TNF-α Gene Knockout in Triple Negative Breast Cancer Cell Line Induces Apoptosis
Abstract
:1. Introduction
2. Results and Discussion
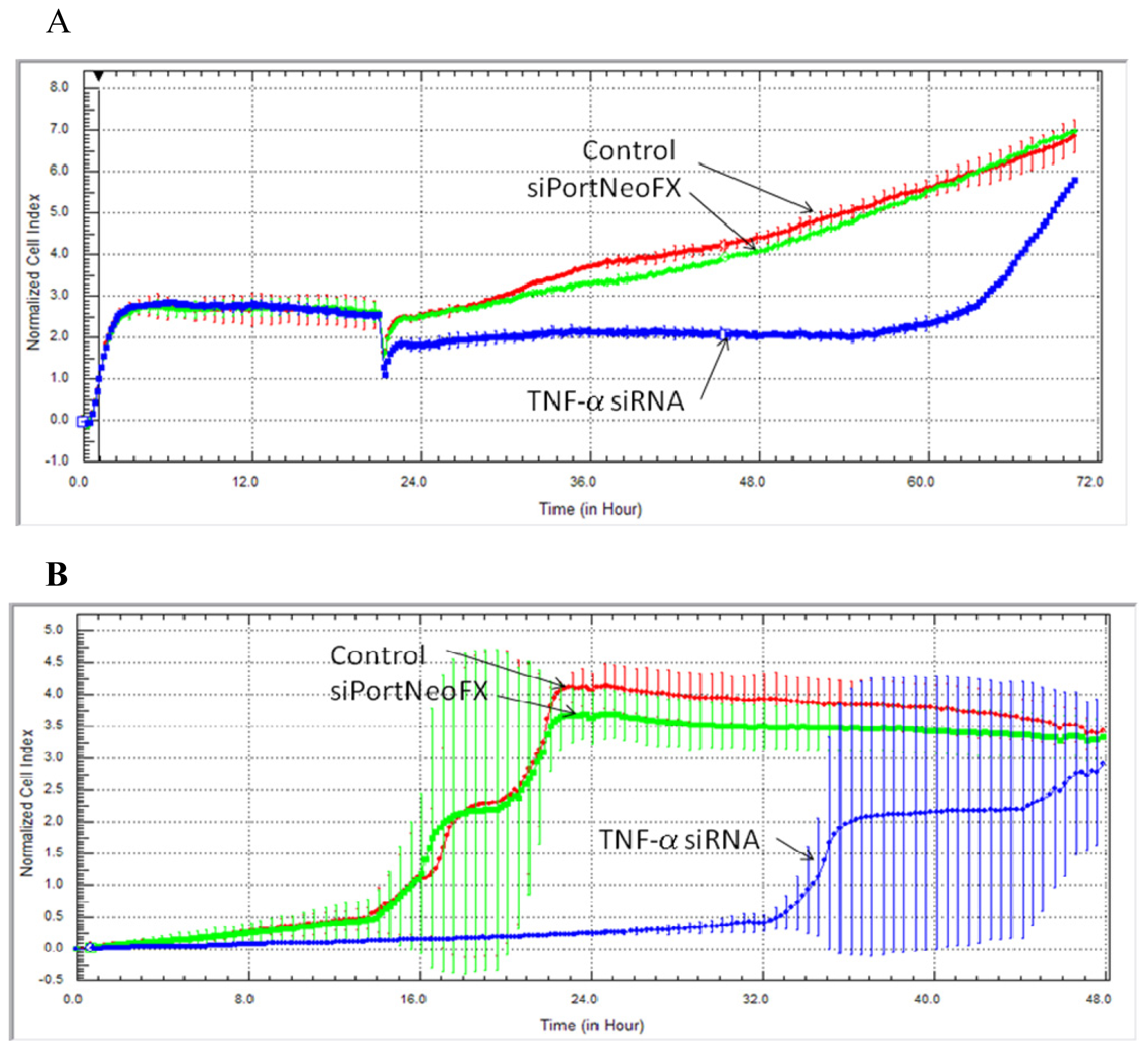
2.1. Cell Survival, Proliferation and Migration after Treatment with siRNA-TNF-α
2.2. Apoptosis/Necrosis in the Hs578T Cell Line after Blocking the Expression of TNF-α
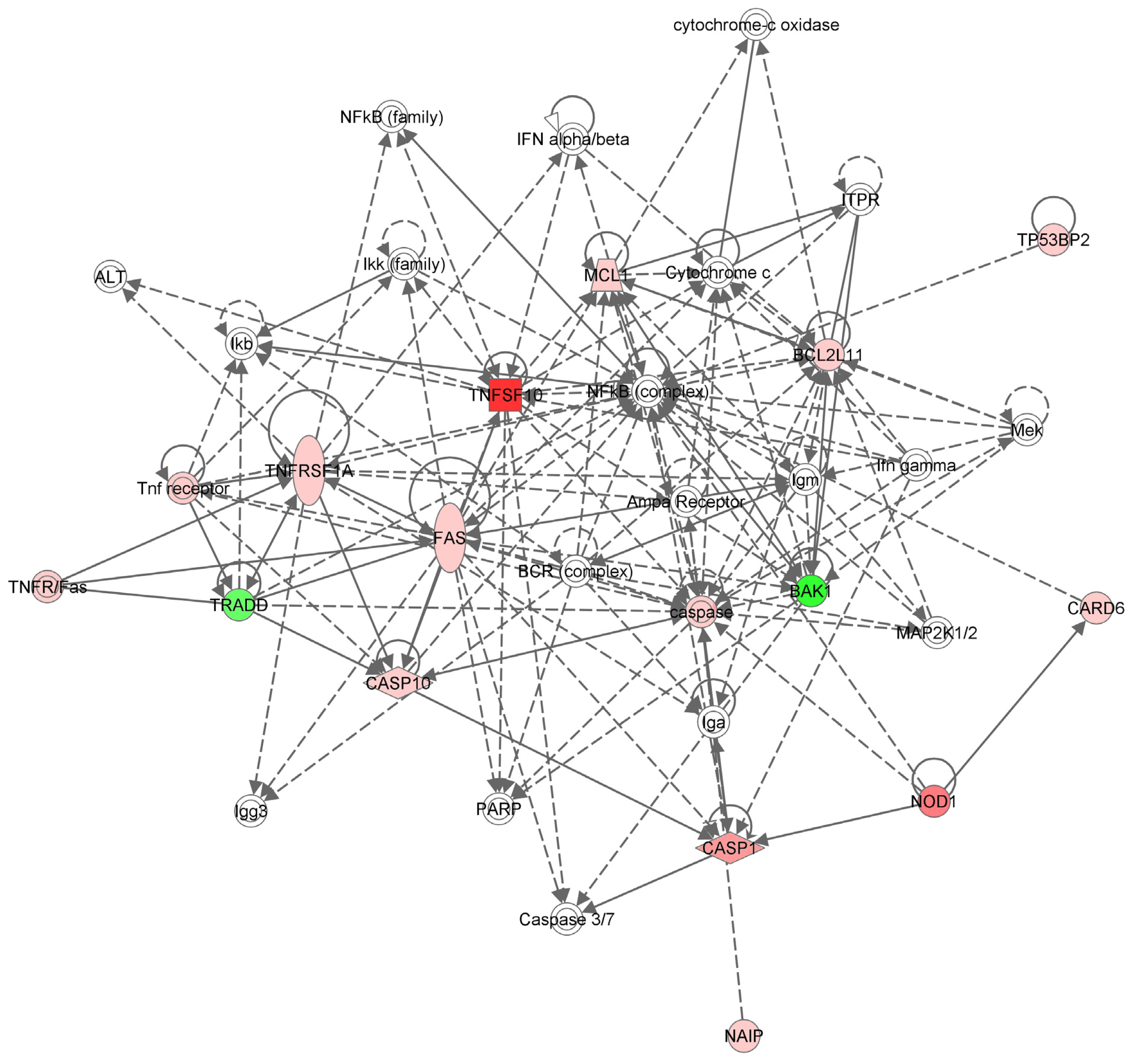
2.3. Apoptotic Gene Expression Profile in the Hs578T Cell Line after TNF-α Gene Inhibition
3. Experimental Section
3.1. Cell Culture and Treatment
3.2. Dynamic Monitoring of Hs578T Cell Proliferation and Cell Migration Using the xCELLigence RTCA System
3.3. Apoptosis by Flow Cytometry
3.4. Gene Evaluation with RT2Profiler™ PCRarray Technology
3.5. Network Analysis
4. Conclusions
Supplementary Information
ijms-14-00411-s001.pdfAcknowledgments
- Conflict of InterestThe authors have no conflict of interest to declare.
