Comparative Analysis of Osteogenic/Chondrogenic Differentiation Potential in Primary Limb Bud-Derived and C3H10T1/2 Cell Line-Based Mouse Micromass Cultures
Abstract
:1. Introduction
2. Results
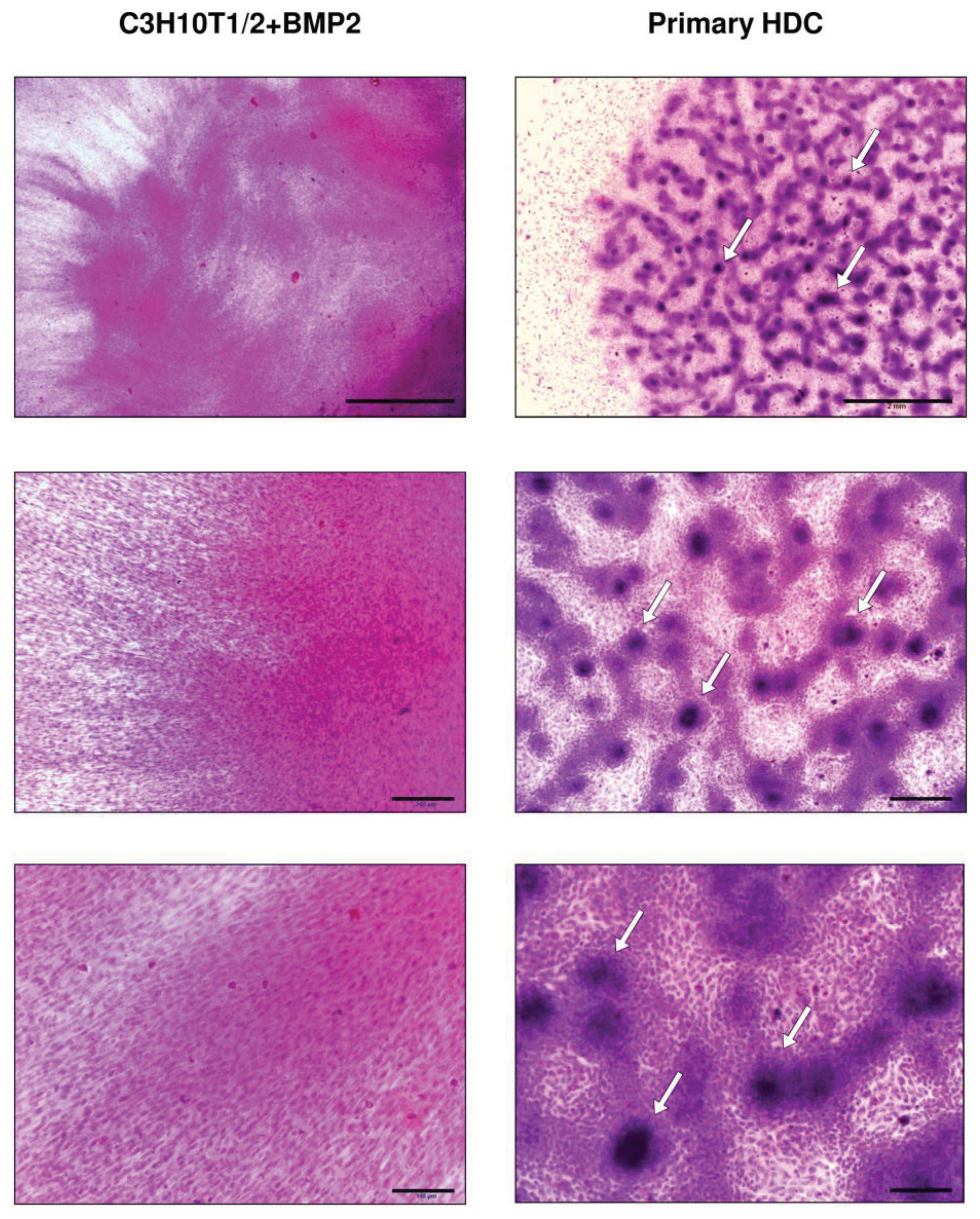
2.1. BMP-2 Overexpressing C3H10T1/2-Derived and Embryonic Limb Bud-Derived Micromass Cultures Show Different Morphology
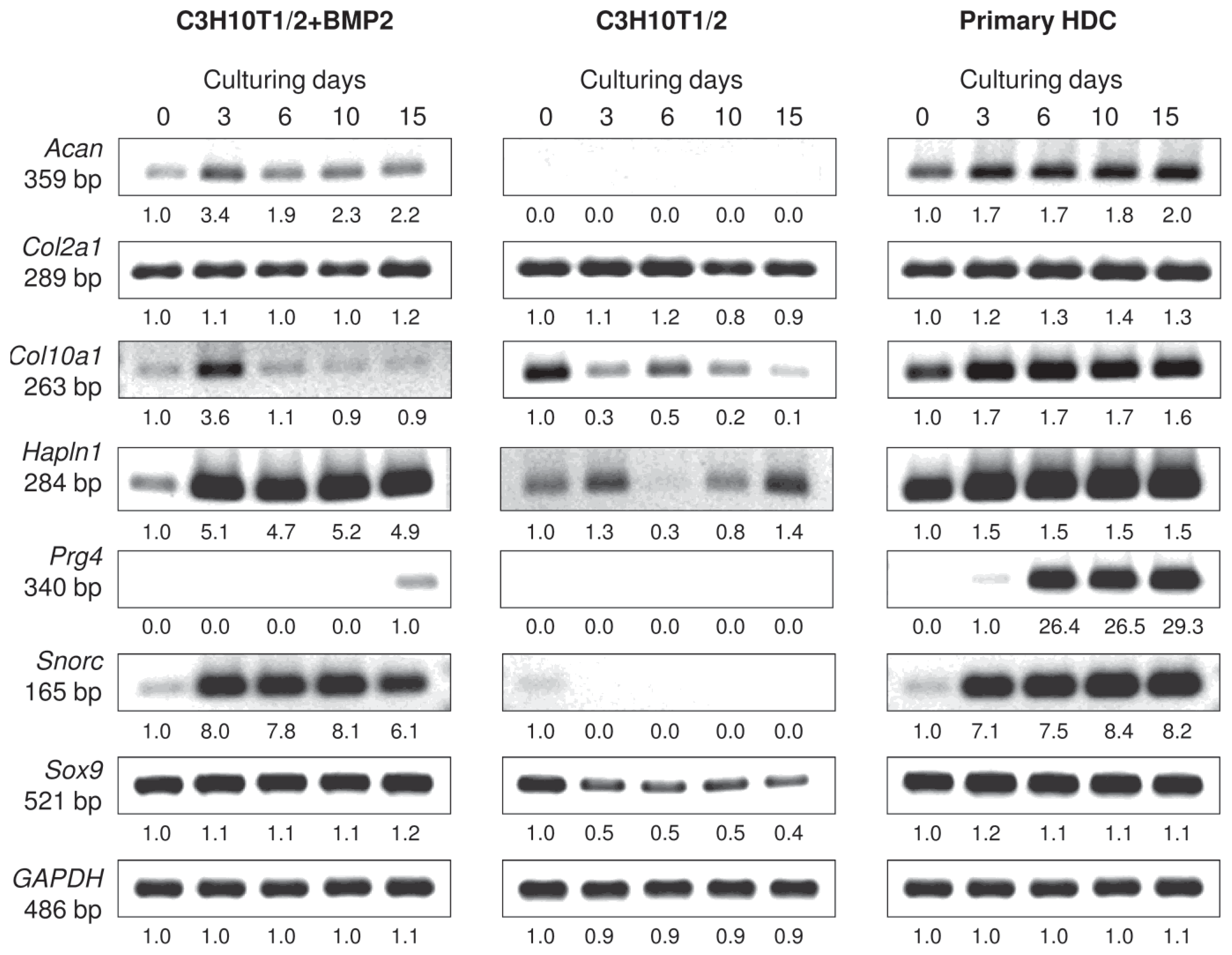
2.2. Micromass Cultures Established from either BMP-2 Overexpressing C3H10T1/2 Cells or Primary Embryonic Limb Bud-Derived Cells Undergo Chondrogenic Differentiation
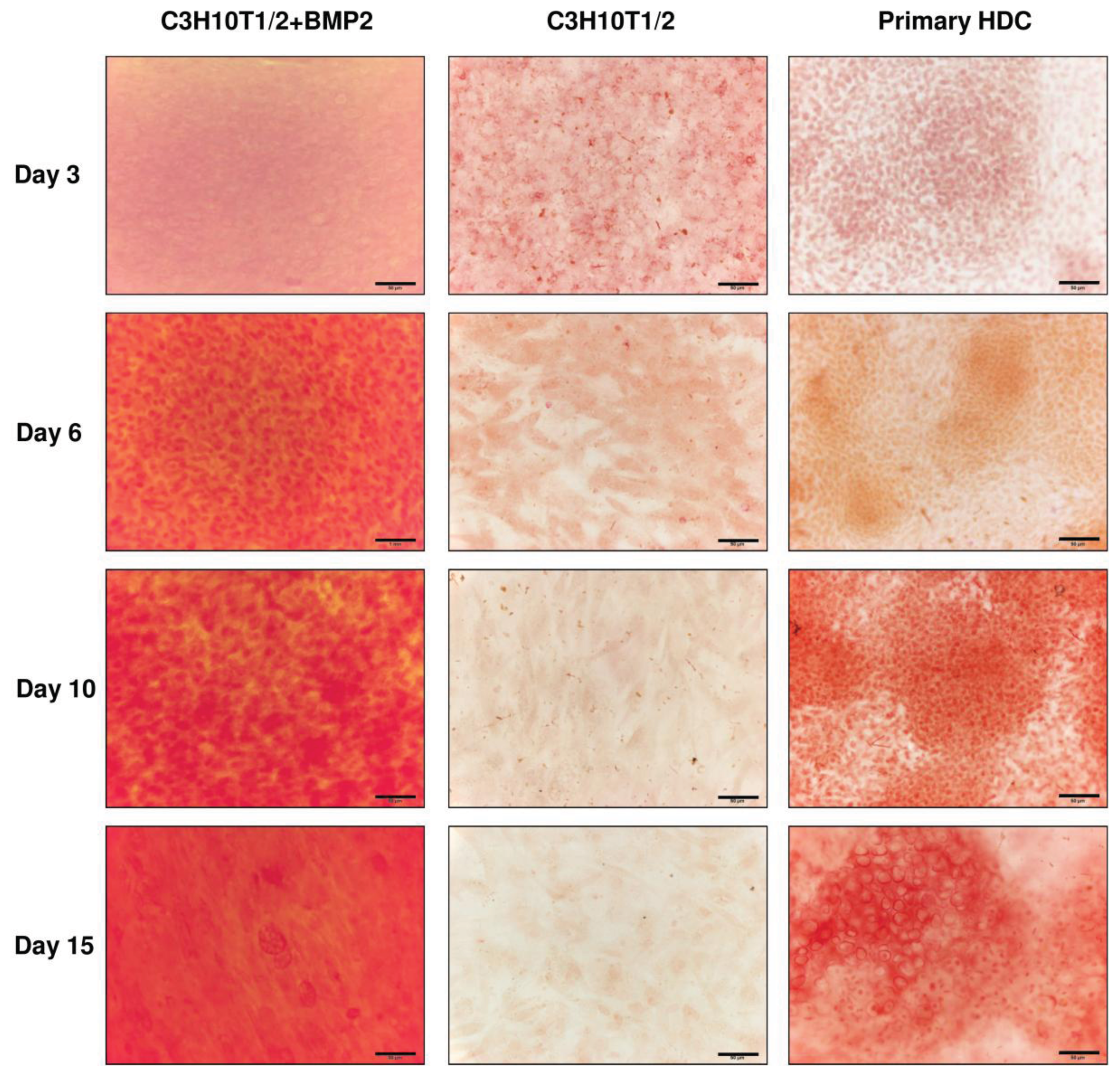
2.3. Matrix Calcification and Osteogenic Differentiation of C3H10T1/2 Cell Line or Primary Embryonic Limb Bud-Derived Micromass Cultures
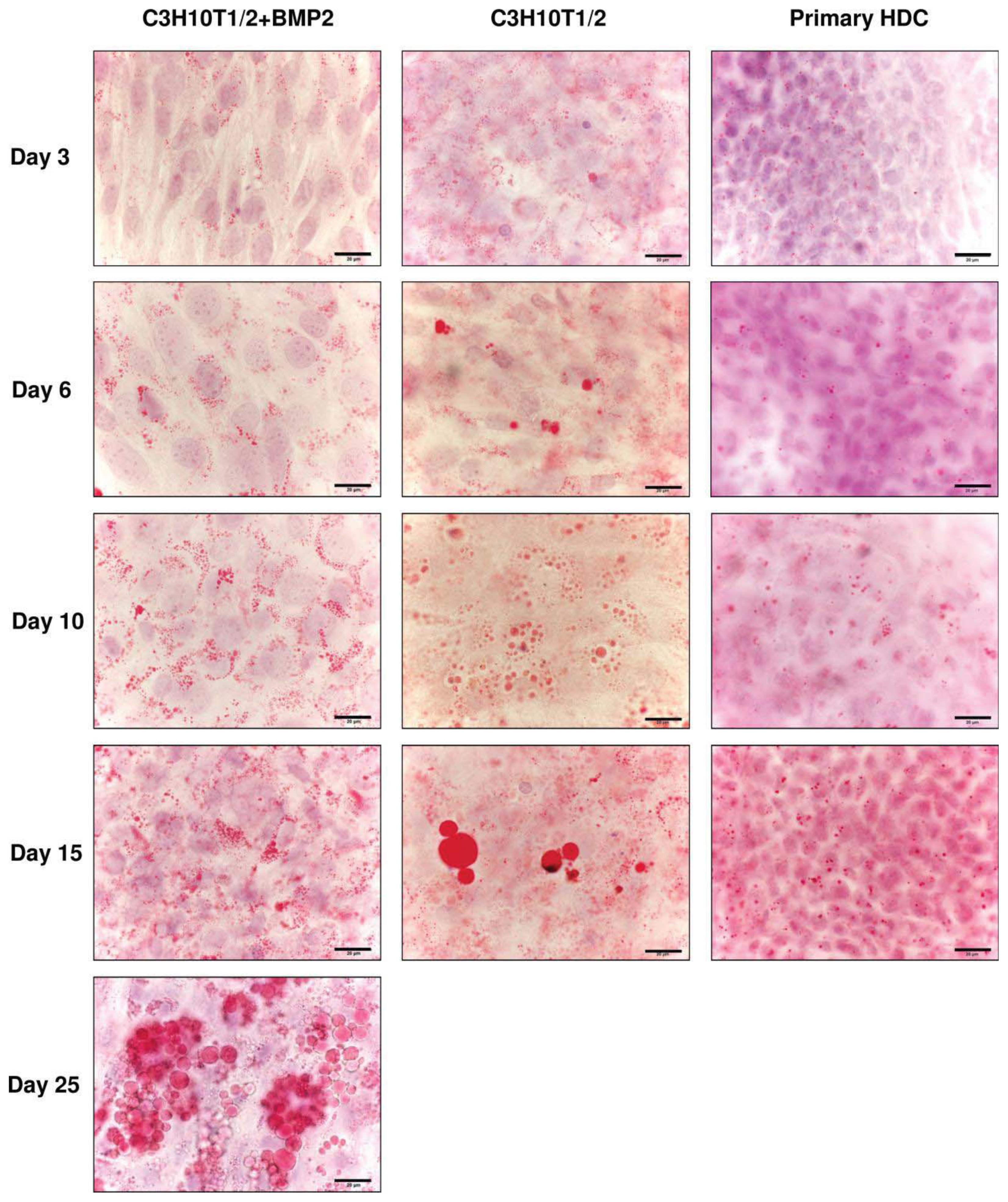
2.4. Differentiation towards the Adipogenic Lineage Is also Characteristic of the Micromass Models Studied
2.5. mRNA Transcripts of Pluripotency Factors Are Detectable Even at Later Stages in Micromass Cultures
3. Discussion
4. Experimental Section
4.1. Cell Culturing
4.1.1. Micromass Cultures Established from C3H10T1/2 Cells
4.1.2. Primary Embryonic Mesenchymal Micromass Cultures
4.2. mRNA Expression Analysis Using Reverse Transcription Followed by PCR
4.3. Histological Analysis of Micromass Cultures
4.3.1. Investigation of Cellular Morphology of Micromass Cultures by Conventional Haematoxylin and Eosin Staining
4.3.2. Assessment of Chondrogenic Differentiation by Low-pH Metachromatic Staining with Dimethyl-Methylene Blue
4.3.3. Assessment of Matrix Mineralisation by Alizarin Red S Staining
4.3.4. Assessment of Lipid Accumulation by Staining with Oil Red O
5. Conclusions
Supplementary Information
ijms-14-16141-s001.pdfAcknowledgments
Conflict of Interest
Abbreviations
| AP | alkaline phosphatase |
| BMP | bone morphogenic protein |
| CMF-PBS | calcium and magnesium free PBS |
| DMEM | Dulbecco’s modified essential medium |
| DMMB | dimethyl-methylene blue |
| dNTP | deoxy nucleotide triphosphate |
| ECM | extracellular matrix |
| EDTA | ethylene diamine tetra-acetic acid |
