Differentiated Epithelial- and Mesenchymal-Like Phenotypes in Subcutaneous Mouse Xenografts Using Diffusion Weighted-Magnetic Resonance Imaging
Abstract
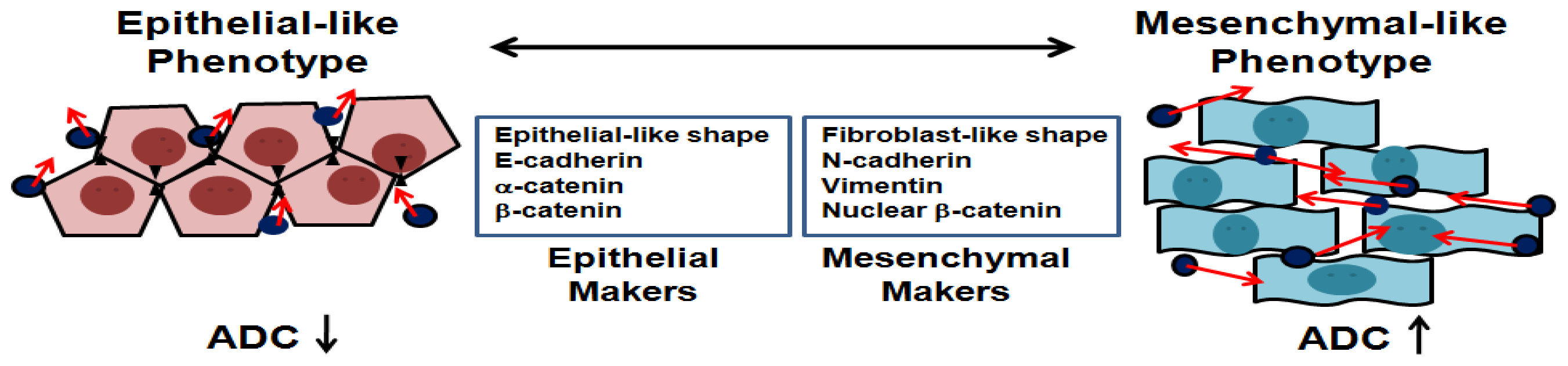
:1. Introduction
2. Results
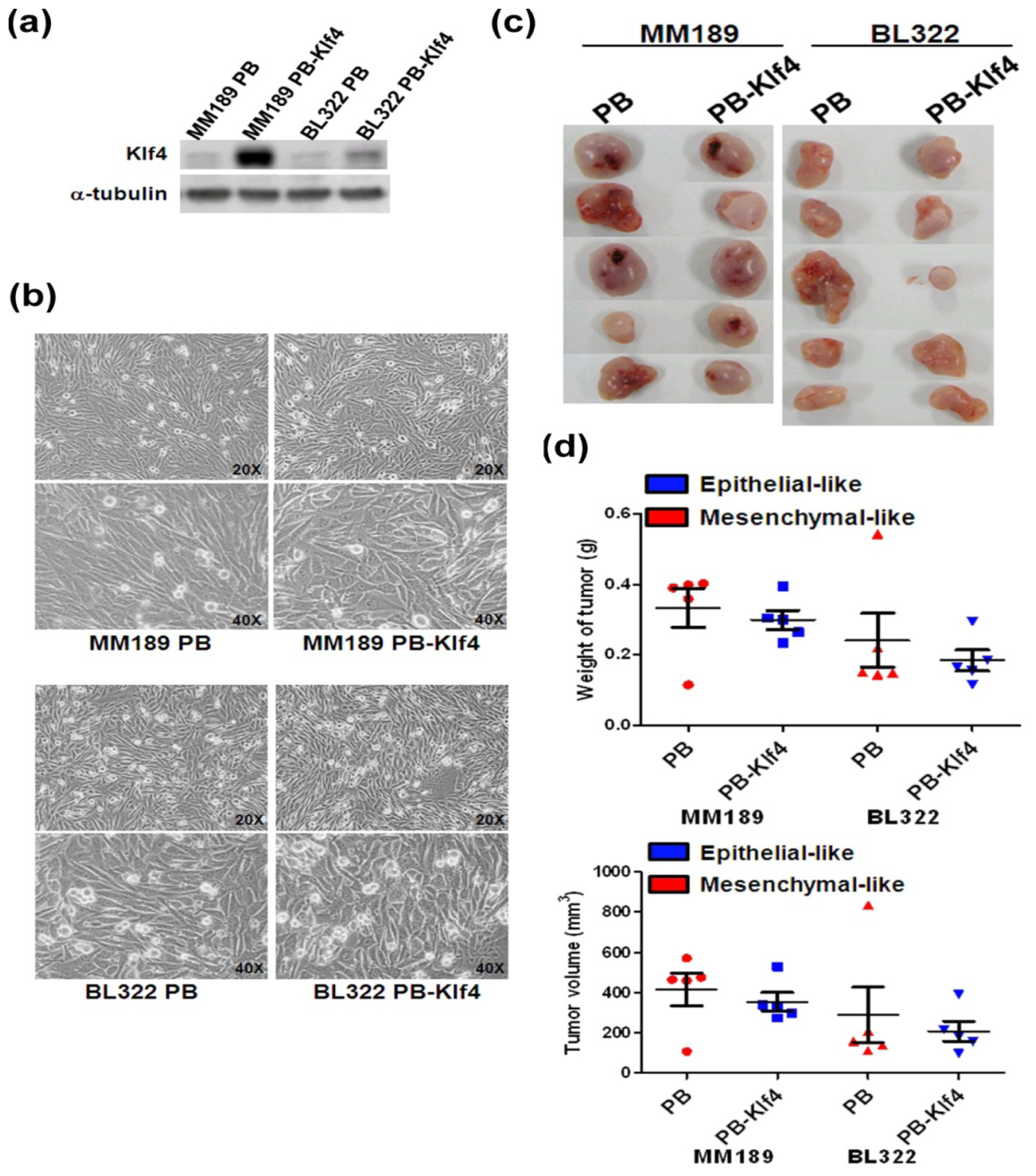
2.1. Epithelial- or Mesenchymal-Like Xenografted Tumors Generated from Kruppel-Like Factor 4 (Klf4) or Vector Control Expressing Mesenchymal-Like Cells
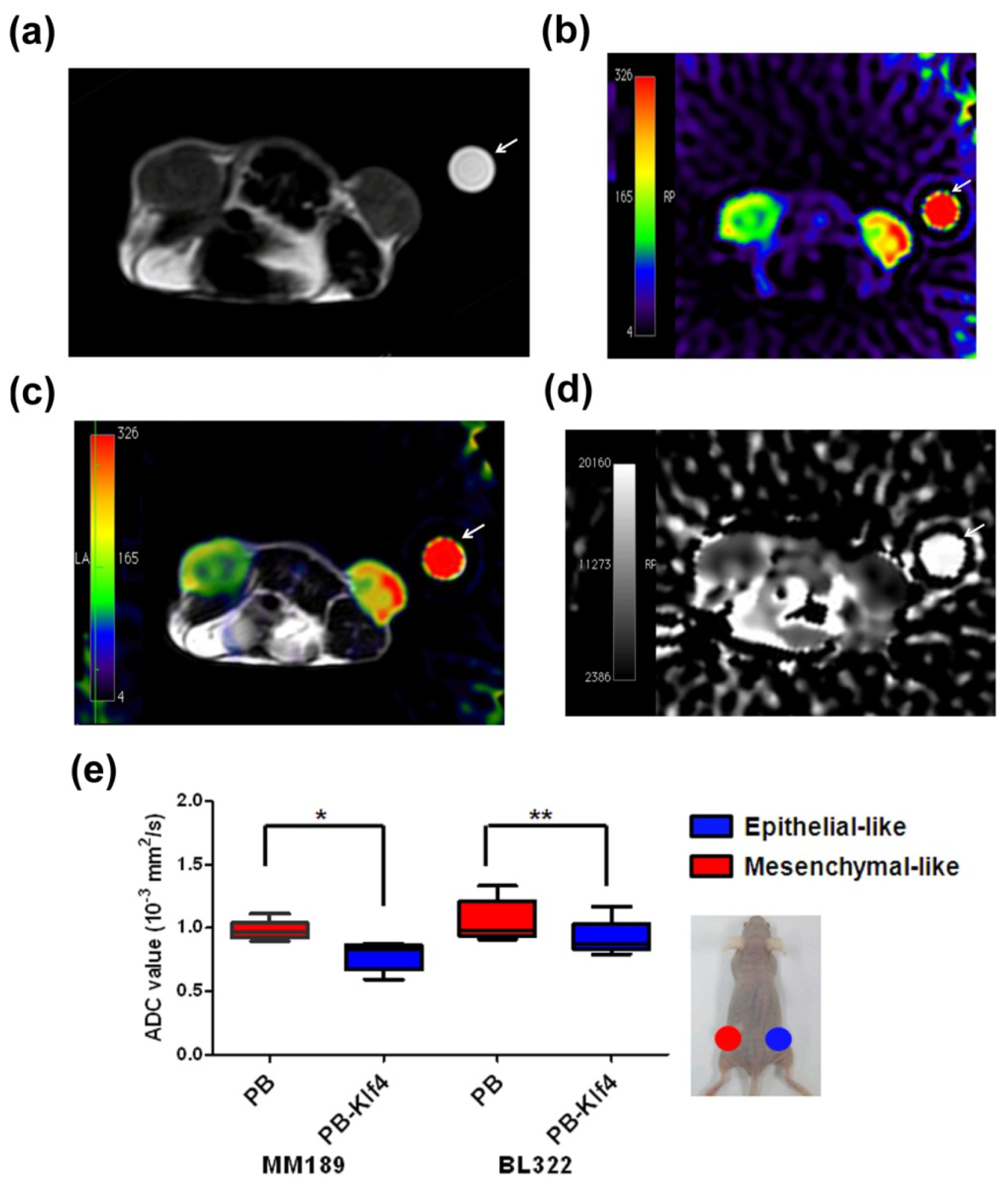
2.2. Mesenchymal-Like Tumors Have Higher ADC Values than Those of Corresponding Epithelial-Like Tumors
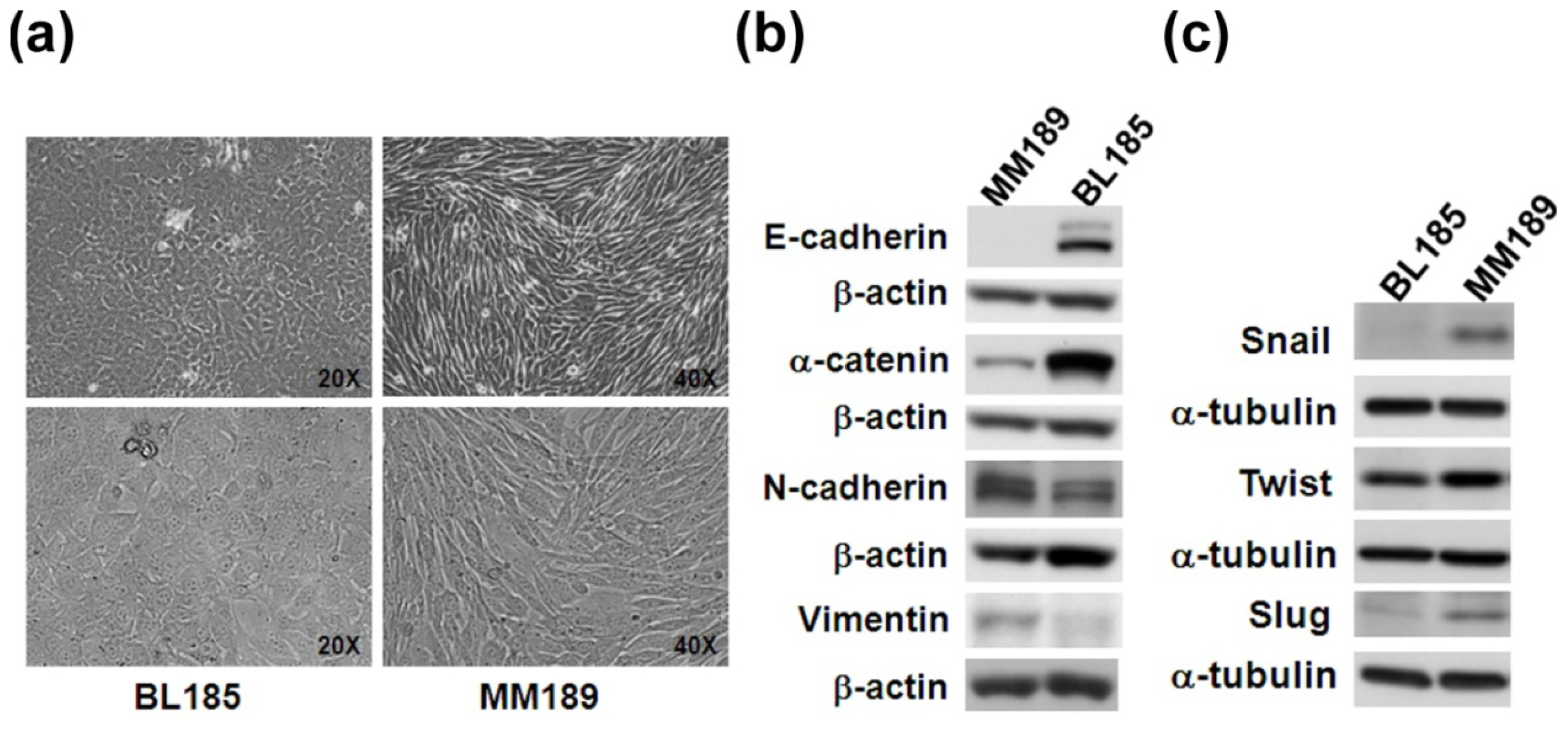
2.3. Epithelial- or Mesenchymal-Like Phenotypes in Two Cancer Cell Lines
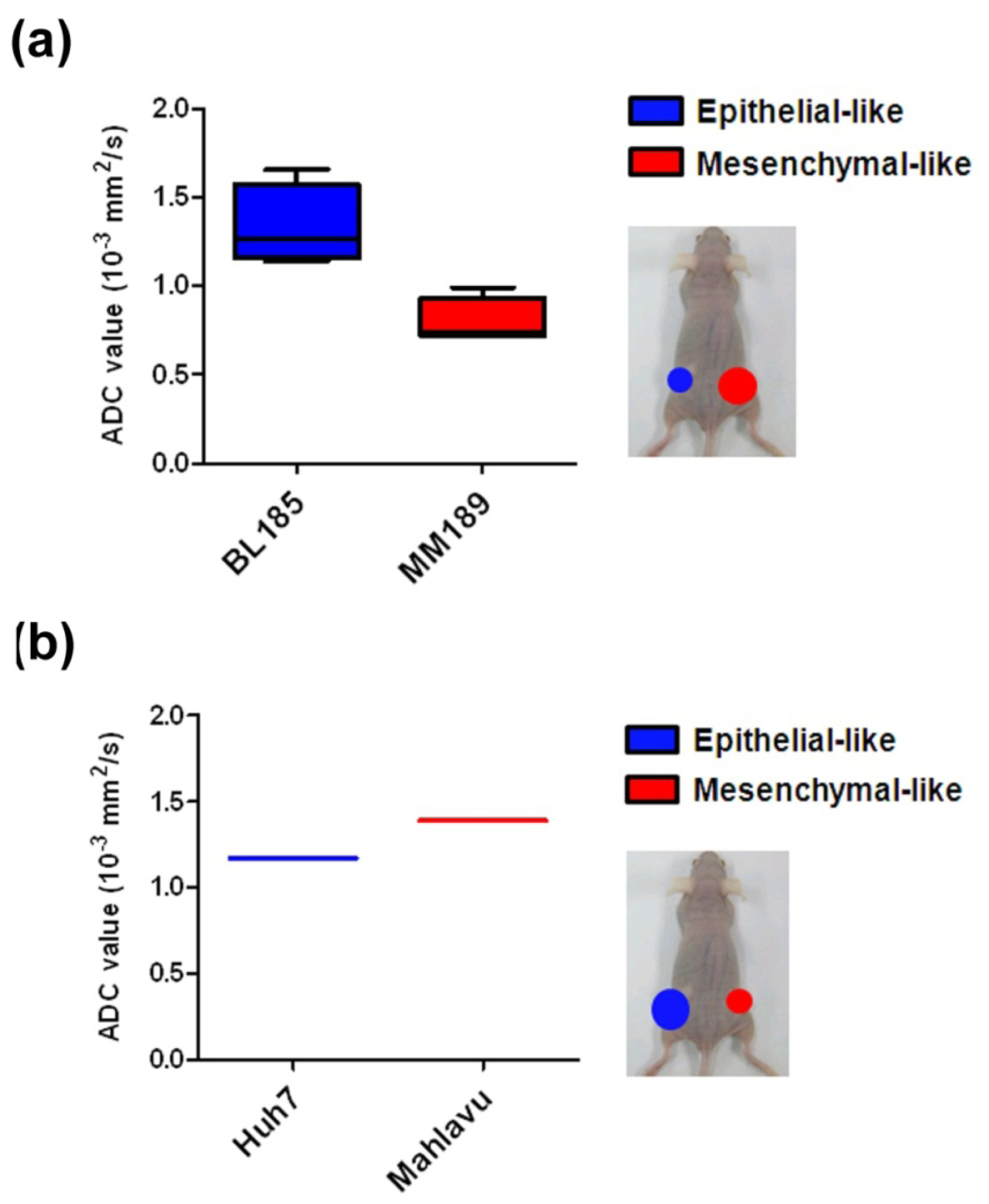
2.4. Diverse ADC Values in Epithelial- or Mesenchymal-Like Xenografted Tumors Generated from Epithelial- or Mesenchymal-Like Cancer Cell Lines
2.5. Similar Tumor Cellularity in Epithelial- or Mesenchymal-Like Xenografted Tumors
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Cell Lines
4.3. Cell Morphology
4.4. Western Blot
4.5. Xenografted Tumor Models
4.6. DW-MRI
4.7. Post-Processing Workstation
4.8. Histology and Analysis of Tumor Cellularity
4.9. Statistical Analysis
5. Conclusions

Acknowledgments
Abbreviations
| TE | echo time |
| TR | repetition time |
| ADC | apparent diffusion coefficient |
| DW-MRI | diffusion weighted magnetic resonance imaging |
| EPI | echo-planar imaging |
| PROPELLER | periodically rotated overlapping parallel lines with enhanced reconstruction |
| FOV | field of view |
| ROI | region of interest |
| EMT | epithelial mesenchymal transition |
| MET | mesenchymal epithelial transition. |
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar]
