bacterium Fusobacterium nucleatum strain ATCC 25586. J. Bacteriol 2002, 184, 2005–2018. [Google Scholar]
- Chew, J.; Zilm, P.S.; Fuss, J.M.; Gully, N.J. A proteomic investigation of Fusobacterium nucleatum alkaline-induced biofilms. BMC Microbiol. 2012, 12. [Google Scholar] [CrossRef]
- Skar, C.K.; Kruger, P.G.; Bakken, V. Characterisation and subcellular localisation of the GroEL-like and DnaK-like proteins isolated from Fusobacterium nucleatum ATCC 10953. Anaerobe 2003, 9, 305–312. [Google Scholar]
- Oscarsson, J.; Karched, M.; Thay, B.; Chen, C.; Asikainen, S. Proinflammatory effect in whole blood by free soluble bacterial components released from planktonic and biofilm cells. BMC Microbiol. 2008, 8. [Google Scholar] [CrossRef]
- Lee, H.R.; Jun, H.K.; Kim, H.D.; Lee, S.H.; Choi, B.K. Fusobacterium nucleatum GroEL induces risk factors of atherosclerosis in human microvascular endothelial cells and apoE(−/−) mice. Mol. Oral Microbiol 2012, 27, 109–123. [Google Scholar]
- Shaniztki, B.; Hurwitz, D.; Smorodinsky, N.; Ganeshkumar, N.; Weiss, E.I. Identification of a Fusobacterium nucleatum PK1594 galactose-binding adhesin which mediates coaggregation with periopathogenic bacteria and hemagglutination. Infect. Immun 1997, 65, 5231–5237. [Google Scholar]
- Liu, P.F.; Shi, W.; Zhu, W.; Smith, J.W.; Hsieh, S.L.; Gallo, R.L.; Huang, C.M. Vaccination targeting surface FomA of Fusobacterium nucleatum against bacterial co-aggregation: implication for treatment of periodontal infection and halitosis. Vaccine 2010, 28, 3496–3505. [Google Scholar]
- Zilm, P.S.; Mira, A.; Bagley, C.J.; Rogers, A.H. Effect of alkaline growth pH on the expression of cell envelope proteins in Fusobacterium nucleatum. Microbiology 2010, 156, 1783–1794. [Google Scholar]
- Schaible, U.E.; Kaufmann, S.H. Iron and microbial infection. Nat. Rev. Microbiol 2004, 2, 946–953. [Google Scholar]
- Shoji, M.; Shibata, Y.; Shiroza, T.; Yukitake, H.; Peng, B.; Chen, Y.Y.; Sato, K.; Naito, M.; Abiko, Y.; Reynolds, E.C.; et al. Characterization of hemin-binding protein 35 (HBP35) in Porphyromonas gingivalis: Its cellular distribution, thioredoxin activity and role in heme utilization. BMC Microbiol. 2010, 10. [Google Scholar] [CrossRef]
- Xu, X.; Kolodrubetz, D. Construction and analysis of hemin binding protein mutants in the oral pathogen Treponema denticola. Res. Microbiol 2002, 153, 569–577. [Google Scholar]
- Rhodes, E.R.; Menke, S.; Shoemaker, C.; Tomaras, A.P.; McGillivary, G.; Actis, L.A. Iron acquisition in the dental pathogen Actinobacillus actinomycetemcomitans: What does it use as a source and how does it get this essential metal? Biometals 2007, 20, 365–377. [Google Scholar]
- Al-Qutub, M.N.; Braham, P.H.; Karimi-Naser, L.M.; Liu, X.; Genco, C.A.; Darveau, R.P. Hemin-dependent modulation of the lipid A structure of Porphyromonas gingivalis lipopolysaccharide. Infect. Immun 2006, 74, 4474–4485. [Google Scholar]
- Amarasinghe, J.J.; Scannapieco, F.A.; Haase, E.M. Transcriptional and translational analysis of biofilm determinants of Aggregatibacter actinomycetemcomitans in response to environmental perturbation. Infect. Immun 2009, 77, 2896–2907. [Google Scholar]