| ERK | extracellular signal-regulated kinase |
| ESC | embryonic stem cell |
| FABP | fatty acid binding protein |
| FCS | foetal calf serum |
| FGF | fibroblast growth factor |
| GDF | growth and differentiation factor |
| HD | high density |
| HDC | high density culture |
| HE | haematoxylin-eosin |
| hMSC | human mesenchymal stem cell |
| IGF | insulin-like growth factor |
| MAPK | mitogen activated protein kinase |
| N-CAM | neuronal cell adhesion molecule |
| OC | osteocalcin |
| OP | osteopontin |
| Osx | osterix |
| PBS | phosphate buffered saline |
| PPAR gamma | peroxisome proliferator-activated receptor gamma |
| RT-PCR | reverse transcription followed by polymerase chain reaction |
| TGF-β | transforming growth factor beta |
References
- Delise, A.M.; Tuan, R.S. Analysis of N-cadherin function in limb mesenchymal chondrogenesis in vitro. Dev. Dyn 2002, 225, 195–204. [Google Scholar]
- Denker, A.E.; Haas, A.R.; Nicoll, S.B.; Tuan, R.S. Chondrogenic differentiation of murine C3H10T1/2 multipotential mesenchymal cells: I. Stimulation by bone morphogenetic protein-2 in high-density micromass cultures. Differentiation 1999, 64, 67–76. [Google Scholar]
- Lee, J.W.; Kim, Y.H.; Kim, S.H.; Han, S.H.; Hahn, S.B. Chondrogenic differentiation of mesenchymal stem cells and its clinical applications. Yonsei Med. J 2004, 45, 41–47. [Google Scholar]
- Horton, W.A.; Hall, J.G.; Hecht, J.T. Achondroplasia. Lancet 2007, 370, 162–172. [Google Scholar]
- Ahrens, P.B.; Solursh, M.; Reiter, R.S. Stage-related capacity for limb chondrogenesis in cell culture. Dev. Biol 1977, 60, 69–82. [Google Scholar]
- Oh, C.D.; Chang, S.H.; Yoon, Y.M.; Lee, S.J.; Lee, Y.S.; Kang, S.S.; Chun, J.S. Opposing role of mitogen-activated protein kinase subtypes, erk-1/2 and p38, in the regulation of chondrogenesis of mesenchymes. J. Biol. Chem 2000, 275, 5613–5619. [Google Scholar]
- Murakami, S.; Kan, M.; McKeehan, W.L.; de Crombrugghe, B. Up-regulation of the chondrogenic Sox9 gene by fibroblast growth factors is mediated by the mitogen-activated protein kinase pathway. Proc. Natl. Acad. Sci. USA 2000, 97, 1113–1118. [Google Scholar]
