1. Introduction
Eggs are an important part of human diet [
1] and contain high amounts of cholesterol. The average egg contains 213 mg of cholesterol [
2], which is approximately twice the amount of cholesterol that is found in butter and freeze-dried meat products and about 5–10 times more cholesterol than that found in most dairy products [
3]. In general, an increase in the concentration of blood cholesterol is widely recognized as a risk factor for coronary artery disease [
4]. Hence, the consumption of table eggs has decreased in many developed countries during the last 4 decades due to these perceptions about cholesterol [
1]. However, some studies [
5,
6] have reported that healthy individuals can consume 1 egg per day without affecting their blood cholesterol levels or increasing their risk of cardiovascular disease.
Numerous studies have demonstrated the ability of biopolymers such as chitosan, pectin, guar gum, xanthan gum, or modified starch to function as dietary fibers by lowering blood cholesterol concentrations and reducing lipid absorption [
7]. In a previous study, Lairon
et al. reported that dietary fibers can alter the breakup and coalescence of lipid droplets in the stomach and small intestine, thereby altering the surface area of emulsified lipids exposed to digestive enzymes [
8]. Hur
et al. reported that dietary fibers from various sources can bind to bile acids as well as mixed micelle components; they also explained particle disruption of the micellization process, which leads to reduced micellar solubilization of lipids [
7]. For instance, cationic chitosan can bind to the surface of anionic lipid droplets, which are stabilized by bile salt or phospholipids, and reduce lipase activity by preventing contact between lipase and emulsified lipid substrates [
9]. However, how biopolymer encapsulation influences the digestion of total lipids and cholesterol in egg yolk remains to be elucidated. Thus, the purpose of this study was to determine the effects of biopolymer encapsulation on the digestion of total lipids and cholesterol present in egg yolk using an
in vitro human digestion model.
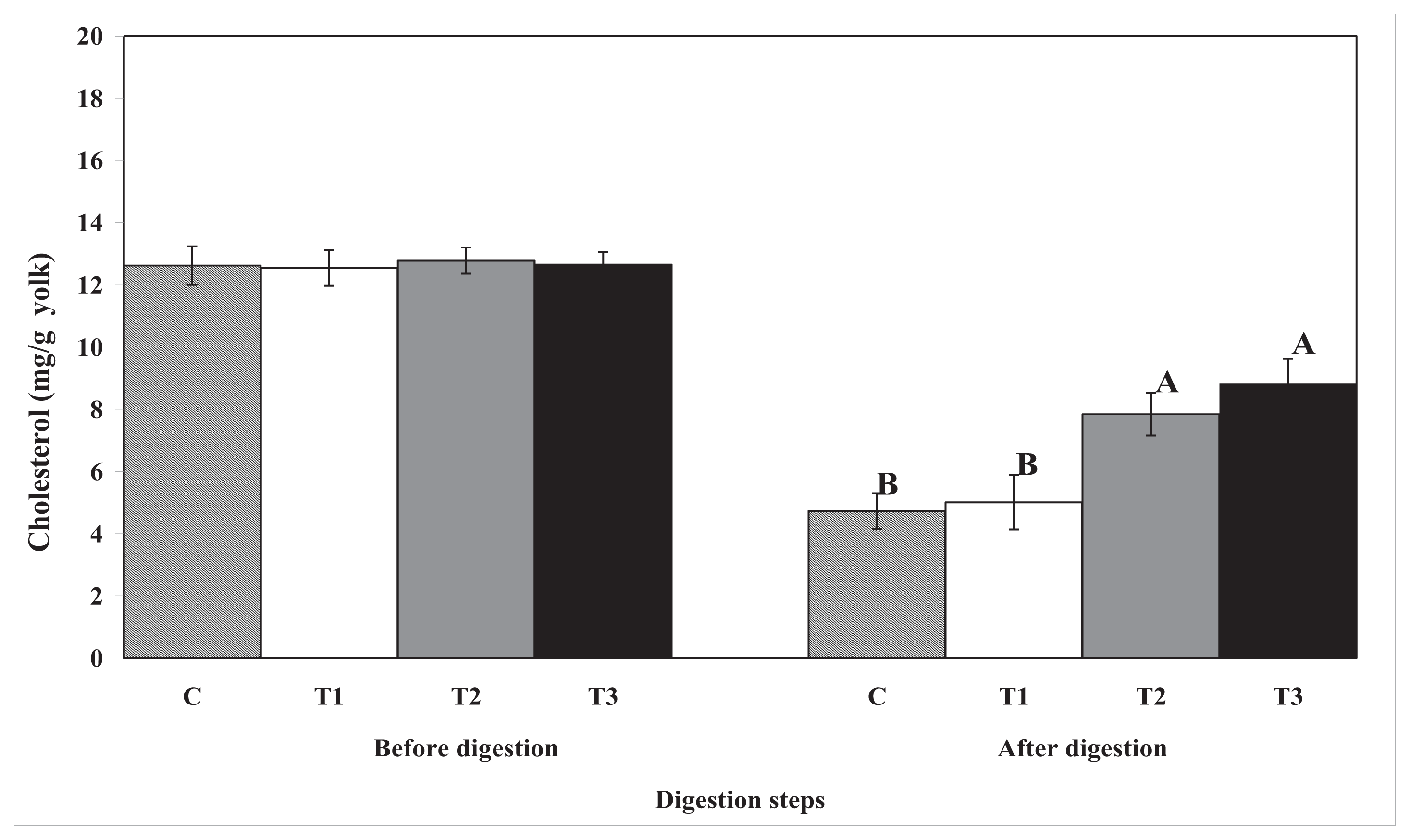
3. Discussion
A number of