- Fransvea, E.; Angelotti, U.; Antonaci, S.; Giannelli, G. Blocking transforming growth factor-β up-regulates E-cadherin and reduces migration and invasion of hepatocellular carcinoma cells. Hepatology 2008, 47, 1557–1566. [Google Scholar]
- Lee, T.K.; Poon, R.T.; Yuen, A.P.; Ling, M.T.; Kwok, W.K.; Wang, X.H.; Wong, Y.C.; Guan, X.Y.; Man, K.; Chau, K.L.; Fan, S.T. Twist overexpression correlates with hepatocellular carcinoma metastasis through induction of epithelial-mesenchymal transition. Clin. Cancer Res 2006, 12, 5369–5376. [Google Scholar]
- Thiery, J.P. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar]
- Levayer, R.; Lecuit, T. Breaking down EMT. Nat. Cell Biol 2008, 10, 757–759. [Google Scholar]
- Huber, M.A.; Kraut, N.; Beug, H. Molecular requirements for epithelial-mesenchymal transition during tumor progression. Curr. Opin. Cell Biol 2005, 17, 548–558. [Google Scholar]
- Roussos, E.T.; Keckesova, Z.; Haley, J.D.; Epstein, D.M.; Weinberg, R.A.; Condeelis, J.S. AACR special conference on epithelial-mesenchymal transition and cancer progression and treatment. Cancer Res 2010, 70, 7360–7364. [Google Scholar]
- Yang, J.; Mani, S.A.; Donaher, J.L.; Ramaswamy, S.; Itzykson, R.A.; Come, C.; Savagner, P.; Gitelman, I.; Richardson, A.; Weinberg, R.A. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 2004, 117, 927–939. [Google Scholar]
- Pautler, R.G.; Mouse, MRI. Concepts and applications in physiology. Physiology 2004, 19, 168–175. [Google Scholar]
- Serrano, F.; Deshazer, M.; Smith, K.D.; Ananta, J.S.; Wilson, L.J.; Pautler, R.G. Assessing transneuronal dysfunction utilizing manganese-enhanced MRI (MEMRI). Magn. Reson. Med 2008, 60, 169–175. [Google Scholar]
