Roles of Rho GTPases in Intracellular Transport and Cellular Transformation
Abstract
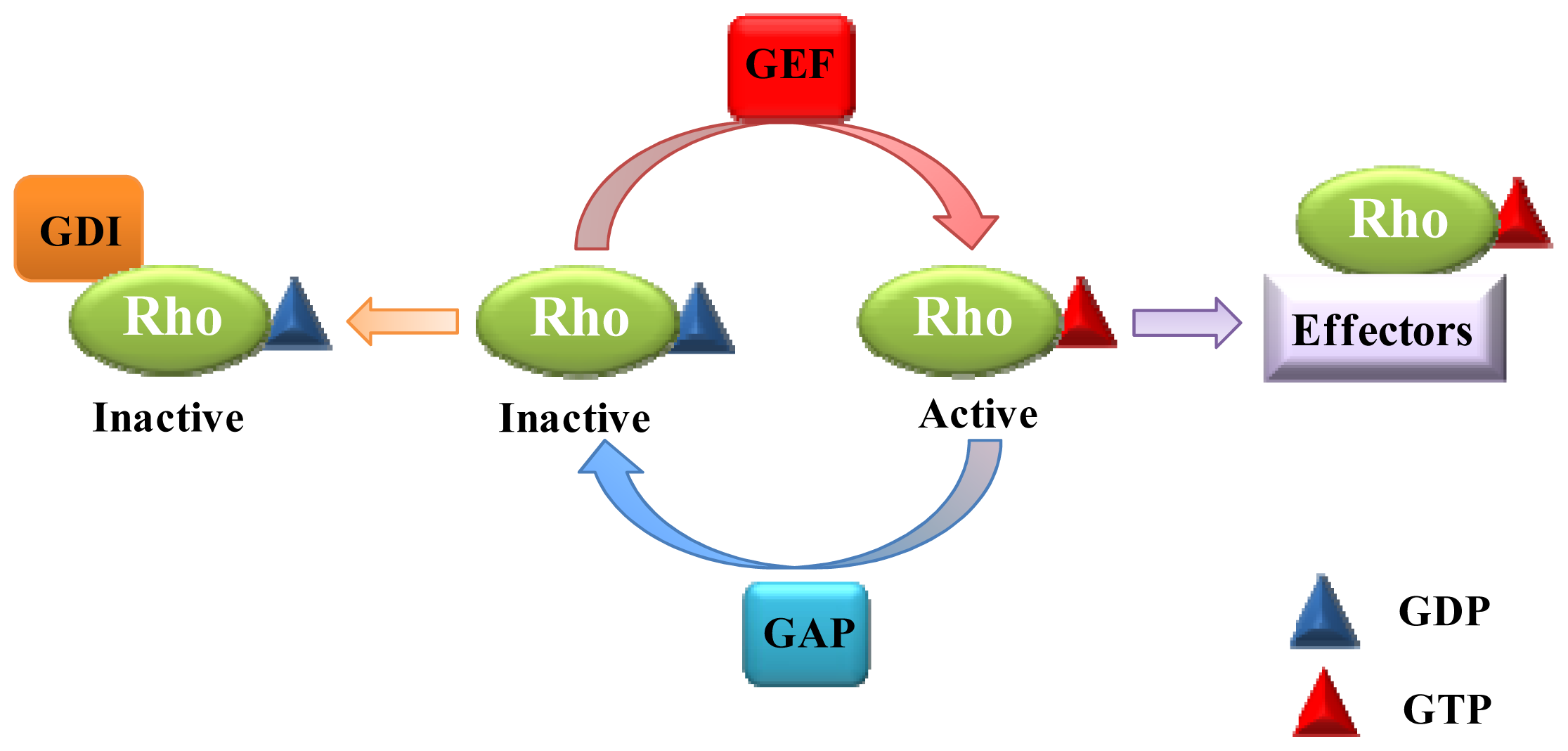
:1. Introduction
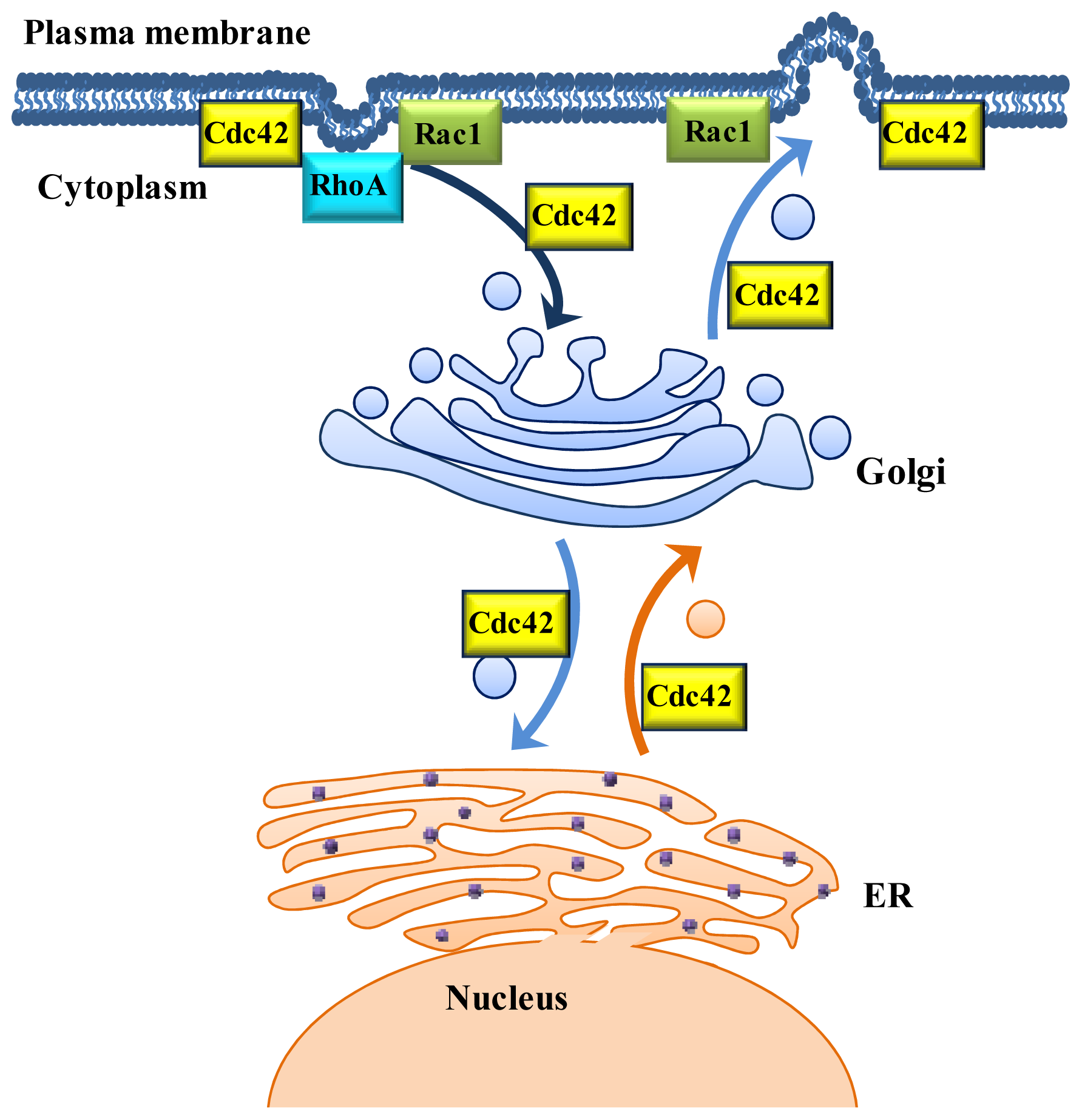
2. Role of Rho GTPases in Vesicular Transport
2.1. Cdc42: A Regulator of Vesicle Trafficking
2.2. Rac1: Involvement in Endocytic and Exocytic Transport
2.3. RhoA Mainly Participates in the Regulation of Endocytic Transport
3. Rho GTPases Regulate Viral Proteins and Genome Transport in Host Cells
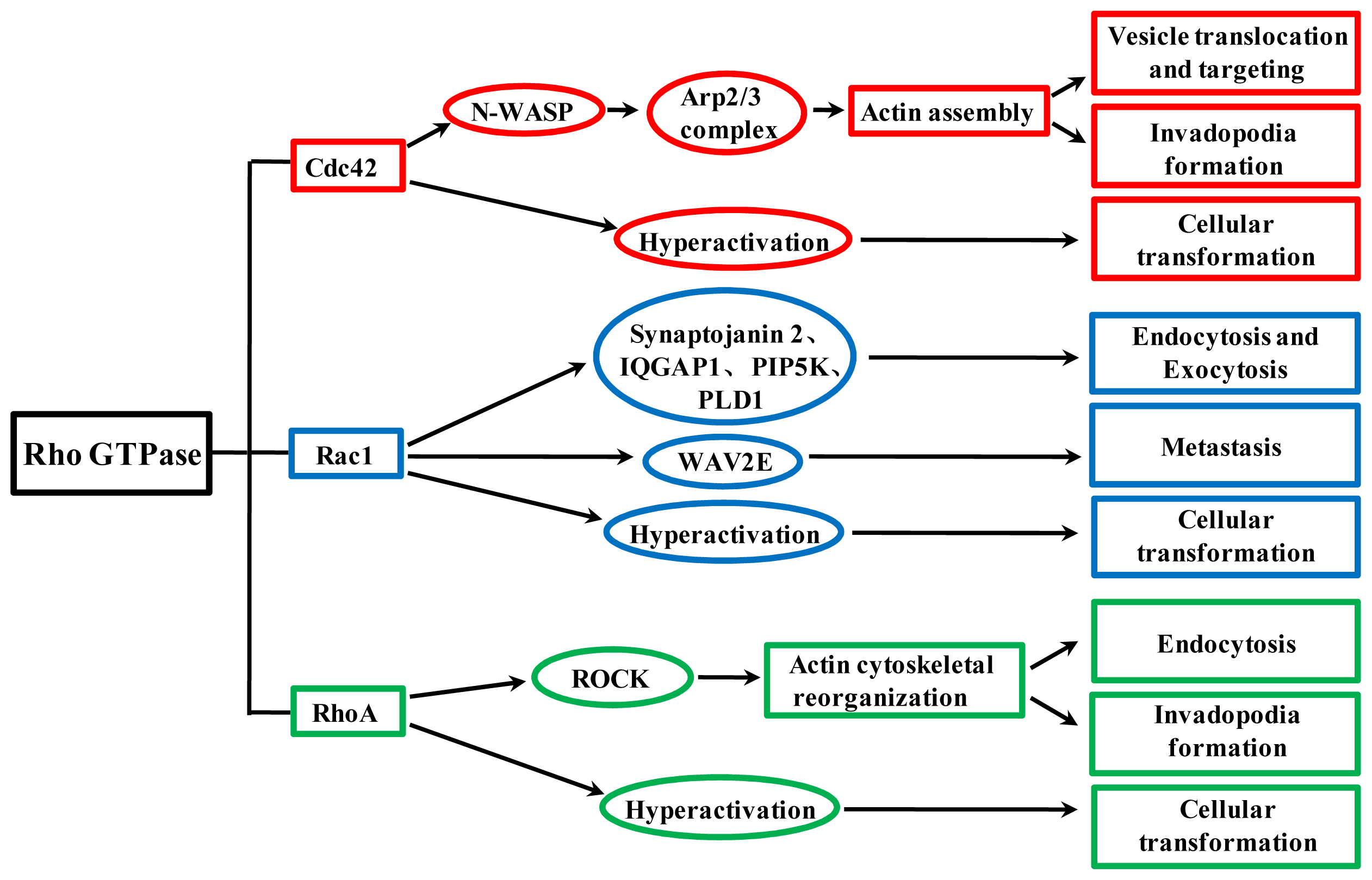
4. Cdc42, Rac1 and RhoA Contribute to Cellular Transformation and Regulate Tumor Invasion and Metastasis
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Heasman, S.J.; Ridley, A.J. Mammalian Rho GTPases: New insights into their functions from in vivo studies. Nat. Rev. Mol. Cell Biol 2008, 9, 690–701. [Google Scholar]
- Hall, A. Rho GTPases and the actin cytoskeleton. Science 1998, 279, 509–514. [Google Scholar]
- Tapon, N.; Hall, A. Rho, Rac and Cdc42 GTPases regulate the organization of the actin cytoskeleton. Curr. Opin. Cell Biol 1997, 9, 86–92. [Google Scholar]
- Boguski, M.S.; McCormick, F. Proteins regulating Ras and its relatives. Nature 1993, 366, 643–654. [Google Scholar]
- Symons, M.; Settleman, J. Rho family GTPases: More than simple switches. Trends Cell Biol 2000, 10, 415–419. [Google Scholar]
- Jaffe, A.B.; Hall, A. Rho GTPases: Biochemistry and biology. Annu. Rev. Cell Dev. Biol 2005, 21, 247–269. [Google Scholar]
- Symons, M.; Rusk, N. Control of vesicular trafficking by Rho GTPases. Curr. Biol 2003, 13, 409–418. [Google Scholar]
- Wu, H.; Rossi, G.; Brennwald, P. The ghost in the machine: small GTPases as spatial regulators of exocytosis. Trends Cell Biol 2008, 18, 397–404. [Google Scholar]
- Delprato, A. Topological and functional properties of the small GTPases protein interaction network. PLoS One 2012, 7, e44882. [Google Scholar]
- Sheen, V.L.; Ganesh, V.S.; Topcu, M.; Sebire, G.; Bodell, A.; Hill, R.S.; Grant, P.E.; Shugart, Y.Y.; Imitola, J.; Khoury, S.J. Mutations in ARFGEF2 implicate vesicle trafficking in neural progenitor proliferation and migration in the human cerebral cortex. Nat. Genet 2003, 36, 69–76. [Google Scholar]
- Jayadev, S.; Petranka, J.G.; Cheran, S.K.; Biermann, J.A.; Barrett, J.C.; Murphy, E. Reduced capacitative calcium entry correlates with vesicle accumulation and apoptosis. J. Biol. Chem 1999, 274, 8261–8268. [Google Scholar]
- Egea, G.; Lázaro-Diéguez, F.; Vilella, M. Actin dynamics at the Golgi complex in mammalian cells. Curr. Opin. Cell Biol 2006, 18, 168–178. [Google Scholar]
- Smythe, E.; Ayscough, K.R. Actin regulation in endocytosis. J. Cell Sci 2006, 119, 4589–4598. [Google Scholar]
- Lanzetti, L. Actin in membrane trafficking. Curr. Opin. Cell Biol 2007, 19, 453–458. [Google Scholar]
- Ho, H.Y.H.; Rohatgi, R.; Lebensohn, A.M.; Li, J.; Gygi, S.P.; Kirschner, M.W. Toca-1 mediates Cdc42-dependent actin nucleation by activating the N-WASP-WIP complex. Cell 2004, 118, 203–216. [Google Scholar]
- Ridley, A.J.; Schwartz, M.A.; Burridge, K.; Firtel, R.A.; Ginsberg, M.H.; Borisy, G.; Parsons, J.T.; Horwitz, A.R. Cell migration: integrating signals from front to back. Science 2003, 302, 1704–1709. [Google Scholar]
- Cerione, R.A. Cdc42: New roads to travel. Trends Cell Biol 2004, 14, 127–132. [Google Scholar]
- Gupta, G.P.; Massagué, J. Cancer metastasis: Building a framework. Cell 2006, 127, 679–695. [Google Scholar]
- Ridley, A.J. Rho proteins: linking signaling with membrane trafficking. Traffic 2001, 2, 303–310. [Google Scholar]
