Keratin K15 as a Biomarker of Epidermal Stem Cells
Abstract
:1. Introduction
What Are Stem Cells?
2. Evolution of Epidermal Stem Cell Research
Epidermal Stem Cell Markers
3. K15—A Marker of Stem Cells
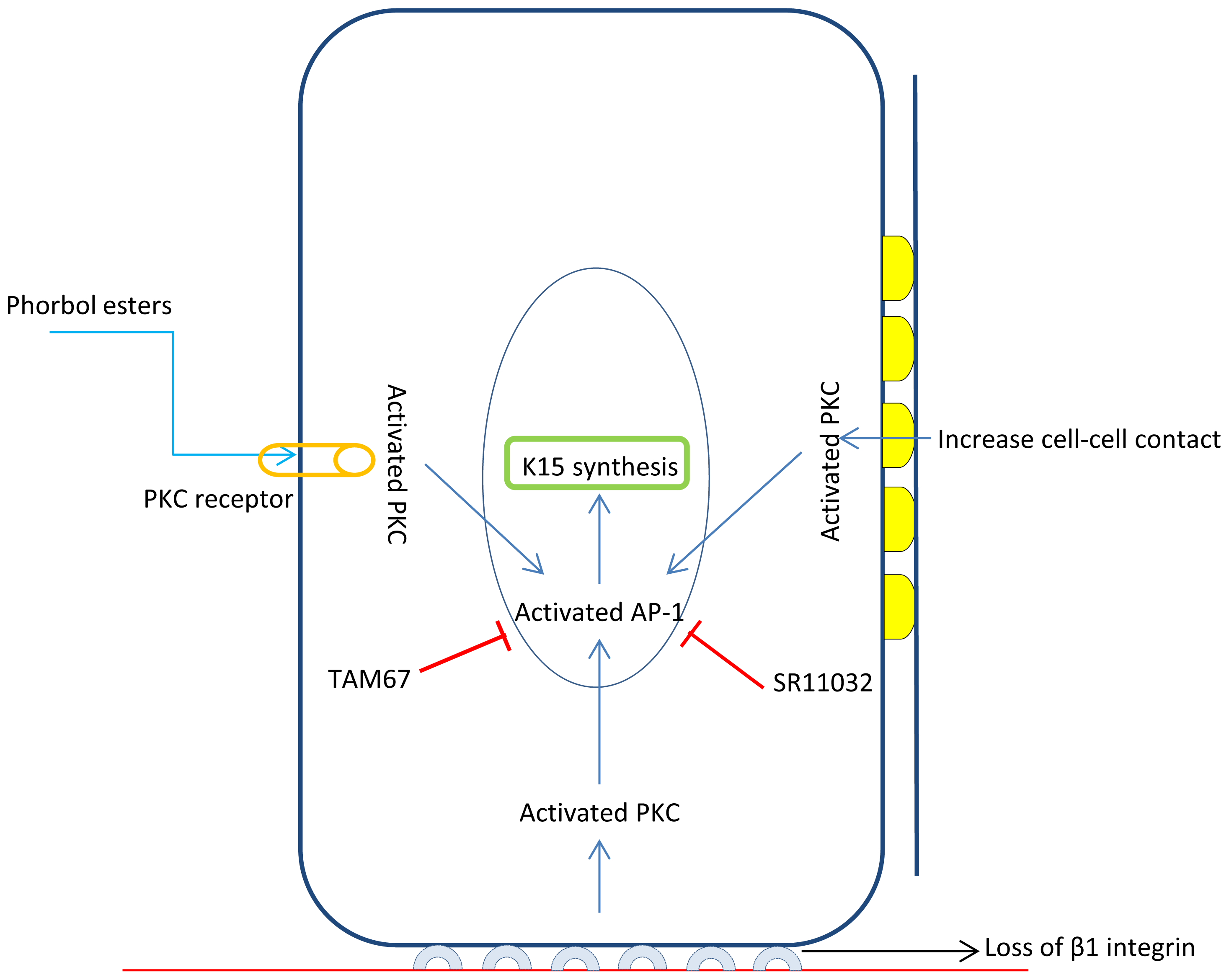
3.1. Functions of Keratin K15
3.2. K15 Is a Target of Stem Cell Specific Transcription Factor FOXM1
3.3. Is K15 a Reliable Stem Cell Marker?
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Wobus, A.M.; Boheler, K.R. Embryonic stem cells: Prospects for developmental biology and cell therapy. Physiol. Rev 2005, 85, 635–678. [Google Scholar]
- Hall, P.A.; Watt, F.M. Stem cells: The generation and maintenance of cellular diversity. Development 1989, 106, 619–633. [Google Scholar]
- Clayton, E.; Doupe, D.P.; Klein, A.M.; Winton, D.J.; Simons, B.D.; Jones, P.H. A single type of progenitor cell maintains normal epidermis. Nature 2007, 446, 185–189. [Google Scholar]
- Mascre, G.; Dekoninck, S.; Drogat, B.; Youssef, K.K.; Brohee, S.; Sotiropoulou, P.A.; Simons, B.D.; Blanpain, C. Distinct contribution of stem and progenitor cells to epidermal maintenance. Nature 2012, 489, 257–262. [Google Scholar]
- Tang, X.H.; Scognamiglio, T.; Gudas, L.J. Basal stem cells contribute to squamous cell carcinomas in the oral cavity. Carcinogenesis 2013, 34, 1158–1164. [Google Scholar]
- Lyle, S.; Christofidou-Solomidou, M.; Liu, Y.; Elder, D.E.; Albelda, S.; Cotsarelis, G. The C8/144B monoclonal antibody recognizes cytokeratin 15 and defines the location of human hair follicle stem cells. J. Cell Sci 1998, 111, 3179–3188. [Google Scholar]
- Ma, D.R.; Yang, E.N.; Lee, S.T. A review: The location, molecular characterisation and multipotency of hair follicle epidermal stem cells. Ann. Acad. Med. Singap 2004, 33, 784–788. [Google Scholar]
- Inoue, K.; Aoi, N.; Sato, T.; Yamauchi, Y.; Suga, H.; Eto, H.; Kato, H.; Araki, J.; Yoshimura, K. Differential expression of stem-cell-associated markers in human hair follicle epithelial cells. Lab. Invest 2009, 89, 844–856. [Google Scholar]
- Jih, D.M.; Lyle, S.; Elenitsas, R.; Elder, D.E.; Cotsarelis, G. Cytokeratin 15 expression in trichoepitheliomas and a subset of basal cell carcinomas suggests they originate from hair follicle stem cells. J. Cutan. Pathol 1999, 26, 113–118. [Google Scholar]
- Mackenzie, J.C. Ordered structure of the stratum corneum of mammalian skin. Nature 1969, 222, 881–882. [Google Scholar]
- Mackenzie, I.C. Relationship between mitosis and the ordered structure of the stratum corneum in mouse epidermis. Nature 1970, 226, 653–655. [Google Scholar]