in vitro studies have shown that biopolymers can alter lipid or cholesterol digestion. Dietary fibers can decrease serum cholesterol concentrations by reducing the amount of ingested (exogenous) cholesterol that is adsorbed [
10]. Lairon
et al. reported that some soluble fibers that form viscous solutions drastically reduce the rate of lipid emulsification, with a noticeable decrease in fat lipolysis [
11]. Moreover, dietary fiber from various sources can bind to bile acids, mixed micelle components such as monoacylglycerols, free fatty acids, or free cholesterol. This explains the partial disruption of the micellization process, which leads to reduced micellar solubilization of lipid moieties and the reduction in intestinal uptake of lipid moieties and cholesterol [
11–
13]. In this study, biopolymer-encapsulated egg yolk had lower digestion rates of total lipids and cholesterol in the
in vitro human digestion model. Moreover, lipase activity was reduced by biopolymer encapsulation. These results indicate that biopolymers, especially pectin and chitosan, easily encapsulate egg yolk lipids and that they can reduce lipase activity. Consequently, lipid and cholesterol digestion was prevented in the
in vitro human digestion model. In addition, biopolymer encapsulation may interfere with the absorption of total lipids (with cholesterol) into the dialysis tubing membrane (to simulate the villi in the small intestine); or, they may interfere with the ability of lipase to access the lipids contained within them. Beysseriat
et al. also reported that the ability of dietary fibers to reduce cholesterol digestion and/or absorption through this mechanism depends on their ability to promote droplet aggregation or to adsorb to lipid droplet surfaces [
9]. This depends strongly on the electrical charge, molecular weight, and structure of the fibers. For example, cationic chitosan can bind to the surface of anionic lipid droplets that are stabilized by bile salts and/or phospholipids and reduce lipase activity by preventing lipase from coming into contact with the emulsified lipid substrate [
9]. The negatively charged lipase may have acted as a bridge between the positively charged chitosan-coated droplets, causing them to clump together, and the interaction of chitosan with lipase may have caused some charge neutralization of the lipid droplets [
14].