- Vega, F.M.; Ridley, A.J. Rho GTPases in cancer cell biology. FEBS Lett 2008, 582, 2093–2101. [Google Scholar]
- Ridley, A.J. Rho GTPases and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell Biol 2006, 16, 522–529. [Google Scholar]
- Wu, D.; Asiedu, M.; Wei, Q. MyoGEF regulates the invasion activity of MDA-MB-231 breast cancer cells through activation of RhoA and RhoC. Oncogene 2009, 28, 2219–2230. [Google Scholar]
- Wu, H.; Brennwald, P. The function of two Rho family GTPases is determined by distinct patterns of cell surface localization. Mol. Cell. Biol 2010, 30, 5207–5217. [Google Scholar]
- Adamo, J.E.; Rossi, G.; Brennwald, P. The Rho GTPase Rho3 has a direct role in exocytosis that is distinct from its role in actin polarity. Mol. Biol. Cell 1999, 10, 4121–4133. [Google Scholar]
- Chiang, S.H.; Baumann, C.A.; Kanzaki, M.; Thurmond, D.C.; Watson, R.T.; Neudauer, C.L.; Macara, I.G.; Pessin, J.E.; Saltiel, A.R. Insulin-stimulated GLUT4 translocation requires the CAP-dependent activation of TC10. Nature 2001, 410, 944–948. [Google Scholar]
- Cheng, J.; Wang, H.; Guggino, W.B. Regulation of cystic fibrosis transmembrane regulator trafficking and protein expression by a Rho family small GTPase TC10. J. Biol. Chem 2005, 280, 3731–3739. [Google Scholar]
- Mitchell, T.; Lo, A.; Logan, M.R.; Lacy, P.; Eitzen, G. Primary granule exocytosis in human neutrophils is regulated by Rac-dependent actin remodeling. Am. J. Physiol. Cell Physiol 2008, 295, 1354–1365. [Google Scholar]
- Abdel-Latif, D.; Steward, M.; Macdonald, D.L.; Francis, G.A.; Dinauer, M.C.; Lacy, P. Rac2 is critical for neutrophil primary granule exocytosis. Blood 2004, 104, 832–839. [Google Scholar]
- Gad, A.K.B.; Aspenström, P. Rif proteins take to the RhoD: Rho GTPases at the crossroads of actin dynamics and membrane trafficking. Cell. Signal 2010, 22, 183–189. [Google Scholar]
- Rondanino, C.; Rojas, R.; Ruiz, W.G.; Wang, E.; Hughey, R.P.; Dunn, K.W.; Apodaca, G. RhoB-dependent modulation of postendocytic traffic in polarized Madin-Darby canine kidney cells. Traffic 2007, 8, 932–949. [Google Scholar]
- Wherlock, M.; Gampel, A.; Futter, C.; Mellor, H. Farnesyltransferase inhibitors disrupt EGF receptor traffic through modulation of the RhoB GTPase. J. Cell Sci 2004, 117, 3221–3231. [Google Scholar]
- Huang, M.; Prendergast, G. RhoB in cancer suppression. Histol. Histopathol 2006, 21, 213–218. [Google Scholar]
- Iiizumi, M.; Bandyopadhyay, S.; Pai, S.K.; Watabe, M.; Hirota, S.; Hosobe, S.; Tsukada, T.; Miura, K.; Saito, K.; Furuta, E. RhoC promotes metastasis via activation of the Pyk2 pathway in prostate cancer. Cancer Res 2008, 68, 7613–7620. [Google Scholar]
- Matos, P.; Jordan, P. Increased Rac1b expression sustains colorectal tumor cell survival. Mol. Cancer Res 2008, 6, 1178–1184. [Google Scholar]
- Singh, A.; Karnoub, A.E.; Palmby, T.R.; Lengyel, E.; Sondek, J.; Der, C.J. Rac1b, a tumor associated, constitutively active Rac1 splice variant, promotes cellular transformation. Oncogene 2004, 23, 9369–9380. [Google Scholar]
- Hartwell, L.H.; Culotti, J.; Pringle, J.R.; Reid, B.J. Genetic control of the cell division cycle in yeast. Science 1974, 183, 46–51. [Google Scholar]
- Johnson, D.I.; Pringle, J.R. Molecular characterization of CDC42, a Saccharomyces cerevisiae gene involved in the development of cell polarity. J. Cell Biol 1990, 111, 143–152. [Google Scholar]
- Kroschewski, R.; Hall, A.; Mellman, I. Cdc42 controls secretory and endocytic transport to the basolateral plasma membrane of MDCK cells. Nat. Cell Biol 1999, 1, 8–13. [Google Scholar]
