Quorum Sensing and Phytochemicals
Abstract
:1. Introduction
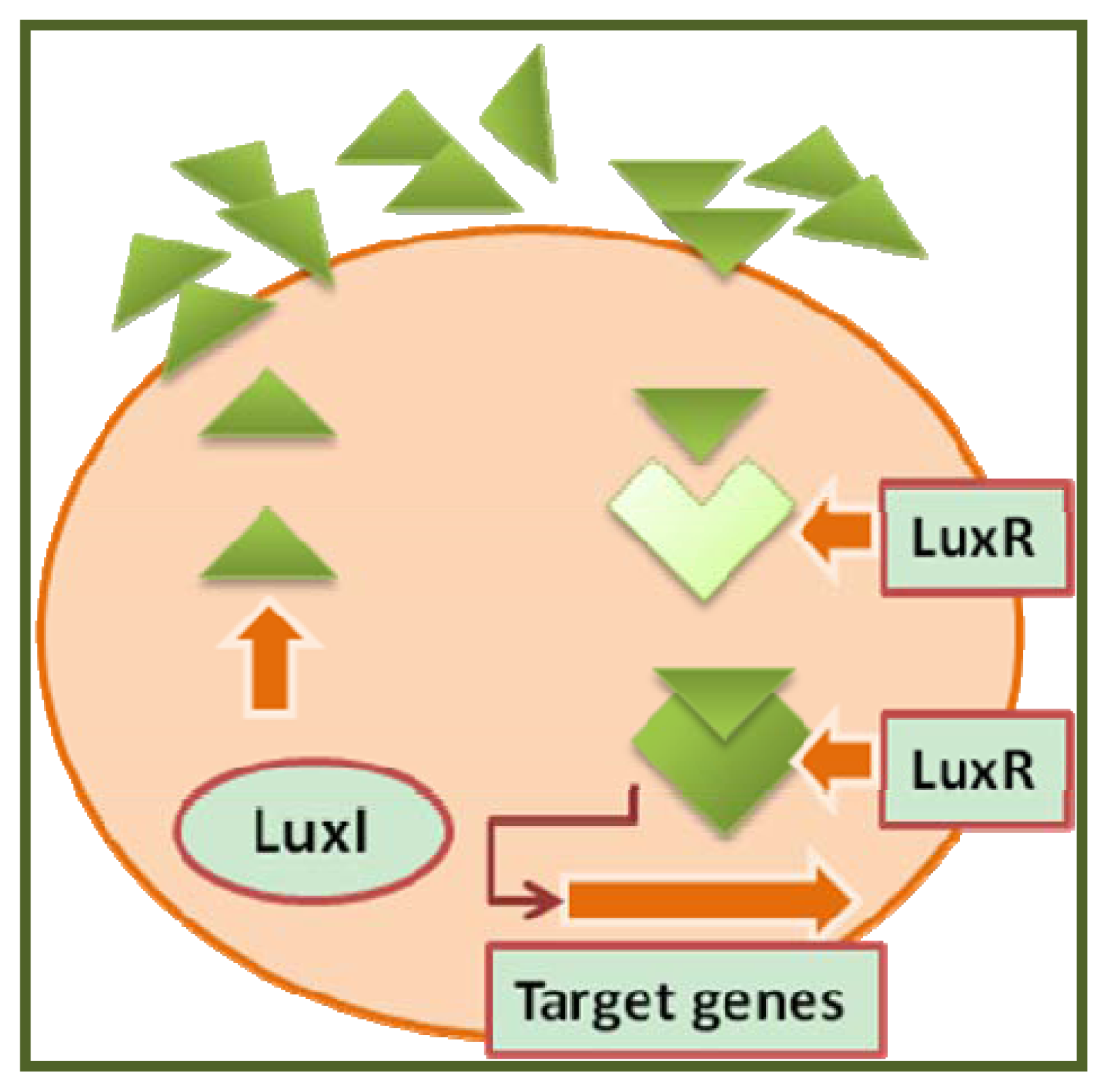
- The autoinducers;
- The signal synthase;
- The signal receptor;
- The signal response regulator;
- The regulated genes (which form the so-called QS regulon).
