In Vitro and in Vivo Evaluation of Lactoferrin-Conjugated Liposomes as a Novel Carrier to Improve the Brain Delivery
Abstract
:1. Introduction
2. Results and Discussion
2.1. Conjugation of Lactoferrin (Lf) to PEGylated liposome (PL)
2.2. Characterization of PL[99mTc] and Lf-PL[99mTc]
2.3. Stability Study
2.4. In Vitro Cell Uptake Study for Evaluation of BBB Penetration Potential
2.5. Pharmacokinetic Study
2.6. Biodistribution Study
2.7. Prospective Development of Lf-PL
3. Experimental Section
3.1. Materials
3.2. Preparation of Maleimide Functional PEGylated Liposome (PL)
3.3. Preparation of Lactoferrin Modified PEGylated Liposome (Lf-PL)
3.4. Preparation of 99mTc-BMEDA Complex, PL[99mTc], and Lf-PL[99mTc]
3.5. Stability Study
3.6. Cell Culture and Animals
3.7. In Vitro Cell Uptake Study for Evaluation of BBB Penetration Potential
3.8. Pharmacokinetic Study
3.9. Biodistribution Study
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Cecchelli, R.; Berezowski, V.; Lundquist, S.; Culot, M.; Renftel, M.; Dehouck, M.P.; Fenart, L. Modelling of the blood-brain barrier in drug discovery and development. Nat. Rev. Drug. Discov 2007, 6, 650–661. [Google Scholar]
- Pardridge, W.M. Drug targeting to the brain. Pharm. Res 2007, 24, 1733–1744. [Google Scholar]
- Calvo, P.; Gouritin, B.; Chacun, H.; Desmaele, D.; D’Angelo, J.; Noel, J.P.; Georgin, D.; Fattal, E.; Andreux, J.P.; Couvreur, P. Long-circulating PEGylated polycyanoacrylate nanoparticles as new drug carrier for brain delivery. Pharm. Res 2001, 18, 1157–1166. [Google Scholar]
- Baker, E.N.; Baker, H.M. Molecular structure, binding properties and dynamics of lactoferrin. Cell. Mol. Life Sci 2005, 62, 2531–2539. [Google Scholar]
