Modification of Pectin and Hemicellulose Polysaccharides in Relation to Aril Breakdown of Harvested Longan Fruit
Abstract
:1. Introduction
2. Results and Discussion
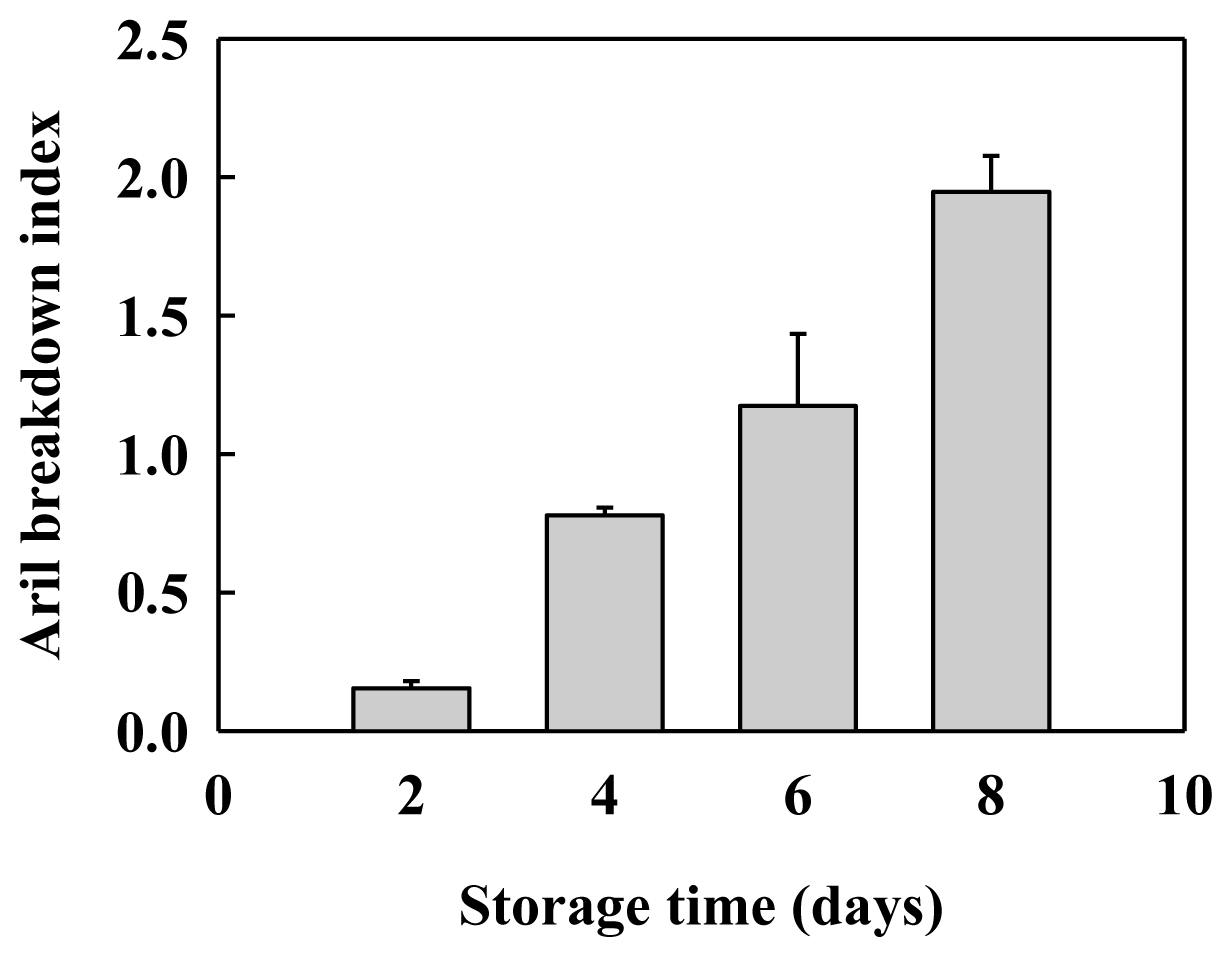
2.1. Aril Breakdown and Cell Wall Material
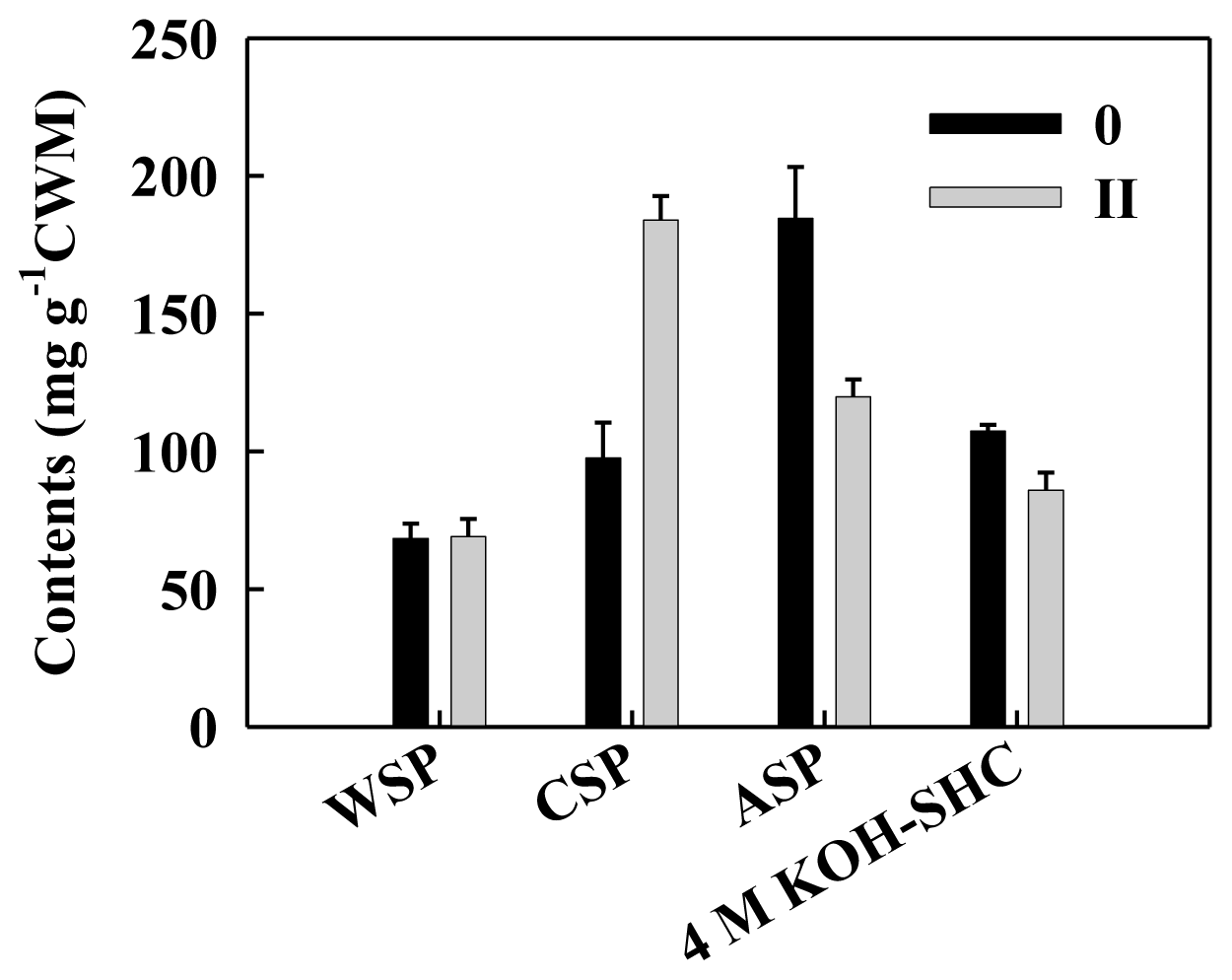
2.2. Cell Wall Polysaccharides
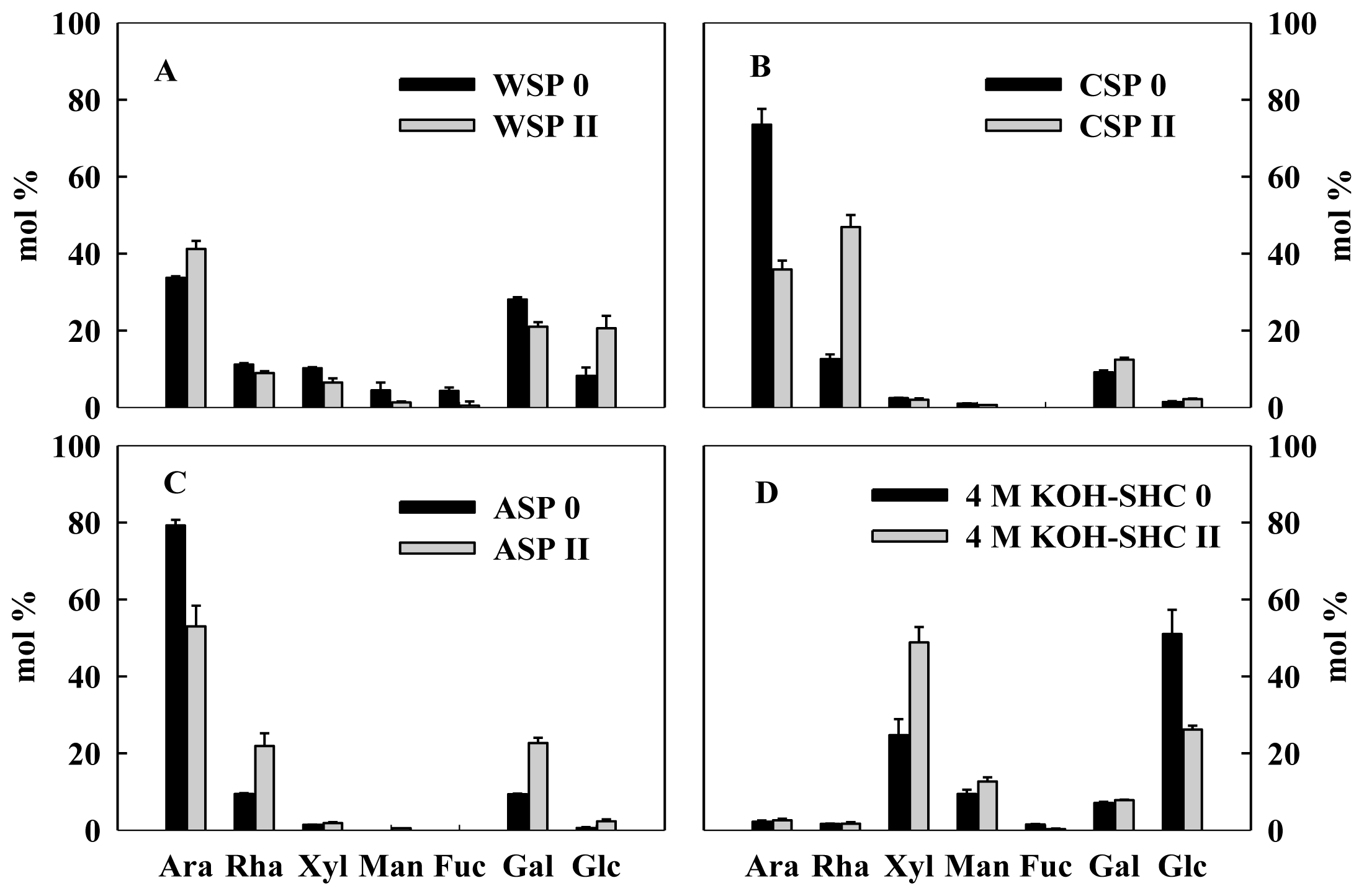
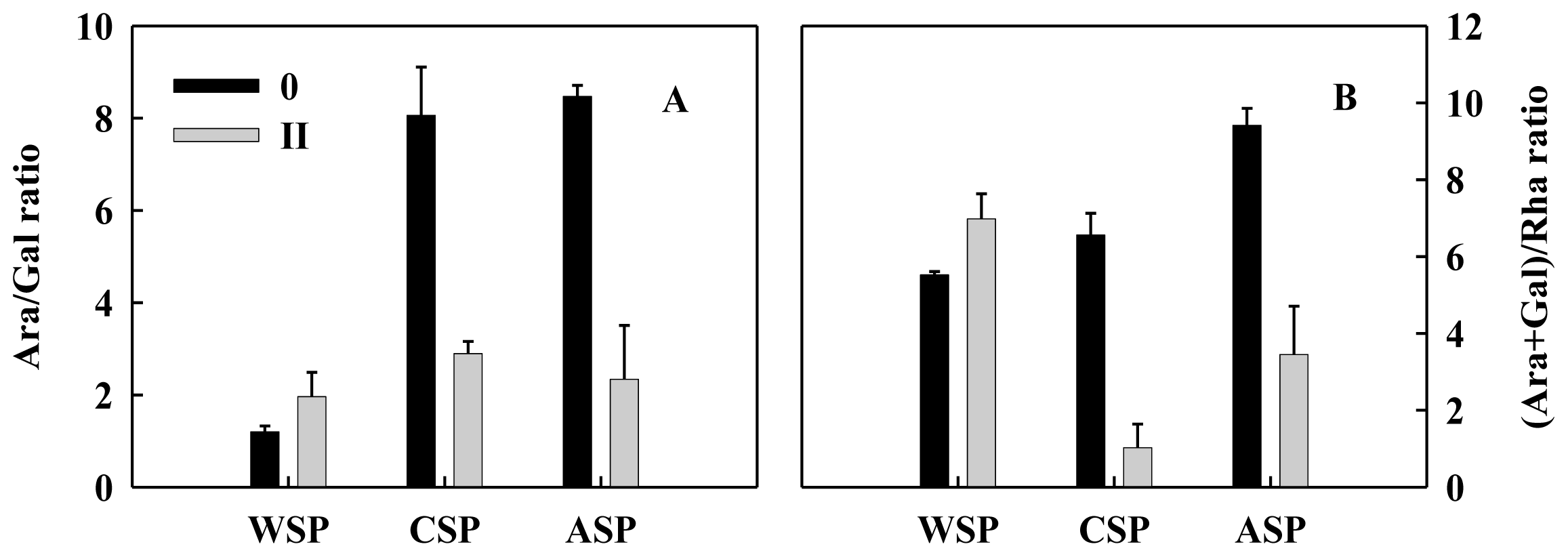
2.3. Monosaccharide Compositions of Pectin and Hemicellulose Polysaccharides Fractions
2.4. Molecular Mass Profile of Pectin and Hemicellulose Polysaccharide Fractions
3. Experimental Section
3.1. Plant Materials
3.2. Evaluation of Aril Breakdown
3.3. Preparation of Cell Wall Material (CWM)
3.4. Fractionation of Pectin and Hemicellulose Polysaccharides
3.5. Analysis of Monosaccharide Compositions
3.6. Gel-Permeation Chromatography
3.7. Data Handling
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jiang, Y.M.; Zhang, Z.Q.; Joyce, D.C.; Ketsa, S. Postharvest biology and handling of longan fruit (Dimocarpus longan Lour.). Postharvest Biol. Technol 2002, 26, 241–252. [Google Scholar]
- Duan, X.W.; Zhang, H.Y.; Zhang, D.D.; Sheng, J.F.; Lin, H.T.; Jiang, Y.M. Role of hydroxyl radical in modification of cell wall polysaccharides and aril breakdown during senescence of harvested longan fruit. Food Chem 2011, 128, 203–207. [Google Scholar]
- Willats, W.G.T.; McCartney, L.; Mackie, W.; Knox, J.P. Pectin: Cell biology and prospects for functional analysis. Plant Mol. Biol 2001, 47, 9–27. [Google Scholar]
- Prasanna, V.; Prabha, T.N.; Tharanathan, R.N. Fruit ripening phenomena—An overview. Crit. Rev. Food Sci 2007, 47, 1–19. [Google Scholar]