- Ellis, S.; Mellor, H. Regulation of endocytic traffic by Rho family GTPases. Trends Cell Biol 2000, 10, 85–88. [Google Scholar]
- Garrett, W.S.; Chen, L.M.; Kroschewski, R.; Ebersold, M.; Turley, S.; Trombetta, S.; Galán, J.E.; Mellman, I. Developmental control of endocytosis in dendritic cells by Cdc42. Cell 2000, 102, 325–334. [Google Scholar]
- Sabharanjak, S.; Sharma, P.; Parton, R.G.; Mayor, S. GPI-anchored proteins are delivered to recycling endosomes via a distinct cdc42-regulated, clathrin-independent pinocytic pathway. Dev. Cell 2002, 2, 411–423. [Google Scholar]
- Chadda, R.; Howes, M.T.; Plowman, S.J.; Hancock, J.F.; Parton, R.G.; Mayor, S. Cholesterol-sensitive Cdc42 activation regulates actin polymerization for endocytosis via the GEEC pathway. Traffic 2007, 8, 702–717. [Google Scholar]
- Mayor, S.; Pagano, R.E. Pathways of clathrin-independent endocytosis. Nat. Rev. Mol. Cell Biol 2007, 8, 603–612. [Google Scholar]
- Cory, G.O.C.; Cramer, R.; Blanchoin, L.; Ridley, A.J. Phosphorylation of the WASP-VCA domain increases its affinity for the Arp2/3 complex and enhances actin polymerization by WASP. Mol. Cell 2003, 11, 1229–1239. [Google Scholar]
- Bu, W.; Lim, K.B.; Yu, Y.H.; Chou, A.M.; Sudhaharan, T.; Ahmed, S. Cdc42 interaction with N-WASP and Toca-1 regulates membrane tubulation, vesicle formation and vesicle motility: implications for endocytosis. PLoS One 2010, 5, e12153. [Google Scholar]
- Wu, W.J.; Erickson, J.W.; Lin, R.; Cerione, R.A. The γ-subunit of the coatomer complex binds Cdc42 to mediate transformation. Nature 2000, 405, 800–804. [Google Scholar]
- Luna, A.; Matas, O.B.; Martínez-Menárguez, J.A.; Mato, E.; Durán, J.M.; Ballesta, J.; Way, M.; Egea, G. Regulation of protein transport from the Golgi complex to the endoplasmic reticulum by CDC42 and N-WASP. Mol. Biol. Cell 2002, 13, 866–879. [Google Scholar]
- Hehnly, H.; Longhini, K.M.; Chen, J.L.; Stamnes, M. Retrograde Shiga toxin trafficking is regulated by ARHGAP21 and Cdc42. Mol. Biol. Cell 2009, 20, 4303–4312. [Google Scholar]
- Egorov, M.V.; Capestrano, M.; Vorontsova, O.A.; di Pentima, A.; Egorova, A.V.; Mariggiò, S.; Ayala, M.I.; Tetè, S.; Gorski, J.L.; Luini, A. Faciogenital dysplasia protein (FGD1) regulates export of cargo proteins from the golgi complex via Cdc42 activation. Mol. Biol. Cell 2009, 20, 2413–2427. [Google Scholar]
- Rohatgi, R.; Ma, L.; Miki, H.; Lopez, M.; Kirchhausen, T.; Takenawa, T.; Kirschner, M.W. The interaction between N-WASP and the Arp2/3 complex links Cdc42-dependent signals to actin assembly. Cell 1999, 97, 221–231. [Google Scholar]
- Chen, J.L.; Fucini, R.V.; Lacomis, L.; Erdjument-Bromage, H.; Tempst, P.; Stamnes, M. Coatomer-bound Cdc42 regulates dynein recruitment to COPI vesicles. J. Cell Biol 2005, 169, 383–389. [Google Scholar]
- Chen, J.L.; Lacomis, L.; Erdjument-Bromage, H.; Tempst, P.; Stamnes, M. Cytosol-derived proteins are sufficient for Arp2/3 recruitment and ARF/coatomer-dependent actin polymerization on Golgi membranes. FEBS Lett 2004, 566, 281–286. [Google Scholar]
- Harris, K.P.; Tepass, U. Cdc42 and vesicle trafficking in polarized cells. Traffic 2010, 11, 1272–1279. [Google Scholar]
- Malacombe, M.; Ceridono, M.; Calco, V.; Chasserot-Golaz, S.; McPherson, P.S.; Bader, M.F.; Gasman, S. Intersectin-1L nucleotide exchange factor regulates secretory granule exocytosis by activating Cdc42. EMBO J 2006, 25, 3494–3503. [Google Scholar]