- Fillebeen, C.; Descamps, L.; Dehouck, M.P.; Fenart, L.; Benaissa, M.; Spik, M.; Cecchelli, R.; Pierce, A. Receptor-mediated transcytosis of lactoferrin through the blood-brain barrier. J. Biol. Chem 1999, 274, 7011–7017. [Google Scholar]
- Ji, B.; Maeda, J.; Higuchi, M.; Inoue, K.; Akita, H.; Harashima, H.; Suhara, T. Pharmacokinetics and brain uptake of lactoferrin in rats. Life Sci 2006, 78, 851–855. [Google Scholar]
- Huang, R.; Ke, W.; Liu, Y.; Jiang, C.; Pei, Y. The use of lactoferrin as a ligand for targeting the polyamidoamine-based gene delivery system to the brain. Biomaterials 2008, 29, 238–246. [Google Scholar]
- Hu, K.; Li, J.; Shen, Y.; Lu, W.; Gao, X.; Zhang, Q.; Jiang, X. Lactoferrin-conjugated PEG-PLA nanoparticles with improved brain delivery: in vitro and in vivo evaluations. J. Control. Release 2009, 134, 55–61. [Google Scholar]
- Xie, H.; Zhu, Y.; Jiang, W.; Zhou, Q.; Yang, H.; Gu, N.; Zhang, Y.; Xu, H.; Xu, H.; Yang, X. Lactoferrin-conjugated superparamagnetic iron oxide nanoparticles as a specific MRI contrast agent for detection of brain glioma in vivo. Biomaterials 2011, 32, 495–502. [Google Scholar]
- Pang, Z.; Feng, L.; Hua, R.; Chen, J.; Gao, H.; Pan, S.; Jiang, X.; Zhang, P. Lactoferrin-conjugated biodegradable polymersome holding doxorubicin and tetrandrine for chemotherapy of glioma rats. Mol. Pharm 2010, 7, 1995–2005. [Google Scholar]
- Faucheux, B.A.; Nillesse, N.; Damier, P.; Spik, G.; Mouatt-Prigent, A.; Pierce, A.; Leveugle, B.; Kubis, N.; Hauw, J.J.; Agid, Y. Expression of lactoferrin receptors is increased in the mesencephalon of patients with Parkinson disease. Proc. Natl. Acad. Sci. USA 1995, 92, 9603–9607. [Google Scholar]