- Potten, C.S. Epidermal cell production rates. J. Invest. Dermatol 1975, 65, 488–500. [Google Scholar]
- Lajtha, L.G. Stem cell concepts. Nouvelle Revue Francaise D’Hematologie 1979, 21, 59–65. [Google Scholar]
- Bickenbach, J.R. Identification and behavior of label-retaining cells in oral mucosa and skin. J. Dent. Res 1981, 60, 1611–1120. [Google Scholar]
- Bickenbach, J.R.; McCutecheon, J.; Mackenzie, I.C. Rate of loss of tritiated thymidine label in basal cells in mouse epithelial tissues. Cell Tissue Kinet 1986, 19, 325–333. [Google Scholar]
- Cotsarelis, G.; Sun, T.T.; Lavker, R.M. Label-retaining cells reside in the bulge area of pilosebaceous unit: Implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 1990, 61, 1329–1337. [Google Scholar]
- Ghadially, R. 25 years of epidermal stem cell research. J. Invest. Dermatol 2012, 132, 797–810. [Google Scholar]
- Abbas, O.; Bhawan, J. Expression of stem cell markers nestin and cytokeratin 15 and 19 in cutaneous malignancies. J. Eur. Acad. Dermatol. Venereol 2011, 25, 311–316. [Google Scholar]
- Lavker, R.M.; Sun, T.T. Epidermal stem cells: Properties, markers, and location. Proc. Natl. Acad. Sci. USA 2000, 97, 13473–13475. [Google Scholar]
- Watt, F.M. Epidermal stem cells: Markers, patterning and the control of stem cell fate. Philos. Trans. R. Soc. Lond. Ser. B 1998, 353, 831–837. [Google Scholar]
- Webb, A.; Li, A.; Kaur, P. Location and phenotype of human adult keratinocyte stem cells of the skin. Differentiation 2004, 72, 387–395. [Google Scholar]
- Trempus, C.S.; Morris, R.J.; Bortner, C.D.; Cotsarelis, G.; Faircloth, R.S.; Reece, J.M.; Tennant, R.W. Enrichment for living murine keratinocytes from the hair follicle bulge with the cell surface marker CD34. J. Invest. Dermatol 2003, 120, 501–511. [Google Scholar]
- Jaks, V.; Barker, N.; Kasper, M.; van Es, J.H.; Snippert, H.J.; Clevers, H.; Toftgard, R. Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat. Genet 2008, 40, 1291–1299. [Google Scholar]
- Snippert, H.J.; Haegebarth, A.; Kasper, M.; Jaks, V.; van Es, J.H.; Barker, N.; van de Wetering, M.; van den Born, M.; Begthel, H.; Vries, R.G.; et al. Lgr6 marks stem cells in the hair follicle that generate all cell lineages of the skin. Science 2010, 327, 1385–1389. [Google Scholar]
- Patel, II.; Harrison, W.J.; Kerns, J.G.; Filik, J.; Wehbe, K.; Carmichael, P.L.; Scott, A.D.; Philpott, M.P.; Frogley, M.D.; Cinque, G.; et al. Isolating stem cells in the inter-follicular epidermis employing synchrotron radiation-based Fourier-transform infrared microspectroscopy and focal plane array imaging. Anal. Bioanal. Chem 2012, 404, 1745–1758. [Google Scholar]
