Yeast α-Glucosidase Inhibitory Phenolic Compounds Isolated from Gynura medica Leaf
Abstract
:1. Introduction
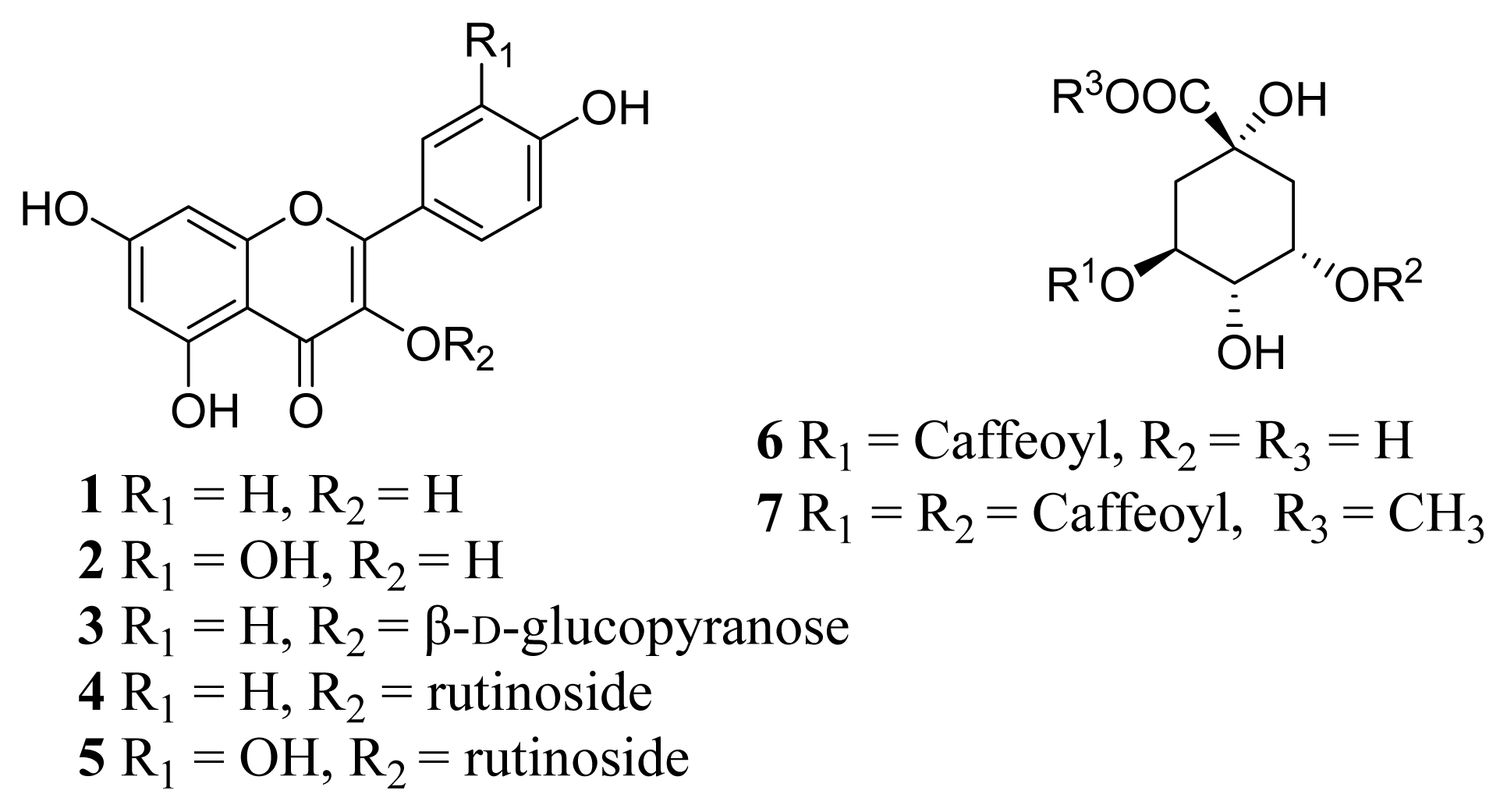
2. Results and Discussion
3. Experimental Section
3.1. Plant Material
3.2. General Experimental Procedures
3.3. Extraction of G. medica Leaf
3.4. Yeast α-Glucosidase Inhibitory Assay
4. Conclusions
Conflict of Interest
References
- Yang, Y.K.; Wu, J.K. A new species of Gynura cass. (Compositae). Chin. Acad. Med. Mag. Org 2004, 4, 55–58. [Google Scholar]
- Jin, J.S.; Kang, F.X.; Ni, Z.D.; Fang, J. Effects of aquatic extract of Gynura medica on anti-hyperglycemia and improving insulin secretion. Anhui Med. Pharm. J 2009, 13, 256–258. [Google Scholar]
- Dong, M.Z.; Shu, W.W. Study on hypoglycemic effects of Gynura medica extraction. Strait Pharm. J 2008, 20, 25–27. [Google Scholar]
- Fu, X.; Zhou, S.; Wan, C. Antioxidant constituents of Gynura medica. China Pharm 2011, 22, 644–645. [Google Scholar]
- Liu, W.; Yu, Y.; Yang, R.; Wan, C.; Xu, B.; Cao, S. Optimization of total flavonoid compound extraction from Gynura medica leaf using response surface methodology and chemical composition analysis. Int. J. Mol. Sci 2010, 11, 4750–4763. [Google Scholar]
- Chou, S.; Chuang, L.; Lee, S. Hypoglycemic constituents of Gynura divaricata subsp. formosana. Nat. Prod. Commun 2012, 7, 221–222. [Google Scholar]
- Deng, Y.; Chen, Y.; Zhang, W.; Chen, B.; Qiu, X.; He, L.; Mu, L.; Yang, C.; Chen, R. Polysaccharide from Gynura divaricata modulates the activities of intestinal disaccharidases in streptozotocin-induced diabetic rats. Brit. J. Nutr 2011, 106, 1323–1329. [Google Scholar]
- Gansau, J.A.; Chin, L.E.E.P.; Embi, N.; Sidek, H.M. Hypoglycemic effects of Gynura procumbens fractions on streptozotocin-induced diabetic rats involved phosphorylation of GSK3β (Ser-9) in liver. Sains Malays 2012, 41, 969–975. [Google Scholar]
- Hassan, Z.; Ahmad, M. Hypoglycemic activity of the aqueous extract of Gynura procumbens (Lour). Merr. Aust. J. Med. Herbal 2004, 16, 116–120. [Google Scholar]
- Hassan, Z.; Yam, M.F.; Ahmad, M.; Yusof, A.P.M. Antidiabetic properties and mechanism of action of Gynura procumbens water extract in streptozotocin-induced diabetic rats. Molecules 2010, 15, 9008–9023. [Google Scholar]
- Li, W.L.; Ren, B.R.; Min, Z.; Hu, Y.; Lu, C.G.; Wu, J.L.; Chen, J.; Sun, S. The anti-hyperglycemic effect of plants in genus Gynura cass. Am. J. Chin. Med 2009, 37, 961–966. [Google Scholar]
