Melatonin Receptor Genes in Vertebrates
Abstract
:1. Introduction
2. Expression of Melatonin Receptors
3. Polymorphisms of Human Melatonin Receptor and GPR50 Genes
4. Evolution of Melatonin Receptor Genes in Vertebrates
5. Melatonin Receptors: A Perspective
Acknowledgments
Conflict of Interest
References
- Sundaresan, N.R.; Marcus Leo, M.D.; Subramani, J.; Anish, D.; Sudhagar, M.; Ahmed, K.A.; Saxena, M.; Tyagi, J.S.; Sastry, K.V.; Saxena, V.K. Expression analysis of melatonin receptor subtypes in the ovary of domestic chicken. Vet. Res. Commun 2009, 33, 49–56. [Google Scholar]
- Jones, C.; Helfer, G.; Brandstätter, R. Melatonin receptor expression in the zebra fish brain and peripheral tissues. Chronobiol. Int 2012, 29, 189–202. [Google Scholar]
- Li, D.Y.; Zhang, L.; Smith, D.G.; Xu, H.L.; Liu, Y.P.; Zhao, X.L.; Wang, Y.; Zhu, Q. Genetic effects of melatonin receptor genes on chicken reproductive traits. Czech. J. Anim. Sci 2013, 58, 58–64. [Google Scholar]
- Hardeland, R.; Cardinali, D.P.; Srinivasan, V.; Spence, D.W.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin—A pleiotropic, orchestrating regulator molecule. Progr. Neurobiol 2011, 93, 350–384. [Google Scholar]
- Dufourny, L.; Levasseur, A.; Migaud, M.; Callebaut, I.; Pontarotti, P.; Malpaux, B.; Monget, P. GPR50 is the mammalian ortholog of Mel1c: Evidence of rapid evolution in mammals. BMC Evol. Biol 2008, 8, 105. [Google Scholar]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T.; Mahle, C.D.; Kolakowski, L.F., Jr. Cloning of a melatonin-related receptor from human pituitary. FEBS Lett. 1996, 386, 219–224. [Google Scholar]
- Gubitz, A.K.; Reppert, S.M. Assignment of the melatonin-related receptor to human chromosome X (GPR50) and mouse chromosome X (GPR50). Genomics 1999, 55, 248–251. [Google Scholar]
- Hamouda, H.O.; Chen, P.; Levoye, A.; Sözer-Topçular, N.; Daulat, A.M.; Guillaume, J.L.; Ravid, R.; Savaskan, E.; Ferry, G.; Boutin, J.A. Detection of the human GPR50 orphan seven transmembrane protein by polyclonal antibodies mapping different epitopes. J. Pineal. Res 2007, 43, 10–15. [Google Scholar]
- Levoye, A.; Dam, J.; Ayoub, M.A.; Guillaume, J.-L.; Couturier, C.; Delagrange, P.; Jockers, R. The orphan GPR50 receptor specifically inhibits MT1 melatonin receptor function through heterodimerization. EMBO J 2006, 25, 3012–3023. [Google Scholar]
- Doolen, S.; Krause, D.; Dubocovich, M.; Duckles, S. Melatonin mediates two distinct responses in vascular smooth muscle. Eur. J. Pharmacol 1998, 345, 67–69. [Google Scholar]
- Dubocovich, M.; Yun, K.; Al-ghoul, W.; Benloucif, S.; Masana, M. Selective MT2 melatonin receptor antagonists block melatonin-mediated phase advances of circadian rhythms. FASEB J 1998, 12, 1211. [Google Scholar]
- Bordt, S.; McKeon, R.; Li, P.; Witt-Enderby, P.; Melan, M. N1E-115mouse neuroblastoma cells express MT1 melatonin receptors and produce neurites in response to melatonin. Biochim. Biophys. Acta 2001, 1499, 257–264. [Google Scholar]