- Bennett, A.B.; Labavitch, J.M. Ethylene and ripening-regulated expression and function of fruit cell wall modifying proteins. Plant Sci 2008, 175, 130–136. [Google Scholar]
- Zhu, Q.G.; Zhang, Z.K.; Rao, J.P.; Huber, D.J.; Lv, J.Y.; Hou, Y.L.; Song, K.H. Identification of xyloglucan endotransglucosylase/hydrolase genes (XTHs) and their expression in persimmon fruit as influenced by 1-methylcyclopropene and gibberellic acid during storage at ambient temperature. Food Chem 2013, 138, 471–477. [Google Scholar]
- Cheng, G.P.; Duan, X.W.; Shi, J.; Lu, W.J.; Luo, Y.B.; Jiang, W.B.; Jiang, Y.M. Effects of reactive oxygen species on cellular wall disassembly of banana fruit during ripening. Food Chem 2008, 109, 319–324. [Google Scholar]
- Lo Scalzo, R.; Forni, E.; Lupi, D.; Giudetti, G.; Testoni, A. Changes of pectic composition of ‘Annurca’ apple fruit after storage. Food Chem 2005, 93, 521–530. [Google Scholar]
- Cheng, G.P.; Duan, X.W.; Jiang, Y.M.; Sun, J.; Yang, S.Y.; Yang, B.; He, S.G.; Liang, H.; Luo, Y.B. Modification of hemicellulose polysaccharides during ripening of postharvest banana fruit. Food Chem 2009, 115, 43–47. [Google Scholar]
- Duan, X.W.; Cheng, G.P.; Yang, E.; Yi, C.; Ruenroengklin, N.; Lu, W.J.; Luo, Y.B.; Jiang, Y.M. Modification of pectin polysaccharides during ripening of postharvest banana fruit. Food Chem 2008, 111, 144–149. [Google Scholar]
- Manrique, G.D.; Lajolo, F.M. Cell-wall polysaccharide modifications during postharvest ripening of papaya fruit (Carica papaya). Postharvest Biol. Technol 2004, 33, 11–26. [Google Scholar]