- Leveugle, B.; Spik, G.; Perl, D.P.; Bouras, C.; Fillit, H.M.; Hof, P.R. The iron-binding protein lactotransferrin is present in pathologic lesions in a variety of neurodegenerative disorders: a comparative immunohistochemical analysis. Brain Res 1994, 650, 20–31. [Google Scholar]
- Iwamaru, Y.; Shimizu, Y.; Imamura, M.; Murayama, Y.; Endo, R.; Tagawa, Y.; Ushiki-Kaku, Y.; Takenouchi, T.; Kitani, H.; Mohri, S.; et al. Lactoferrin induces cell surface retention of prion protein and inhibits prion accumulation. J. Neurochem 2008, 107, 636–646. [Google Scholar]
- Torchilin, V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug. Discov 2005, 4, 145–160. [Google Scholar]
- Chen, H.; Tang, L.; Qin, Y.; Yin, Y.; Tang, J.; Tang, W.; Sun, X.; Zhang, Z.; Liu, J.; He, Q. Lactoferrin-modified procationic liposomes as a novel drug carrier for brain delivery. Eur. J. Pharm. Sci 2010, 40, 94–102. [Google Scholar]
- Chen, H.; Qin, Y.; Zhang, Q.; Jiang, W.; Tang, L.; Liu, J.; He, Q. Lactoferrin modified doxorubicin-loaded procationic liposomes for the treatment of gliomas. Eur. J. Pharm. Sci 2011, 44, 164–173. [Google Scholar]
- Zhang, Y.; Schlachetzki, F.; Zhang, Y.F.; Boado, R.J.; Pardridge, W.M. Normalization of striatal tyrosine hydroxylase and reversal of motor impairment in experimental parkinsonism with intravenous nonviral gene therapy and a brain-specific promoter. Hum. Gene Ther 2004, 15, 339–350. [Google Scholar]
- Huwyler, J.; Wu, D.; Pardridge, W.M. Brain drug delivery of small molecules using immunoliposomes. Proc. Natl. Acad. Sci. USA 1996, 93, 14164–14169. [Google Scholar]
- Brown, R.C.; Morris, A.P.; O’Neil, R.G. Tight junction protein expression and barrier properties of immortalized mouse brain microvessel endothelial cells. Brain Res 2007, 1130, 17–30. [Google Scholar]
