Plant bZIP Transcription Factors Responsive to Pathogens: A Review
Abstract
:1. Introduction
2. Response to Pathogens in Plants: Signaling and bZIP Transcription Factors
3. bZIPs and cis-Elements Related to Pathogen Elicitors
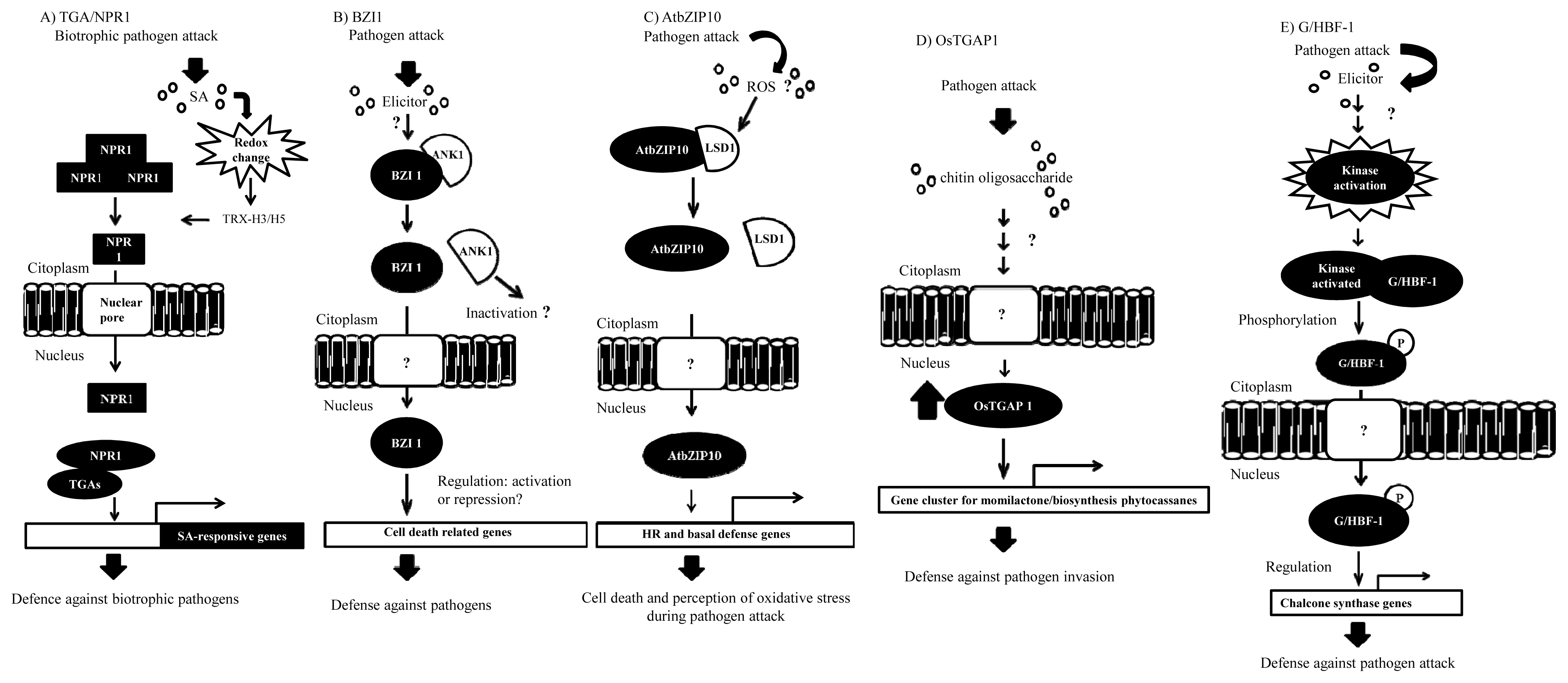
4. Functional Interactions of bZIPs with Other Proteins in Defense Responses
5. bZIPs Responsive to Pathogens in Agronomic Crops
6. Conclusions
Acknowledgments
Conflict of Interest
References
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys 2005, 444, 139–158. [Google Scholar]
- Cheong, Y.H.; Moon, B.C.; Kim, J.K.; Kim, C.Y.; Kim, M.C.; Kim, I.H.; Park, C.Y.; Kim, J.C.; Park, B.O.; Koo, S.C.; et al. BWMK1, a rice mitogen-activated protein kinase, locates in the nucleus and mediates pathogenesis-related gene expression by activation of a transcription factor. Plant Physiol 2003, 132, 1961–1972. [Google Scholar]
- Udvardi, M.K.; Kakar, K.; Wandrey, M.; Montanari, O.; Murray, J.; Andriankaja, A.; Zhang, J.; Benedito, V.; Hofer, J.M.I.; Chueng, F.; et al. Legume transcription factors: Global regulators of plant development and response to the environment. Plant Physiol 2007, 144, 538–549. [Google Scholar]
- Liao, Y.; Zou, H.F.; Wei, W.; Hao, Y.J.; Tian, A.G.; Huang, J.; Liu, Y.F.; Zhang, J.S.; Chen, S.Y. Soybean GmbZIP44, GmbZIP62 and GmbZIP78 genes function as negative regulator of ABA signaling and confer salt and freezing tolerance in transgenic Arabidopsis. Planta 2008, 228, 225–240. [Google Scholar]
- Singh, K.; Foley, R.C.; Oñate-Sánchez, L. Transcription factors in plant defense and stress responses. Curr. Opin. Plant Biol 2002, 5, 430–436. [Google Scholar]
- Pandey, S.P.; Somssich, I.E. The role of WRKY transcription factors in plant immunity. Plant Physiol 2009, 150, 1648–1655. [Google Scholar]
- Jakoby, M.; Weisshaar, M.J.B.; Dröge-Laser, W.; Vicente-Carbajosa, J.; Tiedemann, J.; Kroj, T.; Parcy, F. bZIP transcription factors in Arabidopsis. Trends Plant Sci 2002, 7, 106–111. [Google Scholar]
- Kuhlmann, M.; Horvay, K.; Stathmann, A.; Heinekamp, T.; Fischer, U.; Böttner, S.; Dröge-Laser, W. The alpha-helical D1 domain of the bZIP transcription factor BZI-1 interacts with the ankyrin-repeat protein ANK1, and is essential for BZI-1 function, both in auxin signaling and pathogen response. J. Biol. Chem 2003, 278, 8786–8794. [Google Scholar]
- Sato, F.; Katajima, S.; Koyama, T.; Yamada, Y. Ethylene-induced gene expression of osmotin-like protein, a neutral isoform of tobacco PR-5, is mediated by the AGCCGCC cis-sequence. Plant Cell Physiol 1996, 37, 249–255. [Google Scholar]