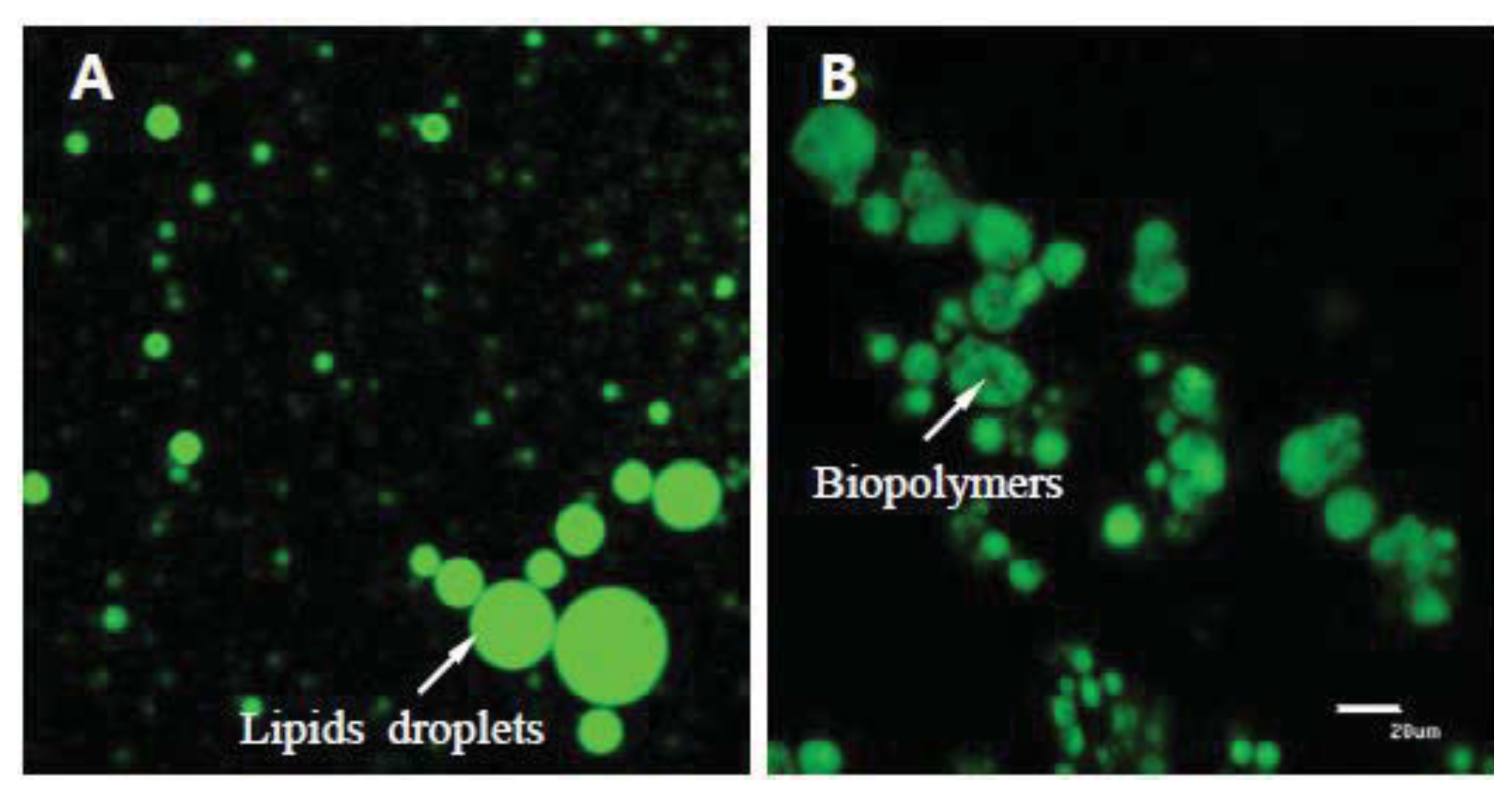
Figure 8 shows that biopolymers have the ability to aggregate lipid droplets. This aggregation would decrease the transport rate of enzymes to the lipid surfaces, as well as the transport rate of lipid digestion products present within micelles to the gastrointestinal walls. Thereby, the digestion and absorption rate of total lipids and cholesterol would be reduced.