2. Quorum System in Gram Positive Bacteria
3. Antiquorum Sensing Activity of Phytochemicals
4. Conclusions
Conflict of Interest
References
- Bassler, B.L. Small talk: Cell-to-cell communication in bacteria. Cell 2002, 109, 421–424. [Google Scholar]
- Reading, N.C.; Sperandio, V. Quorum sensing: The many languages of bacteria. FEMS Microbiol. Lett 2006, 254, 1–11. [Google Scholar]
- McClean, K.H.; Winson, M.K.; Fish, L.; Taylor, A.; Chhabra, S.R.; Camara, M.; Daykin, M.; Lamb, J.H.; Swift, S.; Bycroft, B.W.; et al. Quorum sensing and Chromobacterium violaceum: Exploitation of violacein production and inhibition for the detection of N-acylhomoserine lactones. Microbiology 1997, 143, 3703–3711. [Google Scholar]
- Miller, M.B.; Bassler, B.L. Quorum sensing in bacteria. Annu. Rev. Microbiol 2001, 55, 165–199. [Google Scholar]
- Chernin, L.S.; Winson, M.K.; Thompson, J.M.; Haran, S.; Bycroft, B.W.; Chet, I.; Williams, P.; Gordon, S.; Stewart, A.B. Chitinolytic activity in Chromobacterium violaceum: Substrate analysis and regulation by quorum sensing. J. Bacteriol 1998, 180, 4435–4441. [Google Scholar]
- Smid, E.J.; Lacroix, C. Microbe-microbe interactions in mixed culture food fermentations. Curr. Opin. Biotechnol 2013, 24, 148–154. [Google Scholar]
- Skandamis, P.N.; Nychas, G.-J. Quorum sensing in the context of food microbiology. Appl. Environ. Microbiol 2012, 78, 5473–5482. [Google Scholar]
- Ammor, M.S.; Michaelidis, C.; Nychas, G.J.E. Insights into the role of quorum sensing in food spoilage. J. Food Protect 2008, 171, 1510–1525. [Google Scholar]
- Musk, D.J., Jr; Hergenrother, P.J. Chemical countermeasures for the control of bacterial biofilms: Effective compounds and promising targets. Curr. Med. Chem 2006, 13, 2163–2177. [Google Scholar]
- Adonizio, A.L.; Downum, K.; Bennett, B.C.; Mathee, K. Antiquorum sensing activity of medicinal plants in southern Florida. J. Ethnopharmacol 2006, 105, 427–435. [Google Scholar]
- Vattem, D.A.; Mihalik, K.; Crixell, S.H.; McClean, R.J.C. Dietary phytochemicals as quorum sensing inhibitors. Fitoterapia 2007, 78, 302–310. [Google Scholar]
- Cos, P.; Vlietink, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro “proof of concept”. J. Ethnopharmacol 2006, 106, 290–302. [Google Scholar]
- Hentzer, M.; Givskov, M. Pharmacological inhibition of quorum sensing for the treatment of chronic bacterial infections. J. Clin. Invest 2003, 112, 1300–1307. [Google Scholar]
- Lewis, K.; Ausubel, F.M. Prospects of plant derived antibacterials. Nat. Biotechnol 2006, 24, 1504–1507. [Google Scholar]
- Kumar, V.P; Chauhan, N.S.; Rajani, H.P.M. Search for antibacterial and antifungal agents from selected Indian medicinal plants. J. Ethnopharmacol 2006, 107, 182–188. [Google Scholar]
- Mowrey, D.B. The Scientific Validation of Herbal Medicine; McGraw-Hill: New York, NY, USA, 1990; pp. 789–799. [Google Scholar]
- Rios, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol 2005, 100, 80–84. [Google Scholar]
- Huber, B.; Eberl, L.; Feucht, W.; Polster, J. Influence of polyphenols on bacterial biofilm formation and quorum-sensing. Z. Naturforsch 2003, 58, 879–884. [Google Scholar]