- Dechambre, S.; Duprez, T.; Lecouvet, F.; Raftopoulos, C.; Gosnard, G. Diffusion-weighted MRI postoperative assessment of an epidermoid tumour in the cerebellopontine angle. Neuroradiology 1999, 41, 829–831. [Google Scholar]
- Dorenbeck, U.; Butz, B.; Schlaier, J.; Bretschneider, T.; Schuierer, G.; Feuerbach, S. Diffusion-weighted echo-planar MRI of the brain with calculated ADCs: A useful tool in the differential diagnosis of tumor necrosis from abscess? J. Neuroimaging 2003, 13, 330–338. [Google Scholar]
- Dorenbeck, U.; Butz, B.; Schlaier, J.; Bretschneider, T.; Schuierer, G.; Feuerbach, S. Diffusion-weighted MR imaging of experimental brain tumors in rats. Magma 1995, 3, 13–20. [Google Scholar]
- Guo, Y.; Cai, Y.Q.; Cai, Z.L.; Gao, Y.G.; An, N.Y.; Ma, L.; Mahankali, S.; Gao, J.H. Differentiation of clinically benign and malignant breast lesions using diffusion-weighted imaging. J. Magn. Reson. Imaging 2002, 16, 172–178. [Google Scholar]
- Hosseinzadeh, K.; Schwarz, S.D. Endorectal diffusion-weighted imaging in prostate cancer to differentiate malignant and benign peripheral zone tissue. J. Magn. Reson. Imaging 2004, 20, 654–661. [Google Scholar]
- Le Bihan, D.; Breton, E.; Lallemand, D.; Aubin, M.L.; Vignaud, J.; Laval-Jeantet, M. Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 1988, 168, 497–505. [Google Scholar]
- Wang, Y.; Chen, Z.E.; Yaghmai, V.; Nikolaidis, P.; McCarthy, R.J.; Merrick, L.; Miller, F.H. Diffusion-weighted MR imaging in pancreatic endocrine tumors correlated with histopathologic characteristics. J. Magn. Reson. Imaging 2011, 33, 1071–1079. [Google Scholar]
