Oenothein B Suppresses Lipopolysaccharide (LPS)-Induced Inflammation in the Mouse Brain
Abstract
:1. Introduction
2. Results
2.1. Effect of Oenothein B on the Abnormal Behavior Induced by LPS
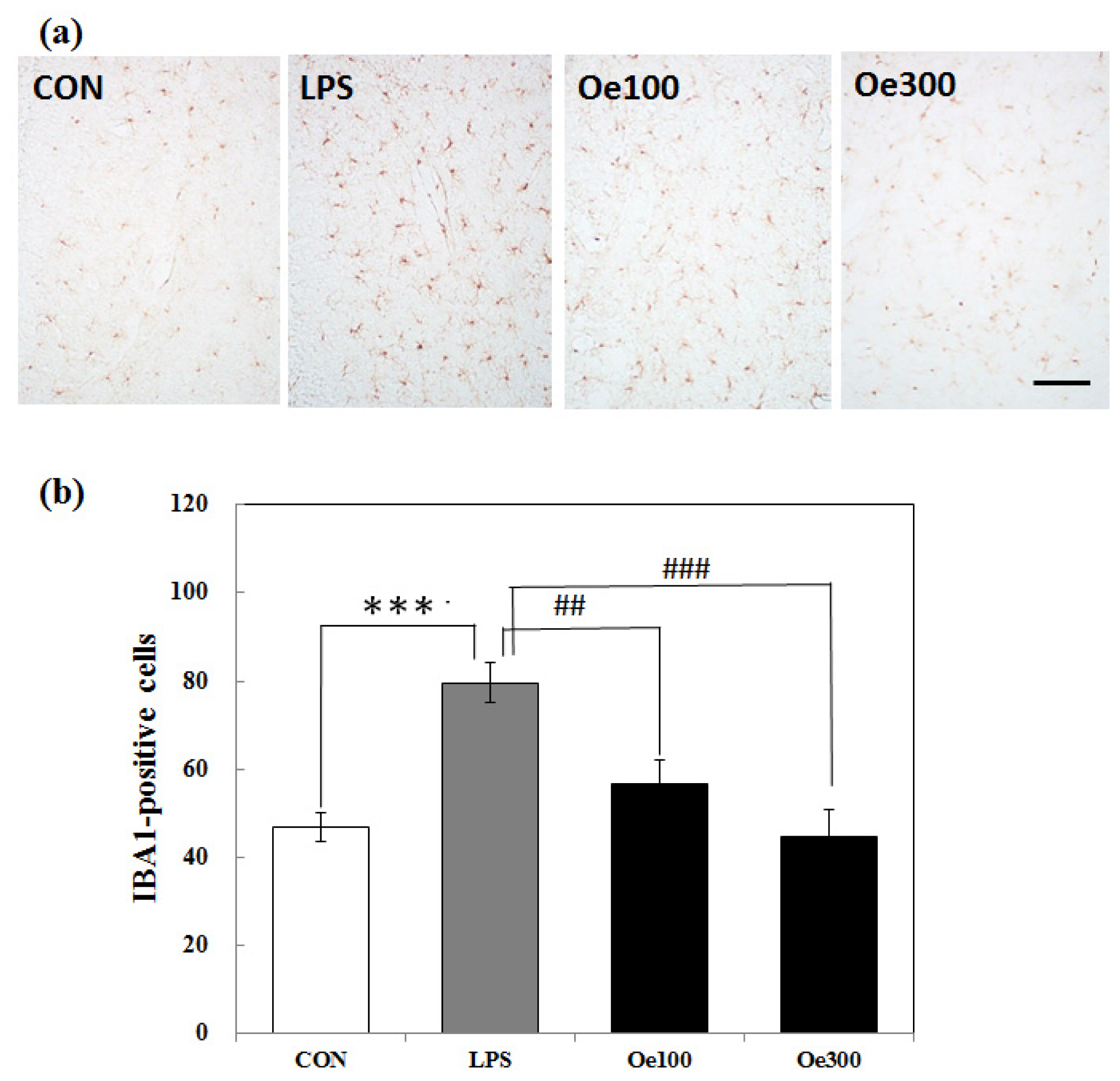
2.2. Effect of Oenothein B on the Microglial Activation Induced by LPS
2.3. Effect of Oenothein B on the COX-2 Expression Induced by LPS
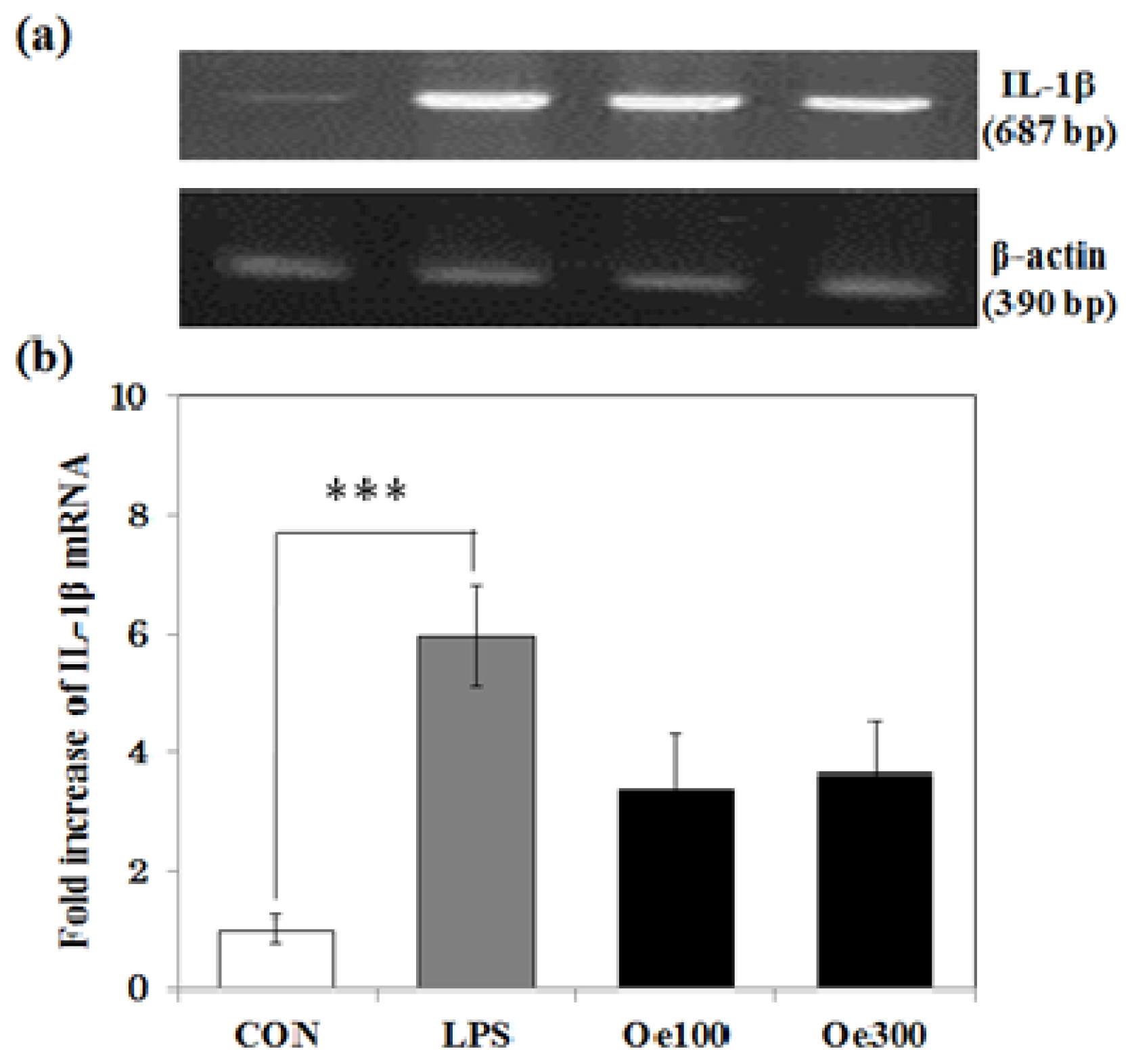
2.4. Effect of Oenothein B on the IL-1β mRNA Expression Induced by LPS
3. Discussion
4. Experimental Section
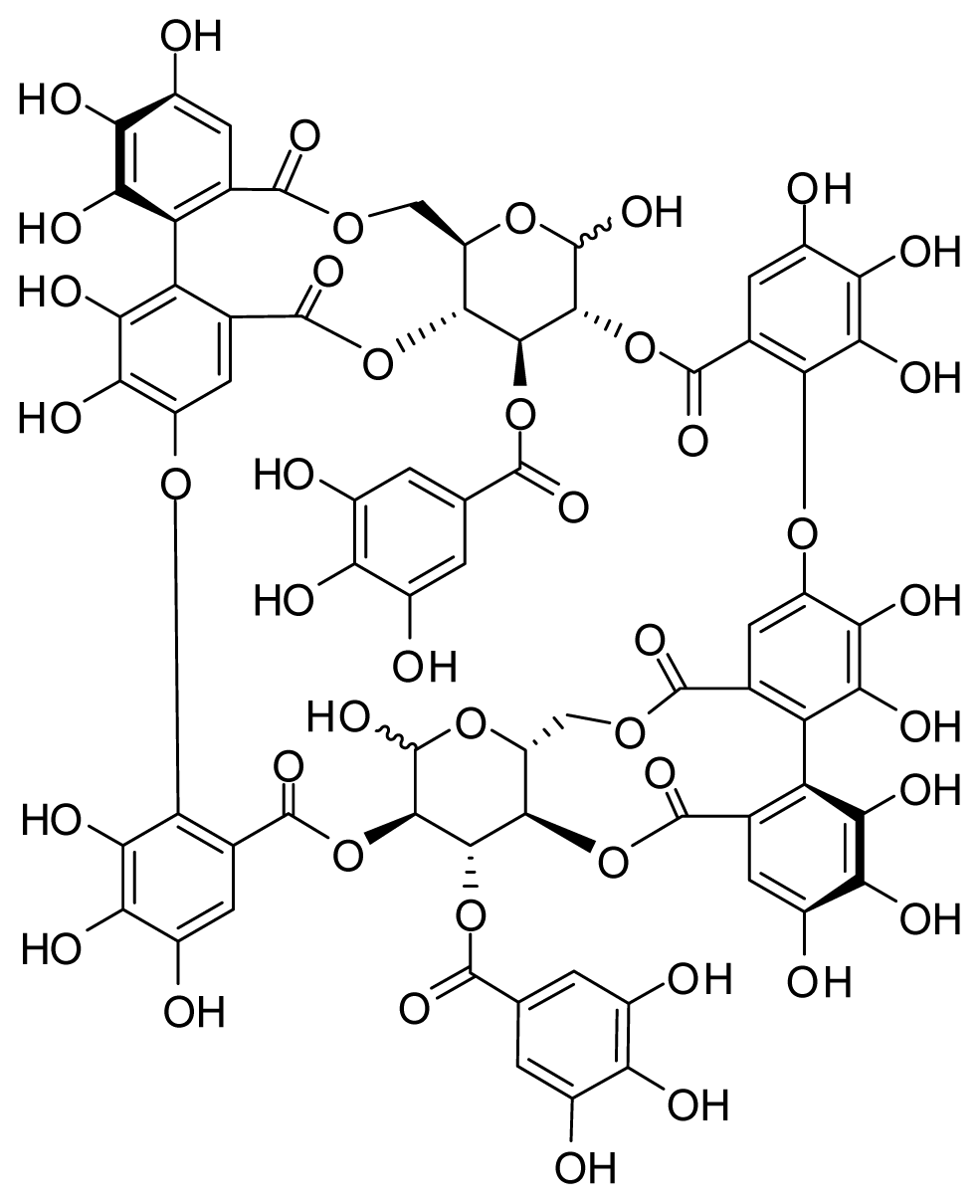
4.1. Preparation of Oenothein B
4.2. Animals
4.3. Oenothein B Treatment
4.4. LPS Treatment
4.5. Open-Field Test
4.6. Immunohistochemistry
4.7. RT-PCR Procedures
4.8. Statistical Analysis
5. Conclusions
References
- Amakura, Y.; Yoshimura, M.; Sugimoto, N.; Yamazaki, T.; Yoshida, T. Marker constituents of the natural antioxidant Eucalyptus leaf extract for the evaluation of food additives. Biosci. Biotechnol. Biochem 2009, 73, 1060–1065. [Google Scholar]
- Ducrey, B.; Marston, A.; Göhring, S.; Hartmann, R.W.; Hostettmann, K. Inhibition of 5 alpha-reductase and aromatase by the ellagitannins oenothein A and oenothein B from Epilobium species. Planta Med 1997, 63, 111–114. [Google Scholar]
- Yoshida, T.; Chou, T.; Matsuda, M.; Yasuhara, T.; Yazaki, K.; Hatano, T.; Nitta, A.; Okuda, T. Woodfordin D and oenothein A, trimeric hydrolyzable tannins of macro-ring structure with antitumor actrivity. Chem. Pharm. Bull 1991, 39, 1157–1162. [Google Scholar]
- Tyler, V.E. Some recent advances in herbal medicines. Pharm. Int 1986, 17, 161–169. [Google Scholar]
- Vitalone, A.; McColl, J.; Thoma, D.; Costa, L.G.; Tita, B. Characterization of the effect of Epilobium extracts on human cell proliferation. Pharmacology 2003, 69, 79–87. [Google Scholar]