- Dubocovich, M.; Masana, M.; Iacob, S.; Sauri, D. Melatonin receptor antagonists that differentiate between the human Mel1a and Mel1b recombinant subtypes are used to assess the pharmacological profile of the rabbit retina ML1 presynaptic heteroreceptor. Naunyn-Schmiedeberg’s Arch. Pharmacol 1997, 355, 365–375. [Google Scholar]
- Barrett, P.; Conway, S.; Jockers, R.; Strosberg, A.; Guardiola-Lemaitre, B.; Delagrange, P.; Morgan, P. Cloning and functional analysis of a polymorphic variant of the ovine Mel 1a melatonin receptor. Biochim. Biophys. Acta 1997, 1356, 299–307. [Google Scholar]
- Clemens, J.; Jarzynka, M.; Witt-Enderby, P. Down-regulation of mt1 melatonin receptors in rat ovary following estrogen exposure. Life Sci 2001, 69, 27–35. [Google Scholar]
- Roth, J.; Kim, B.; Lin, W.; Cho, M. Melatonin promotes osteoblast differentiation and bone formation. J. Biol. Chem 1999, 274, 22041. [Google Scholar]
- Witt-Enderby, P.; Bennett, J.; Jarzynka, M.; Firestine, S.; Melan, M. Melatonin receptors and their regulation: Biochemical and structural mechanisms. Life Sci 2003, 72, 2183–2198. [Google Scholar]
- Reiter, R.J. The melatonin rhythm: Both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev 1991, 12, 151–180. [Google Scholar]
- Natesan, A.K.; Cassone, V.M. Melatonin receptor mRNA localization and rhythmicity in the retina of the domestic chick, Gallus domesticus. Vis. Neurosci 2002, 19, 265–274. [Google Scholar]
- Rada, J.; Wiechmann, A. Melatonin receptors in chick ocular tissues: Implications for a role of melatonin in ocular growth regulation. Investig. Ophthalmol. Vis. Sci 2006, 47, 25. [Google Scholar]
- Reiter, R.J.; Poeggeler, B.; Tan, D.-X.; Chen, L.-D.; Manchester, L.C.; Guerrero, J.M. Antioxidant capacity of melatonin: A novel action not requiring a receptor. Neuroendocrinol. Lett 1993, 15, 103–116. [Google Scholar]
- Jan, J.E.; Reiter, R.J.; Wong, P.K.; Bax, M.C.; Ribary, U.; Wasdell, M.B. Melatonin has membrane receptor-independent hypnotic action on neurons: An hypothesis. J. Pineal Res 2011, 50, 233–240. [Google Scholar]
- Reiter, R.J.; Tan, D.-X.; Qi, W.; Manchester, L.C.; Karbownik, M.; Calvo, J.R. Pharmacology and physiology of melatonin in the reduction of oxidative stress in vivo. Biol. Signals Recept 2000, 9, 160–171. [Google Scholar]
- Reiter, R.J.; Tan, D.-X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci 2007, 52, 11–28. [Google Scholar]
- Benitez-King, G.; Anton-Tay, F. Calmodulin mediates melatonin cytoskeletal effects. Cell Mol. Life Sci 1993, 49, 635–641. [Google Scholar]
- Hardeland, R. Melatonin: Signaling mechanisms of a pleiotropic agent. BioFactors 2009, 35, 183–192. [Google Scholar]
- Okano, T.; Fukada, Y. Phototransduction cascade and circadian oscillator in chicken pineal gland. J. Pineal. Res 1997, 22, 145–151. [Google Scholar]