- Wang, H.; Cheng, L.; Zhang, X.; Wang, D.; Guo, A.; Gao, Y.; Ye, H. Renal cell carcinoma: Diffusion-weighted MR imaging for subtype differentiation at 3.0 T. Radiology 2010, 257, 135–143. [Google Scholar]
- Patterson, D.M.; Padhani, A.R.; Collins, D.J. Technology insight: Water diffusion MRI—A potential new biomarker of response to cancer therapy. Nat. Clin. Pract. Oncol 2008, 5, 220–233. [Google Scholar]
- Lin, Z.S.; Chu, H.; Yen, Y.; Lewis, B.C.; Chen, Y. Kruppel-like factor 4, a tumor suppressor in hepatocellular carcinoma cells reverts epithelial mesenchymal transition by suppressing slug expression. PLoS One 2012, 7, e43593. [Google Scholar]
- Chen, Y.W.; Klimstra, D.S.; Mongeau, M.E.; Tatem, J.L.; Boyartchuk, V.; Lewis, B.C. Loss of p53 and Ink4a/Arf cooperate in a cell autonomous fashion to induce metastasis of hepatocellular carcinoma cells. Cancer Res 2007, 67, 7589–7596. [Google Scholar]
- Yang, M.H.; Chen, C.L.; Chau, G.Y.; Chiou, S.H.; Su, C.W.; Chou, T.Y.; Peng, W.L.; Wu, J.C. Comprehensive analysis of the independent effect of twist and snail in promoting metastasis of hepatocellular carcinoma. Hepatology 2009, 50, 1464–1474. [Google Scholar]
- Deng, J.; Virmani, S.; Young, J.; Harris, K.; Yang, G.Y.; Rademaker, A.; Woloschak, G.; Omary, R.A.; Larson, A.C. Diffusion-weighted PROPELLER MRI for quantitative assessment of liver tumor necrotic fraction and viable tumor volume in VX2 rabbits. J. Magn. Reson. Imaging 2008, 27, 1069–1076. [Google Scholar]
- Thompson, E.W.; Haviv, I. The social aspects of EMT-MET plasticity. Nat. Med 2011, 17, 1048–1049. [Google Scholar]
- Garber, K. Epithelial-to-mesenchymal transition is important to metastasis, but questions remain. J. Natl. Cancer Inst 2008, 100, 232–233. [Google Scholar]