- Amarasinghe, J.J.; Connell, T.D.; Scannapieco, F.A.; Haase, E.M. Novel iron-regulated and Fur-regulated small regulatory RNAs in Aggregatibacter actinomycetemcomitans. Mol. Oral Microbiol 2012, 27, 327–349. [Google Scholar]
- Smalley, J.W.; Birss, A.J.; McKee, A.S.; Marsh, P.D. Haemin-restriction influences haemin-binding, haemagglutination and protease activity of cells and extracellular membrane vesicles of Porphyromonas gingivalis W50. FEMS Microbiol. Lett 1991, 69, 63–67. [Google Scholar]
- Hughes, D.T.; Sperandio, V. Inter-kingdom signalling: Communication between bacteria and their hosts. Nat. Rev. Microbiol 2008, 6, 111–120. [Google Scholar]
- Roberts, A.; Matthews, J.B.; Socransky, S.S.; Freestone, P.P.; Williams, P.H.; Chapple, I.L. Stress and the periodontal diseases: Effects of catecholamines on the growth of periodontal bacteria in vitro. Oral Microbiol. Immunol 2002, 17, 296–303. [Google Scholar]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L., Jr. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar]
- Saito, T.; Inagaki, S.; Sakurai, K.; Okuda, K.; Ishihara, K. Exposure of P. gingivalis to noradrenaline reduces bacterial growth and elevates ArgX protease activity. Arch. Oral Biol 2011, 56, 244–250. [Google Scholar]
- Mariotti, A. Sex steroid hormones and cell dynamics in the periodontium. Crit. Rev. Oral Biol. Med 1994, 5, 27–53. [Google Scholar]
- Armitage, G.C. Development of a classification system for periodontal diseases and conditions. Ann. Periodontol 1999, 4, 1–6. [Google Scholar]
- Jonsson, R.; Howland, B.E.; Bowden, G.H. Relationships between periodontal health, salivary steroids, and Bacteroides intermedius in males, pregnant and non-pregnant women. J. Dent. Res 1988, 67, 1062–1069. [Google Scholar]
- Figuero, E.; Carrillo-de-Albornoz, A.; Herrera, D.; Bascones-Martinez, A. Gingival changes during pregnancy: I. Influence of hormonal variations on clinical and immunological parameters. J. Clin. Periodontol 2010, 37, 220–229. [Google Scholar]
- Gursoy, M.; Gursoy, U.K.; Sorsa, T.; Pajukanta, R.; Könönen, E. High salivary estrogen and risk of developing pregnancy gingivitis. J. Periodontol. 2012. [Google Scholar] [CrossRef]
- Lopatin, D.E.; Kornman, K.S.; Loesche, W.J. Modulation of immunoreactivity to periodontal disease-associated microorganisms during pregnancy. Infect. Immun 1980, 28, 713–718. [Google Scholar]
- Lapp, C.A.; Thomas, M.E.; Lewis, J.B. Modulation by progesterone of interleukin-6 production by gingival fibroblasts. J. Periodontol 1995, 66, 279–284. [Google Scholar]
- Rodriguez, E.; Lopez, R.; Paez, A.; Masso, F.; Montano, L.F. 17Beta-estradiol inhibits the adhesion of leukocytes in TNF-alpha stimulated human endothelial cells by blocking IL-8 and MCP-1 secretion, but not its transcription. Life Sci 2002, 71, 2181–2193. [Google Scholar]
- Shu, L.; Guan, S.M.; Fu, S.M.; Guo, T.; Cao, M.; Ding, Y. Estrogen modulates cytokine expression in human periodontal ligament cells. J. Dent. Res 2008, 87, 142–147. [Google Scholar]
- Jensen, J.; Liljemark, W.; Bloomquist, C. The effect of female sex hormones on subgingival plaque. J. Periodontol 1981, 52, 599–602. [Google Scholar]