In an animal study, Fernandez reported that pectin was found to be more effective than guar gum in guinea pigs [
15], whereas Moundras
et al. reported that guar gum was found to be more effective than pectin or gum arabic in rats [
16]. In this study, chitosan was found to be more effective for the inhibition of total lipid and cholesterol digestion including the inhibition of lipase activity than other biopolymers during
in vitro human digestion. This result may be due to the higher electrical charge of chitosan and due to the inhibitory effect of lipase activity. Cellulose had lesser effect on the inhibition of total lipid and cholesterol digestion during
in vitro human digestion, even though cellulose inhibited lipase activity. This may be because cellulose has a weak electrical charge and, consequently, has no aggregation effect with lipids. In an
in vitro study, Mun
et al. reported that chitosan reduced the amount of fatty acids released because lipid droplets were surrounded by cationic chitosan layers or because the lipids droplets were trapped within large chitosan aggregates [
17]. They also suggested that chitosan formed a protective layer around the droplets and that it promoted extensive droplet flocculation, both of which inhibited the ability of lipase to interact with the fat inside the droplets. Thus, we assume that the digestion of total lipids and cholesterol would be further reduced in chitosan-encapsulated egg yolks than in those encapsulated with other biopolymers.
Another possible mechanism for the inhibition of lipid digestion is, presumably, the increase in solution viscosity due to biopolymer encapsulation, which causes a reduction in the diffusion of molecules (such as lipase and other enzymes or mixed micelles) and other species in the gastrointestinal tract. As mentioned above, this reduction in diffusion by increasing the solution viscosity would decrease the transport rate of enzymes to the lipid surfaces, thereby reducing the digestion and absorption rates of lipids. Meyer and Doty reported that a high viscosity of the contents of the small intestine may delay lipid digestion, promoting absorption in a more distal part of the small intestine [
18]. An increase in solution viscosity is responsible for a decrease in the transit time of ingested food in the gastrointestinal tract. Therefore, an increase in solution viscosity causes a decrease in the transport rate of lipid digestion products present within micelles to the gastrointestinal walls. Khan
et al. [
19] also reported that biopolymers may interfere with the formation of micelles and/or lower the diffusion rate of bile acid and cholesterol-containing micelles through the bolus, consequently diminishing the uptake of cholesterol and bile acids. In our previous study, dietary biopolymers increased the viscosity of the contents of the small intestine [
7]. Thus, total lipid and cholesterol digestion in egg yolks can be reduced by biopolymer encapsulation because biopolymers were increased the viscosity of egg yolks in this study (data are not shown). In general, the differences between dietary fibers such as molecular weight and hydrophobicity cause differences in their physicochemical properties such as water solubility, viscosity enhancement, opacity, surface activity, and binding capacity [
20]. These differences can cause significant alterations in their effectiveness in reducing cholesterol digestion by interfering with the various physiological processes during digestion and absorption. Yamaguchi
et al. reported that low molecular weight pectins are more effective in lowering cholesterol than high molecular weight pectins [
21]. This has been attributed to the reduction in bile acid binding that occurs when the molecular weight of pectin falls below a critical value [
22]. Therefore, the effect of biopolymer encapsulation on total lipid and cholesterol digestion in egg yolk would be largely influenced by the conditions of the biopolymers such as molecular weight, hydrophobicity, viscosity, and pH. Thus, further research is needed to find the most effective biopolymer.
4. Experimental Section
4.1. Materials
Potassium chloride, sodium sulfate, sodium hydrogen carbonate, hydrogen chloride, potassium phosphate monobasic, magnesium chloride, hexane, trichloroacetic acid, ether, and ethanol were purchased from Fisher Scientific chemical company (Pittsburgh, PA, USA). Cellulose, pectin, chitosan, bicarbonate, potassium thiocyanate, sodium phosphate dibasic, sodium phosphate monobasic, sodium chloride, calcium chloride, ammonium chloride, urea, glucose sigma, glucuronic acid, glucosamine, α-amylase, uric acid, mucin, bovine serum albumin, pepsin, pancreatin, lipase, bile salt extraction, Nile red, 5α-cholestane, thiobarbituric acid, butylated hydroxyanisole, and phenolphthalein were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). Pyridine, bis-[trimethylsilyl] trifluoroacetamide, and trimethylchlorosilane were purchased from Supelco Co. (St. Louis, MO, USA).