- Riedel, K.; Kothe, M.; Kramer, B.; Saeb, W.; Gotschlich, A.; Ammendola, A.; Eberl, L. Computer-aided design of agents that inhibit the cep quorum-sensing system of Burkholderia cenocepacia. Antimicrob. Agents Chemother 2006, 50, 318–323. [Google Scholar]
- Sarabhai, S.; Sharma, P.; Capalash, N. Ellagic acid derivatives from Terminalia chebula Retz. downregulate the expression of quorum sensing genes to attenuate Pseudomonas aeruginosa PAO1 virulence. PLoS One 2013, 8, e53441. [Google Scholar]
- Cragg, G.M.; Newman, D.J.; Snader, K.M. Natural products in drug discovery and development. J. Nat. Prod 1997, 60, 52–60. [Google Scholar]
- Gao, M.; Teplitski, M.; Robinson, J.B.; Bauer, W.D. Production of substances by Medicago truncatula that affect bacterial quorum sensing. Mol. Plant Microbe Interact 2003, 16, 827–834. [Google Scholar]
- Teplitski, M.; Robinson, J.B.; Bauer, W.D. Plants secrete substances that mimic bacterial N-acyl homoserine lactone signal activities and affect population density-dependent behaviors in associated bacteria. Mol. Plant Microbe Interact 2000, 13, 637–648. [Google Scholar]
- Al-Hussaini, R.; Mahasneh, A.M. Microbial growth and quorum sensing antagonist activities of herbal plants extracts. Molecules 2009, 14, 3425–3435. [Google Scholar]
- Brackman, G.; Hillaert, U.; van Calenbergh, S.; Nelis, H.J.; Coenye, T. Use of quorum sensing inhibitors to interfere with biofilm formation and development in Burkholderia multivorans and Burkholderia cenocepacia. Res. Microbiol 2009, 160, 144–151. [Google Scholar]
- Vikram, A.; Jesudhasan, P.R.; Jayaprakasha, G.K.; Pillai, B.S.; Patil, B.S. Grapefruit bioactive limonoids modulate E. coli O157:H7 TTSS and biofilm. Int. J. Food Microbiol 2010, 140, 109–116. [Google Scholar]
- De Nys, R.; Wright, A.D.; König, G.M.; Sticher, O. New halogenated furanones from the marine alga Delisea pulchra. Tetrahedron 1993, 49, 11213–11220. [Google Scholar]
- Givskov, M.; de Nys, R.; Manefield, M.; Gram, L.; Maximilien, R.; Eberl, L.; Molin, S.; Steinberg, P.D.; Kjelleberg, S. Eukaryotic interference with homoserine lactone-mediated prokaryotic signaling. J. Bacteriol 1996, 178, 6618–6622. [Google Scholar]
- Manefield, M.; Rasmussen, T.B.; Henzter, M.; Andersen, J.B.; Steinberg, P.; Kjelleberg, S.; Givskov, M. Halogenated furanones inhibit quorum sensing through accelerated LuxR turnover. Microbiology 2002, 148, 1119–1127. [Google Scholar]
- Coates, A.; Hu, Y.; Bax, R.; Page, C. The future challenges facing the development of new antimicrobial drugs. Nat. Rev. Drug Discov 2002, 1, 895–910. [Google Scholar]
- Martin, C.A.; Hoven, A.D.; Cook, A.M. Therapeutic frontiers: Preventing and treating infectious diseases by inhibiting bacterial quorum sensing. Eur. J. Clin. Microbiol 2008, 27, 635–642. [Google Scholar]
- Kalia, C.V. Quorum sensing inhibitors: An overview. Biotechnol. Adv 2013, 31, 224–245. [Google Scholar]
- Chevrot, R.; Rosen, R.; Haudecoeur, E.; Cirou, A.; Shelp, B.J.; Ron, E.; Faure, D. GABA controls the level of quorum-sensing signal in Agrobacterium tumefaciens. Proc. Natl. Acad. Sci. USA 2006, 103, 7460–7464. [Google Scholar]