- Korf, H.W.; Schomerus, C.; Stehle, J.H. The pineal organ, its hormone melatonin, and the photoneuroendocrine system. Adv. Anat. Embryol. Cell Biol 1998, 146, 1–100. [Google Scholar]
- Malpaux, B.; Migaud, M.; Tricoire, H.; Chemineau, P. Biology of mammalian photoperiodism and the critical role of the pineal gland and melatonin. J. Biol. Rhythms 2001, 16, 336–347. [Google Scholar]
- Drew, J.; Barrett, P.; Mercer, J.; Moar, K.; Canet, E.; Delagrange, P.; Morgan, P. Localization of the melatonin-related receptor in the rodent brain and peripheral tissues. J. Neuroendocrinol 2001, 13, 453–458. [Google Scholar]
- Ekmekcioglu, C. Melatonin receptors in humans: Biological role and clinical relevance. Biomed. Pharmacother 2006, 60, 97–108. [Google Scholar]
- Morgan, P.; Barrett, P.; Howell, H.; Helliwell, R. Melatonin receptors: Localization, molecular pharmacology and physiological significance. Neurochem. Int 1994, 24, 101–146. [Google Scholar]
- Sugden, D.; Davidson, K.; Hough, K.; Teh, M. Melatonin, melatonin receptors and melanophores: A moving story. Pigment. Cell Res 2004, 17, 454–460. [Google Scholar]
- Adachi, A.; Natesan, A.; Whitfield-Rucker, M.; Weigum, S.; Cassone, V. Functional melatonin receptors and metabolic coupling in cultured chick astrocytes. Glia 2002, 39, 268–278. [Google Scholar]
- Soares, J.M.; Masana, M.I.; Erşahin, Ç.; Dubocovich, M.L. Functional melatonin receptors in rat ovaries at various stages of the estrous cycle. J. Pharmacol. Exp. Ther. 2003, 306, 694. [Google Scholar]
- Drew, J.E.; Barrett, P.; Williams, L.M.; Conway, S.; Morgan, P.J. The ovine melatonin-related receptor: Cloning and preliminary distribution and binding studies. J. Neuroendocrinol 1998, 10, 651–661. [Google Scholar]
- Ivanova, E.A.; Bechtold, D.A.; Dupré, S.M.; Brennand, J.; Barrett, P.; Luckman, S.M.; Loudon, A.S. Altered metabolism in the melatonin-related receptor (GPR50) knockout mouse. Am. J. Physiol. Endocrinol. Metab 2008, 294, E176–E182. [Google Scholar]
- Barrett, P.; Ivanova, E.; Graham, E.S.; Ross, A.W.; Wilson, D.; Plé, H.; Mercer, J.G.; Ebling, F.J.; Schuhler, S.; Dupré, S.M. Photoperiodic regulation of cellular retinoic acid-binding protein 1, GPR50 and nestin in tanycytes of the third ventricle ependymal layer of the Siberian hamster. J. Endocrinol 2006, 191, 687–698. [Google Scholar]
- Grünewald, E.; Kinnell, H.L.; Porteous, D.J.; Thomson, P.A. GPR50 interacts with neuronal NOGO-A and affects neurite outgrowth. Mol. Cell Neurosci 2009, 42, 363–371. [Google Scholar]
- Li, J.; Hand, L.E.; Meng, Q.J.; Loudon, A.S.; Bechtold, D.A. GPR50 interacts with TIP60 to modulate glucocorticoid receptor signalling. PLoS One 2011, 6, e23725. [Google Scholar]
- Gillette, M.U.; Mitchell, J.W. Signaling in the suprachiasmatic nucleus: Selectively responsive and integrative. Cell Tissue Res 2002, 309, 99–107. [Google Scholar]