- Katagiri, F.; Chua, N.H. Plant transcription factors: Present knowledge and future challenges. Trends Genet 1992, 8, 22–27. [Google Scholar]
- Mateo, A.; Muhlenbock, P.; Rusterucci, C.; Chang, C.C.; Miszalski, Z.; Karpinska, B.; Parker, J.E.; Mullineaux, P.M.; Karpinski, S. LESION SIMULATING DISEASE 1 is required for acclimation to conditions that promote excess excitation energy. Plant Physiol 2004, 136, 2818–2830. [Google Scholar]
- Kaminaka, H.; Näke, C.; Epple, P.; Dittgen, J.; Schütze, K.; Chaban, C.; Holt, B.F., 3rd; Merkle, T.; Schäfer, E.; Harter, K.; Dangl, J.L. bZIP10-LSD1 antagonism modulates basal defense and cell death in Arabidopsis following infection. EMBO J. 2006, 25, 4400–4411. [Google Scholar]
- Després, C.; DeLong, C.; Glaze, S.; Liu, E.; Fobert, P.R. The Arabidopsis NPR1/NIM1 protein enhances the DNA binding activity of a subgroup of the TGA family of bZIP transcription factors. Plant Cell 2000, 12, 279–290. [Google Scholar]
- Karlowski, W.M.; Hirsch, A.M. The over-expression of an alfalfa RING-H2 gene induces pleiotropic effects on plant growth and development. Plant Mol. Biol 2003, 52, 121–133. [Google Scholar]
- Van Verk, M.C.; Neeleman, L.; Bol, J.F.; Linthorst, H.J. Tobacco transcription factor NtWRKY12 interacts with TGA2.2 in vitro and in vivo. Front. Plant Sci 2011, 2, 32. [Google Scholar]
- Dröge-Laser, W.; Kaiser, A.; Lindsay, W.P.; Halkier, B.A.; Loake, G.J.; Doerner, P.; Dixon, R.; Lamb, C. Rapid stimulation of a soybean protein-serine kinase that phosphorylates a novel bZIP DNA-binding protein, G/HBF-1, during the induction of early transcription-dependent defenses. EMBO J 1997, 16, 726–738. [Google Scholar]
- Foster, R.; Izawa, T.; Chua, N.H. Plant bZIP proteins gather at ACGT elements. FASEB J 1994, 8, 192–200. [Google Scholar]
- Tak, H.; Mhatre, M. Cloning and molecular characterization of a putative bZIP transcription factor VvbZIP23 from Vitis vinifera. Protoplasma 2012, 250, 333–345. [Google Scholar]
- Lee, S.J.; Lee, M.Y.; Yi, S.Y.; Oh, S.K.; Choi, S.H.; Her, N.H.; Choi, D.; Min, B.W.; Yang, S.G.; Harn, C.H. PPI1: A novel pathogen-induced basic region-leucine zipper (bZIP) transcription factor from pepper. Mol. Plant Microbe Interact 2002, 15, 540–548. [Google Scholar]
- Okada, A.; Okada, K.; Miyamoto, K.; Koga, J.; Shibuya, N.; Nojiri, H.; Yamane, H. OsTGAP1, a bZIP transcription factor, coordinately regulates the inductive production of diterpenoidphytoalexins in rice. J. Biol. Chem 2009, 284, 26510–26518. [Google Scholar]