- Cowin, P.; Rowlands, T.M.; Hatsell, S.J. Cadherins and catenins in breast cancer. Curr. Opin. Cell Biol 2005, 17, 499–508. [Google Scholar]
- Junghans, D.; Haas, I.G.; Kemler, R. Mammalian cadherins and protocadherins: About cell death, synapses and processing. Curr. Opin. Cell Biol 2005, 17, 446–452. [Google Scholar]
- Yauch, R.L.; Januario, T.; Eberhard, D.A.; Cavet, G.; Zhu, W.; Fu, L.; Pharm, T.Q.; Soriano, R.; Stinson, J.; Seshagiri, S.; et al. Epithelial versus mesenchymal phenotype determines in vitro sensitivity and predicts clinical activity of erlotinib in lung cancer patients. Clin. Cancer Res 2005, 11, 8686–8698. [Google Scholar]
- Wijnhoven, B.P.; Dinjens, W.N.; Pignatelli, M. E-cadherin-catenin cell-cell adhesion complex and human cancer. Br. J. Surg 2000, 87, 992–1005. [Google Scholar]
- Garcia, S.; Martini, F.; De Micco, C.; Andrac, L.; Hardwigsen, J.; Sappa, P.; Lavaut, M.N.; Le Treut, Y.P.; Charpin, C. Immunoexpression of E-cadherin and beta-catenin correlates to survival of patients with hepatocellular carcinomas. Int. J. Oncol 1998, 12, 443–447. [Google Scholar]
- Suzuki, H.; Masuda, N.; Shimura, T.; Araki, K.; Kobayashi, T.; Tsutsumi, S.; Asao, T.; Kuwano, H. Nuclear beta-catenin expression at the invasive front and in the vessels predicts liver metastasis in colorectal carcinoma. Anticancer Res 2008, 28, 1821–1830. [Google Scholar]
- Stephen, R.M.; Pagel, M.D.; Brown, K.; Baker, A.F.; Meuillet, E.J.; Gillies, R.J. Monitoring the development of xenograft triple-negative breast cancer models using diffusion-weighted magnetic resonance imaging. Exp. Biol. Med 2012, 237, 1273–1280. [Google Scholar]