4.2. Biopolymer Encapsulation Preparation
The experiments were performed by 5 replications from 10 different egg yolk samples. Eggs were purchased from the local market. Chitosan (10 wt%) was dissolved in acetate buffer solutions (100 mM acetic acid: sodium acetate, pH 3.0, 0–150 mM NaCl). Pectin and cellulose (10 wt% each) were dissolved in phosphate buffer solutions (2 M monobasic sodium phosphate and 2 M dibasic sodium phosphate, pH 7.0). These solutions were stirred for 12 h and then mixed for 3 h using a magnetic stirrer. During mixing, 1 mL of Tween 20 (0.1%, pH 7.6) was added dropwise to reduce surface tension and enhance encapsulation formation. The biopolymer encapsulation was prepared by mixing a final volume of 10 wt% biopolymer solution and egg yolk together for 1 h using a bio-homogenizer. The mixture was continuously stirred for 15 min using power ultrasound at a frequency of 10 MHz (final volume: whole egg yolk mixed with 1% biopolymers). This process was aimed at developing a coating layer around the lipophilic egg yolk. Encapsulation of biopolymers and egg yolk was confirmed using confocal microscopy (
Figure 8). The molecular characteristics of biopolymers are listed in
Table 1.
4.3. In Vitro Human Digestion
An
in vitro human digestion model that simulated the mouth, stomach, and small intestine was used in this study, which was a modified version of that described by Versantvoort
et al. [
23].
- (1)
Initial system: The initial egg yolk samples encapsulated with 1% biopolymers.
- (2)
Mouth: 10 g of initial egg yolk samples was mixed with 12 mL of simulated saliva solution (pH 6.8) and then stirred for 5 min at 37 °C.
- (3)
Stomach: 24 mL of simulated gastric juice (pH 2) was then added, and the mixture was stirred for 2 h at 37 °C.
- (4)
Small intestine: 24 mL of duodenal juice, 12 mL of bile juice, and 2 mL of HCO3 solution (pH 6.5 to 7) was then added. The total solution was placed in a 250 mL flask, and then the dialysis tubing (molecular weight cutoff of 50,000, flat width 34 mm, thickness 18 μm, Membrane Filtration Products, Inc. Seguin, TX, USA) containing 10 mL of phosphate buffer (pH 7) was placed in a 250 mL flask, and the mix was stirred for 2 h at 37 °C.
The compositions of the simulated saliva, gastric, duodenal, and bile juices are listed in
Table 2. During
in vitro human digestion, the samples were swirled (60 rpm) on a shaking water bath to simulate the motility of the GI tract (Model 3582, Labline Instruments, Inc., Melrose Park, IL, USA).
4.4. Total Lipid and Cholesterol Contents
Total lipids were extracted with chloroform and methanol as described by Folch
et al. [
24]. Cholesterol was determined by the modified method of Russo and others [
25]. Briefly, extracted lipid (50 mg) was added into a 50 mL tube with 10 mL of saponification reagent (30% KOH and ethanol with the ratio of 6:94) and 0.5 mL internal standard (2 mg 5
α-cholestane/sample), and then capped and incubated for 1 h at 60 °C. After cooling the sample, 8 mL of deionized distilled water and 3 mL hexane were added and mixed thoroughly and allowed to separate. The top layer (hexane layer) was removed and dried in scintillation vials, and 100 μL of bis-[trimethylsilyl]trifluoroacetamide + 1% trimethylchlorosilane (Supelco Co., Bellefonte, PA, USA) and 200 μL of pyridine were added and mixed. The samples were left to set overnight and then analyzed by gas chromatography (Agilent 6890). A ramped oven temperature condition (180 °C for 2.5 min, increased to 230 °C at 2.5 °C/min, then held at 230 °C for 7.5 min) was used. Temperatures of both the inlet and detector were 280 °C. Helium was the carrier gas at linear flow of 1.1 mL/min. Detector (flame ion detector) air, H
2, and make-up gas (He) flows were 350, 35, and 43 mL/min, respectively.