- Gasman, S.; Chasserot-Golaz, S.; Malacombe, M.; Way, M.; Bader, M.F. Regulated exocytosis in neuroendocrine cells: A role for subplasmalemmal Cdc42/N-WASP-induced actin filaments. Mol. Biol. Cell 2004, 15, 520–531. [Google Scholar]
- Matas, O.B.; Fritz, S.; Luna, A.; Egea, G. Membrane trafficking at the ER/Golgi interface: Functional implications of RhoA and Rac1. Eur. J. Cell Biol 2005, 84, 699–707. [Google Scholar]
- Lamaze, C.; Chuang, T.H.; Terlecky, L.J.; Bokoch, G.M.; Schmid, S.L. Regulation of receptor-mediated endocytosis by Rho and Rac. Nature 1996, 382, 177–179. [Google Scholar]
- Oikawa, T.; Yamaguchi, H.; Itoh, T.; Kato, M.; Ijuin, T.; Yamazaki, D.; Suetsugu, S.; Takenawa, T. PtdIns (3, 4, 5) P3 binding is necessary for WAVE2-induced formation of lamellipodia. Nat. Cell Biol 2004, 6, 420–426. [Google Scholar]
- Wertheimer, E.; Gutierrez-Uzquiza, A.; Rosemblit, C.; Lopez-Haber, C.; Sosa, M.S.; Kazanietz, M.G. Rac signaling in breast cancer: A tale of GEFs and GAPs. Cell. Signal 2012, 24, 353–362. [Google Scholar]
- Balamatsias, D.; Kong, A.M.; Waters, J.E.; Sriratana, A.; Gurung, R.; Bailey, C.G.; Rasko, J.E.J.; Tiganis, T.; Macaulay, S.L.; Mitchell, C.A. Identification of P-Rex1 as a novel Rac1-guanine nucleotide exchange factor (GEF) that promotes actin remodeling and GLUT4 protein trafficking in adipocytes. J. Biol. Chem 2011, 286, 43229–43240. [Google Scholar]
- Malecz, N.; McCabe, P.C.; Spaargaren, C.; Qiu, R.G.; Chuang, Y.; Symons, M. Synaptojanin 2, a novel Rac1 effector that regulates clathrin-mediated endocytosis. Curr. Biol 2000, 10, 1383–1386. [Google Scholar]
- Izumi, G.; Sakisaka, T.; Baba, T.; Tanaka, S.; Morimoto, K.; Takai, Y. Endocytosis of E-cadherin regulated by Rac and Cdc42 small G proteins through IQGAP1 and actin filaments. J. Cell Biol 2004, 166, 237–248. [Google Scholar]
- Vidal-Quadras, M.; Gelabert-Baldrich, M.; Soriano-Castell, D.; Lladó, A.; Rentero, C.; Calvo, M.; Pol, A.; Enrich, C.; Tebar, F. Rac1 and calmodulin interactions modulate dynamics of ARF6-dependent endocytosis. Traffic 2011, 12, 1879–1896. [Google Scholar]
- Van den Bout, I.; Divecha, N. PIP5K-driven PtdIns (4, 5) P2 synthesis: regulation and cellular functions. J. Cell Sci 2009, 122, 3837–3850. [Google Scholar]
- Doussau, F.; Gasman, S.; Humeau, Y.; Vitiello, F.; Popoff, M.; Boquet, P.; Bader, M.F.; Poulain, B. A Rho-related GTPase is involved in Ca2+-dependent neurotransmitter exocytosis. J. Biol. Chem 2000, 275, 7764–7770. [Google Scholar]
- Li, Q.; Ho, C.S.; Marinescu, V.; Bhatti, H.; Bokoch, G.M.; Ernst, S.A.; Holz, R.W.; Stuenkel, E.L. Facilitation of Ca2+-dependent exocytosis by Rac1-GTPase in bovine chromaffin cells. J. Physiol 2004, 550, 431–445. [Google Scholar]
- Bi, Y.; Williams, J.A. A role for Rho and Rac in secretagogue-induced amylase release by pancreatic acini. Am. J. Physiol. Cell Physiol 2005, 289, 22–32. [Google Scholar]
- Bretscher, M.S.; Aguado-Velasco, C. EGF induces recycling membrane to form ruffles. Curr. Biol 1998, 8, 721–724. [Google Scholar]
- Momboisse, F.; Ory, S.; Ceridono, M.; Calco, V.; Vitale, N.; Bader, M.F.; Gasman, S. The Rho guanine nucleotide exchange factors intersectin 1L and β-Pix control calcium-regulated exocytosis in neuroendocrine PC12 cells. Cell. Mol. Neurobiol 2010, 30, 1327–1333. [Google Scholar]
- Momboisse, F.; Lonchamp, E.; Calco, V.; Ceridono, M.; Vitale, N.; Bader, M.F.; Gasman, S. beta PIX-activated Rac1 stimulates the activation of phospholipase D, which is associated with exocytosis in neuroendocrine cells. J. Cell Sci 2009, 122, 798–806. [Google Scholar]