- Raber-Durlacher, J.E.; van Steenbergen, T.J.; van der Velden, U.; de Graaff, J.; Abraham-Inpijn, L. Experimental gingivitis during pregnancy and post-partum: Clinical, endocrinological, and microbiological aspects. J. Clin. Periodontol 1994, 21, 549–558. [Google Scholar]
- Carrillo-de-Albornoz, A.; Figuero, E.; Herrera, D.; Bascones-Martinez, A. Gingival changes during pregnancy: II. Influence of hormonal variations on the subgingival biofilm. J. Clin. Periodontol 2010, 37, 230–240. [Google Scholar]
- Kornman, K.S.; Loesche, W.J. Effects of estradiol and progesterone on Bacteroides melaninogenicus and Bacteroides gingivalis. Infect. Immun 1982, 35, 256–263. [Google Scholar]
| Stimuli | Effect | Species | References |
|---|---|---|---|
| Elevated temperature | Proteases ↓ | Porphyromonas gingivalis | [16] |
| Fimbrial proteins ↓ | [15] | ||
| TLR4 activating lipid-A ↑ | [18] | ||
| Oxidative stress | ATP production ↓ | Fusobacterium nucleatum | [25] |
| Chaperones ClpB, DnaK ↑ | [25] | ||
| Heat shock protein HtpG ↑ | [25] | ||
| Transcription repressor HrcA ↑ | [25] | ||
| Oxidative stress | Chaperones ClpB, DnaK ↑ | Porphyromonas gingivalis | [26] |
| Heat shock protein HtpG ↑ | [26] | ||
| superoxide dismutase ↑ | [26] | ||
| Inflammatory cytokine IL-1β | Biofilm formation ↑ | Aggregatibacter actinomycetemcomitans | [65] |
| Metabolism ↓ | [65] | ||
| Alkaline pH | Co-adhesion ↑ | Fusobacterium nucleatum | [74] |
| Biofilm formation ↑ | [74] | ||
| Flavodoxin ↑ | [75] | ||
| Glucose storage ↑ | [77] | ||
| Lactate production ↑ | [77] | ||
| Protein synthesis enzymes ↓ | [77] | ||
| Glutamate dehydrogenase ↑ | [77] | ||
| PPI and GroEL ↑ | [75,77] | ||
| FomA adhesion isoforms ↑ | [77] | ||
| ATP synthesis proteins ↓ | [83] | ||
| Butyrate-acetoacetate CoA transferase ↓ | [83] | ||
| Surface antigens Omp IP ↑ | [83] | ||
| Iron-limitation | EPS (pgaC) ↑ | Aggregatibacter actinomycetemcomitans | [89] |
| Fimbrial (tadV) ↑ | [89] | ||
| LPS (rmlB) ↑ | [89] | ||
| Biofilm formation ↑ | [89] | ||
| Iron limitation | Iron uptake ↑ | Porphyromonas gingivalis | [9] |
| Iron storage ↓ | [9] | ||
| Oxidative stress response ↓ | [9] | ||
| Biofilm formation ↑ | [9] | ||
| Host cell invasion ↑ | [9] | ||
| High hemin concentration | Proteases ↓ | Porphyromonas gingivalis | [91] |
| Vesicles ↓ | [91] | ||
| TLR4 inactivating lipid A ↑ | [88] | ||
| Noradrenaline | Growth ↓ | Porphyromonas gingivalis | [93] |
| Arg-gingipain B ↑ | [95] | ||
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pöllänen, M.T.; Paino, A.; Ihalin, R. Environmental Stimuli Shape Biofilm Formation and the Virulence of Periodontal Pathogens. Int. J. Mol. Sci. 2013, 14, 17221-17237. https://doi.org/10.3390/ijms140817221
Pöllänen MT, Paino A, Ihalin R. Environmental Stimuli Shape Biofilm Formation and the Virulence of Periodontal Pathogens. International Journal of Molecular Sciences. 2013; 14(8):17221-17237. https://doi.org/10.3390/ijms140817221
Chicago/Turabian StylePöllänen, Marja T., Annamari Paino, and Riikka Ihalin. 2013. "Environmental Stimuli Shape Biofilm Formation and the Virulence of Periodontal Pathogens" International Journal of Molecular Sciences 14, no. 8: 17221-17237. https://doi.org/10.3390/ijms140817221