4.5. Free Fatty Acid Content
Free fatty acid content was determined by the modified method of AOAC [
26]. Free fatty acid contents were weighed by titrimetry. Briefly, 5 g of sample was weighed into a 50-mL test tube and homogenized with 15 mL of deionized distilled water using a Polytron homogenizer (IKA, Model T25, Staufen, Germany) for 10 s at the highest speed. Then, 2 mL of egg yolk homogenate was transferred to a 300 mL flask, and 100 mL of ether/ethanol solution (ether:ethanol, 1:1,
v/
v) was added. Several drops of phenolphthalein were added, and the free fatty acids were titrated with 0.1 M KOH. Free fatty acid (KOH
/g) = 5.611 ×
A ×
F/Sample weight (g),
A: volume (mL) of 0.1 M KOH solution;
F: titer of KOH.
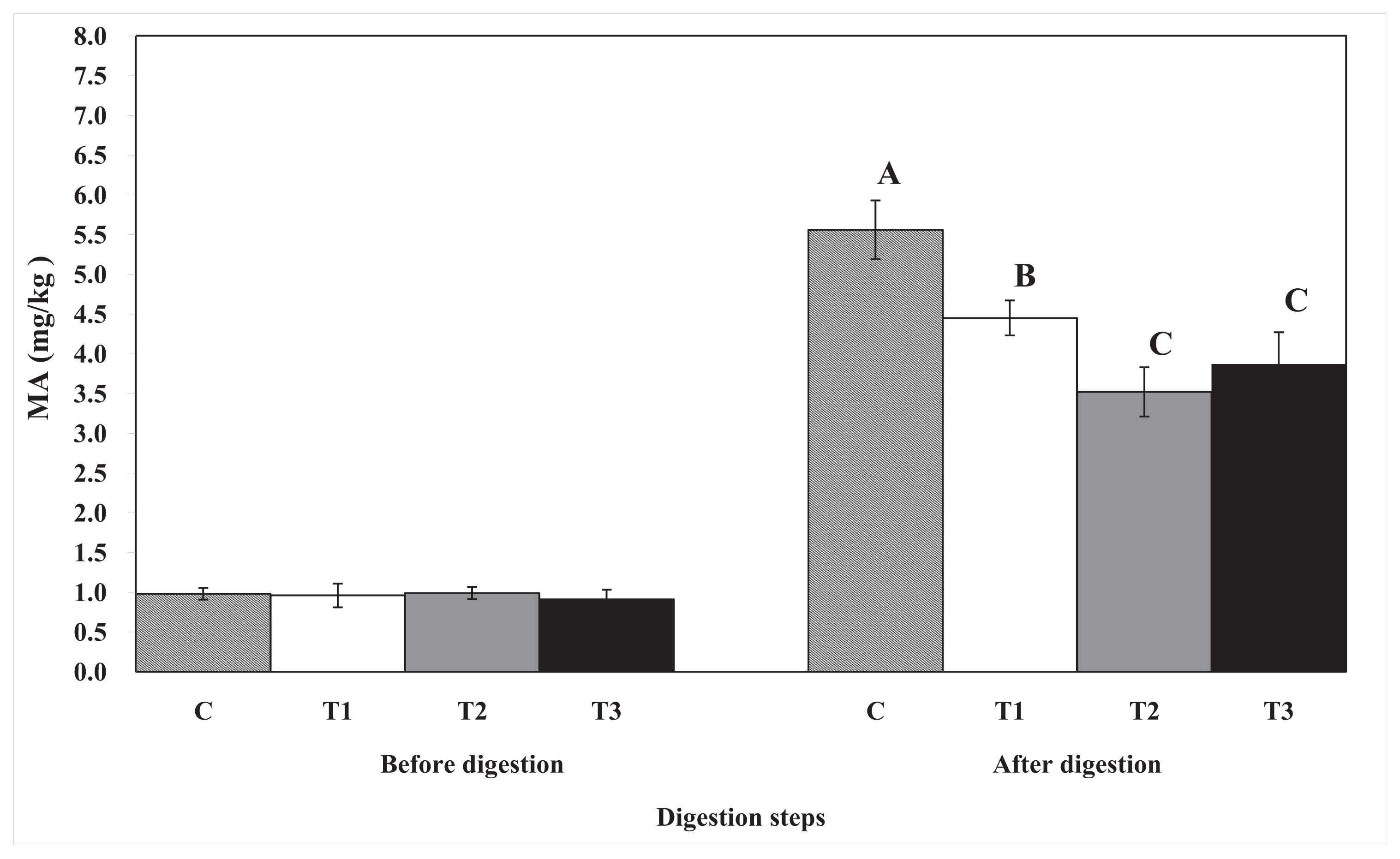
4.6. Thiobarbituric Acid-Reactive Substances
TBARS were determined by the modified method of Buege and Aust [
27]. Briefly, 5 g of egg yolk sample was weighed into a 50-mL test tube and homogenized with 15 mL of deionized distilled water using a Polytron homogenizer for 10 s at the highest speed (before digestion samples only). Then, 1 mL of egg yolk homogenate was transferred to a disposable test tube (3 × 100 mm), and butylated hydroxyanisole (50 μL, 10%) and thiobarbituric acid/trichloroacetic acid (TBA/TCA) (2 mL) were added. The mixture was vortexed and then incubated in a boiling water bath for 15 min to develop color. The sample was cooled in cold water for 5 min, vortexed again, and centrifuged for 15 min at 2000×
g. The absorbance of the resulting supernatant solution was determined at 531 nm against a blank containing 1 mL of DDW and 2 mL of TBA/TCA solution. The amounts of TBARS were expressed as milligrams of malondialdehyde (MA) per kilogram of egg yolk samples.
4.7. Lipase Activity
Lipase activity was determined by the modified method of Gooda Sahib
et al. [
28]. Porcine lipase was dissolved in 0.01 M Tris-HCl buffer (25 units
/mL).
In vitro digested solutions were dissolved in 0.01 M Tris-HCl buffer at different concentrations (7.81–250 ppm). Olive oil (10%
v/v) was mixed with Arabic gum (10 g) mixture (10%
w/v in 0.1 M), (1.57 g) Tris-HCl buffer, pH 2, 0.5 M (2.92 g) NaCl, and 20 mM (0.02 g) CaCl