- Yoshioka, H.; Hayama, H.; Tatsuki, M.; Nakamura, Y. Cell wall modifications during softening in melting type peach “Akatsuki” and non-melting type peach “Mochizuki”. Postharvest Biol. Technol 2011, 60, 100–110. [Google Scholar]
- Raffo, M.D.; Ponce, N.M.A.; Sozzi, G.O.; Stortz, C.A.; Vicente, A.R. Changes on the cell wall composition of tree-ripened “Bartlett” pears (Pyrus communis L.). Postharvest Biol. Technol 2012, 73, 72–79. [Google Scholar]
- Ponce, N.M.A.; Ziegler, V.H.; Stortz, C.A.; Sozzi, G.O. Compositional changes in cell wall polysaccharides from Japanese plum (Prunus salicina Lindl.) during growth and on-tree ripening. J. Agric. Food Chem 2010, 58, 2562–2570. [Google Scholar]
- Lunn, D.; Phan, T.D.; Tucker, G.A.; Lycett, G.W. Cell wall composition of tomato fruit changes during development and inhibition of vesicle trafficking is associated with reduced pectin levels and reduced softening. Plant Physiol. Biochem 2013, 66, 91–97. [Google Scholar]
- Nishiyama, K.; Guis, M.; Rose, J.K.C.; Kubo, Y.; Bennett, K.A.; Lu, W.J.; Kato, K.; Ushijima, K.; Nakano, R.; Inaba, A.; et al. Ethylene regulation of fruit softening and cell wall disassembly in Charentais melon. J. Exp. Bot 2007, 58, 1281–1290. [Google Scholar]
- Lahaye, M.; Falourd, X.; Quemener, B.; Ralet, M.C.; Howad, W.; Dirlewanger, E.; Arus, P. Cell wall polysaccharide chemistry of peach genotypes with contrasted textures and other fruit traits. J. Agric. Food Chem 2012, 60, 6594–6605. [Google Scholar]
- Wu, F.W.; Zhang, D.D.; Zhang, H.Y.; Jiang, G.Q.; Su, X.G.; Qu, H.X.; Jiang, Y.M.; Duan, X.W. Physiological and biochemical response of harvested plum fruit to oxalic acid during ripening or shelf-life. Food Res. Int 2011, 44, 1299–1305. [Google Scholar]