- Zhang, H.B.; Wang, L.H.; Zhang, L.H. Genetic control of quorum-sensing signal turnover in Agrobacterium tumefaciens. Proc. Natl. Acad. Sci. USA 2002, 99, 4638–4643. [Google Scholar]
- Ni, N.; Choudhary, G.; Li, M.; Wang, B. Pyrogallol and its analogs can antagonize bacterial quorum sensing in Vibrio harveyi. Bioorg. Med. Chem. Lett 2008, 18, 1567–1572. [Google Scholar]
- Mathesius, U.; Mulders, S.; Gao, M.; Teplitski, M.; Caetano-Anolles, G.; Rolfe, B.G.; Bauer, W.D. Extensive and specific responses of a eukaryote to bacterial quorum-sensing signals. Proc. Natl. Acad. Sci. USA 2003, 100, 1444–1449. [Google Scholar]
- Rudrappa, T.; Bais, H.P. Curcumin, a known phenolic from Curcuma longa, attenuates the virulence of Pseudomonas aeruginosa PAO1 in whole plant and animal pathogenicity models. J. Agric. Food Chem 2008, 56, 1955–1962. [Google Scholar]
- Fratianni, F.; Coppola, R.; Nazzaro, F. Phenolic composition and antimicrobial and antiquorum sensing activity of an ethanolic extract of peels from the apple cultivar Annurca. J. Med. Food 2011, 14, 957–963. [Google Scholar]
- Fratianni, F.; de Giulio, A.; Sada, A.; Nazzaro, F. Biochemical characteristics and biological properties of annurca apple cider. J. Med. Food 2012, 15, 18–23. [Google Scholar]
- Brackman, G.; Defoirdt, T.; Miyamoto, C.; Bossier, P.; van Calenbergh, S.; Nelis, H.; Coenye, T. Cinnamaldehyde and cinnamaldehyde derivatives reduce virulence in Vibrio spp. by decreasing the DNA-binding activity of the quorum sensing response regulator LuxR. BMC Microbiol 2008, 8, 149–162. [Google Scholar]
- Niu, C.; Afre, S.; Gilbert, E.S. Sub-inhibitory concentrations of cinnamaldehyde interfere with quorum sensing. Lett. Appl. Microbiol 2006, 43, 489–494. [Google Scholar]
- Girennavar, B.; Cepeda, M.L.; Soni, K.A.; Vikram, A.; Jesudhasan, P.; Jayaprakasha, G.K.; Pillai, S.D.; Patil, B.S. Grapefruit juice and its furocoumarins inhibits autoinducer signaling and biofilm formation in bacteria. Int. J. Food Microbiol 2008, 125, 204–208. [Google Scholar]
- Vikram, A.; Jesudhasan, P.R.; Jayaprakasha, G.K.; Pillai, S.D.; Patil, B.S. Citrus limonoids interfere with Vibrio harveyi cell-cell signaling and biofilm formation by modulating the response regulator LuxO. Microbiology 2011, 157, 99–110. [Google Scholar]
- Truchado, P.; Giménez-Bastida, J.-A.; Larrosa, M.; Castro-Ibáñez, I.; Espín, J.C.; Tomás-Barberán, F.A.; García-Conesa, M.T.; Allende, A. Inhibition of quorum sensing (QS) in Yersinia enterocolitica by an orange extract rich in glycosylated flavanones. J. Agric. Food Chem 2012, 60, 8885–8894. [Google Scholar]
- Vandeputte, O.M.; Kiendrebeogo, M.; Rasamiravaka, T.; Stévigny, C.; Duez, P.; Rajaonson, S.; Diallo, B.; Mol, A.; Baucher, M.; El Jazir, M. The flavanone naringenin reduces the production of quorum sensing-controlled virulence factors in Pseudomonas aeruginosa PAO1. Microbiology 2011, 157, 2120–2132. [Google Scholar]