- Chen, Y.; Yang, Z.; Meng, M.; Zhao, Y.; Dong, N.; Yan, H.; Liu, L.; Ding, M.; Peng, H.B.; Shao, F. Cullin mediates degradation of RhoA through evolutionarily conserved BTB adaptors to control actin cytoskeleton structure and cell movement. Mol. Cell 2009, 35, 841–855. [Google Scholar]
- Wheeler, A.P.; Ridley, A.J. Why three Rho proteins? RhoA, RhoB, RhoC, and cell motility. Exp. Cell Res 2004, 301, 43–49. [Google Scholar]
- Yu, C.; Nwabuisi-Heath, E.; Laxton, K.; LaDu, M.J. Endocytic pathways mediating oligomeric Aβ42 neurotoxicity. Mol. Neurodegener 2010, 5, 19. [Google Scholar]
- Vögler, O.; Krummenerl, P.; Schmidt, M.; Jakobs, K.H.; Van Koppen, C.J. RhoA-sensitive trafficking of muscarinic acetylcholine receptors. J. Pharmacol. Exp. Ther 1999, 288, 36–42. [Google Scholar]
- Chen, N.X.; Chen, X.; O’Neill, K.D.; Atkinson, S.J.; Moe, S.M. RhoA/Rho kinase (ROCK) alters fetuin-A uptake and regulates calcification in bovine vascular smooth muscle cells (BVSMC). Am. J. Physiol. Renal. Physiol 2010, 299, 674–680. [Google Scholar]
- Lamaze, C.; Dujeancourt, A.; Baba, T.; Lo, C.G.; Benmerah, A.; Dautry-Varsat, A. Interleukin 2 receptors and detergent-resistant membrane domains define a clathrin-independent endocytic pathway. Mol. Cell 2001, 7, 661–671. [Google Scholar]
- Khandelwal, P.; Ruiz, W.G.; Apodaca, G. Compensatory endocytosis in bladder umbrella cells occurs through an integrin-regulated and RhoA-and dynamin-dependent pathway. EMBO J 2010, 29, 1961–1975. [Google Scholar]
- Riento, K.; Ridley, A.J. Rocks: multifunctional kinases in cell behaviour. Nat. Rev. Mol. Cell Biol 2003, 4, 446–456. [Google Scholar]
- Maekawa, M.; Ishizaki, T.; Boku, S.; Watanabe, N.; Fujita, A.; Iwamatsu, A.; Obinata, T.; Ohashi, K.; Mizuno, K.; Narumiya, S. Signaling from Rho to the actin cytoskeleton through protein kinases ROCK and LIM-kinase. Science 1999, 285, 895–898. [Google Scholar]
- Bishop, A.L.; Hall, A. Rho GTPases and their effector proteins. Biochem. J 2000, 348, 241–255. [Google Scholar]
- Shen, D.W.; Pouliot, L.M.; Gillet, J.P.; Ma, W.; Johnson, A.C.; Hall, M.D.; Gottesman, M.M. The transcription factor GCF2 is an upstream repressor of the small GTPase RhoA, regulating membrane protein trafficking, sensitivity to doxorubicin, and resistance to cisplatin. Mol. Pharm 2012, 9, 1822–1833. [Google Scholar]
- Lundmark, R.; Doherty, G.J.; Howes, M.T.; Cortese, K.; Vallis, Y.; Parton, R.G.; McMahon, H.T. The GTPase-activating protein GRAF1 regulates the CLIC/GEEC endocytic pathway. Curr. Biol 2008, 18, 1802–1808. [Google Scholar]
- Doherty, G.J.; Lundmark, R. GRAF1-dependent endocytosis. Biochem. Soc. Trans 2009, 37, 1061–1065. [Google Scholar]
- Pathak, R.; Delorme-Walker, V.D.; Howell, M.C.; Anselmo, A.N.; White, M.A.; Bokoch, G.M.; DerMardirossian, C. The microtubule-associated Rho activating factor GEF-H1 interacts with exocyst complex to regulate vesicle traffic. Dev. Cell 2012, 23, 397–411. [Google Scholar]
- Johnson, J.L.; Monfregola, J.; Napolitano, G.; Kiosses, W.B.; Catz, S.D. Vesicular trafficking through cortical actin during exocytosis is regulated by the Rab27a effector JFC1/Slp1 and the RhoA-GTPase-activating protein Gem-interacting protein. Mol. Biol. Cell 2012, 23, 1902–1916. [Google Scholar]
- Wang, S.; Li, H.; Chen, Y.; Wei, H.; Gao, G.F.; Liu, H.; Huang, S.; Chen, J.L. Transport of influenza virus neuraminidase (NA) to host cell surface is regulated by ARHGAP21 and Cdc42 proteins. J. Biol. Chem 2012, 287, 9804–9816. [Google Scholar]