2 using a homogenizer. Then, 0.2 mL of lipase solution (25 units) was allowed to react with 0.5 mL of
in vitro digested solution for 30 min at 4 °C. Reconstituted substrate emulsion (2 mL) was then added, and the mixture was incubated for 30 min at 37 °C. The reaction was stopped using acetone and ethanol mixture (1:1/
v:v) and titrated with 0.02 M NaOH to pH 9.4. Titrations were carried out using an automatic titrator (785 DMP Titrino, Metrohm, Herisau, Switzerland).
4.8. Confocal Laser Scanning Microscopy
The total lipid content and biopolymer encapsulation were analyzed through confocal laser scanning microscopy. A confocal scanning fluorescence microscope (Carl Zeiss, LSM 5 Live, GmbH, Jena, Germany) with a 20× objective lens was used to capture confocal images. Nile red (a lipid fluorescent dye) was excited with a 488-nm argon laser line. The fluorescence emitted from the sample was monitored using a fluorescence detector (543 nm) with a pinhole size of 150 μm. The resulting images consisted of 512 × 512 pixels, with a pixel size of 414 nm and a pixel dwell time of 5 s.
4.9. Particle Size
The particle size of the sample was measured using a laser light scattering instrument (Mastersizer X, Malvern Instruments Ltd., Malvern, UK). This instrument is based on diffraction of a monochromatic beam of laser light (λ = 632.8 nm) when it is scattered by the droplets in a dilute egg yolk. The instrument measures the angular dependence of the intensity of laser light diffraction and finds the particle size that gives the best fit to the experimental measurements and predictions based on light scattering theory. The mean particle size was reported as the surface-weighted mean diameter, d32 (= ∑nidi3 ∑nidi2), where ni is the number of particles with diameter di.
4.10. Statistics
The effect of biopolymer encapsulation on the digestion of total lipids and cholesterol in egg yolks during in vitro human digestion were analyzed using SAS software (SAS Inst. Inc., Cary, NC, USA) by the generalized linear model procedure. The Student-Newman-Keuls multiple range test was used to compare differences between means.