- Brummell, D.A. Cell wall disassembly in ripening fruit. Funct. Plant Biol 2006, 33, 103–119. [Google Scholar]
- Deng, Y.; Wu, Y.; Li, Y.F. Changes in firmness, cell wall composition and cell wall hydrolases of grapes stored in high oxygen atmospheres. Food Res. Int 2005, 38, 769–776. [Google Scholar]
- Missang, C.E.; Maingonnat, J.F.; Renard, C.; Audergon, J.M. Apricot cell wall composition: Relation with the intra-fruit texture heterogeneity and impact of cooking. Food Chem 2012, 133, 45–54. [Google Scholar]
- Wakabayashi, K. Changes in cell wall polysaccharides during fruit ripening. J. Plant Res 2000, 113, 231–237. [Google Scholar]
- Brummell, D.A.; Dal Cin, V.; Crisosto, C.H.; Labavitch, J.M. Cell wall metabolism during maturation, ripening and senescence of peach fruit. J. Exp. Bot 2004, 55, 2029–2039. [Google Scholar]
- Popper, Z.A.; Fry, S.C. Widespread occurrence of covalent linkage between xyloglucan and acidic polysaccarides in suspension-cultured angiosperm cells. Ann. Bot 2005, 96, 91–99. [Google Scholar]
- Thompson, J.E.; Fry, S.C. Evidence for covalaent linkage between xyloglucan and acidic pectins in suspension-cultured rose cells. Planta 2000, 211, 275–286. [Google Scholar]
- Gross, K.C.; Sams, C.E. Changes in cell-wall neutral sugar composition during fruit ripenin—A species survey. Phytochemistry 1984, 23, 2457–2461. [Google Scholar]
- Renard, C.; Ginies, C. Comparison of the cell wall composition for flesh and skin from five different plums. Food Chem 2009, 114, 1042–1049. [Google Scholar]