- Michel, M.; Torok, N.; Godbout, M.J.; Lussier, M.; Gaudreau, P.; Royal, A.; Germain, L. Keratin 19 as a biochemical marker of skin stem cells in vivo and in vitro: Keratin 19 expressing cells are differentially localized in function of anatomic sites, and their number varies with donor age and culture stage. J. Cell Sci 1996, 109, 1017–1028. [Google Scholar]
- Waseem, A.; Dogan, B.; Tidman, N.; Alam, Y.; Purkis, P.; Jackson, S.; Lalli, A.; Machesney, M.; Leigh, I.M. Keratin 15 expression in stratified epithelia: Downregulation in activated keratinocytes. J. Invest. Dermatol 1999, 112, 362–369. [Google Scholar]
- Liu, Y.; Lyle, S.; Yang, Z.; Cotsarelis, G. Keratin 15 promoter targets putative epithelial stem cells in the hair follicle bulge. J. Invest. Dermatol 2003, 121, 963–968. [Google Scholar]
- Morris, R.J.; Liu, Y.; Marles, L.; Yang, Z.; Trempus, C.; Li, S.; Lin, J.S.; Sawicki, J.A.; Cotsarelis, G. Capturing and profiling adult hair follicle stem cells. Nat. Biotechnol 2004, 22, 411–417. [Google Scholar]
- Ohyama, M.; Terunuma, A.; Tock, C.L.; Radonovich, M.F.; Pise-Masison, C.A.; Hopping, S.B.; Brady, J.N.; Udey, M.C.; Vogel, J.C. Characterization and isolation of stem cell-enriched human hair follicle bulge cells. J. Clin. Invest 2006, 116, 249–260. [Google Scholar]
- Garza, L.A.; Yang, C.C.; Zhao, T.; Blatt, H.B.; Lee, M.; He, H.; Stanton, D.C.; Carrasco, L.; Spiegel, J.H.; Tobias, J.W.; et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Invest 2011, 121, 613–622. [Google Scholar]
- Lloyd, C.; Yu, Q.C.; Cheng, J.; Turksen, K.; Degenstein, L.; Hutton, E.; Fuchs, E. The basal keratin network of stratified squamous epithelia: Defining K15 function in the absence of K14. J. Cell Biol 1995, 129, 1329–1344. [Google Scholar]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol 2008, 129, 705–733. [Google Scholar]
- Porter, R.M.; Lunny, D.P.; Ogden, P.H.; Morley, S.M.; McLean, W.H.; Evans, A.; Harrison, D.L.; Rugg, E.L.; Lane, E.B. K15 expression implies lateral differentiation within stratified epithelial basal cells. Lab. Invest 2000, 80, 1701–1710. [Google Scholar]
- Zhan, Q.; Signoretti, S.; Whitaker-Menezes, D.; Friedman, T.M.; Korngold, R.; Murphy, G.F. Cytokeratin15-positive basal epithelial cells targeted in graft-versus-host disease express a constitutive antiapoptotic phenotype. J. Invest. Dermatol 2007, 127, 106–115. [Google Scholar]
- Kose, O.; Lalli, A.; Kutulola, A.O.; Odell, E.W.; Waseem, A. Changes in the expression of stem cell markers in oral lichen planus and hyperkeratotic lesions. J. Oral Sci 2007, 49, 133–139. [Google Scholar]