- Koh, D.M.; Scurr, E.; Collins, D.J.; Pirgon, A.; Kanber, B.; Karanjia, N.; Brown, G.; Leach, M.O.; Husband, J.E. Colorectal hepatic metastases: Quantitative measurements using single-shot echo-planar diffusion-weighted MR imaging. Eur. Radiol 2006, 16, 1898–1905. [Google Scholar]
- Wagner, M.; Maggiori, L.; Ronot, M.; Paradis, V.; Vilgrain, V.; Panis, Y.; Van Beers, B.E. Diffusion-weighted and T2-weighted MR imaging for colorectal liver metastases detection in a rat model at 7 T: A comparative study using histological examination as reference. Eur. Radiol 2013, 23, 2156–2164. [Google Scholar]
- Kim, C.K.; Park, B.K.; Han, J.J.; Kang, T.W.; Lee, H.M. Diffusion-weighted imaging of the prostate at 3 T for differentiation of malignant and benign tissue in transition and peripheral zones: Preliminary results. J. Comput. Assist. Tomogr 2007, 31, 449–454. [Google Scholar]
- Kumar, V.; Jagannathan, N.R.; Kumar, R.; Thulkar, S.; Gupta, S.D.; Dwivedi, S.N.; Hemal, A.K.; Gupta, N.P. Apparent diffusion coefficient of the prostate in men prior to biopsy: Determination of a cut-off value to predict malignancy of the peripheral zone. NMR Biomed 2007, 20, 505–511. [Google Scholar]
- Koppenol, W.H.; Bounds, P.L.; Dang, C.V. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 2011, 11, 325–337. [Google Scholar]
- Seppet, E.; Gruno, M.; Peetsalu, A.; Gizatullina, Z.; Nguyen, H.P.; Vielhaber, S.; Wussling, M.H.P.; Trumbeckaite, S.; Arandarcikaite, O.; Jerzembeck, D.; et al. Mitochondria and energetic depression in cell pathophysiology. Int. J. Mol. Sci 2009, 10, 2252–2303. [Google Scholar]
- Byun, B.H.; Noh, W.C.; Lim, I.; Lee, S.S.; Cho, A.R.; Park, J.A.; Kim, K.M.; Kim, H.A.; Kim, E.K.; Kim, B.I.; Choi, C.W.; Lim, S.M. A new method for apparent diffusion coefficient measurement using sequential F-FDG PET and MRI: Correlation with histological grade of invasive ductal carcinoma of the breast. Ann. Nucl. Med 2013. [Google Scholar] [CrossRef]