- Seghatoleslami, M.R.; Roman-Blas, J.A.; Rainville, A.M.; Modaressi, R.; Danielson, K.G.; Tuan, R.S. Progression of chondrogenesis in C3H10T1/2 cells is associated with prolonged and tight regulation of ERK1/2. J. Cell. Biochem 2003, 88, 1129–1144. [Google Scholar]
- Daumer, K.M.; Tufan, A.C.; Tuan, R.S. Long-term in vitro analysis of limb cartilage development: Involvement of Wnt signaling. J. Cell. Biochem 2004, 93, 526–541. [Google Scholar]
- Hosseini-Farahabadi, S.; Geetha-Loganathan, P.; Fu, K.; Nimmagadda, S.; Yang, H.J.; Richman, J.M. Dual functions for WNT5A during cartilage development and in disease. Matrix Biol 2013, 32, 252–264. [Google Scholar]
- Lee, Y.S.; Chuong, C.M. Activation of protein kinase A is a pivotal step involved in both BMP-2- and cyclic AMP-induced chondrogenesis. J. Cell. Physiol 1997, 170, 153–165. [Google Scholar]
- Solursh, M.; Reiter, R.; Ahrens, P.B.; Pratt, R.M. Increase in levels of cyclic AMP during avian limb chondrogenesis in vitro. Differentiation 1979, 15, 183–186. [Google Scholar]
- Widelitz, R.B.; Jiang, T.X.; Murray, B.A.; Chuong, C.M. Adhesion molecules in skeletogenesis: II. Neural cell adhesion molecules mediate precartilaginous mesenchymal condensations and enhance chondrogenesis. J. Cell. Physiol 1993, 156, 399–411. [Google Scholar]
- Zakany, R.; Bako, E.; Felszeghy, S.; Hollo, K.; Balazs, M.; Bardos, H.; Gergely, P.; Modis, L. Okadaic acid-induced inhibition of protein phosphatase 2A enhances chondrogenesis in chicken limb bud micromass cell cultures. Anat. Embryol 2001, 203, 23–34. [Google Scholar]
- Cash, D.E.; Bock, C.B.; Schughart, K.; Linney, E.; Underhill, T.M. Retinoic acid receptor alpha function in vertebrate limb skeletogenesis: A modulator of chondrogenesis. J. Cell. Biol 1997, 136, 445–457. [Google Scholar]
- Hdud, I.M.; El-Shafei, A.A.; Loughna, P.; Barrett-Jolley, R.; Mobasheri, A. Expression of transient receptor potential vanilloid (TRPV) channels in different passages of articular chondrocytes. Int. J. Mol. Sci 2012, 13, 4433–4445. [Google Scholar]