- Stehle, J.H.; von Gall, C.; Korf, H.W. Melatonin: A clock-output, a clock-input. J. Neuroendocrinol 2003, 15, 383–389. [Google Scholar]
- Pévet, P.; Challet, E. Melatonin: Both master clock output and internal time-giver in the circadian clocks network. J. Physiol. Paris 2011, 105, 170–182. [Google Scholar]
- Hardeland, R.; Madrid, J.A.; Tan, D.X.; Reiter, R.J. Melatonin, the circadian multioscillator system and health: The need for detailed analyses of peripheral melatonin signaling. J. Pineal. Res 2012, 52, 139–166. [Google Scholar]
- Oprea-Ilies, G.; Haus, E.; Sackett-Lundeen, L.; Liu, Y.; McLendon, L.; Busch, R.; Adams, A.; Cohen, C. Expression of melatonin receptors in triple negative breast cancer (TNBC) in African American and Caucasian women: Relation to survival. Breast Cancer Res. Treat 2013, 137, 677–687. [Google Scholar]
- Deming, S.L.; Lu, W.; Beeghly-Fadiel, A.; Zheng, Y.; Cai, Q.; Long, J.; Shu, X.O.; Gao, Y.-T.; Zheng, W. Melatonin pathway genes and breast cancer risk among Chinese women. Breast Cancer Res. Treat 2012, 132, 1–7. [Google Scholar]
- Jablonska, K.; Pula, B.; Zemla, A.; Owczarek, T.; Wojnar, A.; Rys, J.; Ambicka, A.; Podhorska-Okolow, M.; Ugorski, M.; Dziegiel, P. Expression of melatonin receptor MT1 in cells of human invasive ductal breast carcinoma. J. Pineal. Res 2012, 54, 334–345. [Google Scholar]
- Wu, Y.H.; Ursinus, J.; Zhou, J.N.; Scheer, F.A.; Ai-Min, B.; Jockers, R.; van Heerikhuize, J.; Swaab, D.F. Alterations of melatonin receptors MT1 and MT2 in the hypothalamic suprachiasmatic nucleus during depression. J. Affect. Disord 2013, 148, 357–367. [Google Scholar]
- Natarajan, R.; Einarsdottir, E.; Riutta, A.; Hagman, S.; Raunio, M.; Mononen, N.; Lehtimaki, T.; Elovaara, I. Melatonin pathway genes are associated with progressive subtypes and disability status in multiple sclerosis among Finnish patients. J. Neuroimmunol 2012, 250, 106–110. [Google Scholar]
- Nagorny, C.; Lyssenko, V. Tired of diabetes genetics? Circadian rhythms and diabetes: The MTNR1B story? Curr. Diab. Rep 2012, 12, 667–672. [Google Scholar]
- McKenna, J.T.; Christie, M.A.; Jeffrey, B.A.; McCoy, J.G.; Lee, E.; Connolly, N.P.; Ward, C.P.; Strecker, R.E. Chronic ramelteon treatment in a mouse model of Alzheimer’s disease. Arch. Ital. Biol 2012, 150, 5–14. [Google Scholar]
- Wang, X.; Sirianni, A.; Pei, Z.; Cormier, K.; Smith, K.; Jiang, J.; Zhou, S.; Wang, H.; Zhao, R.; Yano, H.; et al. The melatonin MT1 receptor axis modulates mutant Huntingtin-mediated toxicity. J. Neurosci 2011, 31, 14496–14507. [Google Scholar]
- Nemeth, C.; Humpeler, S.; Kallay, E.; Mesteri, I.; Svoboda, M.; Rogelsperger, O.; Klammer, N.; Thalhammer, T.; Ekmekcioglu, C. Decreased expression of the melatonin receptor 1 in human colorectal adenocarcinomas. J. Biol. Regul. Homeostat. Agents 2011, 25, 531–542. [Google Scholar]