- Huang, R.Q.; Ke, W.L.; Qu, Y.H.; Zhu, J.H.; Pei, Y.Y.; Jiang, C. Characterization of lactoferrin receptor in brain endothelial capillary cells and mouse brain. J. Biomed. Sci 2007, 14, 121–128. [Google Scholar]
- Suzuki, Y.A.; Lonnerdal, B. Baculovirus expression of mouse lactoferrin receptor and tissue distribution in the mouse. Biometals 2004, 17, 301–309. [Google Scholar]
- Suzuki, Y.A.; Lopez, V.; Lonnerdal, B. Mammalian lactoferrin receptors: structure and function. Cell. Mol. Life Sci 2005, 62, 2560–2575. [Google Scholar]
- Huang, R.; Han, L.; Li, J.; Ren, F.; Ke, W.; Jiang, C.; Pei, Y. Neuroprotection in a 6-hydroxydopamine-lesioned Parkinson model using lactoferrin-modified nanoparticles. J. Gene Med 2009, 11, 754–763. [Google Scholar]
- Huang, R.; Ke, W.; Han, L.; Liu, Y.; Shao, K.; Jiang, C.; Pei, Y. Lactoferrin-modified nanoparticles could mediate efficient gene delivery to the brain in vivo. Brain Res. Bull 2010, 81, 600–604. [Google Scholar]
- Huang, F.Y.J.; Lee, T.W.; Kao, C.H.; Chang, C.H.; Zhang, X.; Lee, W.Y.; Chen, W.J.; Wang, S.C.; Lo, J.M. Imaging, autoradiography, and biodistribution of 188Re-labeled PEGylated nanoliposome in orthotopic glioma bearing rat model. Cancer Biother. Radiopharm 2011, 26, 717–725. [Google Scholar]
- Bartlett, G.R. Phosphorus assay in column chromatography. J. Biol. Chem 1959, 234, 466–468. [Google Scholar]
- Jue, R.; Lambert, J.M.; Pierce, L.R.; Traut, R.R. Addition of sulfhydryl groups to escherichia coli ribosomes by protein modification with 2-Iminothiolane (Methyl 4-Mercaptobutyrimidate). Biochemistry 1978, 17, 5399–5406. [Google Scholar]
- Hansen, C.B.; Kao, G.Y.; Moase, E.H.; Zalipsky, S.; Allen, T.M. Attachment of antibodies to sterically stabilized liposomes: evaluation, comparison and optimization of coupling procedures. Biochim. Biophys. Acta 1995, 1239, 133–144. [Google Scholar]