- Orellana, S.; Yañez, M.; Espinoza, A.; Verdugo, I.; González, E.; Ruiz-Lara, S.; Casaretto, J.A. The transcription factor SlAREB1 confers drought, salt stress tolerance and regulates biotic and abiotic stress-related genes in tomato. Plant Cell Environ 2010, 33, 2191–2208. [Google Scholar]
- Chisholm, S.T.; Coaker, G.; Day, B.; Stakawicz, B.J. Host-microbe interactions: Shaping the evolution of the plant immune response. Cell 2006, 124, 803–814. [Google Scholar]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar]
- DebRoy, S.; Thilmony, R.; Kwack, Y.B.; Nomura, K.; He, S.Y. A family of conserved bacterial effectors inhibits salicylic acid-mediated basal immunity and promotes disease necrosis in plants. Proc. Natl. Acad. Sci. USA 2004, 101, 9927–9932. [Google Scholar]
- Dempsey, D.A.; Shah, J.; Klessig, D.F. Salicylic acid and disease resistance in plants. Crit. Rev. Plant Sci 1999, 18, 547–575. [Google Scholar]
- Durner, J.; Shah, J.; Klessig, D.F. Salicylic acid and disease resistance in plants. Trends Plant Sci 1997, 2, 266–274. [Google Scholar]
- Klessig, D.F.; Malamy, J. The salicylic acid signal in plants. Plant Mol. Biol 1994, 26, 1439–1458. [Google Scholar]
- Loake, G.; Grant, M. Salicylic acid in plant defence—The players and protagonists. Curr. Opin. Plant Biol 2007, 10, 466–472. [Google Scholar]
- Shah, J. The salicylic acid loop in plant defence. Curr. Opin. Plant Biol 2003, 6, 365–371. [Google Scholar]
- Howe, G.A.; Jander, G. Plant immunity to insect herbivores. Annu. Rev. Plant Biol 2008, 59, 41–66. [Google Scholar]
- Pieterse, C.M.J.; van der Does, D.; Zamioudis, C.; Leon-Reyes, A.; van Wees, S.C.M. Hormonal modulation of plant immunity. Annu. Rev. Cell Dev. Biol 2012, 28, 489–521. [Google Scholar]
- Resende, M.L.V.; Salgado, S.M.L.; Chaves, Z.M. Espécies ativas de oxigênio na resposta de defesa de plantas a patógenos. Fitopatol. Bras 2003, 28, 123–130. [Google Scholar]
- Lee, S.C.; Choi, H.W.; Hwang, I.S.; Choi, D.S.; Hwang, B.K. Functional roles of the pepper pathogen-induced bZIP transcription factor, CAbZIP1, in enhanced resistance to pathogen infection and environmental stresses. Planta 2006, 224, 1209–1225. [Google Scholar]
- Lebel, E.; Heifetz, P.; Thorne, L.; Uknes, S.; Ryals, J.; Ward, E. Functional analysis of regulatory sequences controlling PR-1 gene expression in Arabidopsis. Plant J 1998, 16, 223–233. [Google Scholar]
- Zhang, Y.; Tessaro, M.J.; Lassner, M.; Li, X. Knockout analysis of Arabidopsis transcription factors TGA2, TGA5, and TGA6 reveals their redundant and essential roles in systemic acquired resistance. Plant Cell 2003, 15, 2647–2653. [Google Scholar]