- Hui, E.K.W.; Barman, S.; Tang, D.H.P.; France, B.; Nayak, D.P. YRKL sequence of influenza virus M1 functions as the L domain motif and interacts with VPS28 and Cdc42. J. Virol 2006, 80, 2291–2308. [Google Scholar]
- Li, E.; Stupack, D.; Bokoch, G.M.; Nemerow, G. R. Adenovirus endocytosis requires actin cytoskeleton reorganization mediated by Rho family GTPases. J. Virol 1998, 72, 8806–8812. [Google Scholar]
- Sanlioglu, S.; Benson, P.K.; Yang, J.; Atkinson, E.M.; Reynolds, T.; Engelhardt, J.F. Endocytosis and nuclear trafficking of adeno-associated virus type 2 are controlled by Rac1 and phosphatidylinositol-3 kinase activation. J. Virol 2000, 74, 9184–9196. [Google Scholar]
- Nonnenmacher, M.; Weber, T. Adeno-associated virus 2 infection requires endocytosis through the CLIC/GEEC pathway. Cell Host Microbe 2011, 10, 563–576. [Google Scholar]
- Kalia, M.; Khasa, R.; Sharma, M.; Nain, M.; Vrati, S. Japanese encephalitis virus infects neuronal cells through a clathrin-independent endocytic mechanism. J. Virol 2013, 87, 148–162. [Google Scholar]
- Hoppe, S.; Schelhaas, M.; Jaeger, V.; Liebig, T.; Petermann, P.; Knebel-Mörsdorf, D. Early herpes simplex virus type 1 infection is dependent on regulated Rac1/Cdc42 signalling in epithelial MDCKII cells. J. Gen. Virol 2006, 87, 3483–3494. [Google Scholar]
- Naranatt, P.P.; Krishnan, H.H.; Smith, M.S.; Chandran, B. Kaposi’s sarcoma-associated herpesvirus modulates microtubule dynamics via RhoA-GTP-diaphanous 2 signaling and utilizes the dynein motors to deliver its DNA to the nucleus. J. Virol 2005, 79, 1191–1206. [Google Scholar]
- Raghu, H.; Sharma-Walia, N.; Veettil, M.V.; Sadagopan, S.; Caballero, A.; Sivakumar, R.; Varga, L.; Bottero, V.; Chandran, B. Lipid rafts of primary endothelial cells are essential for Kaposi's sarcoma-associated herpesvirus/human herpesvirus 8-induced phosphatidylinositol 3-kinase and RhoA-GTPases critical for microtubule dynamics and nuclear delivery of viral DNA but dispensable for binding and entry. J. Virol 2007, 81, 7941–7959. [Google Scholar]
- Loomis, R.J.; Holmes, D.A.; Elms, A.; Solski, P.A.; Der, C.J.; Su, L. Citron kinase, a RhoA effector, enhances HIV-1 virion production by modulating exocytosis. Traffic 2006, 7, 1643–1653. [Google Scholar]
- Kolesnikova, L.; Bohil, A.B.; Cheney, R.E.; Becker, S. Budding of Marburgvirus is associated with filopodia. Cell. Microbiol 2006, 9, 939–951. [Google Scholar]
- Greene, W.; Gao, S.J. Actin dynamics regulate multiple endosomal steps during Kaposi’s sarcoma-associated herpesvirus entry and trafficking in endothelial cells. PLoS Pathog 2009, 5, e1000512. [Google Scholar]
- Quetglas, J.I.; Hernáez, B.; Galindo, I.; Muñoz-Moreno, R.; Cuesta-Geijo, M.A.; Alonso, C. Small Rho GTPases and cholesterol biosynthetic pathway intermediates in African swine fever virus infection. J. Virol 2012, 86, 1758–1767. [Google Scholar]
- Palazzo, A.F.; Cook, T.A.; Alberts, A.S.; Gundersen, G.G. mDia mediates Rho-regulated formation and orientation of stable microtubules. Nat. Cell Biol 2001, 3, 723–729. [Google Scholar]
- Arakawa, Y.; Cordeiro, J.V.; Way, M. F11L-mediated inhibition of RhoA-mDia signaling stimulates microtubule dynamics during vaccinia virus infection. Cell Host Microbe 2007, 1, 213–226. [Google Scholar]
- Arakawa, Y.; Cordeiro, J.V.; Schleich, S.; Newsome, T.P.; Way, M. The release of vaccinia virus from infected cells requires RhoA-mDia modulation of cortical actin. Cell Host Microbe 2007, 1, 227–240. [Google Scholar]