- Krogh, A.; Larsson, B.È.; von Heijne, G.; Sonnhammer, E.L.L. Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 2001, 305, 567–580. [Google Scholar]
- Thomson, P.; Wray, N.; Thomson, A.; Dunbar, D.; Grassie, M.; Condie, A.; Walker, M.; Smith, D.; Pulford, D.; Muir, W. Sex-specific association between bipolar affective disorder in women and GPR50, an X-linked orphan G protein-coupled receptor. Mol. Psychiat 2004, 10, 470–478. [Google Scholar]
- Delavest, M.; Even, C.; Benjemaa, N.; Poirier, M.-F.; Jockers, R.; Krebs, M.-O. Association of the intronic rs2072621 polymorphism of the X-linked GPR50 gene with affective disorder with seasonal pattern. Eur. Psychiat 2012, 27, 369–371. [Google Scholar]
- Chaste, P.; Clement, N.; Mercati, O.; Guillaume, J.L.; Delorme, R.; Botros, H.G.; Pagan, C.; Périvier, S.; Scheid, I.; Nygren, G.; et al. Identification of pathway-biased and deleterious melatonin receptor mutants in autism spectrum disorders and in the general population. PLoS One 2010, 5, e11495. [Google Scholar]
- Bonnefond, A.; Clément, N.; Fawcett, K.; Yengo, L.; Vaillant, E.; Guillaume, J.L.; Dechaume, A.; Payne, F.; Roussel, R.; Czernichow, S.; et al. Rare MTNR1B variants impairing melatonin receptor 1B function contribute to type 2 diabetes. Nat. Genet 2012, 44, 297–301. [Google Scholar]
- Hardeland, R. Melatonin in aging and disease—multiple consequences of reduced secretion, options and limits of treatment. Ag. Dis 2012, 3, 194–225. [Google Scholar]
- Weaver, D.R.; Liu, C.; Reppert, S.M. Nature’s knockout: The Mel1b receptor is not necessary for reproductive and circadian responses to melatonin in Siberian hamsters. Mol. Endocrinol 1996, 10, 1478–1487. [Google Scholar]
- Sethi, S.; Adams, W.; Pollock, J.; Witt-Enderby, P.A. C-terminal domains within human MT1 and MT2 melatonin receptors are involved in internalization processes. J. Pineal. Res 2008, 45, 212–218. [Google Scholar]
- Guillaume, J.L.; Daulat, A.M.; Maurice, P.; Levoye, A.; Migaud, M.; Brydon, L.; Malpaux, B.; Borg-Capra, C.; Jockers, R. The PDZ protein mupp1 promotes Gi coupling and signaling of the Mt1 melatonin receptor. J. Biol. Chem 2008, 283, 16762–16771. [Google Scholar]
- Witt-Enderby, P.A.; Jarzynka, M.J.; Krawitt, B.J.; Melan, M.A. Knoch-down of RGS4 and beta tubulin in CHO cells expressing the human MT1 melatonin receptor prevents melatonin-induced receptor desensitization. Life Sci 2004, 75, 2703–2715. [Google Scholar]
- Dupré, S.M.; Dardente, H.; Birnie, M.J.; Loudon, A.S.; Lincoln, G.A.; Hazzlerigg, D.G. Evidence for RGS4 modulation of melatonin and thyrotrophin signalling pathways in the pars tuberalis. J. Neuroendocrinol 2011, 23, 725–732. [Google Scholar]
- Ji, M.; Zhao, W.J.; Dong, L.D.; Miao, Y.; Yang, X.L.; Sun, X.H.; Wang, Z. RGS2 and RGS4 modulate melatonin-induced potentiation of glycine currents in rat retinal ganglion cells. Brain Res 2011, 1411, 1–8. [Google Scholar]
