Fluctuation of Global Gene Expression by Endogenous miRNA Response to the Introduction of an Exogenous miRNA
Abstract
:1. Introduction
2. Results and Discussion
2.1. Microarray Profiling of the Reduced Expression of Exo-miRNA Target Genes
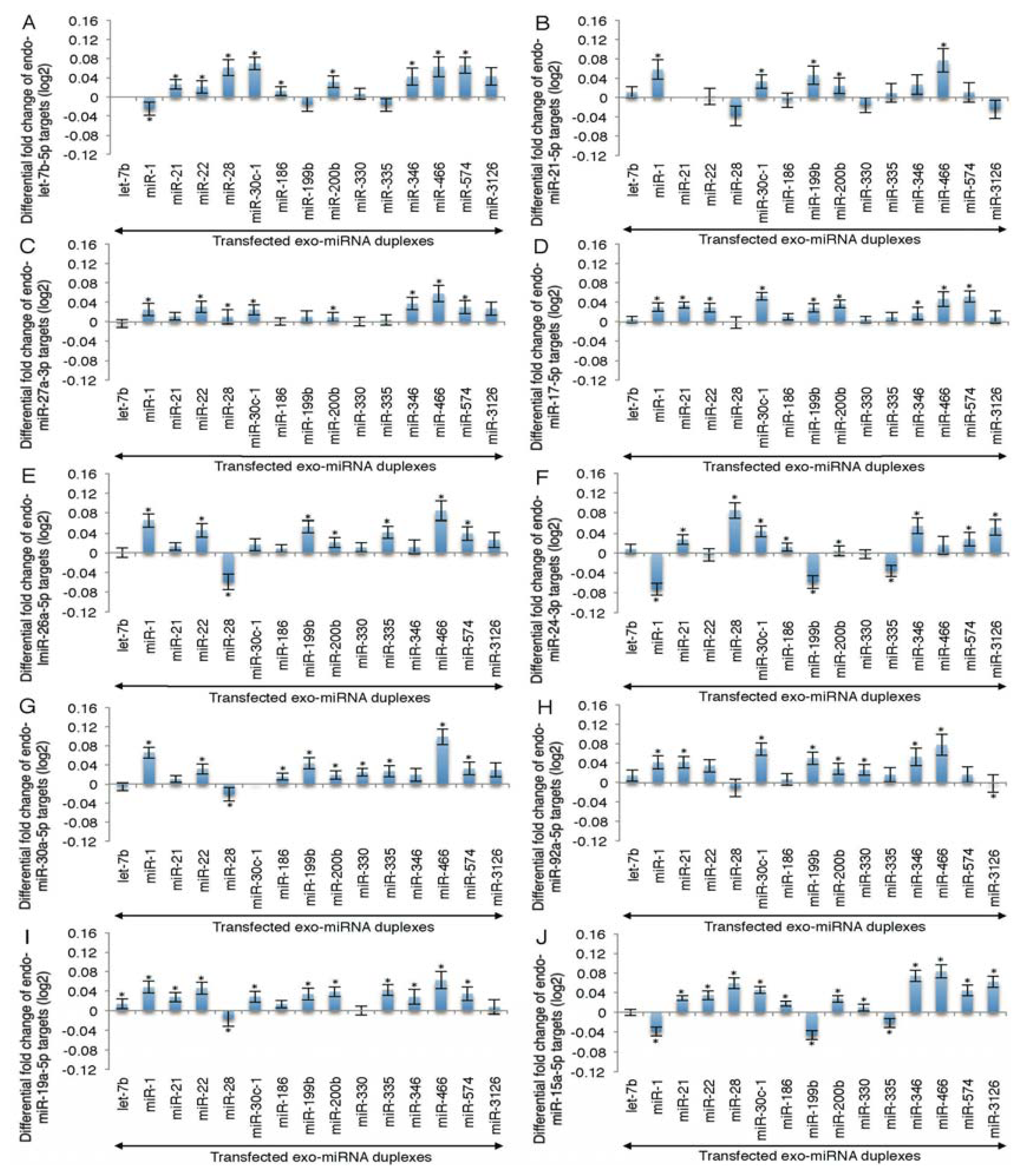
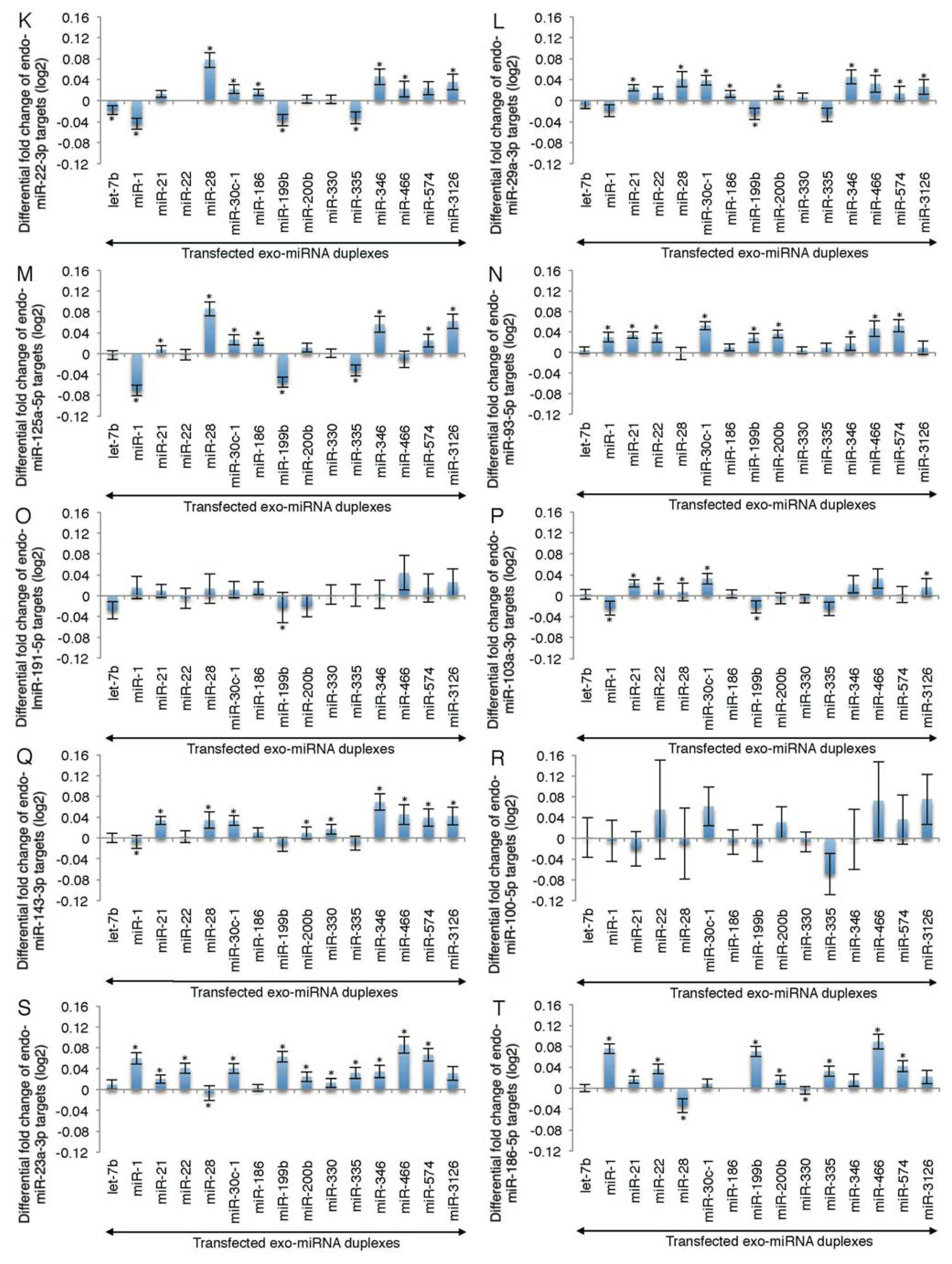
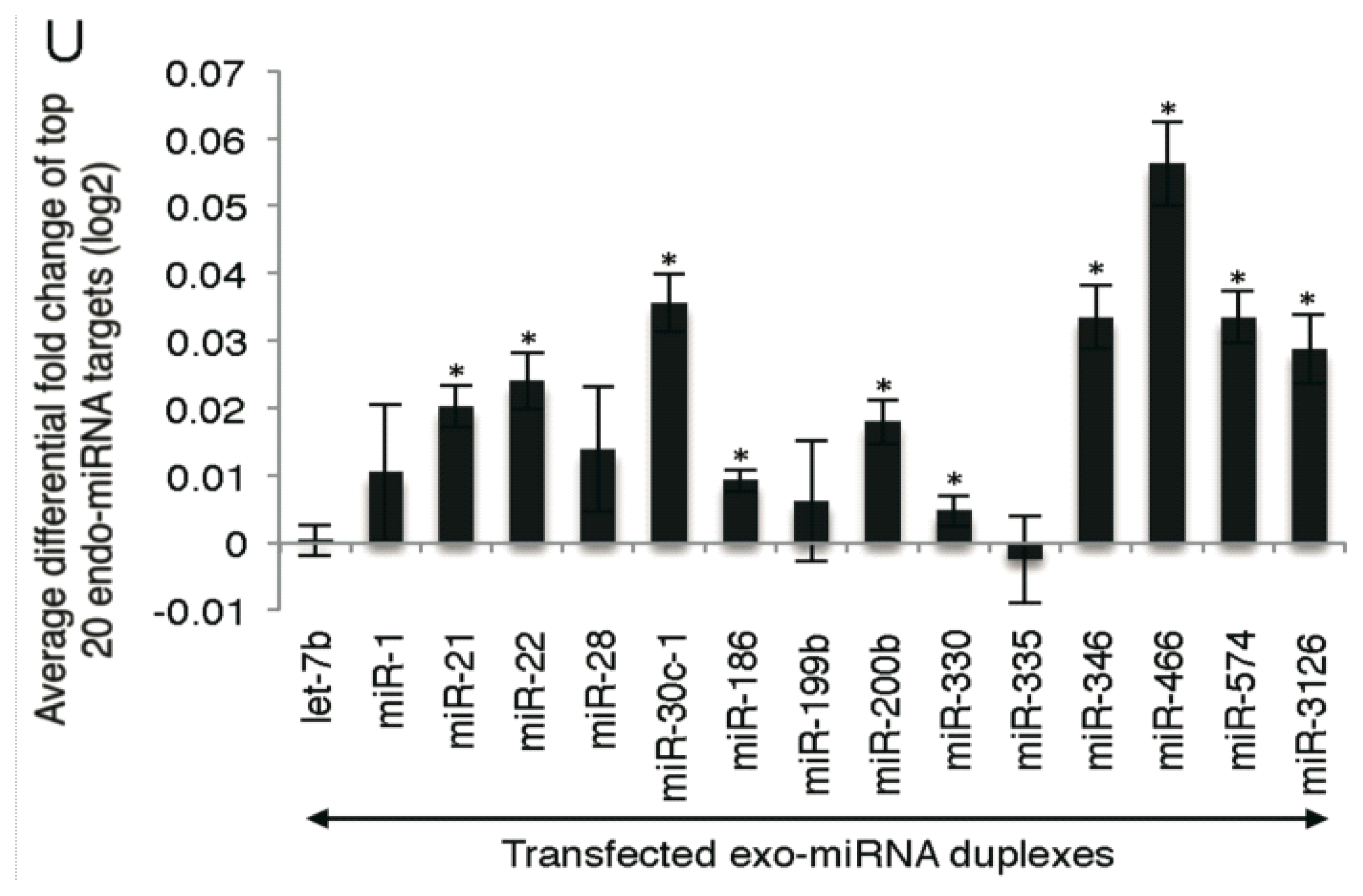
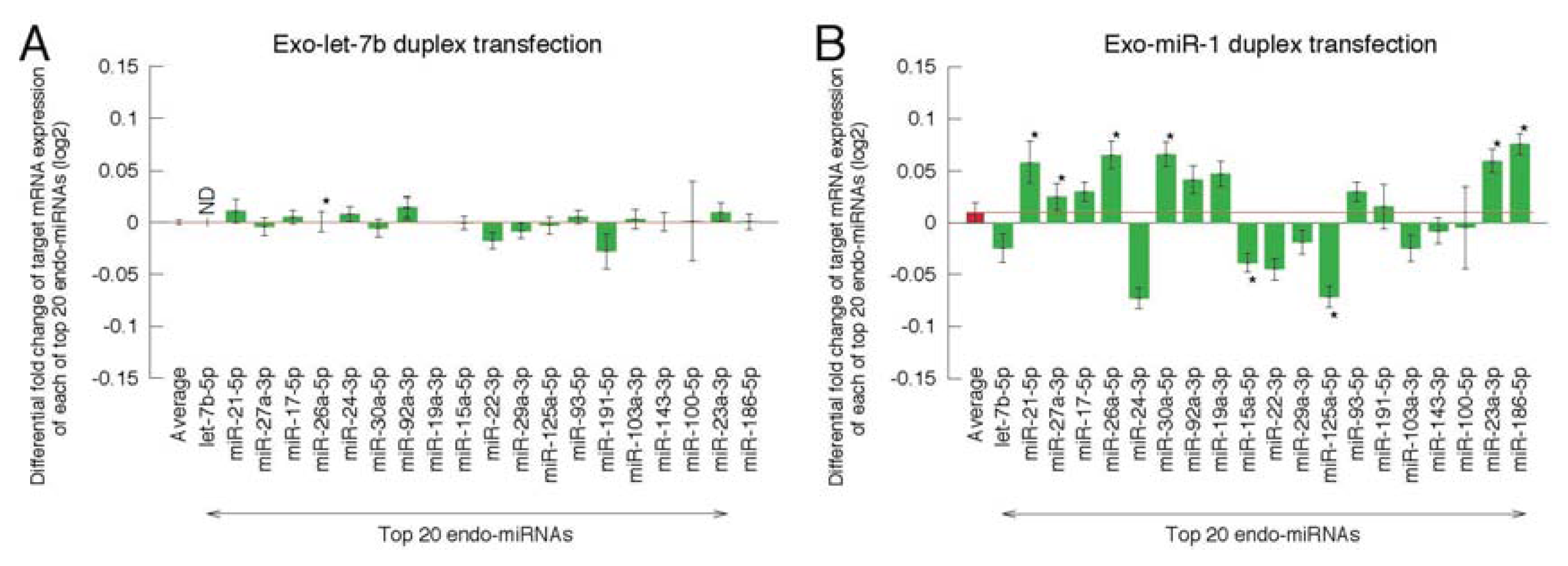
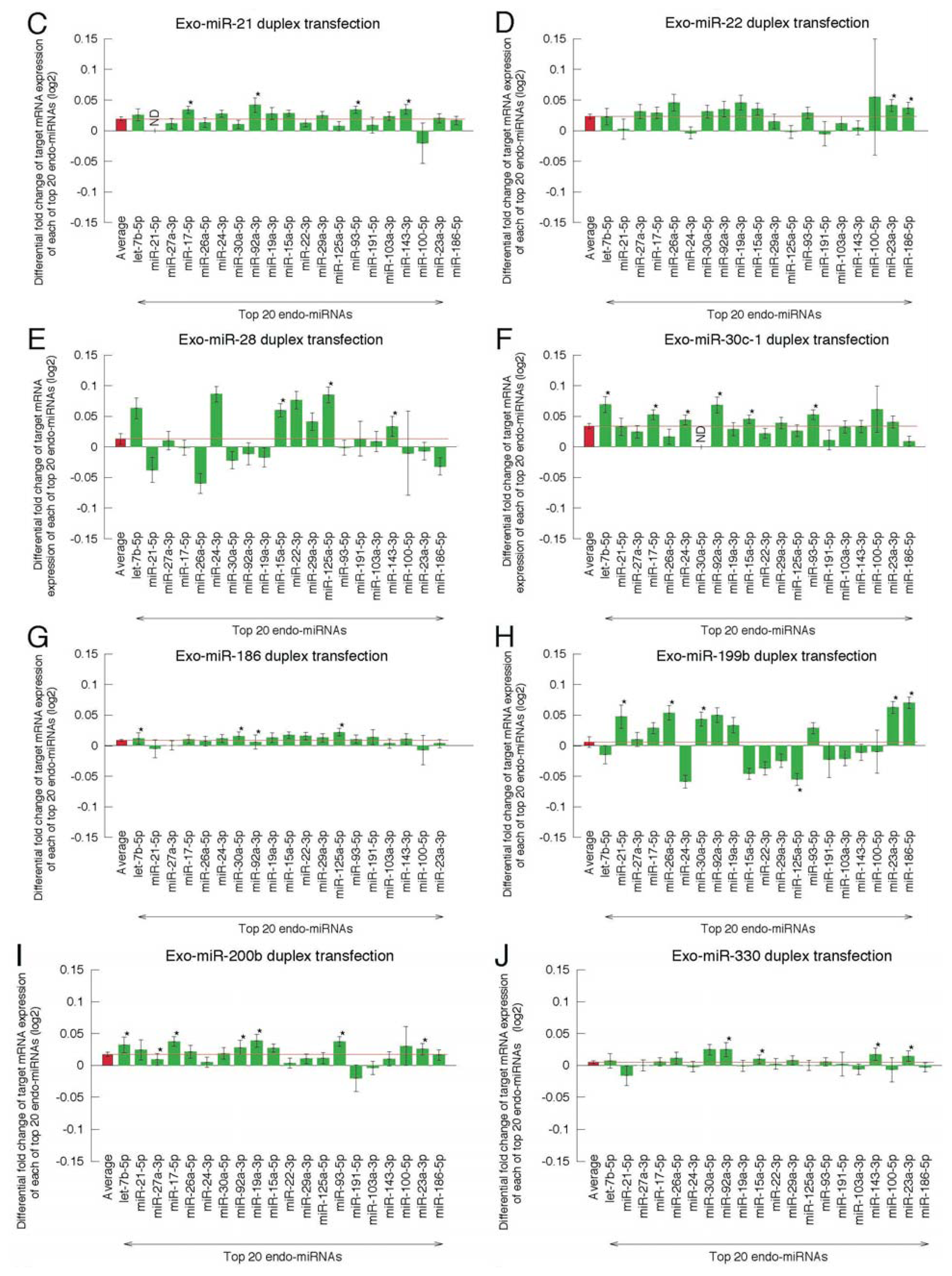
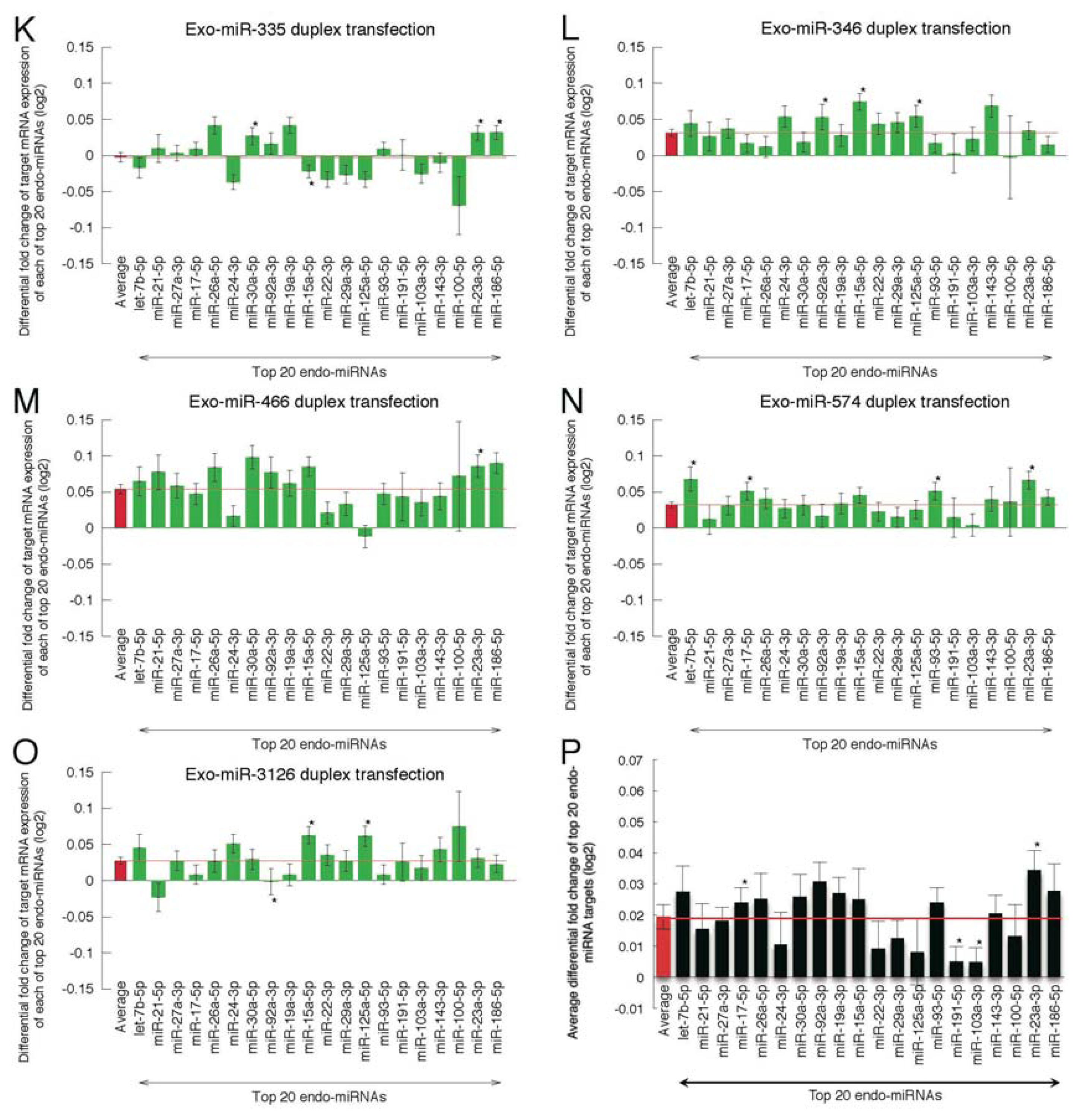
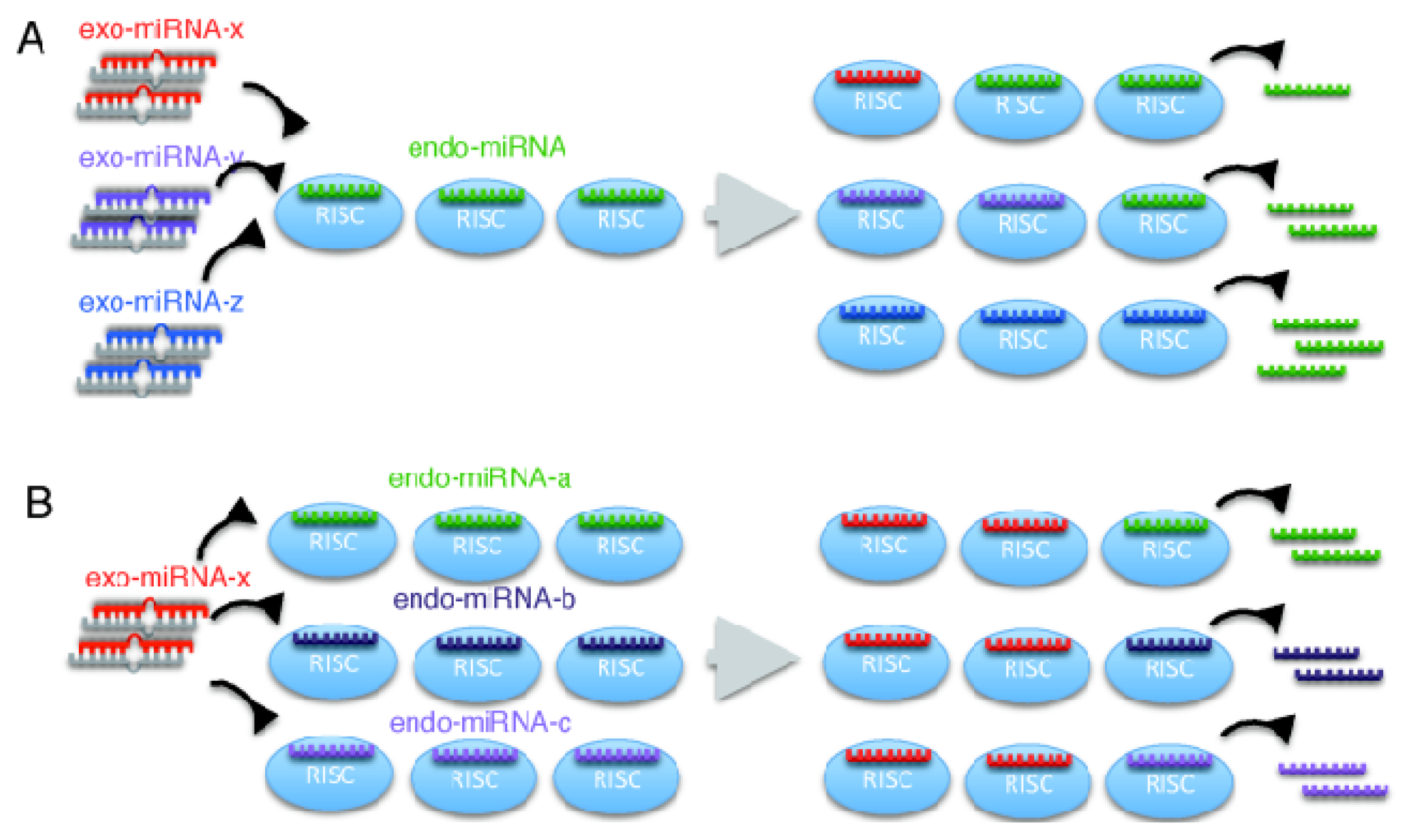
2.2. Microarray Profiling of the Increased Expression of Endo-miRNA Target Genes Resulting from the Introduction of Exo-miRNAs
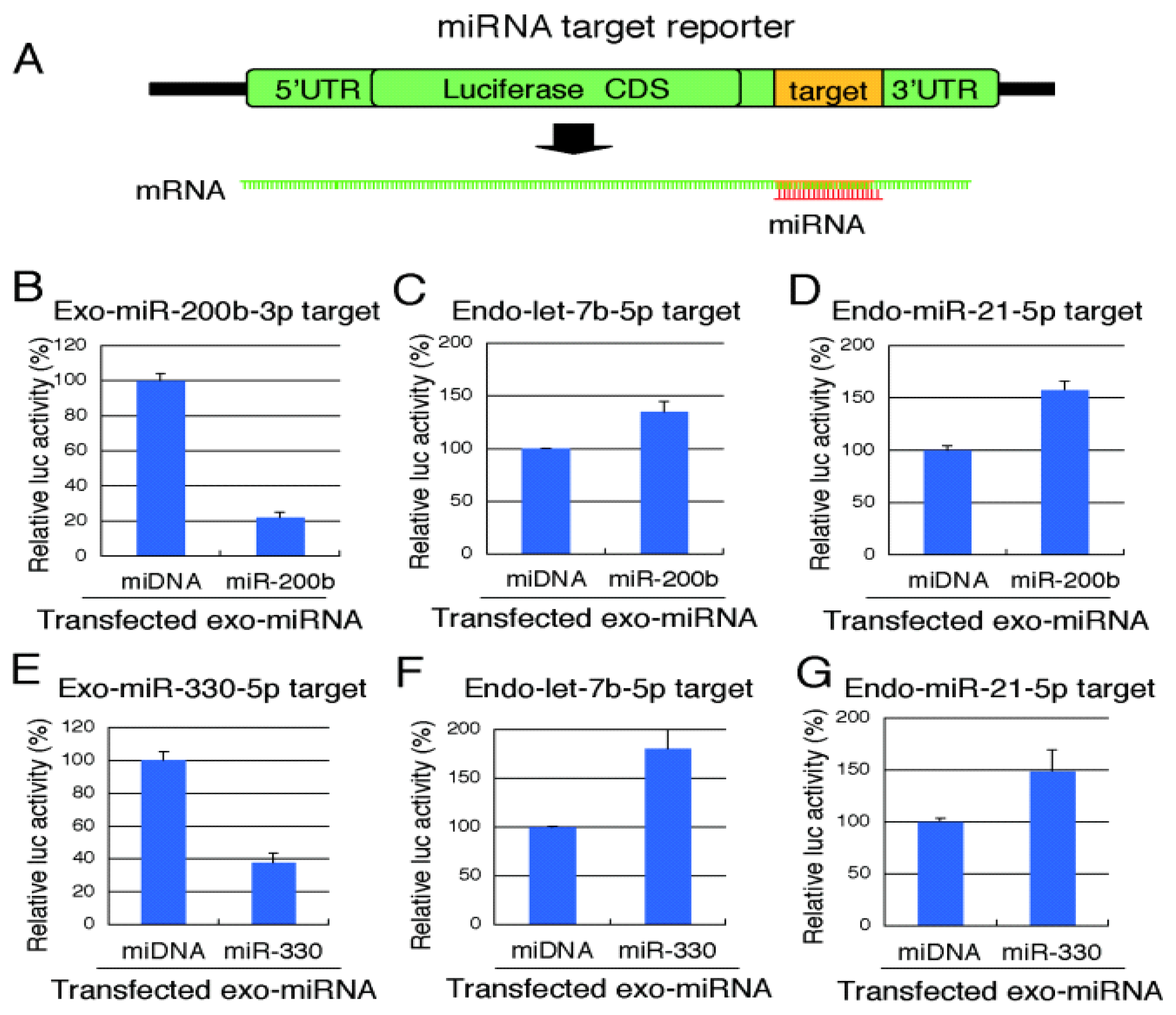
2.3. Reporter Analysis of Exo-miRNA and Endo-miRNA Target Expression in Cells Transfected with Exo-miRNA
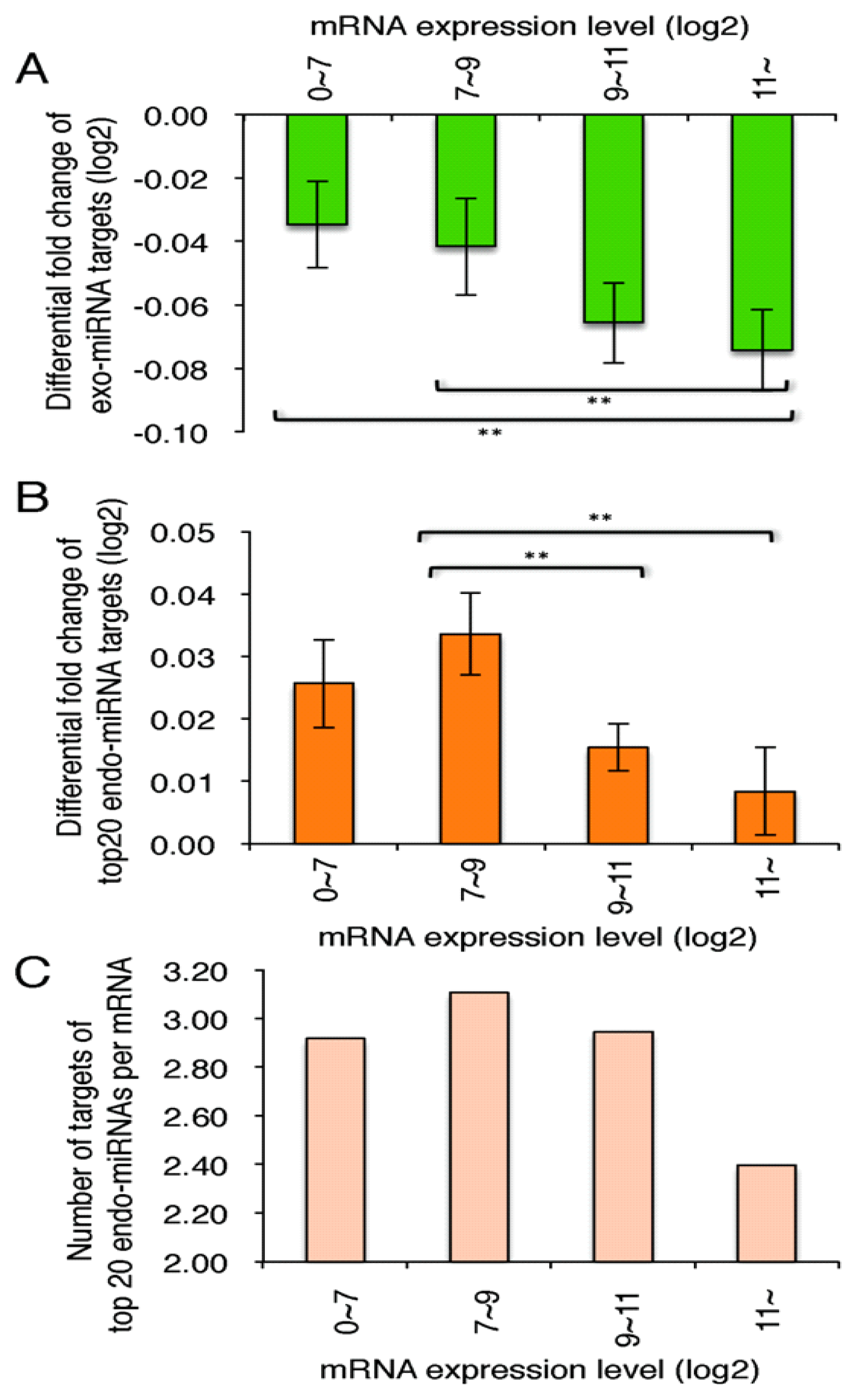
2.4. Exo-miRNA Targets with Short 3′ UTRs Are Efficiently Downregulated and Endo-miRNA Targets with Long 3′UTRs Are Efficiently Upregulated by the Introduction of Exo-miRNA Duplex