- Valderrama, F.; Cordeiro, J.V.; Schleich, S.; Frischknecht, F.; Way, M. Vaccinia virus-induced cell motility requires F11L-mediated inhibition of RhoA signaling. Science 2006, 311, 377–381. [Google Scholar]
- Bos, J.L.; Rehmann, H.; Wittinghofer, A. GEFs and GAPs: Critical elements in the control of small G proteins. Cell 2007, 129, 865–877. [Google Scholar]
- Stengel, K.; Zheng, Y. Cdc42 in oncogenic transformation, invasion, and tumorigenesis. Cell. Signal 2011, 23, 1415–1423. [Google Scholar]
- Lin, R.; Cerione, R.A.; Manor, D. Specific contributions of the small GTPases Rho, Rac, and Cdc42 to Dbl transformation. J. Biol. Chem 1999, 274, 23633–23641. [Google Scholar]
- Tcherkezian, J.; Lamarche-Vane, N. Current knowledge of the large RhoGAP family of proteins. Biol. Cell 2007, 99, 67–86. [Google Scholar]
- Liao, Y.C.; Lo, S.H. Deleted in liver cancer-1 (DLC-1): A tumor suppressor not just for liver. Int. J. Biochem. Cell Biol 2008, 40, 843–847. [Google Scholar]
- Yagi, R.; Tanaka, M.; Sasaki, K.; Kamata, R.; Nakanishi, Y.; Kanai, Y.; Sakai, R. ARAP3 inhibits peritoneal dissemination of scirrhous gastric carcinoma cells by regulating cell adhesion and invasion. Oncogene 2010, 30, 1413–1421. [Google Scholar]
- Li, Z.; Chang, Z.; Chiao, L.J.; Kang, Y.A.; Xia, Q.; Zhu, C.; Fleming, J.B.; Evans, D.B.; Chiao, P.J. TrkBT1 induces liver metastasis of pancreatic cancer cells by sequestering Rho GDP dissociation inhibitor and promoting RhoA activation. Cancer Res 2009, 69, 7851–7859. [Google Scholar]
- Theodorescu, D.; Sapinoso, L.; Conaway, M.; Oxford, G.; Hampton, G.; Frierson, H. Reduced expression of metastasis suppressor RhoGDI2 is associated with decreased survival for patients with bladder cancer. Clin. Cancer. Res 2004, 10, 3800–3806. [Google Scholar]
- Lin, Q.; Fuji, R.N.; Yang, W.; Cerione, R.A. RhoGDI is required for Cdc42-mediated cellular transformation. Curr. Biol 2003, 13, 1469–1479. [Google Scholar]
- Qiu, R.G.; Chen, J.; Kirn, D.; McCormick, F.; Symons, M. An essential role for Rac in Ras transformation. Nature 1995, 374, 457–459. [Google Scholar]
- Qiu, R.G.; Chen, J.; McCormick, F.; Symons, M. A role for Rho in Ras transformation. Proc. Natl. Acad. Sci. USA 1995, 92, 11781–11785. [Google Scholar]
- Qiu, R.G.; Abo, A.; McCormick, F.; Symons, M. Cdc42 regulates anchorage-independent growth and is necessary for Ras transformation. Mol. Cell. Biol 1997, 17, 3449–3458. [Google Scholar]
- Stengel, K.R.; Zheng, Y. Essential role of Cdc42 in Ras-induced transformation revealed by gene targeting. PLoS One 2012, 7, e37317. [Google Scholar]
- Yang, H.W.; Shin, M.G.; Lee, S.; Kim, J.R.; Park, W.S.; Cho, K.H.; Meyer, T.; Do Heo, W. Cooperative activation of PI3K by Ras and Rho family small GTPases. Mol. Cell 2012, 47, 281–290. [Google Scholar]
- Liu, J.F.; Chevet, E.; Kebache, S.; Lemaitre, G.; Barritault, D.; Larose, L.; Crepin, M. Functional Rac-1 and Nck signaling networks are required for FGF-2-induced DNA synthesis in MCF-7 cells. Oncogene 1999, 18, 6425–6433. [Google Scholar]
- Baranwal, S.; K Alahari, S. Rho GTPase effector functions in tumor cell invasion and metastasis. Curr. Drug Targets 2011, 12, 1194–1201. [Google Scholar]
- Ye, D.Z.; Field, J. PAK signaling in cancer. Cell Logist 2012, 2, 105–116. [Google Scholar]
- Wu, W.J.; Tu, S.; Cerione, R.A. Activated Cdc42 sequesters c-Cbl and prevents EGF receptor degradation. Cell 2003, 114, 715–725. [Google Scholar]