- Maurice, P.; Daulat, A.M.; Turecek, R.; Ivankova-Susankova, K.; Zamponi, F.; Kamal, M.; Clement, N.; Guillaume, J.L.; Bettler, B.; Galès, C.; et al. Molecular organization and dynamics of the melatonin MT1 receptor/RSG20/Gi protein complex reveal asymmetry of receptor dimers for RGS and Gi coupling. EMBO J 2010, 29, 3646–3659. [Google Scholar]
- Vrajová, M.; Peková, S.; Horáček, J.; Höschl, C. The effects of siRNA-mediated RGS4 gene silencing on the whole genome transcription profile: Implications for schizophrenia. Neuroendocrinol. Lett 2011, 32, 246–252. [Google Scholar]
- Chenna, R.; Sugawara, H.; Koike, T.; Lopez, R.; Gibson, T.J.; Higgins, D.G.; Thompson, J.D. Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 2003, 31, 3497–3500. [Google Scholar]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol 2011, 28, 2731–2739. [Google Scholar]
- Zuckerkandl, E.; Pauling, L. Evolutionary Divergence and Convergence in Proteins. In Evolving Genes and Proteins; Bryson, V., Vogel, H.J., Eds.; Academic Press: New York, NY, USA, 1965; pp. 97–165. [Google Scholar]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar]
- Ebisawa, T.; Karne, S.; Lerner, M.R.; Reppert, S.M. Expression cloning of a high-affinity melatonin receptor from Xenopus dermal melanophores. Proc. Natl. Acad. Sci. USA 1994, 91, 6133–6137. [Google Scholar]
- Reppert, S.M.; Weaver, D.R.; Cassone, V.M.; Godson, C.; Kolakowski, L.F., Jr. Melatonin receptors are for the birds: Molecular analysis of two receptor subtypes differentially expressed in chick brain. Neuron 1995, 15, 1003–1015. [Google Scholar]
- Nei, M. Phylogenetic analysis in molecular evolutionary genetics. Annu. Rev. Genet 1996, 30, 371–403. [Google Scholar]
- Liu, C.; Weaver, D.R.; Jin, X.; Shearman, L.P.; Pieschl, R.L.; Gribkoff, V.K.; Reppert, S.M. Molecular dissection of two distinct actions of melatonin on the suprachiasmatic circadian clock. Neuron 1997, 19, 91. [Google Scholar]
- Weaver, D.R.; Reppert, S.M. The Mel1a melatonin receptor gene is expressed in human suprachiasmatic nuclei. Neuroreport 1996, 8, 109–112. [Google Scholar]
- Hunt, A.E.; Al-Ghoul, W.M.; Gillette, M.U.; Dubocovich, M.L. Activation of MT2 melatonin receptors in rat suprachiasmatic nucleus phase advances the circadian clock. Am. J. Physiol. 2001, 280, C110–C118. [Google Scholar]
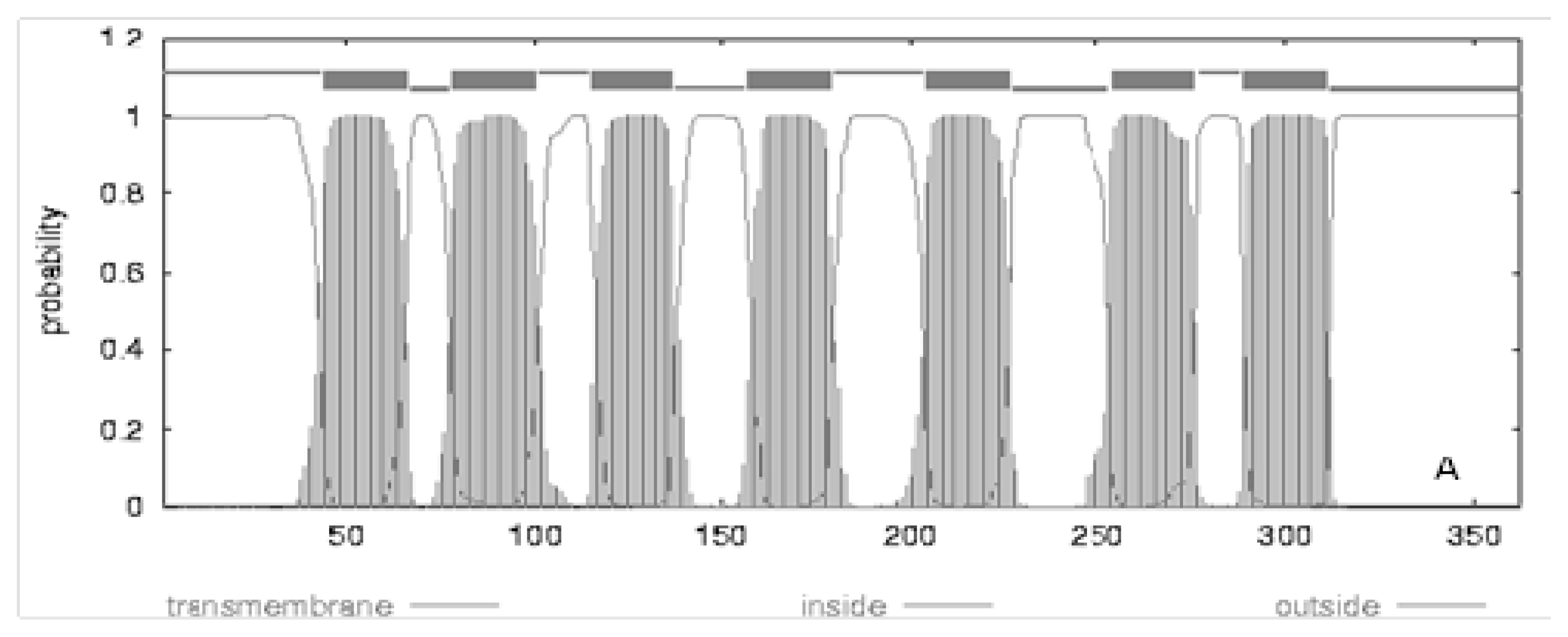
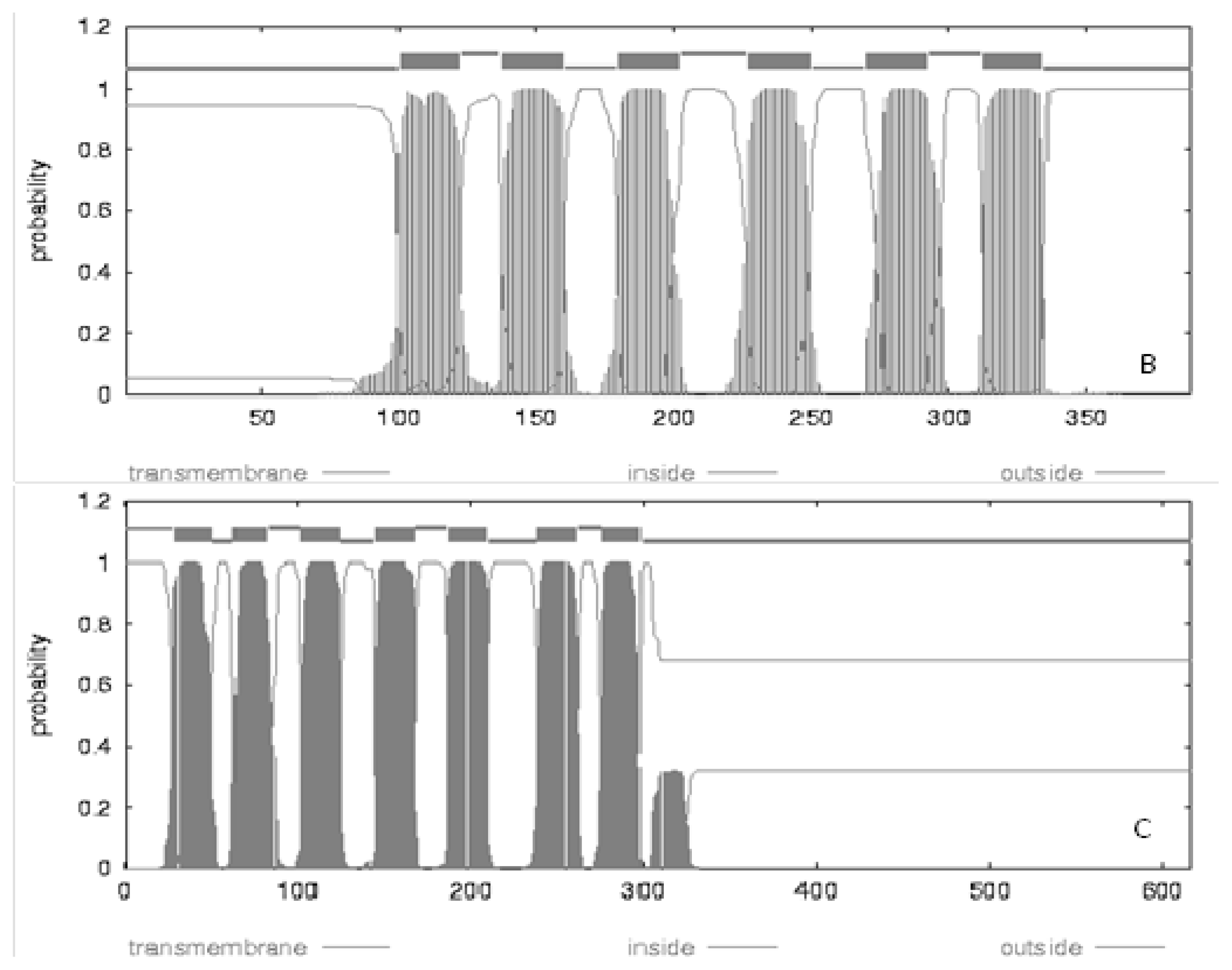
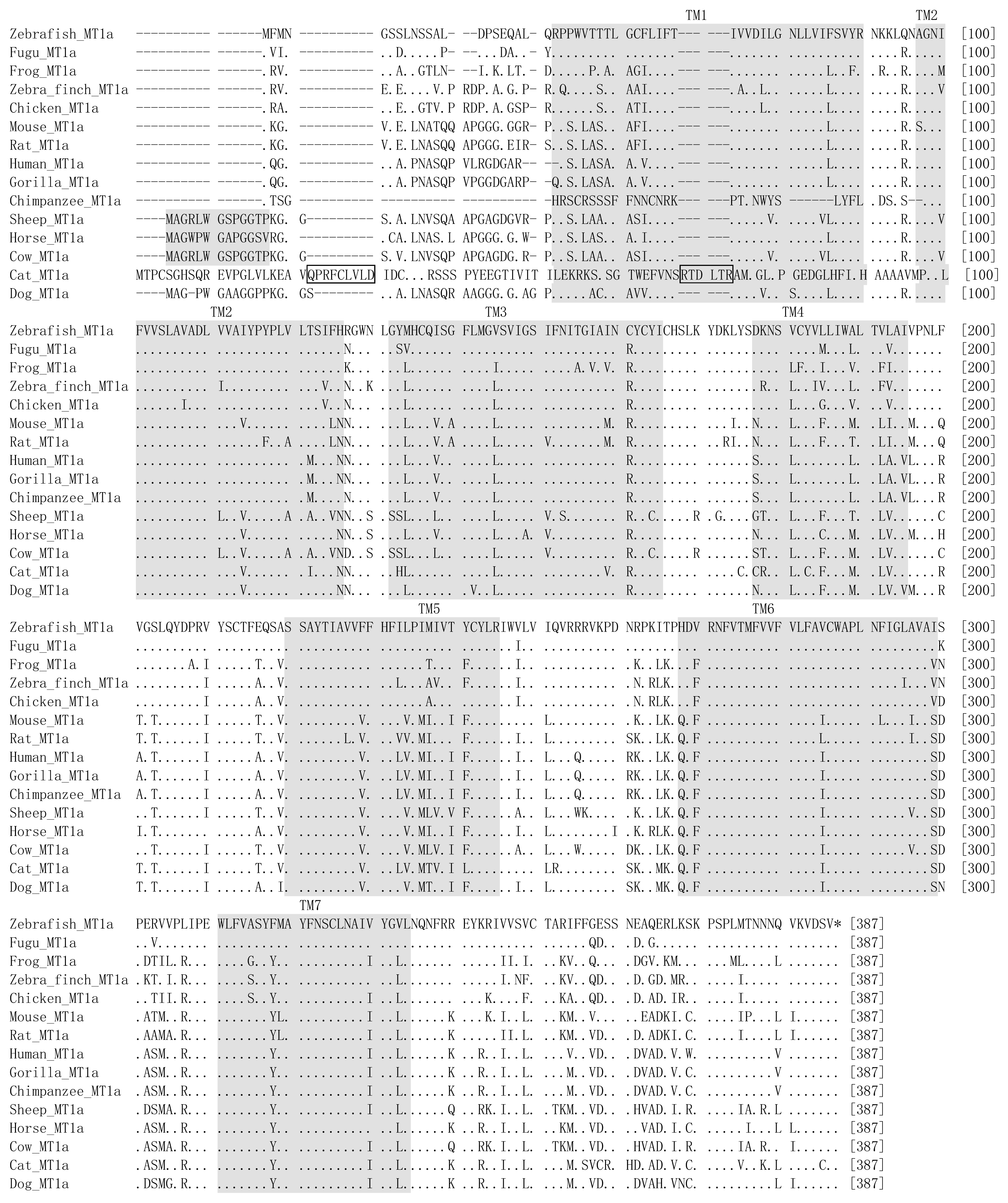
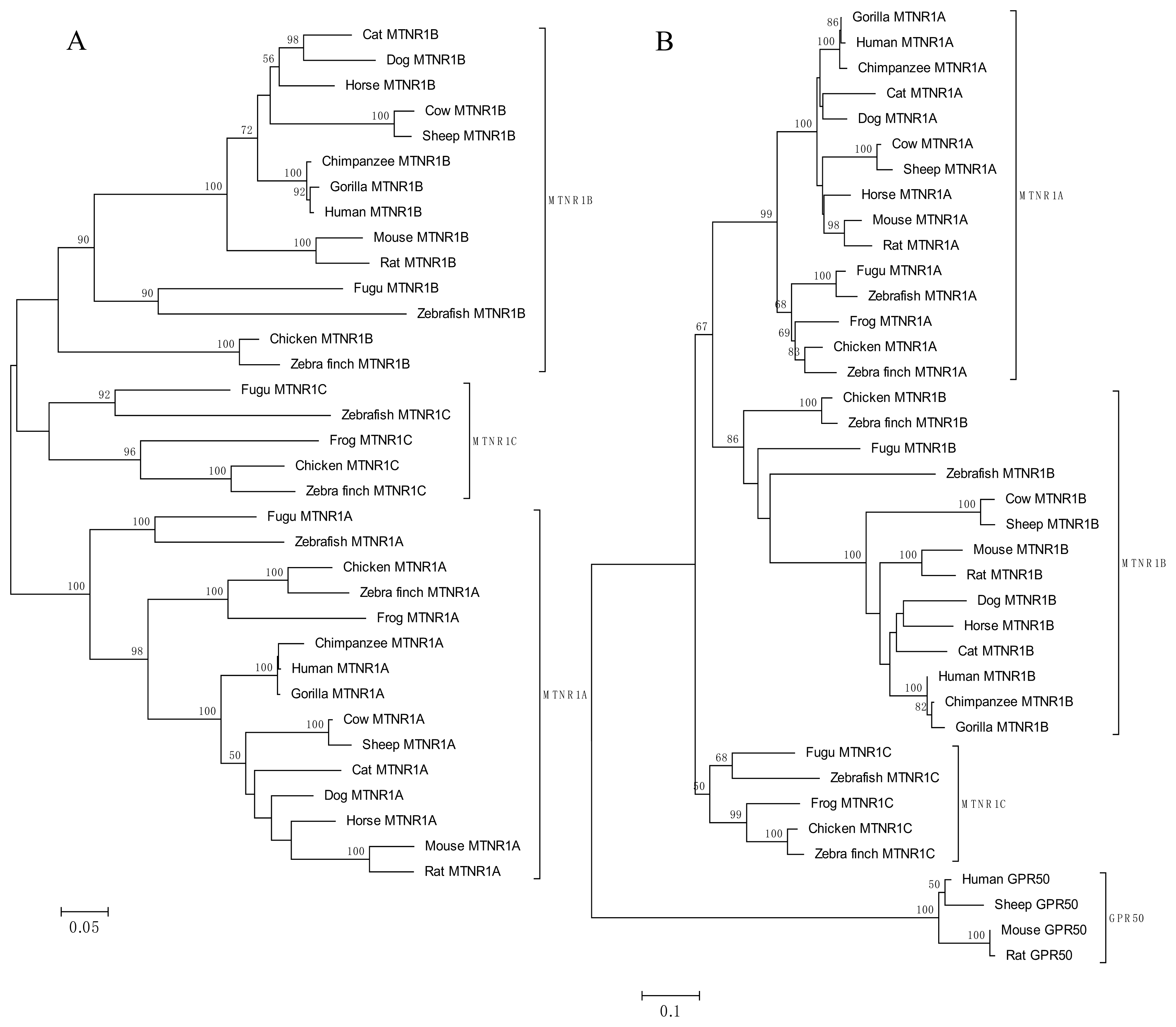
| Location | Gene | Length | Amino acids length | Synonymous sites | Missense sites | Frame shift sites | |
|---|---|---|---|---|---|---|---|
| Human | Chr: 4 | MTNR1A | 1053 bp | 351 | 21 | 27 | 0 |
| Chr: 11 | MTNR1B | 1089 bp | 363 | 18 | 50 | 0 | |
| Chr: X | GPR50 | 1854 bp | 618 | 9 | 21 | 3 |
| Species | Mel-1a GenBank ID | Mel-1b GenBank ID | Mel-1c GenBank ID | GPR50 GenBank ID | |