- Pontiggia, L.; Biedermann, T.; Meuli, M.; Widmer, D.; Bottcher-Haberzeth, S.; Schiestl, C.; Schneider, J.; Braziulis, E.; Montano, I.; Meuli-Simmen, C.; et al. Markers to evaluate the quality and self-renewing potential of engineered human skin substitutes in vitro and after transplantation. J. Invest. Dermatol 2009, 129, 480–490. [Google Scholar]
- Leube, R.E.; Bader, B.L.; Bosch, F.X.; Zimbelmann, R.; Achtstaetter, T.; Franke, W.W. Molecular characterization and expression of the stratification-related cytokeratins 4 and 15. J. Cell Biol 1988, 106, 1249–1261. [Google Scholar]
- Bloor, B.K.; Seddon, S.V.; Morgan, P.R. Gene expression of differentiation-specific keratins (K4, K13, K1 and K10) in oral non-dysplastic keratoses and lichen planus. J. Oral Pathol. Med 2000, 29, 376–384. [Google Scholar]
- Coulombe, P.A.; Kerns, M.L.; Fuchs, E. Epidermolysis bullosa simplex: A paradigm for disorders of tissue fragility. J. Clin. Invest 2009, 119, 1784–1793. [Google Scholar]
- Rugg, E.L.; Leigh, I.M. The keratins and their disorders. Am. J. Med. Genet. Part C 2004, 131C, 4–11. [Google Scholar]
- Rugg, E.L.; McLean, W.H.; Lane, E.B.; Pitera, R.; McMillan, J.R.; Dopping-Hepenstal, P.J.; Navsaria, H.A.; Leigh, I.M.; Eady, R.A. A functional “knockout” of human keratin 14. Genes Dev 1994, 8, 2563–2573. [Google Scholar]
- Peters, B.; Kirfel, J.; Bussow, H.; Vidal, M.; Magin, T.M. Complete cytolysis and neonatal lethality in keratin 5 knockout mice reveal its fundamental role in skin integrity and in epidermolysis bullosa simplex. Mol. Biol. Cell 2001, 12, 1775–1789. [Google Scholar]
- Jonkman, M.F.; Heeres, K.; Pas, H.H.; van Luyn, M.J.; Elema, J.D.; Corden, L.D.; Smith, F.J.; McLean, W.H.; Ramaekers, F.C.; Burton, M.; et al. Effects of keratin 14 ablation on the clinical and cellular phenotype in a kindred with recessive epidermolysis bullosa simplex. J. Invest. Dermatol 1996, 107, 764–769. [Google Scholar]
- Bieniek, R.; Lazar, A.J.; Photopoulos, C.; Lyle, S. Sebaceous tumours contain a subpopulation of cells expressing the keratin 15 stem cell marker. Br. J. Dermatol 2007, 156, 378–380. [Google Scholar]
- Morris, R.J. Keratinocyte stem cells: Targets for cutaneous carcinogens. J. Clin. Invest 2000, 106, 3–8. [Google Scholar]
- Choi, C.W.; Park, H.S.; Kim, Y.K.; Lee, S.H.; Cho, K.H. Elastic fiber staining and cytokeratin 15 expression pattern in trichoepithelioma and basal cell carcinoma. J. Dermatol 2008, 35, 499–502. [Google Scholar]
- Werner, S.; Munz, B. Suppression of keratin 15 expression by transforming growth factor beta in vitro and by cutaneous injury in vivo. Exp. Cell Res 2000, 254, 80–90. [Google Scholar]