2.5. Highly Expressed Exo-miRNA Targets Are Efficiently Downregulated and Endo-miRNA Targets with Low Expression Levels Are Efficiently Upregulated by the Transfection of the Exo-miRNA Duplex
3. Experimental Section
3.1. Cell Culture and miRNA Synthesis
3.2. Construction of Luciferase Reporters
3.3. Microarray Analysis
3.4. Quantitative RT-PCR
4. Conclusions
Supplementary Information
ijms-14-11171-s001.pdfSupplementary Information
ijms-14-11171-s002.pdfAcknowledgments
Conflict of Interest
References
- Rana, T.M. Illuminating the silence: Understanding the structure and function of small RNAs. Nat. Rev. Mol. Cell Biol 2007, 8, 23–36. [Google Scholar]
- Filipowicz, W.; Bhattacharyya, S.N.; Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight? Nat. Rev. Genet 2008, 9, 102–114. [Google Scholar]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar]
- Grimson, A.; Farh, K.K.; Johnston, W.K.; Garrett-Engele, P.; Lim, L.P.; Bartel, D.P. MicroRNA targeting specificity in mammals: Determinants beyond seed pairing. Mol. Cell 2007, 27, 91–105. [Google Scholar]
- Lewis, B.P.; Burge, C.B.; Bartel, D.P. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005, 120, 15–20. [Google Scholar]
- Lim, L.P.; Lau, N.C.; Garrett-Engele, P.; Grimson, A.; Schelter, J.M.; Castle, J.; Bartel, D.P.; Linsley, P.S.; Johnson, J.M. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 2005, 433, 769–773. [Google Scholar]
- Grimm, D.; Streetz, K.L.; Jopling, C.L.; Storm, T.A.; Pandey, K.; Davis, C.R.; Marion, P.; Salazar, F.; Kay, M.A. Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature 2006, 441, 537–541. [Google Scholar]
- Castanotto, D.; Sakurai, K.; Lingeman, R.; Li, H.; Shively, L.; Aagaard, L.; Soifer, H.; Gatignol, A.; Riggs, A.; Rossi, J.J. Combinatorial delivery of small interfering RNAs reduces RNAi efficacy by selective incorporation into RISC. Nucleic Acids Res 2007, 35, 5154–5164. [Google Scholar]
- Stewart, C.K.; Li, J.; Golovan, S.P. Adverse effects induced by short hairpin RNA expression in porcine fetal fibroblasts. Biochem. Biophys. Res. Commun 2008, 370, 113–117. [Google Scholar]