- Vikram, A.; Jayaprakasha, G.K.; Jesudhasan, P.R.; Pillai, S.D.; Patil, B.S. Suppression of bacterial cell-cell signalling, biofilm formation and type III secretion system by citrus flavonoids. J. Appl. Microbiol. 2010, 10(9), 515–527. [Google Scholar]
- Vandeputte, O.M.; Kiendrebeogo, M.; Rajaonson, S.; Diallo, B.; Mol, A.; Jaziri, M.E.; Baucher, M. Identification of catechin as one of the flavonoids from Combretum albiflorum bark extract that reduces the production of quorum-sensing-controlled virulence factors in Pseudomonas aeruginosa PAO1. Appl. Environ. Microbiol 2010, 71, 243–253. [Google Scholar]
- Rasamiravaka, T.; Jedrzejowski, A.; Kiendrebeogo, M.; Rajaonson, S.; Randriamampionona, D.; Rabemanantsoa, S.; Andriantsimahavandy, A.; Rasamindrakotroka, A.; El Jaziri, M.; Vandeputte, O.M. Endemic Malagasy Dalbergia species inhibit quorum sensing in Pseudomonas aeruginosa PAO1. Microbiology 2013. [Google Scholar] [CrossRef]
- Delalande, L.; Faure, D.; Raffoux, A.; Uroz, S.; D’Angelo-Picard, C.; Elasri, M.; Carlier, A.; Berruyer, R.; Petit, A.; Williams, P.; Dessaux, Y. N-hexanoyl-l-homoserine lactone, a mediator of bacterial quorum-sensing regulation, exhibits plant-dependent stability and may be inactivated by germinating Lotus corniculatus seedlings. FEMS Microbiol. Ecol 2005, 52, 13–20. [Google Scholar]
- Fatima, Q.; Zahin, M.; Khan, M.S.A.; Ahmad, I. Modulation of quorum sensing controlled behavior of bacteria by growing seedling, seed and seedling extracts of leguminous plants. Indian J. Microbiol 2010, 50, 238–242. [Google Scholar]
- Gotz, C.; Fekete, A.; Gebefuegi, I.; Forczek, S.T.; Fuksova, K.; Li, X.; Englmann, M.; Gryndler, M.; Hartmann, M.; Matucha, M.; et al. Uptake, degradation and chiral discrimination of N-acyl-d/l-homoserine lactones by barley (Hordeum vulgare) and yam bean (Pachyrhizus erosus) plants. Anal. Bioanal. Chem 2007, 389, 1447–1457. [Google Scholar]
- Yuan, Z.C.; Edlind, M.P.; Liu, P.; Saenkham, P.; Banta, L.M.; Wise, A.A.; Ronzone, E.; Binns, A.N.; Kerr, K.; Nester, E.W. The plant signal salicylic acid shuts down expression of the vir regulon and activates quormone-quenching genes in Agrobacterium. Proc. Natl. Acad. Sci. USA 2007, 104, 11790–11795. [Google Scholar]
- Ren, D.; Zuo, R.; González-Barrios, A.F.; Bedzyk, L.A.; Eldridge, G.R.; Pasmore, M.E.; Wood, T.K. Differential gene expression for investigation of Escherichia coli biofilm inhibition by plant extract ursolic acid. Appl. Environ. Microbiol 2005, 71, 4022–4034. [Google Scholar]
- Musthafa, K.S.; Ravi, A.V.; Annapoorani, A.; Packiavathy, I.S.V.; Pandian, S.K. Evaluation of anti-quorum-sensing activity of edible plants and fruits through inhibition of the N-acyl-homoserine lactone system in Chromobacterium violaceum and Pseudomonas aeruginosa. Chemotherapy 2010, 56, 333–339. [Google Scholar]
- Lee, K.-M.; Lim, J.; Nam, S.; Young Yoon, M.; Kwon, Y.-K.; Jung, B.Y.; Park, Y.J.; Park, S.; Yoon, S.S. Inhibitory effects of broccoli extract on Escherichia coli O157:H7 quorum sensing and in vivo virulence. FEMS Microbiol. Lett 2011, 321, 67–74. [Google Scholar]