References
- Takeshita, F.; Ochiya, T.; Zhang, L.; Hao, C.; Dong, G.; Tong, Z. Analysis of clinical features and outcome of 356 triple-negative breast cancer patients in China. Breast Care 2012, 7, 13–17. [Google Scholar]
- Griffiths, C.L.; Olin, J.L. Triple negative breast cancer: A brief review of its characteristics and treatment options. J. Pharm. Pract 2012, 25, 319–323. [Google Scholar]
- Ossovskaya, V.; Wang, Y.; Budoff, A.; Xu, Q.; Lituev, A.; Potapova, O.; Vansant, G.; Monforte, J.; Daraselia, N. Exploring molecular pathways of triple-negative breast cancer. Genes Cancer 2011, 2, 870–879. [Google Scholar]
- Brown, E.R.; Charles, K.A.; Hoare, S.A.; Rye, R.L.; Jodrell, D.I.; Aird, R.E.; Vora, R.; Prabhakar, U.; Nakada, M.; Corringham, R.E.; et al. A clinical study assessing the tolerability and biological effects of infliximab, a TNF-a inhibitor, in patients with advanced cancer. Ann. Oncol 2008, 19, 1340–1346. [Google Scholar]
- Balkwill, F. TNF-α in promotion and progression of cancer. Cancer Metastasis Rev 2006, 25, 409–416. [Google Scholar]
- Madhusudan, S.; Foster, M.; Muthuramalingam, S.R.; Braybrooke, J.P.; Wilner, S.; Kaur, K.; Han, C.; Hoare, S.; Balkwill, F.; Talbot, D.C.; et al. A phase II study of Etanercept (Enbrel), a tumor necrosis factor inhibitor in patients with metastatic breast cancer. Clin. Cancer Res 2004, 10, 6528–6534. [Google Scholar]
- Hwang, J.R.; Jo, K.; Lee, Y.; Sung, J.B.; Park, Y.W.; Lee, J.H. Upregulation of CD9 in ovarian cancer is related to the induction of TNF-a gene expression and constitutive NF-κB activation. Carcinogenesis 2012, 33, 77–83. [Google Scholar]
- Kulbe, H.; Thompson, R.; Wilson, J.L.; Robinson, S.; Hagemann, T.; Fatah, R.; Gould, D.; Ayhan, A.; Balkwill, F. The inflammatory cytokine tumor necrosis factor-α generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Res 2007, 67, 585–592. [Google Scholar]
- He, S.; Zhang, D.; Cheng, F.; Gong, F.; Guo, Y. Applications of RNA interference in cancer therapeutics as a powerful tool for suppressing gene expression. Mol. Biol. Rep 2009, 36, 2153–2163. [Google Scholar]
- Cuccato, G.; Polynikis, A.; Siciliano, V.; Graziano, M.; di Bernardo, M.; di Bernardo, D. Modeling RNA interference in mammalian cells. BMC Syst. Biol 2011, 5, 19. [Google Scholar]
- Chen, S.H.; Zhaori, G. Potential clinical applications of siRNA technique: Benefits and limitations. Eur. J. Clin. Invest 2010, 41, 221–232. [Google Scholar]
- Lares, M.R.; Rossi, J.J.; Ouellet, D.L. RNAi and small interfering RNAs in human disease therapeutic applications. Trends Biotechnol 2010, 28, 570–579. [Google Scholar]
- Grimm, D.; Kay, M.A. Therapeutic application of RNAi: Is mRNA targeting finally ready for prime time? J. Clin. Invest 2007, 117, 3633–3641. [Google Scholar]
- Simmons, O.; Maples, P.B.; Senzer, N.; Nemunaitis, J. Ewing’s Sarcoma: Development of RNA interference-based therapy for advanced disease. ISRN Oncol 2012, 2012, 247657. [Google Scholar]
- Aagaard, L.; Rossi, J.J. RNAi Therapeutics: Principles, prospects and challenges. Adv. Drug Deliv. Rev 2007, 59, 75–86. [Google Scholar]