- Khan, A.A.; Betel, D.; Miller, M.L.; Sander, C.; Leslie, C.S.; Marks, D.S. Transfection of small RNAs globally perturbs gene regulation by endogenous microRNAs. Nat. Biotechnol 2009, 27, 549–555. [Google Scholar]
- Yi, R.; Qin, Y.; Macara, J.G.; Cullen, B.R. Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev 2003, 17, 3011–3016. [Google Scholar]
- Lund, E.; Guttinger, S.; Calado, A.; Dahlberg, J.E.; Kutay, U. Nuclear export of microRNA precursors. Science 2004, 303, 95–98. [Google Scholar]
- Bohnsack, M.T.; Czaplinski, K.; Gorlich, D. Exportin-5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA 2004, 10, 185–191. [Google Scholar]
- Kim, V.N. MicroRNA precursors in motion; exportin-5 mediates their nuclear export. Trends Cell Biol 2004, 14, 156–159. [Google Scholar]
- Vankoningsloo, S.; de Longueville, F.; Evrard, S.; Rahier, P.; Houbion, A.; Fattaccioli, A.; Gastellier, M.; Remacle, J.; Raes, M.; Ranard, P.; et al. Gene expression silencing with “specific” small interfering RNA goes beyond specificity–a study of key parameters to take into account in the onset of small interfering RNA off-target effects. FEBS J 2008, 275, 2738–2753. [Google Scholar]