- Fratianni, F.; Nazzaro, F.; Marandino, A.; Fusco, M.D.R.; Coppola, R.; de Feo, V.; de Martino, L. Biochemical composition, antimicrobial activities, and anti-quorum-sensing activities of ethanol and ethyl acetate extracts from Hypericum connatum Lam. (Guttiferae). J. Med. Food 2013, 16, 454–459. [Google Scholar]
- Larrosa, M.; García-Conesa, M.T.; Espín, J.C.; Tomás-Barberán, F.A. Ellagitannins, ellagic acid and vascular health. Mol. Asp. Med 2010, 31, 513–539. [Google Scholar]
- Larrosa, M.; González-Sarrías, A.; Yánez-Gascón, M.J.; Selma, M.V.; Azorín-Ortuño, M.; Totia, S.; Tomás-Barberán, F.A.; Dolara, P.; Espín, J.C. Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolics metabolism. J. Nutr. Biochem 2010, 21, 717–725. [Google Scholar]
- Giménez-Bastida, J.A.; Truchado, P.; Larrosa, M.; Espín, J.C.; Tomás-Barberán, F.A.; Allende, A.; García-Conesa, M.T. Urolithins, ellagitannin metabolites produced by colon microbiota, inhibit Quorum Sensing in Yersinia enterocolitica: Phenotypic response and associated molecular changes. Food Chem 2012, 132, 1465–1474. [Google Scholar]
- Fiamegos, Y.C.; Panagiotis, L.; Kastritis, X.; Vassiliki, E.; Han, H.; Bonvin, A.M.J.J.; Vervoort, J.; Lewis, K.; Hamblin, M.R.; Tegos, G.P. Antimicrobial and efflux pump inhibitory activity of caffeoylquinic acids from Artemisia absinthium against Gram-positive pathogenic bacteria. PLoS One 2011, 6, e18127. [Google Scholar]
- Leach, J.E.; Lloyd, L.A.; McGee, J.D.; Hilaire, E.; Wang, X.; Guikema, J.A. Trafficking of Plant Defense Response Compounds. In Delivery and Perception of Pathogen Signals in Plants; Keen, N.T., Mayama, S., Leach, J.E., Tsuyumu, S., Eds.; The American Phytopathological Society (APS) Press: St. Paul, MN, USA, 2007; pp. 1–268. [Google Scholar]
- Feucht, W.; Schwalb, P.; Zinkernagel, V. Complexation of fungal structures with monomeric and prooligomeric flavanols. J. Plant Dis. Prot 2000, 107, 106–110. [Google Scholar]
- Truchado, P.; López-Gálvez, F.; Gil, M.I.; Tomás-Barberán, F.A.; Allende, A. Quorum sensing inhibitory and antimicrobial activities of honeys and the relationship with individual phenolics. Food Chem 2009, 115, 1337–1344. [Google Scholar]
- Al-Shuneigat, J.; Cox, S.D.; Markham, J.L. Effects of a topical essential oil-containing formulation on bio-film-forming coagulase-negative staphylococci. Lett. Appl. Microbiol 2005, 41, 52–55. [Google Scholar]
- Chorianopoulos, N.G.; Giaouris, F.D.; Skendamis, P.N.; Haroutounian, S.A.; Nychas, G.-J. Disinfectant test against monoculture and mixed-culture biofilms composed of technological, spoilage and pathogenic bacteria: Bactericidal effect of essential oil and hydrosol of Satureja thymbra and comparison with standard acid-base sanitizers. J. Appl. Microbiol 2008, 104, 1586–96. [Google Scholar]
- Khan, M.S.; Zahin, M.; Hasan, S.; Husain, F.M.; Ahmad, I. Inhibition of quorum sensing regulated bacterial functions by plant essential oils with special reference to clove oil. Lett. Appl. Microbiol 2009, 49, 354–360. [Google Scholar]
- Zaki, A.A.; Shaaban, M.I.; Hashish, N.E.; Amer, M.A.; Lahloub, M.-F. Assessment of anti-quorum sensing activity for some ornamental and medicinal plants native to Egypt. Sci. Pharm 2013, 81, 251–258. [Google Scholar]