- Padhani, A.R.; Liu, G.; Mu-Koh, D.; Chenevert, T.L.; Thoeny, H.C.; Takahara, T.; Dzik-Jurasz, A.; Ross, B.D.; van Cauteren, M.; Collins, D.; et al. Diffusion-weighted magnetic resonance imaging as a cancer biomarker: Consensus and recommendations. Neoplasia 2009, 11, 102–125. [Google Scholar]
- Zhang, J.L.; Sigmund, E.E.; Chandarana, H.; Rusinek, H.; Chen, Q.; Vivier, P.H.; Taouli, B.; Lee, V.S. Variability of renal apparent diffusion coefficients: Limitations of the monoexponential model for diffusion quantification. Radiology 2010, 254, 783–792. [Google Scholar]
- Cui, Y.; Zhang, X.P.; Sun, Y.S.; Tang, L.; Shen, L. Apparent diffusion coefficient: Potential imaging biomarker for prediction and early detection of response to chemotherapy in hepatic metastases. Radiology 2008, 248, 894–900. [Google Scholar]
- Squillaci, E.; Manenti, G.; Cova, M.; Di Roma, M.; Miano, R.; Palmieri, G.; Simonetti, G. Correlation of diffusion-weighted MR imaging with cellularity of renal tumours. Anticancer Res 2004, 24, 4175–4179. [Google Scholar]
- Yoshikawa, M.I.; Ohsumi, S.; Sugata, S.; Kataoka, M.; Takashima, S.; Mochizuki, T.; Ikura, H.; Imai, Y. Relation between cancer cellularity and apparent diffusion coefficient values using diffusion-weighted magnetic resonance imaging in breast cancer. Radiat. Med 2008, 26, 222–226. [Google Scholar]
- Chen, Y.W.; Paliwal, S.; Draheim, K.; Grossman, S.R.; Lewis, B.C. p19Arf inhibits the invasion of hepatocellular carcinoma cells by binding to C-terminal binding protein. Cancer Res 2008, 68, 476–482. [Google Scholar]