- Giovannoni, J.J.; Dellapenna, D.; Bennett, A.B.; Fischer, R.L. Expression of a chimeric polygalacturonase gene in transgenic rin (ripening inhibitor) tomato fruit results in polyuronide degradation but not fruit softening. Plant Cell 1989, 1, 53–63. [Google Scholar]
- Lazan, H.; Ng, S.Y.; Goh, L.Y.; Ali, Z.M. Papaya beta-galactosidase/galactanase isoforms in differential cell wall hydrolysis and fruit softening during ripening. Plant Physiol. Biochem 2004, 42, 847–853. [Google Scholar]
- Lin, H.T.; Zhao, Y.F.; Xi, Y.F. Changes in cell wall components and cell wall-degrading enzyme activities of postharvest longan fruit during aril breakdown. J. Plant Physiol. Mol. Biol 2007, 33, 137–145. [Google Scholar]
- Tateishi, A. beta-galactosidase and alpha-l-arabinofuranosidase in cell wall modification related with fruit development and softening. J. Jpn. Soc. Hortic. Sci 2008, 77, 329–340. [Google Scholar]
- Banik, M.; Bourgault, R.; Bewley, J.D. Endo-beta-mannanase is present in an inactive form in ripening tomato fruits of the cultivar Walter. J. Exp. Bot 2001, 52, 105–111. [Google Scholar]
- Chin, L.H.; Ali, Z.M.; Lazan, H. Cell wall modifications, degrading enzymes and softening of carambola fruit during ripening. J. Exp. Bot 1999, 50, 767–775. [Google Scholar]
- Ronen, R.; Zauberman, G.; Akerman, M.; Weksler, A.; Rot, I.; Fuchs, Y. Xylanase and xylosidase activities in avocado fruit. Plant Physiol 1991, 95, 961–964. [Google Scholar]
- Schroder, R.; Wegrzyn, T.F.; Sharma, N.N.; Atkinson, R.G. LeMAN4 endo-beta-mannanase from ripe tomato fruit can act as a mannan transglycosylase or hydrolase. Planta 2006, 224, 1091–1102. [Google Scholar]