- Atsumi, T.; Miwa, Y.; Kimata, K.; Ikawa, Y. A chondrogenic cell line derived from a differentiating culture of AT805 teratocarcinoma cells. Cell. Differ. Dev 1990, 30, 109–116. [Google Scholar]
- Grigoriadis, A.E.; Heersche, J.N.; Aubin, J.E. Differentiation of muscle, fat, cartilage, and bone from progenitor cells present in a bone-derived clonal cell population: Effect of dexamethasone. J. Cell. Biol 1988, 106, 2139–2151. [Google Scholar]
- Roy, R.; Kudryashov, V.; Doty, S.B.; Binderman, I.; Boskey, A.L. Differentiation and mineralization of murine mesenchymal C3H10T1/2 cells in micromass culture. Differentiation 2010, 79, 211–217. [Google Scholar]
- Ahrens, M.; Ankenbauer, T.; Schroder, D.; Hollnagel, A.; Mayer, H.; Gross, G. Expression of human bone morphogenetic proteins-2 or -4 in murine mesenchymal progenitor C3H10T1/2 cells induces differentiation into distinct mesenchymal cell lineages. DNA Cell Biol 1993, 12, 871–880. [Google Scholar]
- Gould, R.P.; Day, A.; Wolpert, L. Mesenchymal condensation and cell contact in early morphogenesis of the chick limb. Exp. Cell Res 1972, 72, 325–336. [Google Scholar]
- Kostovic-Knezevic, L.; Bradamante, Z.; Svajger, A. Ultrastructure of elastic cartilage in the rat external ear. Cell. Tissue Res 1981, 218, 149–160. [Google Scholar]
- Edwall-Arvidsson, C.; Wroblewski, J. Characterization of chondrogenesis in cells isolated from limb buds in mouse. Anat. Embryol 1996, 193, 453–461. [Google Scholar]
- Zhang, X.; Ziran, N.; Goater, J.J.; Schwarz, E.M.; Puzas, J.E.; Rosier, R.N.; Zuscik, M.; Drissi, H.; O’Keefe, R.J. Primary murine limb bud mesenchymal cells in long-term culture complete chondrocyte differentiation: TGF-beta delays hypertrophy and PGE2 inhibits terminal differentiation. Bone 2004, 34, 809–817. [Google Scholar]