- Schepetkin, I.A.; Kirpotina, L.N.; Jarissa, L.; Khlebnikov, A.I.; Blaskovich, C.L.; Jutila, M.A.; Quinn, M.T. Immunomodulatory activity of oenothein B isolated from Epilobium angustifolium. J. Immun 2009, 183, 6754–6766. [Google Scholar]
- Tóth, B.H.; Blazics, B.; Kéry, Á. Polyphenol composition and antioxidant capacity of Epilobium species. J. Pharmacol. Biomed. Anal 2009, 49, 26–31. [Google Scholar]
- Kiss, A.K.; Bazylko, A.; Filipek, A.; Granica, S.; Jaszewska, E.; Kiarszys, U.; Kośmider, A.; Piwowarski, J. Oenothein B’s contribution to the anti-inflammatory and anti-oxidant activity of Epilobium sp. Phytomedicine 2011, 18, 557–560. [Google Scholar]
- Schmid, D.; Gruber, M.; Piskaty, C.; Woehs, F.; Renner, A.; Nagy, Z.; Kaltenboeck, A.; Wasserscheid, T.; Bazylko, A.; Kiss, A.K.; et al. Inhibition of NF-κB-dependent cytokine and inducible nitric oxide synthesis by the macrocyclic ellagtannin oenothein B in TLR-stimulated RAW 264.7 macrophages. J. Nat. Prod 2012, 75, 870–875. [Google Scholar]
- Kiss, A.; Kowalski, J.; Melzig, M.F. Induction of neutral endopeptidase activity in PC-3 cells by aqueous extract of Epilobium angustifolium L. and oenothein B. Phytomedicine 2006, 13, 284–289. [Google Scholar]
- Miyamoto, K.; Nomura, M.; Sasakura, M.; Matsui, E.; Koshiura, R.; Furukawa, T.; Hatano, T.; Yoshida, T.; Okuda, T. Antitumor activity of oenothein B, a unique macrocyclic ellagitannin. Jpn. J. Cancer Res 1993, 84, 99–103. [Google Scholar]
- Yoshida, T.; Hatano, T.; Ito, H. Chemistry and function of vegetable polyphenols with high molecular weights. Biofactors 2000, 13, 121–125. [Google Scholar]