- Bao, A.; Goins, B.; Klipper, R.; Negrete, G.; Mahindaratne, M.; Phillips, W.T. A novel liposome radiolabeling method using 99mTc-“SNS/S” complexes: in vitro and in vivo evaluation. J. Pharm. Sci 2003, 92, 1893–1904. [Google Scholar]
- Chen, L.C.; Chang, C.H.; Yu, C.Y.; Chang, Y.J.; Hsu, W.C.; Ho, C.L.; Yeh, C.H.; Luo, T.Y.; Lee, T.W.; Ting, G. Biodistribution, pharmacokinetics and imaging of 188Re-BMEDA-labeled pegylated liposomes after intraperitoneal injection in a C26 colon carcinoma ascites mouse model. Nucl. Med. Biol 2007, 34, 415–423. [Google Scholar]
- Montesano, R.; Pepper, M.S.; Mijhle-Steinlein, U.; Risau, W.; Wagner, E.F.; Orci, L. Increased proteolytic activity is responsible for the aberrant morphogenetic behavior of endothelial cells expressing the middle T oncogene. Cell 1990, 62, 435–445. [Google Scholar]
- Lo, J.M.; Pillai, M.R.; John, C.S.; Troutner, D.E. Radiochemical purity evaluation of rhodium-105 complexes by magnesium oxide. Int. J. Rad. Appl. Instrum. A 1990, 41, 103–105. [Google Scholar]

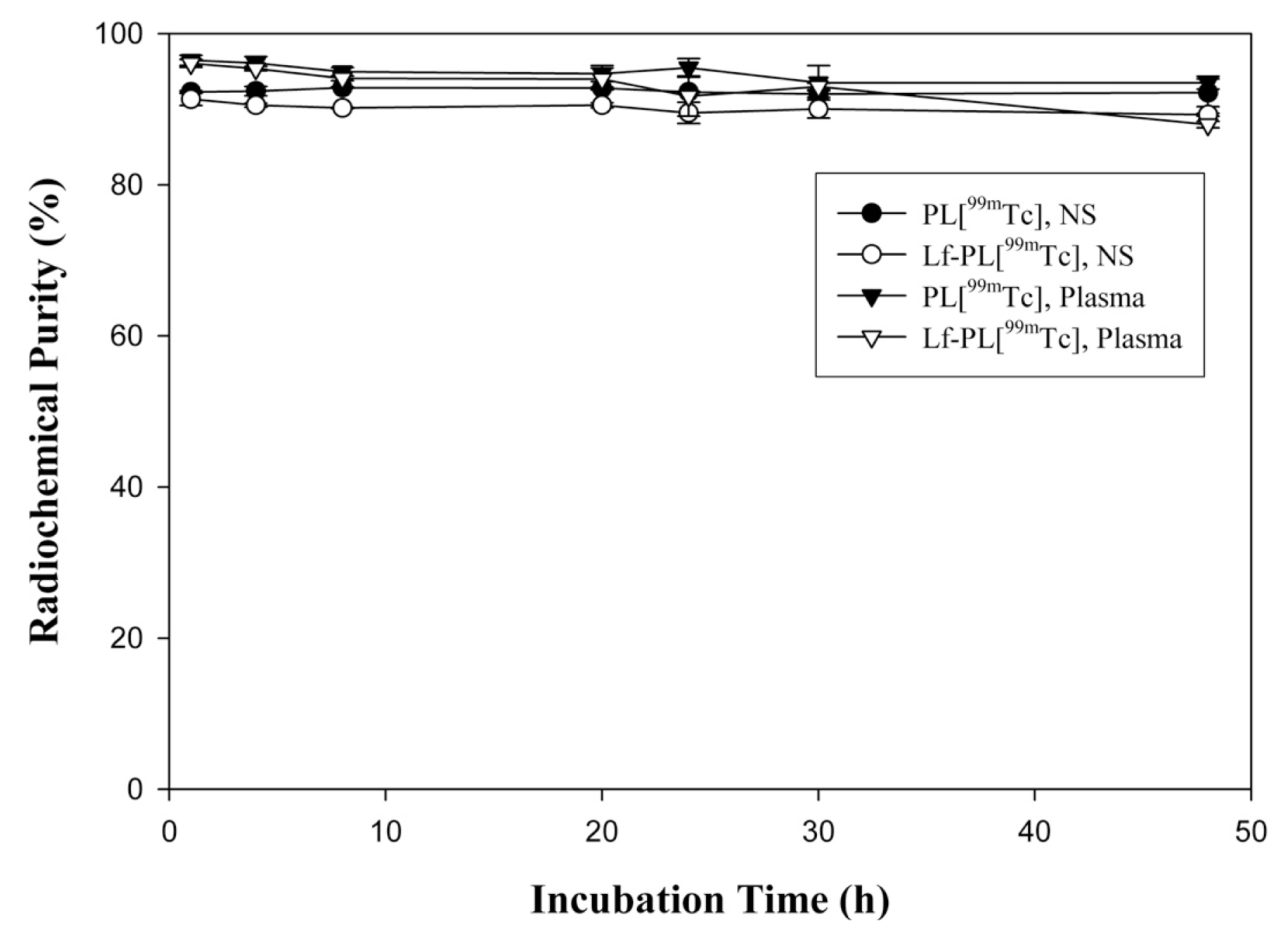

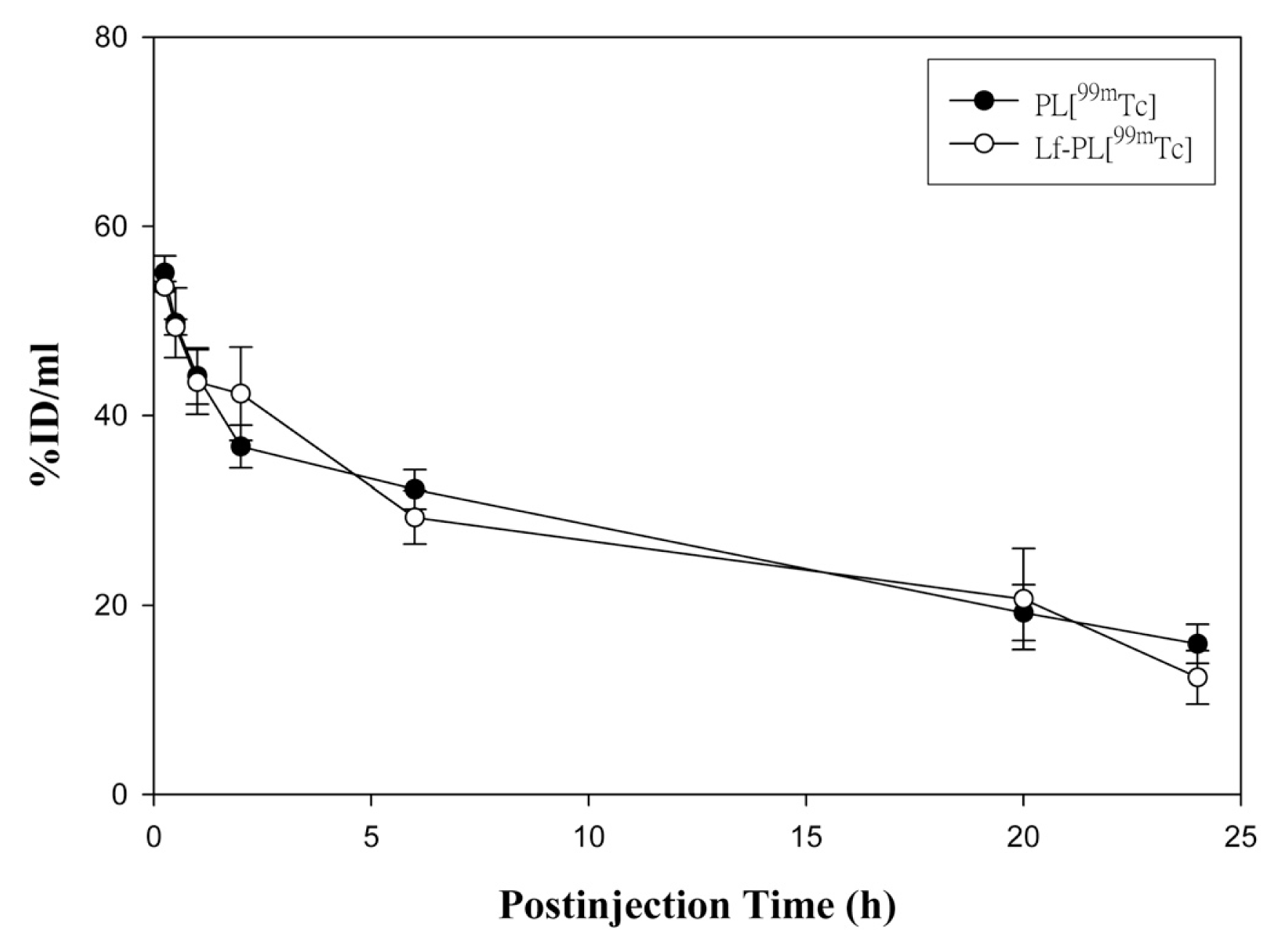

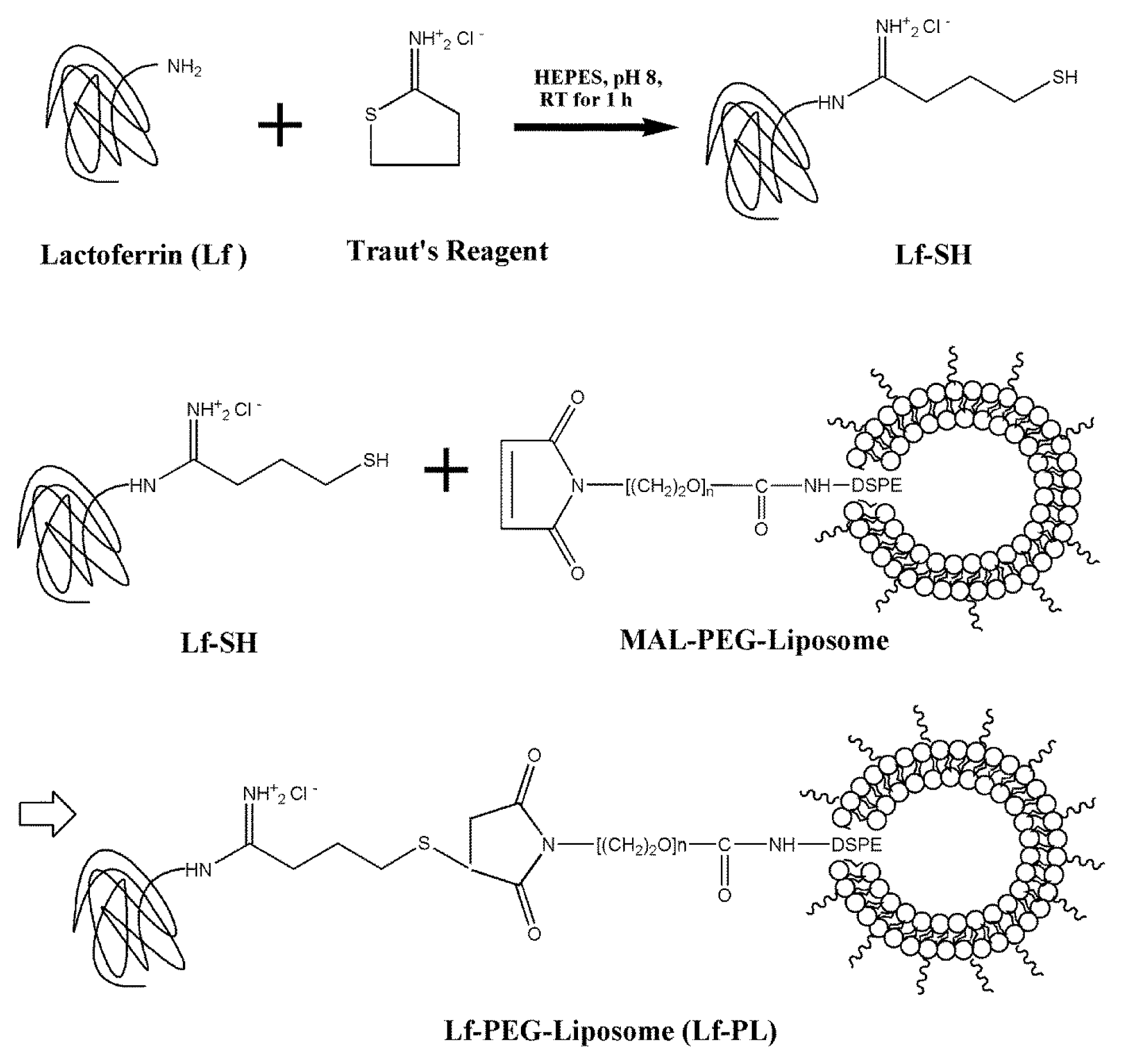
| Parameter | Unit | PL[99mTc] | Lf-PL[99mTc] |
|---|---|---|---|
| 0–24 h | 0–24 h | ||
| T1/2 | h | 18.06 ± 2.09 | 13.88 ± 2.52 |
| MRT | h | 25.23 ± 3.07 | 19.81 ± 4.12 |
| Cmax | %ID/mL | 55.10 ± 1.79 | 53.61 ± 0.55 |
| Cl | mL/h | 0.09 ± 0.01 | 0.11 ± 0.02 |
| AUC(0→24h) | h × %ID/mL | 659.72 ± 58.41 | 653.57 ± 40.84 |
| Organ/tissue | PL[99mTc] | Lf-PL[99mTc] | ||
|---|---|---|---|---|
| 1 h | 2 h | 1 h | 2 h | |
| brain * | 0.69 ± 0.06 | 0.60 ± 0.04 | 1.02 ± 0.06 | 0.81 ± 0.05 |