- Wu, T.; Zhou, X.; Deng, Y.; Jing, Q.; Li, M.; Yuan, L. In vitro studies of Gynura divaricata (L.) DC extracts as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. J. Ethnopharmacol 2011, 136, 305–308. [Google Scholar]
- Liu, X.; Liu, W.; Cao, X. Ultrasound-assisted extraction of total flavonoids from stems and leaves of Gynura divaricata (L.) DC. and its inhibitory activity on α-glucosidase. Food Sci 2012, 33, 134–139. [Google Scholar]
- Gunawan-Puteri, M.D.; Kato, E.; Kawabata, J. α-Amylase inhibitors from an Indonesian medicinal herb, Phyllanthus urinaria. J. Sci. Food Agric 2011, 92, 606–609. [Google Scholar]
- Chen, L.; Li, H.Q.; Song, H.T.; Zhang, G.G. A new cerebroside from Gynura divaricata. Fitoterapia 2009, 80, 517–520. [Google Scholar]
- Hou, W.C.; Lin, R.D.; Lee, T.H.; Huang, Y.H.; Hsu, F.L.; Lee, M.H. The phenolic constituents and free radical scavenging activities of Gynura formosana Kiamnra. J. Sci. Food Agric 2005, 85, 615–621. [Google Scholar]
- Lin, W.Y.; Yen, M.H.; Teng, C.M.; Tsai, I.L.; Chen, I.S. Cerebrosides from the rhizomes of Gynura japonica. J. Chin. Chem. Soc 2004, 51, 1429–1434. [Google Scholar]
- Rana, V.S.; Blazquez, M.A. Chemical constituents of Gynura cusimbua aerial parts. J. Essent. Oil Res 2007, 19, 21–22. [Google Scholar]
- Wan, C.; Yu, Y.; Zhou, S.; Tian, S.; Cao, S. Isolation and identification of phenolic compounds from Gynura divaricata leaves. Pharmacogn. Mag 2011, 7, 101–108. [Google Scholar]
- Bhandari, M.R.; Anurakkun, N.J.; Hong, G.; Kawabata, J. α-Glucosidase and α-amylase inhibitory activities of Nepalese medicinal herb Pakhanbhed (Bergenia ciliata, Haw.). Food Chem 2008, 106, 247–252. [Google Scholar]
- Yao, Y.; Cheng, X.; Wang, L.; Wang, S.; Ren, G. A determination of potential α-glucosidase inhibitors from azuki beans (Vigna angularis). Int. J. Mol. Sci 2011, 12, 6445–6451. [Google Scholar]
- Wang, Z.B.; Jiang, H.; Xia, Y.G.; Yang, B.Y.; Kuang, H.X. α-Glucosidase inhibitory constituents from Acanthopanax senticosus harm leaves. Molecules 2012, 17, 6269–6276. [Google Scholar]
- Green, B.D.; Flatt, P.R.; Bailey, C.J. Dipeptidyl peptidase IV (DPP IV) inhibitors: A newly emerging drug class for the treatment of type 2 diabetes. Diab. Vasc. Dis. Res 2006, 3, 159–165. [Google Scholar]