- Sakagami, H.; Jiang, Y.; Kusama, K.; Atsumi, T.; Ueda, T.; Toguchi, M.; Iwakura, I.; Satoh, H.; Ito, H.; Hatano, T.; et al. Cytotoxic activity of hydrolyzable tannins against human oral tumor cell lines. A possible mechanism. Phytomedicine 2000, 7, 39–47. [Google Scholar]
- Yoshimura, M.; Akiyama, H.; Kondo, K.; Sakata, K.; Matsuoka, H.; Amakura, Y.; Teshima, R.; Yoshida, T. Immunological effects of oenothein B, an ellagitannin dimer, on dendritic cells. Int. J. Mol. Sci 2013, 14, 46–56. [Google Scholar]
- Luheshi, G.N. Cytokines and fever. Mechanisms and sites of action. Ann. N. Y. Acad. Sci 1998, 856, 83–89. [Google Scholar]
- Dinarello, C.A. Cytokines as endogenous pyrogens. J. Infect. Dis 1999, 179, S294–S304. [Google Scholar]
- Kilbourn, R.G.; Belloni, P. Endothelial cell production of nitrogen oxides in response to interferon gamma in combination with tumor necrosis factor, interleukin-1, or endotoxin. J. Natl. Cancer Inst 1990, 82, 772–776. [Google Scholar]
- Rivest, S. Molecular insights on the cerebral innate immune system. Brain Behav. Immun 2003, 17, 13–19. [Google Scholar]
- Matsumura, K.; Kaihatsu, S.; Imai, T.; Terao, A.; Shiraki, T.; Kobayashi, S. Cyclooxygenase in the vagal afferents. Is it involved in the brain prostaglandin response evoked by lipopolysaccharide? Auton. Neurosci 2000, 85, 88–92. [Google Scholar]
- Cao, C.; Matsumura, K.; Yamagata, K.; Watanabe, Y. Induction by lipopolysaccharide of cyclooxygenase-2 mRNA in rat brain; its possible role in the febrile response. Brain Res 1995, 697, 187–196. [Google Scholar]
- Chen, Z.; Jalabi, W.; Shpargel, K.B.; Farabaugh, K.T.; Dutta, R.; Yin, X.; Kidd, G.J.; Bergmann, C.C.; Stohlman, S.A.; Trapp, B.D. Lipopolysaccharide-induced microglial activation and neuroprotection against experimental brain injury is independent of hematogenous TLR4. J. Neurosci 2012, 32, 11706–11715. [Google Scholar]