- Geiger, T.; Sabanay, H.; Kravchenko-Balasha, N.; Geiger, B.; Levitzki, A. Anomalous features of EMT during keratinocyte transformation. PLoS One 2008, 3, e1574. [Google Scholar]
- Biddle, A.; Liang, X.; Gammon, L.; Fazil, B.; Harper, L.J.; Emich, H.; Costea, D.E.; Mackenzie, I.C. Cancer stem cells in squamous cell carcinoma switch between two distinct phenotypes that are preferentially migratory or proliferative. Cancer Res 2011, 71, 5317–5326. [Google Scholar]
- Sakamoto, K.; Aragaki, T.; Morita, K.; Kawachi, H.; Kayamori, K.; Nakanishi, S.; Omura, K.; Miki, Y.; Okada, N.; Katsube, K.; et al. Down-regulation of keratin 4 and keratin 13 expression in oral squamous cell carcinoma and epithelial dysplasia: A clue for histopathogenesis. Histopathology 2011, 58, 531–542. [Google Scholar]
- Troy, T.C.; Arabzadeh, A.; Turksen, K. Re-assessing K15 as an epidermal stem cell marker. Stem Cell Rev 2011, 7, 927–934. [Google Scholar]
- Jackson, B.C.; Carpenter, C.; Nebert, D.W.; Vasiliou, V. Update of human and mouse forkhead box (FOX) gene families. Hum. Genomics 2010, 4, 345–352. [Google Scholar]
- Myatt, S.S.; Lam, E.W. The emerging roles of forkhead box (Fox) proteins in cancer. Nat. Rev. Cancer 2007, 7, 847–859. [Google Scholar]
- Wierstra, I.; Alves, J. FOXM1, a typical proliferation-associated transcription factor. Biol. Chem 2007, 388, 1257–1274. [Google Scholar]
- Xie, Z.; Tan, G.; Ding, M.; Dong, D.; Chen, T.; Meng, X.; Huang, X.; Tan, Y. Foxm1 transcription factor is required for maintenance of pluripotency of P19 embryonal carcinoma cells. Nucleic Acids Res 2010, 38, 8027–8038. [Google Scholar]
- Tompkins, D.H.; Besnard, V.; Lange, A.W.; Keiser, A.R.; Wert, S.E.; Bruno, M.D.; Whitsett, J.A. Sox2 activates cell proliferation and differentiation in the respiratory epithelium. Am. J. Respir. Cell Mol. Biol 2011, 45, 101–110. [Google Scholar]
- Wang, Z.; Park, H.J.; Carr, J.R.; Chen, Y.J.; Zheng, Y.; Li, J.; Tyner, A.L.; Costa, R.H.; Bagchi, S.; Raychaudhuri, P. FoxM1 in tumorigenicity of the neuroblastoma cells and renewal of the neural progenitors. Cancer Res 2011, 71, 4292–4302. [Google Scholar]
- Carr, J.R.; Kiefer, M.M.; Park, H.J.; Li, J.; Wang, Z.; Fontanarosa, J.; DeWaal, D.; Kopanja, D.; Benevolenskaya, E.V.; Guzman, G.; et al. FoxM1 regulates mammary luminal cell fate. Cell Rep 2012, 1, 715–729. [Google Scholar]
- Gemenetzidis, E.; Elena-Costea, D.; Parkinson, E.K.; Waseem, A.; Wan, H.; Teh, M.T. Induction of human epithelial stem/progenitor expansion by FOXM1. Cancer Res 2010, 70, 9515–9526. [Google Scholar]