- Takeshita, F.; Ochiya, T. Therapeutic potential of RNA interference against cancer. Cancer Sci 2006, 97, 689–696. [Google Scholar]
- Petrova, N.S.; Chernikov, I.V.; Meschaninova, M.I.; Dovydenko, I.S.; Venyaminova, A.G.; Zenkova, M.A.; Vlassov, V.V.; Chernolovskaya, E.L. Carrier-free cellular uptake and the gene-silencing activity of the lipophilic siRNAs is strongly affected by the length of the linker between siRNA and lipophilic group. Nucleic Acids Res 2012, 40, 2330–2344. [Google Scholar]
- Zhang, J.D.; Koerner, C.; Bechtel, S.; Bender, C.; Keklikoglou, I.; Schmidt, C.; Irsigler, A.; Ernst, U.; Sahin, O.; Wiemann, S.; et al. Time-resolved human kinome RNAi screen identifies a network regulating mitotic-events as early regulators of cell proliferation. PLoS One 2011, 6, e22176. [Google Scholar]
- Kumar, P.S.; Shiras, A.; Das, G.; Jagtap, J.C.; Prasad, V.; Shastry, P. Differential expression and role of p21cip/waf1 and p27kip1 in TNF-α-induced inhibition of proliferation in human glioma cells. Mol. Cancer 2007, 6, 42. [Google Scholar]
- Hagemann, T.; Wilson, J.; Kulbe, H. TNF-α dependent increased c-Jun and NF-κB activity in tumour cell lines upon co-cultivation with macrophages. J. Immunol 2005, 175, 1197–1205. [Google Scholar]
- Wu, Y.; Zhou, B.P. TNF-a/NF-kB/Snail pathway in cancer cell migration and invasion. Br. J. Cancer 2010, 102, 639–644. [Google Scholar]
- Szlosarek, P.W.; Grimshaw, M.J.; Kulbe, H.; Wilson, J.L.; Wilbanks, G.D.; Burke, F.; Balkwill, F.R. Expression and regulation of tumor necrosis factor alpha in normal and malignant ovarian epithelium. Mol. Cancer Ther 2006, 5, 382–390. [Google Scholar]
- Szlosarek, P.; Charles, K.A.; Balkwill, F.R. Tumour necrosis factor-α as a tumour promoter. Eur. J. Cancer 2006, 42, 745–750. [Google Scholar]
- Egberts, J.H.; Cloosters, V.; Noack, A.; Schniewind, B.; Thon, L.; Klose, S.; Kettler, B.; von Forstner, C.; Kneitz, C.; Tepel, J.; et al. Anti-tumor necrosis factor therapy inhibits pancreatic tumor growth and metastasis. Cancer Res 2008, 68, 1443–1450. [Google Scholar]
- Setlur, S.R.; Royce, T.E.; Sboner, A.; Mosquera, J.M.; Demichelis, F.; Hofer, M.D.; Mertz, K.D.; Gerstein, M.; Rubin, M.A. Integrative microarray analysis of pathways dysregulated in metastatic prostate cancer. Cancer Res 2007, 67, 10296–10303. [Google Scholar]
- Lessard, L.; Karakiewicz, P.I.; Bellon-Gagnon, P.; Alam-Fahmy, M.; Ismail, H.A.; Mes-Masson, A.M.; Saad, F. Nuclear localization of nuclear factor-κB p65 in primary prostate tumors is highly predictive of pelvic lymph node metastases. Clin. Cancer Res 2006, 12, 5741–5745. [Google Scholar]
- Sprowl, J.A.; Reed, K.; Armstrong, S.R.; Lanner, C.; Guo, B.; Kalatskaya, I.; Stein, L.; Hembruff, S.L.; Tam, A.; Parissenti, A.M. Alterations in tumor necrosis factor signaling pathways are associated with cytotoxicity and resistance to taxanes: A study in isogenic resistant tumor cells. Breast Cancer Res 2012, 14, R2. [Google Scholar]