- Taipaleenmaki, H.; Harkness, L.; Chen, L.; Larsen, K.H.; Saamanen, A.M.; Kassem, M.; Abdallah, B.M. The crosstalk between transforming growth factor-beta1 and delta like-1 mediates early chondrogenesis during embryonic endochondral ossification. Stem Cells 2012, 30, 304–313. [Google Scholar]
- Duprez, D.M.; Coltey, M.; Amthor, H.; Brickell, P.M.; Tickle, C. Bone morphogenetic protein-2 (BMP-2) inhibits muscle development and promotes cartilage formation in chick limb bud cultures. Dev. Biol 1996, 174, 448–452. [Google Scholar]
- Hogan, B.L. Bone morphogenetic proteins: Multifunctional regulators of vertebrate development. Genes Dev 1996, 10, 1580–1594. [Google Scholar]
- Wang, E.A.; Israel, D.I.; Kelly, S.; Luxenberg, D.P. Bone morphogenetic protein-2 causes commitment and differentiation in C3H10T1/2 and 3T3 cells. Growth Factors 1993, 9, 57–71. [Google Scholar]
- Haas, A.R.; Tuan, R.S. Chondrogenic differentiation of murine C3H10T1/2 multipotential mesenchymal cells: II. Stimulation by bone morphogenetic protein-2 requires modulation of N-cadherin expression and function. Differentiation 1999, 64, 77–89. [Google Scholar]
- Heinonen, J.; Taipaleenmaki, H.; Roering, P.; Takatalo, M.; Harkness, L.; Sandholm, J.; Uusitalo-Jarvinen, H.; Kassem, M.; Kiviranta, I.; Laitala-Leinonen, T.; et al. Snorc is a novel cartilage specific small membrane proteoglycan expressed in differentiating and articular chondrocytes. Osteoarthritis Cartilage 2011, 19, 1026–1035. [Google Scholar]
- Chen, T.; Hilton, M.J.; Brown, E.B.; Zuscik, M.J.; Awad, H.A. Engineering superficial zone features in tissue engineered cartilage. Biotechnol. Bioeng 2013, 110, 1476–1486. [Google Scholar]
- Eerola, I.; Salminen, H.; Lammi, P.; Lammi, M.; von der Mark, K.; Vuorio, E.; Saamanen, A.M. Type X collagen, a natural component of mouse articular cartilage: Association with growth, aging, and osteoarthritis. Arthritis. Rheum 1998, 41, 1287–1295. [Google Scholar]
- O’Keefe, R.J.; Puzas, J.E.; Loveys, L.; Hicks, D.G.; Rosier, R.N. Analysis of type II and type X collagen synthesis in cultured growth plate chondrocytes by in situ hybridization: Rapid induction of type X collagen in culture. J. Bone Miner. Res 1994, 9, 1713–1722. [Google Scholar]