- An, T.; Hong, D.; Hu, L.; Li, J. Protein Tyrosine Phosphatases 1B Inhibitors from Traditional Chinese Medicine. In Herbs: Challenges in Chemistry and Biology; ACS Symposium Series: Washington, DC, USA, 2006; pp. 143–156. [Google Scholar]
- Huang, T.H.W.; Peng, G.; Kota, B.P.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Anti-diabetic action of Punica granatum flower extract: Activation of PPAR-γ and identification of an active component. Toxicol. Appl. Pharm 2005, 207, 160–169. [Google Scholar]
- Wan, C.; Zheng, X.; Chen, H.; Zou, X.; Song, Z.; Zhou, S.; Qiu, Y. Flavonoid constituents from herbs of Sarcopyramis bodinieri var. delicata. China J. Chin. Mater. Med 2009, 34, 172–174. [Google Scholar]
- Deng, S.; Deng, Z.; Fan, Y.; Peng, Y.; Li, J.; Xiong, D.; Liu, R. Isolation and purification of three flavonoid glycosides from the leaves of Nelumbo nucifera (Lotus) by high-speed counter-current chromatography. J. Chromatogr. B 2009, 877, 2487–2492. [Google Scholar]
- Zhou, Z.H.; Yang, C.R. Chemical constituents of crude green Pu-Er tea in Yunnan. Acta Botanica Yunnanica 2000, 22, 343–350. [Google Scholar]
- Wan, C.; Yuan, T.; Cirello, A.L.; Seeram, N.P. Antioxidant and α-glucosidase inhibitory phenolics isolated from highbush blueberry flowers. Food Chem 2012, 135, 1929–1937. [Google Scholar]
- Li, Y.Q.; Zhou, F.C.; Gao, F.; Bian, J.S.; Shan, F. Comparative evaluation of quercetin, isoquercetin and rutin as inhibitors of α-glucosidase. J. Agric. Food Chem 2009, 57, 11463–11468. [Google Scholar]

| Compounds | IC50 (mg/mL) |
|---|---|
| 1 | >2.0 |
| 2 | 1.67 ± 0.05 |
| 3 | 1.46 ± 0.03 |
| 4 | 0.38 ± 0.03 |
| 5 | 0.10 ± 0.01 |
| 6 | >2.0 |
| 7 | 0.53 ± 0.02 |
| 8 | 0.99 ± 0.02 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tan, C.; Wang, Q.; Luo, C.; Chen, S.; Li, Q.; Li, P. Yeast α-Glucosidase Inhibitory Phenolic Compounds Isolated from Gynura medica Leaf. Int. J. Mol. Sci. 2013, 14, 2551-2558. https://doi.org/10.3390/ijms14022551
Tan C, Wang Q, Luo C, Chen S, Li Q, Li P. Yeast α-Glucosidase Inhibitory Phenolic Compounds Isolated from Gynura medica Leaf. International Journal of Molecular Sciences. 2013; 14(2):2551-2558. https://doi.org/10.3390/ijms14022551
Chicago/Turabian StyleTan, Chao, Qunxing Wang, Chunhua Luo, Sai Chen, Qianyuan Li, and Peng Li. 2013. "Yeast α-Glucosidase Inhibitory Phenolic Compounds Isolated from Gynura medica Leaf" International Journal of Molecular Sciences 14, no. 2: 2551-2558. https://doi.org/10.3390/ijms14022551