- Bose, A.; Teh, M.T.; Hutchison, I.L.; Wan, H.; Leigh, I.M.; Waseem, A. Two mechanisms regulate keratin K15 expression in keratinocytes: Role of PKC/AP-1 and FOXM1 mediated signalling. PLoS One 2012, 7, e38599. [Google Scholar]
- Ghali, L.; Wong, S.T.; Green, J.; Tidman, N.; Quinn, A.G. Gli1 protein is expressed in basal cell carcinomas, outer root sheath keratinocytes and a subpopulation of mesenchymal cells in normal human skin. J. Invest. Dermatol 1999, 113, 595–599. [Google Scholar]
- Teh, M.T.; Wong, S.T.; Neill, G.W.; Ghali, L.R.; Philpott, M.P.; Quinn, A.G. FOXM1 is a downstream target of Gli1 in basal cell carcinomas. Cancer Res 2002, 62, 4773–4780. [Google Scholar]
- Whitbread, L.A.; Powell, B.C. Expression of the intermediate filament keratin gene, K15, in the basal cell layers of epithelia and the hair follicle. Exp. Cell Res 1998, 244, 448–459. [Google Scholar]
- Abbas, O.; Mahalingam, M. Epidermal stem cells: Practical perspectives and potential uses. Br. J. Dermatol 2009, 161, 228–236. [Google Scholar]
- Radoja, N.; Stojadinovic, O.; Waseem, A.; Tomic-Canic, M.; Milisavljevic, V.; Teebor, S.; Blumenberg, M. Thyroid hormones and gamma interferon specifically increase K15 keratin gene transcription. Mol. Cell. Biol 2004, 24, 3168–3179. [Google Scholar]
- Ryle, C.M.; Breitkreutz, D.; Stark, H.J.; Leigh, I.M.; Steinert, P.M.; Roop, D.; Fusenig, N.E. Density-dependent modulation of synthesis of keratins 1 and 10 in the human keratinocyte line HACAT and in ras-transfected tumorigenic clones. Differentiation 1989, 40, 42–54. [Google Scholar]
- Keratin, type I cytoskeletal 15. Available online: http://www.nextprot.org/db/entry/NX_P19012/expression#a-ts-line-320 (accessed on 12 September 2013).
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bose, A.; Teh, M.-T.; Mackenzie, I.C.; Waseem, A. Keratin K15 as a Biomarker of Epidermal Stem Cells. Int. J. Mol. Sci. 2013, 14, 19385-19398. https://doi.org/10.3390/ijms141019385
Bose A, Teh M-T, Mackenzie IC, Waseem A. Keratin K15 as a Biomarker of Epidermal Stem Cells. International Journal of Molecular Sciences. 2013; 14(10):19385-19398. https://doi.org/10.3390/ijms141019385
Chicago/Turabian StyleBose, Amrita, Muy-Teck Teh, Ian C. Mackenzie, and Ahmad Waseem. 2013. "Keratin K15 as a Biomarker of Epidermal Stem Cells" International Journal of Molecular Sciences 14, no. 10: 19385-19398. https://doi.org/10.3390/ijms141019385
APA StyleBose, A., Teh, M.-T., Mackenzie, I. C., & Waseem, A. (2013). Keratin K15 as a Biomarker of Epidermal Stem Cells. International Journal of Molecular Sciences, 14(10), 19385-19398. https://doi.org/10.3390/ijms141019385