- Komori, T. Regulation of osteoblast differentiation by transcription factors. J. Cell. Biochem 2006, 99, 1233–1239. [Google Scholar]
- Stanton, L.A.; Beier, F. Inhibition of p38 MAPK signaling in chondrocyte cultures results in enhanced osteogenic differentiation of perichondral cells. Exp. Cell Res 2007, 313, 146–155. [Google Scholar]
- Matsubara, T.; Kida, K.; Yamaguchi, A.; Hata, K.; Ichida, F.; Meguro, H.; Aburatani, H.; Nishimura, R.; Yoneda, T. BMP2 regulates Osterix through Msx2 and Runx2 during osteoblast differentiation. J. Biol. Chem 2008, 283, 29119–29125. [Google Scholar]
- Shea, C.M.; Edgar, C.M.; Einhorn, T.A.; Gerstenfeld, L.C. BMP treatment of C3H10T1/2 mesenchymal stem cells induces both chondrogenesis and osteogenesis. J. Cell. Biochem 2003, 90, 1112–1127. [Google Scholar]
- Shan, T.; Liu, W.; Kuang, S. Fatty acid binding protein 4 expression marks a population of adipocyte progenitors in white and brown adipose tissues. FASEB J 2013, 27, 277–287. [Google Scholar]
- Tontonoz, P.; Hu, E.; Spiegelman, B.M. Stimulation of adipogenesis in fibroblasts by PPAR gamma 2, a lipid-activated transcription factor. Cell 1994, 79, 1147–1156. [Google Scholar]
- Pringle, S.; de Bari, C.; Dell’Accio, F.; Przyborski, S.; Cooke, M.J.; Minger, S.L.; Grigoriadis, A.E. Mesenchymal differentiation propensity of a human embryonic stem cell line. Cell Prolif 2011, 44, 120–127. [Google Scholar]
- Fodor, J.; Matta, C.; Olah, T.; Juhasz, T.; Takacs, R.; Toth, A.; Dienes, B.; Csernoch, L.; Zakany, R. Store-operated calcium entry and calcium influx via voltage-operated calcium channels regulate intracellular calcium oscillations in chondrogenic cells. Cell. Calcium 2013, 54, 1–16. [Google Scholar]
- ImageJ. Image Processing and Analysis in Java. Available online: http://rsbweb.nih.gov/ij/ (accessed on 8 July 2013).
- Muhammad, H.; Schminke, B.; Miosge, N. Current concepts in stem cell therapy for articular cartilage repair. Expert Opin. Biol. Ther 2013, 13, 541–548. [Google Scholar]