|---|---|---|---|---|---|
| Non-mammals | Zebrafish | NM_131393.1 | NM_131395.1 | NM_001161484.1 | |
| Fugu | AB492764.1 | AB492765.1 | AB492766.1 | ||
| Frog | XP_002940910.1 | U09561.1 | |||
| Chicken | NM_205362.1 | XM_417201.2 | NM_205361.1 | ||
| Zebra finch | NM_001048257.1 | NM_001048258.1 | XM_002193412.1 | ||
| Mammals | Horse | XP_001490221.1 | XM_001917051.1 | ||
| Gorilla gorilla | XM_004040725.1 | XM_004051965.1 | |||
| Cat | XM_003984615.1 | XM_003992620.1 | |||
| Human | NM_005958.3 | NM_005959.3 | NM_004224.3 | ||
| Chimpanzee | XM_526799.2 | XM_522146.4 | |||
| Rat | NM_053676.2 | NM_001100641.1 | NM_001191915.1 | ||
| Dog | XM_540019.3 | XM_844629.2 | |||
| Mouse | NM_008639.2 | NM_145712.2 | NM_010340.1 | ||
| Cow | XM_002698656.1 | NM_001206907.1 | |||
| Sheep | NP_001009725.1 | NM_001130938.1 | NM_001009726.1 | ||
| Zebrafish | Cat | Chicken | Chimpanzee | Cow | Dog | Frog | Fugu | Gorilla | Horse | Human | Mouse | Rat | Sheep | Zebrafinch | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zebrafish | 0.014 | 0.014 | 0.014 | 0.013 | 0.013 | 0.013 | 0.012 | 0.014 | 0.013 | 0.014 | 0.014 | 0.013 | 0.014 | 0.013 | |
| Cat | 0.333 | 0.014 | 0.012 | 0.012 | 0.011 | 0.014 | 0.013 | 0.012 | 0.012 | 0.012 | 0.012 | 0.012 | 0.013 | 0.014 | |
| Chicken | 0.276 | 0.286 | 0.014 | 0.012 | 0.012 | 0.012 | 0.013 | 0.013 | 0.013 | 0.013 | 0.013 | 0.013 | 0.013 | 0.009 | |
| Chimpanzee | 0.302 | 0.182 | 0.289 | 0.013 | 0.012 | 0.014 | 0.013 | 0.009 | 0.012 | 0.008 | 0.012 | 0.012 | 0.013 | 0.014 | |
| Cow | 0.297 | 0.188 | 0.260 | 0.210 | 0.011 | 0.013 | 0.013 | 0.011 | 0.011 | 0.011 | 0.012 | 0.011 | 0.005 | 0.012 | |
| Dog | 0.281 | 0.153 | 0.219 | 0.196 | 0.146 | 0.013 | 0.012 | 0.010 | 0.009 | 0.010 | 0.010 | 0.010 | 0.011 | 0.012 | |
| Frog | 0.287 | 0.310 | 0.211 | 0.315 | 0.271 | 0.258 | 0.013 | 0.013 | 0.013 | 0.013 | 0.013 | 0.013 | 0.014 | 0.012 | |
| Fugu | 0.202 | 0.312 | 0.275 | 0.285 | 0.279 | 0.276 | 0.299 | 0.012 | 0.012 | 0.012 | 0.013 | 0.013 | 0.013 | 0.013 | |
| Gorilla | 0.275 | 0.187 | 0.239 | 0.075 | 0.152 | 0.135 | 0.266 | 0.243 | 0.010 | 0.003 | 0.010 | 0.011 | 0.011 | 0.013 | |
| Horse | 0.274 | 0.175 | 0.235 | 0.200 | 0.140 | 0.100 | 0.262 | 0.267 | 0.139 | 0.010 | 0.011 | 0.010 | 0.011 | 0.013 | |
| Human | 0.276 | 0.187 | 0.239 | 0.074 | 0.152 | 0.138 | 0.268 | 0.244 | 0.007 | 0.137 | 0.011 | 0.011 | 0.011 | 0.013 | |
| Mouse | 0.292 | 0.222 | 0.249 | 0.221 | 0.186 | 0.161 | 0.257 | 0.264 | 0.169 | 0.147 | 0.169 | 0.009 | 0.012 | 0.013 | |
| Rat | 0.289 | 0.233 | 0.250 | 0.219 | 0.181 | 0.164 | 0.258 | 0.268 | 0.166 | 0.152 | 0.166 | 0.082 | 0.011 | 0.013 | |
| Sheep | 0.292 | 0.202 | 0.258 | 0.216 | 0.029 | 0.150 | 0.276 | 0.279 | 0.159 | 0.145 | 0.159 | 0.192 | 0.187 | 0.013 | |
| Zebrafinch | 0.276 | 0.282 | 0.102 | 0.296 | 0.256 | 0.235 | 0.210 | 0.269 | 0.251 | 0.244 | 0.249 | 0.263 | 0.265 | 0.258 |