- O’Connor, J.C.; Lawson, M.A.; André, C.; Moreau, M.; Lestage, J.; Castanon, N.; Kelley, K.W.; Dantzer, R. Lipopolysaccharide-induced depressive-like behavior is mediated by indoleamine 2,3-dioxygenase activation in mice. Mol. Psychiatr 2009, 14, 511–522. [Google Scholar]
- Noble, F.; Rubira, E.; Boulanouar, M.; Palmier, B.; Plotkine, M.; Warnet, J.-M.; Marchand-Leroux, C.; Massicot, F. Acute systemic inflammation induces central mitochondrial damage and mnesic deficit in adult Swiss mice. Neurosci. Lett 2007, 424, 106–110. [Google Scholar]
- Shaw, K.N.; Commins, S.; O’Mara, S.M. Lipopolysaccharide causes deficits in spatial learning in the water maze but not in BDNF expression in the rat dentate gyrus. Behav. Brain Res 2001, 124, 47–54. [Google Scholar]
- Kreutzberg, G.W. Microglia: A sensor for pathological events in the CNS. Trends Neurosci 1996, 19, 312–318. [Google Scholar]
- Vila, M.; Jackson-Lewis, V.; Guégan, C.; Wu, D.C.; Teismann, P.; Choi, D.K.; Tieu, K.; Przedborski, S. The role of glial cells in Parkinson’s disease. Curr. Opin. Neurol 2001, 14, 483–489. [Google Scholar]
- Teismann, P.; Schulz, J.B. Cellular pathology of Parkinson’s disease: Astrocytes, microglia and inflammation. Cell Tissue Res 2004, 318, 149–161. [Google Scholar]
- Okuyama, S.; Minami, S.; Shimada, N.; Makihata, N.; Nakajima, M.; Furukawa, Y. Anti-inflammatory and neuroprotective effect of auraptene, a citrus coumarin, following cerebral global ischemia in mice. Eur. J. Pharm 2013, 699, 118–123. [Google Scholar]