- Gregory, R.I.; Chendrimada, T.P.; Cooch, N.; Shienkhattar, R. Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell 2005, 123, 631–540. [Google Scholar]
- Haase, A.D.; Jaskiewicz, L.; Zhang, H.; Lainé, S.; Sack, R.; Gatignol, A.; Filipowicz, S. TRBP, a regulator of cellular PKR and HIV-1 virus expression, interacts with Dicer and functionas in RNA silencing. EMBO J 2005, 6, 961–967. [Google Scholar]
- Macrae, I.J.; Ma, E.; Zhou, M.; Robinson, C.V.; Doudna, J.A. In vitro reconstitution of the human RISC-loading complex. Proc. Natl. Acad. Sci. USA 2008, 105, 512–517. [Google Scholar]
- Winter, J.; Jung, S.; Keller, S.; Gregory, R.; Diederichs, S. Many roads to maturity: microRNA biogenesis pathways and their regulation. Nat. Cell Biol 2009, 11, 228–234. [Google Scholar]
- Matranga, C.; Tomari, Y.; Shin, C.; Bartal, D.P.; Zamore, P.D. Passenger-strand cleavage facilitates assembly of siRNA into Ago2-containing RNAi enzyme complexes. Cell 2005, 123, 607–620. [Google Scholar]