- Xiao, R.; Chen, J.Y.; Chen, J.W.; Ou, M.; Jiang, Y.M.; Lin, H.T.; Lu, W.J. Expression analysis of endo-1,4-beta-glucanase genes during aril breakdown of harvested longan fruit. J. Sci. Food Agric 2009, 89, 1129–1136. [Google Scholar]
- Fry, S.C. The Growing Plant Cell Wall: Chemical and Metabolic Analysis; Longman: London, UK, 1988. [Google Scholar]
- Selvendran, R.R.; Ryden, P. Isolation and Analysis of Plant Cell Walls. In Methods in Plant Biochemistry; Dey, P.M., Ed.; Academic Press: New York, NY, USA, 1990; Volume 2, pp. 549–579. [Google Scholar]
- Wen, L.R.; Lin, L.Z.; You, L.J.; Yang, B.; Jiang, G.X.; Zhao, M.M. Ultrasound-assited extraction and structural identification of polysaccharides from Isodon lophanthoides var. gerardianus (Bentham) H. Hara. Carbohyd. Polym 2011, 85, 541–547. [Google Scholar]



© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, D.; Zhang, H.; Wu, F.; Li, T.; Liang, Y.; Duan, X. Modification of Pectin and Hemicellulose Polysaccharides in Relation to Aril Breakdown of Harvested Longan Fruit. Int. J. Mol. Sci. 2013, 14, 23356-23368. https://doi.org/10.3390/ijms141223356
Wang D, Zhang H, Wu F, Li T, Liang Y, Duan X. Modification of Pectin and Hemicellulose Polysaccharides in Relation to Aril Breakdown of Harvested Longan Fruit. International Journal of Molecular Sciences. 2013; 14(12):23356-23368. https://doi.org/10.3390/ijms141223356
Chicago/Turabian StyleWang, Duoduo, Haiyan Zhang, Fuwang Wu, Taotao Li, Yuxiang Liang, and Xuewu Duan. 2013. "Modification of Pectin and Hemicellulose Polysaccharides in Relation to Aril Breakdown of Harvested Longan Fruit" International Journal of Molecular Sciences 14, no. 12: 23356-23368. https://doi.org/10.3390/ijms141223356