| Zebrafish | Cat | Chicken | Chimpanzee | Cow | Dog | Fugu | Gorilla | Horse | Human | Mouse | Rat | Sheep | Zebrafinch | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zebrafish | 0.014 | 0.014 | 0.014 | 0.014 | 0.016 | 0.014 | 0.014 | 0.014 | 0.014 | 0.014 | 0.014 | 0.014 | 0.015 | |
| Cat | 0.367 | 0.015 | 0.011 | 0.011 | 0.012 | 0.014 | 0.011 | 0.011 | 0.011 | 0.012 | 0.012 | 0.012 | 0.015 | |
| Chicken | 0.359 | 0.316 | 0.014 | 0.014 | 0.015 | 0.015 | 0.014 | 0.014 | 0.014 | 0.015 | 0.014 | 0.014 | 0.008 | |
| Chimpanzee | 0.361 | 0.138 | 0.307 | 0.011 | 0.013 | 0.015 | 0.004 | 0.011 | 0.003 | 0.013 | 0.013 | 0.011 | 0.014 | |
| Cow | 0.358 | 0.178 | 0.337 | 0.171 | 0.013 | 0.015 | 0.011 | 0.011 | 0.011 | 0.012 | 0.013 | 0.005 | 0.014 | |
| Dog | 0.420 | 0.182 | 0.371 | 0.216 | 0.272 | 0.016 | 0.013 | 0.013 | 0.013 | 0.014 | 0.014 | 0.014 | 0.015 | |
| Fugu | 0.329 | 0.338 | 0.347 | 0.334 | 0.339 | 0.389 | 0.014 | 0.015 | 0.014 | 0.014 | 0.015 | 0.014 | 0.015 | |
| Gorilla | 0.362 | 0.139 | 0.306 | 0.019 | 0.170 | 0.221 | 0.333 | 0.011 | 0.003 | 0.013 | 0.013 | 0.012 | 0.013 | |
| Horse | 0.364 | 0.133 | 0.303 | 0.131 | 0.173 | 0.210 | 0.325 | 0.133 | 0.011 | 0.012 | 0.012 | 0.011 | 0.014 | |
| Human | 0.358 | 0.135 | 0.306 | 0.015 | 0.167 | 0.219 | 0.330 | 0.012 | 0.128 | 0.013 | 0.013 | 0.012 | 0.013 | |
| Mouse | 0.381 | 0.192 | 0.338 | 0.190 | 0.240 | 0.288 | 0.357 | 0.191 | 0.201 | 0.191 | 0.009 | 0.013 | 0.015 | |
| Rat | 0.380 | 0.200 | 0.340 | 0.202 | 0.250 | 0.284 | 0.358 | 0.203 | 0.206 | 0.204 | 0.095 | 0.013 | 0.014 | |
| Sheep | 0.372 | 0.180 | 0.346 | 0.170 | 0.034 | 0.268 | 0.337 | 0.171 | 0.171 | 0.166 | 0.239 | 0.247 | 0.014 | |
| Zebrafinch | 0.370 | 0.317 | 0.071 | 0.311 | 0.338 | 0.376 | 0.352 | 0.311 | 0.305 | 0.307 | 0.330 | 0.336 | 0.343 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, D.Y.; Smith, D.G.; Hardeland, R.; Yang, M.Y.; Xu, H.L.; Zhang, L.; Yin, H.D.; Zhu, Q. Melatonin Receptor Genes in Vertebrates. Int. J. Mol. Sci. 2013, 14, 11208-11223. https://doi.org/10.3390/ijms140611208
Li DY, Smith DG, Hardeland R, Yang MY, Xu HL, Zhang L, Yin HD, Zhu Q. Melatonin Receptor Genes in Vertebrates. International Journal of Molecular Sciences. 2013; 14(6):11208-11223. https://doi.org/10.3390/ijms140611208
Chicago/Turabian StyleLi, Di Yan, David Glenn Smith, Rüdiger Hardeland, Ming Yao Yang, Huai Liang Xu, Long Zhang, Hua Dong Yin, and Qing Zhu. 2013. "Melatonin Receptor Genes in Vertebrates" International Journal of Molecular Sciences 14, no. 6: 11208-11223. https://doi.org/10.3390/ijms140611208
APA StyleLi, D. Y., Smith, D. G., Hardeland, R., Yang, M. Y., Xu, H. L., Zhang, L., Yin, H. D., & Zhu, Q. (2013). Melatonin Receptor Genes in Vertebrates. International Journal of Molecular Sciences, 14(6), 11208-11223. https://doi.org/10.3390/ijms140611208