- Kaneko, T.; Maeda, A.; Takefuji, M.; Aoyama, H.; Nakayama, M.; Kawabata, S.; Kawano, Y.; Iwamatsu, A.; Amano, M.; Kaibuchi, K. Rho mediates endocytosis of epidermal growth factor receptor through phosphorylation of endophilin A1 by Rho-kinase. Genes Cells 2005, 10, 973–987. [Google Scholar]
- Ung, C.Y.; Li, H.; Ma, X.H.; Jia, J.; Li, B.W.; Low, B.C.; Chen, Y.Z. Simulation of the regulation of EGFR endocytosis and EGFR-ERK signaling by endophilin-mediated RhoA-EGFR crosstalk. FEBS Lett 2008, 582, 2283–2290. [Google Scholar]
- Narumiya, S.; Tanji, M.; Ishizaki, T. Rho signaling, ROCK and mDia1, in transformation, metastasis and invasion. Cancer Metast. Rev 2009, 28, 65–76. [Google Scholar]
- Tanji, M.; Ishizaki, T.; Ebrahimi, S.; Tsuboguchi, Y.; Sukezane, T.; Akagi, T.; Frame, M.C.; Hashimoto, N.; Miyamoto, S.; Narumiya, S. mDia1 targets v-Src to the cell periphery and facilitates cell transformation, tumorigenesis, and invasion. Mol. Cell. Biol 2010, 30, 4604–4615. [Google Scholar]
- Albiges-Rizo, C.; Destaing, O.; Fourcade, B.; Planus, E.; Block, M.R. Actin machinery and mechanosensitivity in invadopodia, podosomes and focal adhesions. J. Cell Sci 2009, 122, 3037–3049. [Google Scholar] [Green Version]
- Yamaguchi, H.; Lorenz, M.; Kempiak, S.; Sarmiento, C.; Coniglio, S.; Symons, M.; Segall, J.; Eddy, R.; Miki, H.; Takenawa, T. Molecular mechanisms of invadopodium formation the role of the N-WASP-Arp2/3 complex pathway and cofilin. J. Cell Biol 2005, 168, 441–452. [Google Scholar]
- Yamaguchi, H.; Condeelis, J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. BBA-Mol. Cell Res 2007, 1773, 642–652. [Google Scholar]
- Pignatelli, J.; Tumbarello, D.A.; Schmidt, R.P.; Turner, C.E. Hic-5 promotes invadopodia formation and invasion during TGF-β-induced epithelial-mesenchymal transition. J. Cell Biol 2012, 197, 421–437. [Google Scholar]
- Kurisu, S.; Suetsugu, S.; Yamazaki, D.; Yamaguchi, H.; Takenawa, T. Rac-WAVE2 signaling is involved in the invasive and metastatic phenotypes of murine melanoma cells. Oncogene 2004, 24, 1309–1319. [Google Scholar]
- Sakurai-Yageta, M.; Recchi, C.; Le Dez, G.; Sibarita, J.-B.; Daviet, L.; Camonis, J.; D’Souza-Schorey, C.; Chavrier, P. The interaction of IQGAP1 with the exocyst complex is required for tumor cell invasion downstream of Cdc42 and RhoA. J. Cell Biol 2008, 181, 985–998. [Google Scholar]
- Vishnubhotla, R.; Sun, S.; Huq, J.; Bulic, M.; Ramesh, A.; Guzman, G.; Cho, M.; Glover, S.C. ROCK-II mediates colon cancer invasion via regulation of MMP-2 and MMP-13 at the site of invadopodia as revealed by multiphoton imaging. Lab. Invest 2007, 87, 1149–1158. [Google Scholar]
- Lizárraga, F.; Poincloux, R.; Romao, M.; Montagnac, G.; Le Dez, G.; Bonne, I.; Rigaill, G.; Raposo, G.; Chavrier, P. Diaphanous-related formins are required for invadopodia formation and invasion of breast tumor cells. Cancer Res 2009, 69, 2792–2800. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chi, X.; Wang, S.; Huang, Y.; Stamnes, M.; Chen, J.-L. Roles of Rho GTPases in Intracellular Transport and Cellular Transformation. Int. J. Mol. Sci. 2013, 14, 7089-7108. https://doi.org/10.3390/ijms14047089
Chi X, Wang S, Huang Y, Stamnes M, Chen J-L. Roles of Rho GTPases in Intracellular Transport and Cellular Transformation. International Journal of Molecular Sciences. 2013; 14(4):7089-7108. https://doi.org/10.3390/ijms14047089
Chicago/Turabian StyleChi, Xiaojuan, Song Wang, Yifan Huang, Mark Stamnes, and Ji-Long Chen. 2013. "Roles of Rho GTPases in Intracellular Transport and Cellular Transformation" International Journal of Molecular Sciences 14, no. 4: 7089-7108. https://doi.org/10.3390/ijms14047089