| Gene | Primer | Nucleotide sequence (5′→3′) | GenBank Accession No. | Annealing temperature | Amplicon size (bp) |
|---|---|---|---|---|---|
| 1. Chondrogenic marker genes | |||||
| Sox9 | sense | GTA CCC GCA TCT GCA CAA CG (378–397) | NM_011448 | 62 °C | 521 |
| antisense | GTG GCA AGT ATT GGT CAA ACT CAT T (874–898) | ||||
| Aggrecan core protein (Acan) | sense | CGG GAA GGT TGC TAT GGT G (782–800) | NM_007424.2 | 59 °C | 359 |
| antisense | CCT GTC TGG TTG GCG TGT A (1122–1140) | ||||
| Collagen II (Col2A1) | sense | AAA GAC GGT GAG ACG GGA GC (1900–1919) | NM_001113515 | 63 °C | 289 |
| antisense | GAC CAT CAG TAC CAG GAG TGC C (2167–2188) | ||||
| Hapln1 | sense | GGC TCA GGA ATC CAC AAA (217–234) | BC066853 | 55 °C | 284 |
| antisense | GGA AAG TAA GGG AAC ACC A (482–500) | ||||
| Lubricin (Prg4) | sense | CGA GGT CAT TAT TTC TGG (64–81) | NM_021400 | 51 °C | 340 |
| antisense | TCA TTG GCT CCT GTT TAT (386–403) | ||||
| Snorc | sense | CCC TGT GGA ACG AGC CTA T (101–119) | NM_028473 | 58 °C | 165 |
| antisense | CAA GCG ATC CTC CAT CCT G (247–265) | ||||
| 2. Osteogenic marker genes | |||||
| Alkaline phosphatase (ALPL) | sense | GAA GTC CGT GGG CAT CGT (474–491) | NM_007431 | 59 °C | 346 |
| antisense | CAG TGC GGT TCC AGA CAT AG (801–820) | ||||
| Collagen I (Col1A1) | sense | GGG CGA GTG CTG TGC TTT (237–254) | BC050014 | 62 °C | 388 |
| antisense | GGG ACC CAT TGG ACC TGA A (606–624) | ||||
| Collagen X (Col10A1) | sense | TTC TGG GAT GCC GCT TGT C (1602–1620) | NM_009925 | 61 °C | 263 |
| antisense | TCG TAG GCG TGC CGT TCT T (1846–1864) | ||||
| Osteocalcin | sense | AGC AGG AGG GCA ATA AGG (110–127) | NM_007541 | 57 °C | 165 |
| antisense | CGT AGA TGC GTT TGT AGG C (256–274) | ||||
| Osteopontin | sense | GCT GAA GCC TGA CCC ATC T (126–144) | X51834 | 59 °C | 494 |
| antisense | TCC CGT TGC TGT CCT GAT (602–619) | ||||
| Osterix | sense | CCC TTC CCT CAC TCA TTT CC (271–290) | AF184902 | 59 °C | 424 |
| antisense | CAA CCG CCT TGG GCT TAT (677–694) | ||||
| Runx2 | sense | GGA CGA GGC AAG AGT TTC A (595–613) | NM_001146038 | 58 °C | 249 |