- Chung, D.W.; Yoo, K.-Y.; Hwang, I.K.; Kim, D.W.; Chung, J.Y.; Lee, C.H.; Choi, J.H.; Choi, S.Y.; Youn, H.Y.; Lee, I.S.; et al. Systemic administration of lipopolysaccharide induces cyclooxygenase-2 immunoreactivity in endothelium and increases microglia in the mouse hippocampus. Cell Mol. Neurobiol 2010, 30, 531–541. [Google Scholar]
- Farina, C.; Aloisi, F.; Meinl, E. Astrocytes are active players in cerebral innate immunity. Trends Immunol 2007, 28, 138–145. [Google Scholar]
- Yirmiya, R. Endotoxin produces a depressive-like episode in rats. Brain Res 1996, 711, 163–174. [Google Scholar]
- Frenois, F.; Moreau, M.; O’Connor, J.; Lawson, M.; Micon, C.; Lestage, J.; Kelley, K.W.; Dantzer, R.; Castanon, N. Lipopolysaccharide induces delayed FosB/DeltaFosB immunostaining within the mouse extended amygdala, hippocampus and hypothalamus, that parallel the expression of depressive-like behavior. Psychoneuroendocrinology 2007, 32, 516–531. [Google Scholar]
- Cerda, B.; Thomas-Barberan, F.A.; Espin, J.C. Metabolism of antioxidant and chemopreventive ellagitannins from strawberries, raspberries, walnuts, and oak-aged wine in humans: Identification of biomarkers and individual variability. J. Agric. Food Chem 2005, 53, 227–235. [Google Scholar]
- Ito, H.; Iguchi, A.; Hatano, T. Identification of urinary and intestinal bacterial metabolites of ellagitannin geraniin in rats. J. Agric. Food Chem 2008, 56, 393–400. [Google Scholar]
- Giménez-Bastida, J.A.; Larrosa, M.; González-Sarrías, A.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.T. Intestinal ellagitannin metabolites ameliorate cytokine-induced inflammation and associated molecular markers in human colon fibroblasts. J. Agric. Food Chem 2012, 60, 8866–8876. [Google Scholar]