- Soares, R.; Meireles, M.; Rocha, A.; Pirraco, A.; Obiol, D.; Alonso, E.; Joos, G.; Balogh, G. Maitake (D Fraction) mushroom extract induces apoptosis in breast cancer cells by BAK-1 gene activation. J. Med. Food 2011, 14, 563–572. [Google Scholar]
- Chittenden, T.; Harrington, E.A.; O’Connor, R.; Flemington, C.; Lutz, R.J.; Evan, G.I.; Guild, B.C. Induction of apoptosis by the Bcl-2 homologue Bak. Nature 1995, 374, 733–736. [Google Scholar]
- Montagut, C.; Tusquets, I.; Ferrer, B.; Corominas, J.M.; Bellosillo, B.; Campas, C.; Suarez, M.; Fabregat, X.; Campo, E.; Gascon, P.; et al. Activation of nuclear factor-κB is linked to resistance to neoadjuvant chemotherapy in breast cancer patients. Endocr. Relat. Cancer 2006, 13, 607–616. [Google Scholar]
- Lee, H.Y.; Youn, S.W.; Kim, J.Y.; Park, K.W.; Hwang, C.I.; Park, W.Y.; Oh, B.H.; Park, Y.B.; Walsh, K.; Seo, J.S.; et al. FOXO3a turns the tumor necrosis factor receptor signaling towards apoptosis through reciprocal regulation of c-Jun N-Terminal Kinase and NF-κB. Arterioscler. Thromb. Vasc. Biol 2008, 28, 112–120. [Google Scholar]

| Gene | Gene symbol | Fold regulation | p-Value |
|---|---|---|---|
| tumor necrosis factor (ligand) superfamily, member 10 | TNFSF10 | 4.6203 | 0.000025 |
| nucleotide-binding oligomerization domain containing 1 | NOD1 | 3.2221 | 0.01072 |
| caspase 1, apoptosis-related cysteine peptidase | CASP1 | 1.8851 | 0.007098 |
| Fas (TNF receptor superfamily, member 6) | FAS | 1.7187 | 0.0127 |
| tumor protein p53 binding protein, 2 | TP53BP2 | 1.6911 | 0.013546 |
| NLR family, apoptosis inhibitory protein | NAIP | 1.6756 | 0.01393 |
| myeloid cell leukemia sequence 1 (BCL2-related) | MCL1 | 1.6073 | 0.004842 |
| BCL2-like 11 (apoptosis facilitator) | BCL2L11 | 1.5526 | 0.011312 |
| tumor necrosis factor receptor superfamily, member 1A | TNFRSF1A | 1.4893 | 0.008169 |
| caspase recruitment domain family, member 6 | CARD6 | 1.38 | 0.003481 |
| caspase 10, apoptosis-related cysteine peptidase | CASP10 | 1.3485 | 0.028438 |
| v-raf murine sarcoma viral oncogene homolog B1 | BRAF | 1.2056 | 0.035962 |
| BCL2-antagonist/killer 1 | BAK1 | −2.1967 | 0.012989 |
| tumor necrosis factor | TNF | −1.7719 | 0.015681 |
| TNFRSF1A-associated via death domain | TRADD | −1.346 | 0.043775 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pileczki, V.; Braicu, C.; Gherman, C.D.; Berindan-Neagoe, I. TNF-α Gene Knockout in Triple Negative Breast Cancer Cell Line Induces Apoptosis. Int. J. Mol. Sci. 2013, 14, 411-420. https://doi.org/10.3390/ijms14010411
Pileczki V, Braicu C, Gherman CD, Berindan-Neagoe I. TNF-α Gene Knockout in Triple Negative Breast Cancer Cell Line Induces Apoptosis. International Journal of Molecular Sciences. 2013; 14(1):411-420. https://doi.org/10.3390/ijms14010411
Chicago/Turabian StylePileczki, Valentina, Cornelia Braicu, Claudia D. Gherman, and Ioana Berindan-Neagoe. 2013. "TNF-α Gene Knockout in Triple Negative Breast Cancer Cell Line Induces Apoptosis" International Journal of Molecular Sciences 14, no. 1: 411-420. https://doi.org/10.3390/ijms14010411