| antisense | TGG TGC AGA GTT CAG GGA G (825–843) | ||||
| 3. Adipogenic marker genes | |||||
| Pparg2 | sense | TGC CTA TGA GCA CTT CAC (62–79) | AY208183 | 52 °C | 258 |
| antisense | TGA TCG CAC TTT GGT ATT (302–319) | ||||
| FABP4 | sense | AAA GAA GTG GGA GTG GGC (64–81) | NM_024406 | 58 °C | 173 |
| antisense | CTG TCG TCT GCG GTG ATT (219–236) | ||||
| 4. Pluripotency factors | |||||
| Nanog | sense | GCC CTG ATT CTT CTA CCA (194–211) | AY278951 | 54 °C | 383 |
| antisense | AGA TGC GTT CAC CAG ATA G (558–576) | ||||
| OCT4 (Pou5f1) | sense | GCA CGA GTG GAA AGC AAC (286–303) | NM_013633 | 56 °C | 453 |
| antisense | CGG GCA CTT CAG AAA CAT (721–738) | ||||
| Sox2 | sense | AAC CAG CGC ATG GAC AGC (466–483) | U31967 | 63 °C | 281 |
| antisense | TCG GAC TTG ACC ACA GAG CC (727–746) | ||||
| 5. Control gene | |||||
| GAPDH | sense | TGG CAA AGT GGA GAT TGT TG (69–88) | NM_008084 | 60 °C | 486 |
| antisense | GTC TTC TGG GTG GCA GTG AT (535–554) | ||||
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Takács, R.; Matta, C.; Somogyi, C.; Juhász, T.; Zákány, R. Comparative Analysis of Osteogenic/Chondrogenic Differentiation Potential in Primary Limb Bud-Derived and C3H10T1/2 Cell Line-Based Mouse Micromass Cultures. Int. J. Mol. Sci. 2013, 14, 16141-16167. https://doi.org/10.3390/ijms140816141
Takács R, Matta C, Somogyi C, Juhász T, Zákány R. Comparative Analysis of Osteogenic/Chondrogenic Differentiation Potential in Primary Limb Bud-Derived and C3H10T1/2 Cell Line-Based Mouse Micromass Cultures. International Journal of Molecular Sciences. 2013; 14(8):16141-16167. https://doi.org/10.3390/ijms140816141
Chicago/Turabian StyleTakács, Roland, Csaba Matta, Csilla Somogyi, Tamás Juhász, and Róza Zákány. 2013. "Comparative Analysis of Osteogenic/Chondrogenic Differentiation Potential in Primary Limb Bud-Derived and C3H10T1/2 Cell Line-Based Mouse Micromass Cultures" International Journal of Molecular Sciences 14, no. 8: 16141-16167. https://doi.org/10.3390/ijms140816141