| blood * | 46.82 ± 1.72 | 47.31 ± 1.48 | 34.38 ± 2.87 | 38.30 ± 2.44 |
| bone | 1.67 ± 0.57 | 2.66 ± 0.35 | 1.41 ± 1.25 | 2.43 ± 0.41 |
| kidney | 8.63 ± 0.74 | 9.23 ± 0.26 | 7.56 ± 0.86 | 9.36 ± 0.55 |
| spleen * | 10.64 ± 0.77 | 10.91 ± 0.73 | 34.12 ± 4.91 | 33.00 ± 2.64 |
| liver | 12.60 ± 3.49 | 10.83 ± 2.80 | 9.04 ± 2.89 | 11.15 ± 1.69 |
| lung | 10.05 ± 2.49 | 10.76 ± 2.39 | 10.61 ± 2.32 | 8.51 ± 0.82 |
| heart | 3.56 ± 2.51 | 4.84 ± 0.48 | 3.82 ± 0.46 | 4.15 ± 0.36 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, F.-Y.J.; Chen, W.-J.; Lee, W.-Y.; Lo, S.-T.; Lee, T.-W.; Lo, J.-M. In Vitro and in Vivo Evaluation of Lactoferrin-Conjugated Liposomes as a Novel Carrier to Improve the Brain Delivery. Int. J. Mol. Sci. 2013, 14, 2862-2874. https://doi.org/10.3390/ijms14022862
Huang F-YJ, Chen W-J, Lee W-Y, Lo S-T, Lee T-W, Lo J-M. In Vitro and in Vivo Evaluation of Lactoferrin-Conjugated Liposomes as a Novel Carrier to Improve the Brain Delivery. International Journal of Molecular Sciences. 2013; 14(2):2862-2874. https://doi.org/10.3390/ijms14022862
Chicago/Turabian StyleHuang, Feng-Yun J., Wan-Jou Chen, Wan-Yu Lee, Su-Tang Lo, Te-Wei Lee, and Jem-Mau Lo. 2013. "In Vitro and in Vivo Evaluation of Lactoferrin-Conjugated Liposomes as a Novel Carrier to Improve the Brain Delivery" International Journal of Molecular Sciences 14, no. 2: 2862-2874. https://doi.org/10.3390/ijms14022862
APA StyleHuang, F.-Y. J., Chen, W.-J., Lee, W.-Y., Lo, S.-T., Lee, T.-W., & Lo, J.-M. (2013). In Vitro and in Vivo Evaluation of Lactoferrin-Conjugated Liposomes as a Novel Carrier to Improve the Brain Delivery. International Journal of Molecular Sciences, 14(2), 2862-2874. https://doi.org/10.3390/ijms14022862