- Leuschner, P.J.; Ameres, S.L.; Kueng, S.; Martinez, J. Cleavage of the siRNA passenger strand during RISC assembly in human cells. EMBO Rep 2006, 7, 314–320. [Google Scholar]
- Tolia, N.H.; Joshua-Tor, L. Slicer and the Argonaute. Nat. Chem. Biol 2006, 3, 36–43. [Google Scholar]
- Czech, B.; Hannon, G.J. Small RNA sorting: Matchmaking for Argonautes. Nat. Rev. Genetics 2011, 12, 19–31. [Google Scholar]
- Khvorova, A.; Reynolds, A.; Jayasena, S.D. Functional siRNAs and miRNAs exhibit strand bias. Cell 2003, 115, 209–215. [Google Scholar]
- Schwarz, D.S.; Hutvágner, G.; Du, T.; Xu, A.; Aronin, N.; Zamore, P.D. Asymmetry in the assembly of the RNAi enzyme complex. Cell 2003, 115, 199–205. [Google Scholar]
- Ui-Tei, K.; Naito, Y.; Takahashi, F.; Haraguchi, T.; Ohki-Hamazaki, H.; Juni, A.; Ueda, R.; Saigo, K. Guidelines for the selection of highly effective siRNA sequences for mammalian and chick RNA interference. Nucleic Acids Res 2004, 32, 936–948. [Google Scholar]
- Yoda, M.; Kawamata, T.; Paroo, Z.; Ye, X.; Iwasaki, S.; Liu, Q.; Tomari, Y. ATP-dependent human RISC assembly pathways. Nat. Struct. Mol. Biol 2010, 17, 17–23. [Google Scholar]