- Townsend, K.P.; Praticò, D. Novel therapeutic opportunities for Alzheimer’s disease: Focus on nonsteroidal anti-inflammatory drugs. FASEB J 2005, 19, 1592–1601. [Google Scholar]
- Okuyama, S.; Shimada, N.; Kaji, M.; Morita, M.; Miyoshi, K.; Minami, S.; Amakura, Y.; Yoshimura, M.; Yoshida, T.; Watanabe, S.; Nakajima, M.; Furukawa, Y. Heptamethoxyflavone, a citrus flavonoid, enhances brain-derived neurotrophic factor production and neurogenesis in the hippocampus following cerebral global ischemia in mice. Neurosci. Lett 2012, 528, 190–195. [Google Scholar]


© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Okuyama, S.; Makihata, N.; Yoshimura, M.; Amakura, Y.; Yoshida, T.; Nakajima, M.; Furukawa, Y. Oenothein B Suppresses Lipopolysaccharide (LPS)-Induced Inflammation in the Mouse Brain. Int. J. Mol. Sci. 2013, 14, 9767-9778. https://doi.org/10.3390/ijms14059767
Okuyama S, Makihata N, Yoshimura M, Amakura Y, Yoshida T, Nakajima M, Furukawa Y. Oenothein B Suppresses Lipopolysaccharide (LPS)-Induced Inflammation in the Mouse Brain. International Journal of Molecular Sciences. 2013; 14(5):9767-9778. https://doi.org/10.3390/ijms14059767
Chicago/Turabian StyleOkuyama, Satoshi, Nahomi Makihata, Morio Yoshimura, Yoshiaki Amakura, Takashi Yoshida, Mitsunari Nakajima, and Yoshiko Furukawa. 2013. "Oenothein B Suppresses Lipopolysaccharide (LPS)-Induced Inflammation in the Mouse Brain" International Journal of Molecular Sciences 14, no. 5: 9767-9778. https://doi.org/10.3390/ijms14059767
APA StyleOkuyama, S., Makihata, N., Yoshimura, M., Amakura, Y., Yoshida, T., Nakajima, M., & Furukawa, Y. (2013). Oenothein B Suppresses Lipopolysaccharide (LPS)-Induced Inflammation in the Mouse Brain. International Journal of Molecular Sciences, 14(5), 9767-9778. https://doi.org/10.3390/ijms14059767