- Cao, H.; Glazebrook, J.; Clarke, J.D.; Volko, S.; Dong, X. The Arabidopsis NPR1 gene that controls systemic acquired resistance encodes a novel protein containing ankyrin repeats. Cell 1997, 88, 57–63. [Google Scholar]
- Ryals, J.; Weymann, K.; Lawton, K.; Friedrich, L.; Ellis, D.; Steiner, H.Y.; Johnson, J.; Delaney, T.P.; Jesse, T.; Vos, P.; Uknes, S. The Arabidopsis NIM1 protein shows homology to the mammalian transcription factor inhibitor I kappa B. Plant Cell 1997, 9, 425–439. [Google Scholar]
- Chen, W.; Singh, K.B. The auxin, hydrogen peroxide and salicylic acid induced expression of the Arabidopsis GST6 promoter is mediated in part by an ocs element. Plant J 1999, 19, 667–677. [Google Scholar]
- Uquillas, C.; Letelier, I.; Blanco, F.; Jordana, X.; Holuigue, L. NPR1-independent activation of immediate early salicylic acid-responsive genes in Arabidopsis. Mol. Plant Microbe Interact 2004, 17, 34–42. [Google Scholar]
- Blanco, F.; Garreton, V.; Frey, N.; Dominguez, C.; Perez-Acle, T.; van der Straeten, D.; Jordana, X.; Holuigue, L. Identification of NPR1-dependent and independent genes early induced by salicylic acid treatment in Arabidopsis. Plant Mol. Biol 2005, 59, 927–944. [Google Scholar]
- Hart, C.M.; Nagy, F.; Meins, F., Jr. A 61 bp enhancer element of the tobacco beta-1,3-glucanase B gene interacts with one or more regulated nuclear proteins. Plant Mol. Biol. 1993, 21, 121–131. [Google Scholar]
- Sessa, G.; Meller, Y.; Fluhr, R. A GCC element and a G-box motif participate in ethylene-induced expression of the PRB-1b gene. Plant Mol. Biol 1995, 28, 145–153. [Google Scholar]
- Ohme-Takagi, M.; Shinshi, H. Ethylene-inducible DNA binding proteins that interact with an ethylene-responsive element. Plant Cell 1995, 7, 173–182. [Google Scholar]
- Foley, R.C.; Grossman, C.; Ellis, J.G.; Llewellyn, D.J.; Dennis, E.S.; Peacock, W.J.; Singh, K.B. Isolation of a maize bZIP protein subfamily: Candidates for the ocs-element transcription factor. Plant J 1993, 3, 669–679. [Google Scholar]
- Zhang, B.; Foley, R.C.; Singh, K.B. Isolation and characterization of two related Arabidopsis ocs-element bZIP binding proteins. Plant J 1993, 4, 711–716. [Google Scholar]
- Rieping, M.; Fritz, M.; Prat, S.; Gatz, C. A dominant negative mutant of PG13 suppresses transcription from a cauliflower mosaic virus 35S truncated promoter in transgenic tobacco plants. Plant Cell 1994, 6, 1087–1098. [Google Scholar]
- Feldbrügge, M.; Sprenger, M.; Dinkelbach, M.; Yazaki, K.; Harter, K.; Weisshaar, B. Functional analysis of a light-responsive plantbZIP transcriptional regulator. Plant Cell 1994, 6, 1607–1621. [Google Scholar]