- Szabó, M.A.; Varga, G.Z.; Hohmann, J.; Schelz, Z.; Szegedi, E.; Amaral, L.; Molnár, J. Inhibition of quorum-sensing signals by essential oils. Phytother. Res 2010, 24, 782–786. [Google Scholar]
- Bjarnsholt, T.; Jensen, P.O.; Rasmussen, T.B.; Christophersen, L.; Calum, H.; Hentzer, M.; Hougen, H.P.; Rygaard, J.; Moser, C.; Eberl, L.; et al. Garlic blocks quorum sensing and promotes rapid clearing of pulmonary Pseudomonas aeruginosa infections. Microbiology 2005, 151, 3873–3880. [Google Scholar]
- Choo, J.H.; Rukayadi, Y.; Hwang, J.K. Inhibition of bacterial quorum sensing by vanilla extract. Lett. Appl. Microbiol 2006, 42, 637–641. [Google Scholar]
- Novak, J.S.; Fratamico, P.M. Evaluation of ascorbic acid as a quorum sensing analogue to control growth, sporulation, and enterotoxin production in Clostridium perfringens. J. Food Sci 2004, 69, 72–78. [Google Scholar]
- Otto, M. Quorum-sensing control in Staphylococci—A target for antimicrobial drug therapy? FEMS Microbiol. Lett 2004, 241, 135–141. [Google Scholar]
- Barriuso, J.; Solano, B.; Fray, R.G.; Cámara, M.; Hartmann, A.; Mañero, J.G. Transgenic tomato plants alter quorum sensing in plant growth-promoting rhizobacteria. Plant Biotechnol. J 2008, 6, 442–452. [Google Scholar]

| Molecules | References |
|---|---|
| Gamma aminobutyric acid (GABA) | Chevrot et al., 2006 [33]; Zhang et al., 2002 [34] |
| Pyrogallol | Ni et al., 2008 [35] |
| Curcumin | Rudrappa and Bais, 2008 [37] |
| Cynnamaldheyde | Brackman et al., 2008 [40]; Niu et al., 2006 [41] |
| Furocoumarins | Girennavar et al., 2008 [42] |
| Flavanones, flavonoids, flavonols | Truchado et al., 2012 [44]; Vandeputte et al., 2011 [45]; Vikram et al., 2010 [46]; Vandeputte et al., 2010 [47]; Rasamiravaka et al., 2013 [48]; Leach et al., 2007 [61] |
| Ursolic acid | Ren et al., 2005 [53] |
| Rosmarinic acid | Vattem et al., 2007 [11] |
| Salycilic acid | Yuan et al., 2007 [52] |
| Epigallocatechin gallate, Ellagic acid, Tannic acid | Riedel et al., 2006 [19]; Sarabhai et al., 2013 [20]; Larrosa et al., 2010a [57] |
| Urolithin A and B | Larrosa et al., 2010b [58]; Gimenez-Bastia et al., 2012 [59] |
| 4,5-O-dicaffeoyl quinic acid | Fiamegos et al., 2011 [60] |
| Chlorogenic acid, vanillic acid, proanthocyanidins | Leach et al., 2007 [61]; Feucht et al., 2000 [62] |
| Volatile organic compounds | Chernin et al., 1998 [5] |
| Furanones | Manefield et al., 2002 [29] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nazzaro, F.; Fratianni, F.; Coppola, R. Quorum Sensing and Phytochemicals. Int. J. Mol. Sci. 2013, 14, 12607-12619. https://doi.org/10.3390/ijms140612607
Nazzaro F, Fratianni F, Coppola R. Quorum Sensing and Phytochemicals. International Journal of Molecular Sciences. 2013; 14(6):12607-12619. https://doi.org/10.3390/ijms140612607
Chicago/Turabian StyleNazzaro, Filomena, Florinda Fratianni, and Raffaele Coppola. 2013. "Quorum Sensing and Phytochemicals" International Journal of Molecular Sciences 14, no. 6: 12607-12619. https://doi.org/10.3390/ijms140612607