- Griffiths-Jones, S.; Saini, H.K.; van Dongen, S.; Enright, A.J. MiRBase tools for microRNA genomics. Nucleic Acids Res 2008, 36, D154–D158. [Google Scholar]
- Jackson, A.L.; Burchard, J.; Schelter, J.; Chau, B.N.; Cleary, M.; Lim, L.; Linsley, P.S. Widespread siRNA “off-target” transcript silencing mediated by seed region sequence complementarity. RNA 2006, 12, 1179–1187. [Google Scholar]
- Mayr, C.; Bartel, D.P. Widespread shortening of 3′UTRs by alternative cleavage and polyadenylation activates oncogenes in cancer cells. Cell 2009, 138, 673–684. [Google Scholar]
- Saito, T.; Sætrom, P. Target gene expression levels and competition between transfected and endogenous microRNAs are strong confounding factors in microRNA high-throughput experiments. Silence 2012, 3, 3. [Google Scholar]
- Hubbell, E.; Liu, W.M.; Mei, R. Robust estimation for expression analysis. Bioinformatics 2002, 18, 1585–1592. [Google Scholar]
- Bolstad, B.M.; Irizarry, R.A.; Gautier, L.; Wu, Z. Preprocessing High-Density Oligonucleotide Arrays. In Bioinformatics and Computational Biology Solutions Using R and Bioconductor; Gentleman, R., Carey, V.J., Huber, W., Irizarry, R.A., Dudoit, S., Eds.; Springer: New York, NY, USA, 2005; pp. 13–32. [Google Scholar]


© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nagata, Y.; Shimizu, E.; Hibio, N.; Ui-Tei, K. Fluctuation of Global Gene Expression by Endogenous miRNA Response to the Introduction of an Exogenous miRNA. Int. J. Mol. Sci. 2013, 14, 11171-11189. https://doi.org/10.3390/ijms140611171
Nagata Y, Shimizu E, Hibio N, Ui-Tei K. Fluctuation of Global Gene Expression by Endogenous miRNA Response to the Introduction of an Exogenous miRNA. International Journal of Molecular Sciences. 2013; 14(6):11171-11189. https://doi.org/10.3390/ijms140611171
Chicago/Turabian StyleNagata, Yoshiro, Eigo Shimizu, Naoki Hibio, and Kumiko Ui-Tei. 2013. "Fluctuation of Global Gene Expression by Endogenous miRNA Response to the Introduction of an Exogenous miRNA" International Journal of Molecular Sciences 14, no. 6: 11171-11189. https://doi.org/10.3390/ijms140611171