- Dietrich, R.A.; Delaney, T.P.; Uknes, S.J.; Ward, E.J.; Ryals, J.A.; Dangl, J.L. Arabidopsis mutants simulating disease resistance response. Cell 1994, 77, 565–578. [Google Scholar]
- Dietrich, R.A.; Richberg, M.H.; Schmidt, R.; Dean, C.; Dangl, J.L. A novel zinc-finger protein is encoded by the Arabidopsis lsd1 gene and functions as a negative regulator of plant cell death. Cell 1997, 88, 685–694. [Google Scholar]
- Jabs, T.; Dietrich, R.A.; Dangl, J.L. Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 1996, 273, 1853–1856. [Google Scholar]
- Chern, M.S.; Fitzgerald, H.A.; Yadav, R.C.; Canlas, P.E.; Dong, X.; Ronald, P.C. Evidence for a disease-resistance pathway in rice similar to the NPR1-mediated signaling pathway in Arabidopsis. Plant J 2001, 27, 101–113. [Google Scholar]
- Ellenberger, T.E.; Brandl, C.J.; Struhl, K.; Harrison, S.C. The GCN4 basic region leucine zipper binds DNA as a dimer of uninterrupted alpha helices crystal structure of the protein-DNA complex. Cell 1992, 71, 1223–1237. [Google Scholar]
- Yoshida, K.; Wakamatsu, S.; Sakuta, M. Characterization of SBZ1, a soybean bZIP protein that binds to the chalcone synthase gene promoter. Plant Biotechnol 2008, 25, 131–140. [Google Scholar]
- Lee, S.; Kim, S.Y.; Chung, E.; Joung, Y.H.; Pai, H.S.; Hur, C.G.; Choi, D. EST and microarray analyses of pathogen-responsive genes in hot pepper (Capsicum annuum L.) non-host resistance against soybean pustule pathogen (Xanthomonas axonopodis pv. glycines). Funct. Integr. Genomics 2004, 4, 196–205. [Google Scholar]
- Wei, K.; Chen, J.; Wang, Y.; Chen, Y.; Chen, S.; Lin, Y.; Pan, S.; Zhong, X.; Xie, D. Genome-wide analysis of bZIP-encoding genes in maize. DNA Res 2012, 19, 463–476. [Google Scholar]
- Lopes, M.A.; Hora, B.T., Jr; Dias, C.V.; Santos, G.C.; Gramacho, K.P.; Cascardo, J.C.; Gesteira, A.S.; Micheli, F. Expression analysis of transcription factors from the interaction between cacao and Moniliophthora Perniciosa (Tricholomataceae). Genet. Mol. Res 2010, 9, 1279–1297. [Google Scholar]
- Zhou, Y.L.; Xu, M.R.; Zhao, M.F.; Xie, X.W.; Zhu, L.H.; Fu, B.Y.; Li, Z.K. Genome-wide gene responses in a transgenic rice line carrying the maize resistance gene Rxo1 to the rice bacterial streak pathogen, Xanthomonas oryzae pv. oryzicola. BMC Genomics 2010, 1, 11–78. [Google Scholar]
- Xu, L.; Zhu, L.; Tu, L.; Liu, L.; Yuan, D.; Jin, L.; Long, L.; Zhang, X. Lignin metabolism has a central role in the resistance of cotton to the wilt fungus Verticillium dahliae as revealed by RNA-Seq-dependent transcriptional analysis and histochemistry. J. Exp. Bot 2011, 62, 5607–5621. [Google Scholar]