- Nakabayashi, H.; Taketa, K.; Yamane, T.; Miyazaki, M.; Miyano, K.; Sato, J. Phenotypical stability of a human hepatoma cell line, HuH-7, in long-term culture with chemically defined medium. Gann 1984, 75, 151–158. [Google Scholar]
- Puisieux, A.; Galvin, K.; Troalen, F.; Bressac, B.; Marcais, C.; Galun, E.; Ponchel, F.; Yakicier, C.; Ji, J.; Ozturk, M. Retinoblastoma and p53 tumor suppressor genes in human hepatoma cell lines. FASEB J 1993, 7, 1407–1413. [Google Scholar]
- Rosset, A.; Spadola, L.; Ratib, O. OsiriX: An open-source software for navigating in multidimensional DICOM images. J. Digit. Imaging 2004, 17, 205–216. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, Y.-W.; Pan, H.-B.; Tseng, H.-H.; Chu, H.-C.; Hung, Y.-T.; Yen, Y.-C.; Chou, C.-P. Differentiated Epithelial- and Mesenchymal-Like Phenotypes in Subcutaneous Mouse Xenografts Using Diffusion Weighted-Magnetic Resonance Imaging. Int. J. Mol. Sci. 2013, 14, 21943-21959. https://doi.org/10.3390/ijms141121943
Chen Y-W, Pan H-B, Tseng H-H, Chu H-C, Hung Y-T, Yen Y-C, Chou C-P. Differentiated Epithelial- and Mesenchymal-Like Phenotypes in Subcutaneous Mouse Xenografts Using Diffusion Weighted-Magnetic Resonance Imaging. International Journal of Molecular Sciences. 2013; 14(11):21943-21959. https://doi.org/10.3390/ijms141121943
Chicago/Turabian StyleChen, Ya-Wen, Huay-Ben Pan, Hui-Hwa Tseng, Hsiao-Chien Chu, Yu-Ting Hung, Yi-Chen Yen, and Chen-Pin Chou. 2013. "Differentiated Epithelial- and Mesenchymal-Like Phenotypes in Subcutaneous Mouse Xenografts Using Diffusion Weighted-Magnetic Resonance Imaging" International Journal of Molecular Sciences 14, no. 11: 21943-21959. https://doi.org/10.3390/ijms141121943