| Protein | Organism | cis-Element | Inducers | Function in plant defense | References |
|---|---|---|---|---|---|
| CabZIP1 | Capsicum annuum L. | G-box | ET, SA, MeJA and pathogen infection | Decrease in plant growth rates | [8] |
| OBF PROTEIN | Arabidopsis thaliana | osc element | SA | Induction of PR gene expression | [9,10] |
| AtbZIP10 | Arabidopsis thaliana | G-box and C-box | Uncharacterized | Positive regulator of HR, cell death and basal defense response | [11] |
| TGA MEMBERS | Arabidopsis thaliana | TGACG element | SA | Induction of PR gene expression | [12] |
| rTGA2.1, rTGA2.2, rTGA2.3 | Oryza sativa | PR-1 promoter (in Arabidopsis) | SA | Induction of SA responsive gene expression | [13] |
| BZI-1 | Nicotiana tabacum | ACEs | Uncharacterized | Auxin signaling and plant defense | [14] |
| G/HBF-1 | Glycine max | G-box and H-box | Uncharacterized | Regulation of defense gene expression | [15] |
| SBZ1 | Glycine max | G-box and H-box | Uncharacterized | Regulation of defense gene expression | [16] |
| VvbZIP23 | Vitis vinifera | Uncharacterized | ABA, ET, JA and SA. | Regulation of biotic and abiotic stress responses | [17] |
| PPI1 | Capsicum chinense | ACEs | Pathogen infection | Regulation of defense gene expression | [18] |
| OsTGAP1 | Oryza sativa | TGACG element | Chitin oligosaccharides | Regulation of genes involved in the biosynthesis of diterpenoids | [19] |
| SlAREB1 | Solanum lycopersicum | ABRE (ABA-responsive element) | ABA | Pathogens response | [20] |
| RT42C09 | Theobroma cacao | Uncharacterized | Pathogen infection | Pathogens response | [21] |
| RT57A09 | Theobroma cacao | Uncharacterized | Pathogen infection | Pathogens response | [21] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alves, M.S.; Dadalto, S.P.; Gonçalves, A.B.; De Souza, G.B.; Barros, V.A.; Fietto, L.G. Plant bZIP Transcription Factors Responsive to Pathogens: A Review. Int. J. Mol. Sci. 2013, 14, 7815-7828. https://doi.org/10.3390/ijms14047815
Alves MS, Dadalto SP, Gonçalves AB, De Souza GB, Barros VA, Fietto LG. Plant bZIP Transcription Factors Responsive to Pathogens: A Review. International Journal of Molecular Sciences. 2013; 14(4):7815-7828. https://doi.org/10.3390/ijms14047815
Chicago/Turabian StyleAlves, Murilo S., Silvana P. Dadalto, Amanda B. Gonçalves, Gilza B. De Souza, Vanessa A. Barros, and Luciano G. Fietto. 2013. "Plant bZIP Transcription Factors Responsive to Pathogens: A Review" International Journal of Molecular Sciences 14, no. 4: 7815-7828. https://doi.org/10.3390/ijms14047815




