Evaluation of Anti-Inflammatory Drug-Conjugated Silicon Quantum Dots: Their Cytotoxicity and Biological Effect
Abstract
:1. Introduction
2. Results and Discussion
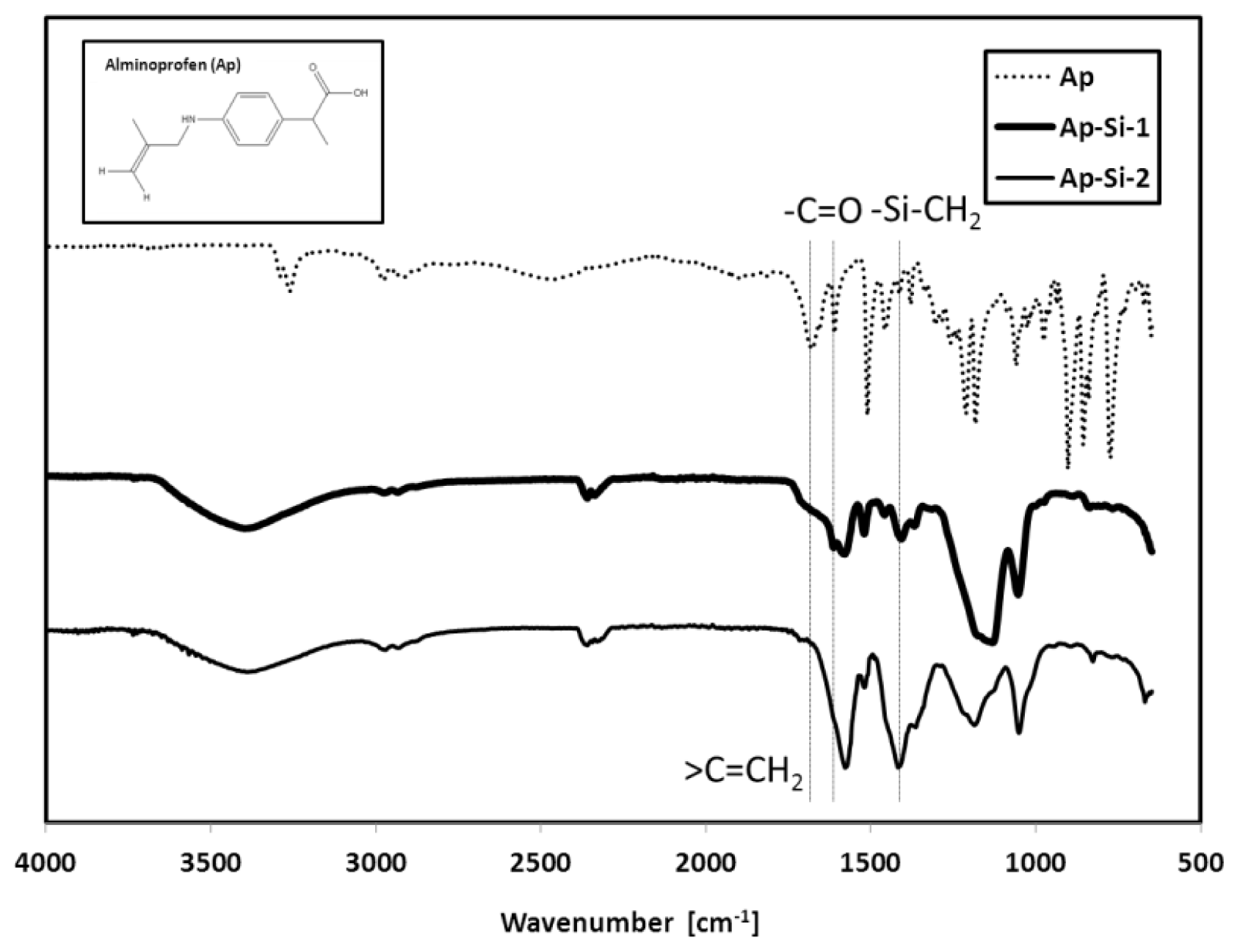
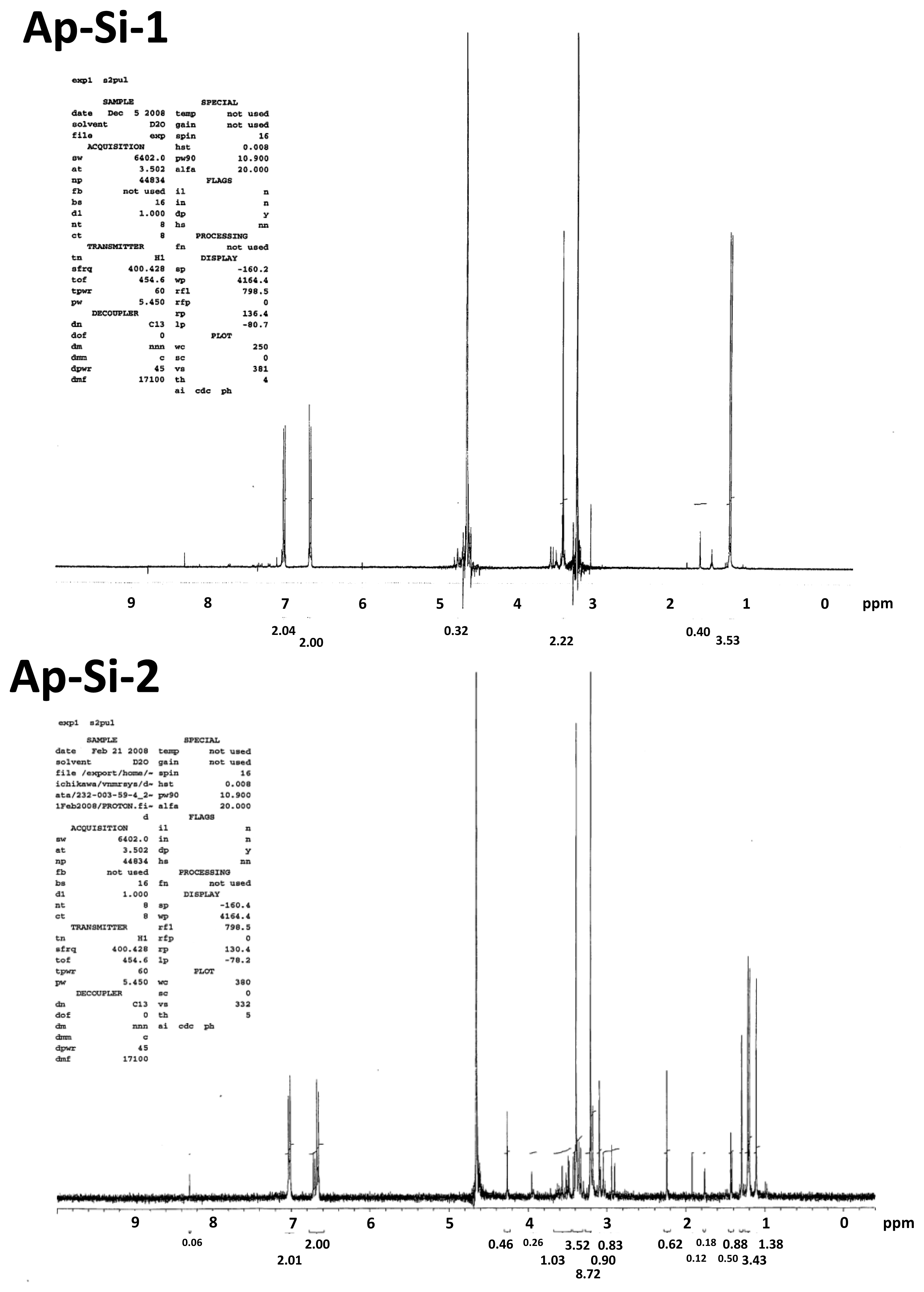
2.1. Characteristics of Si-QDs
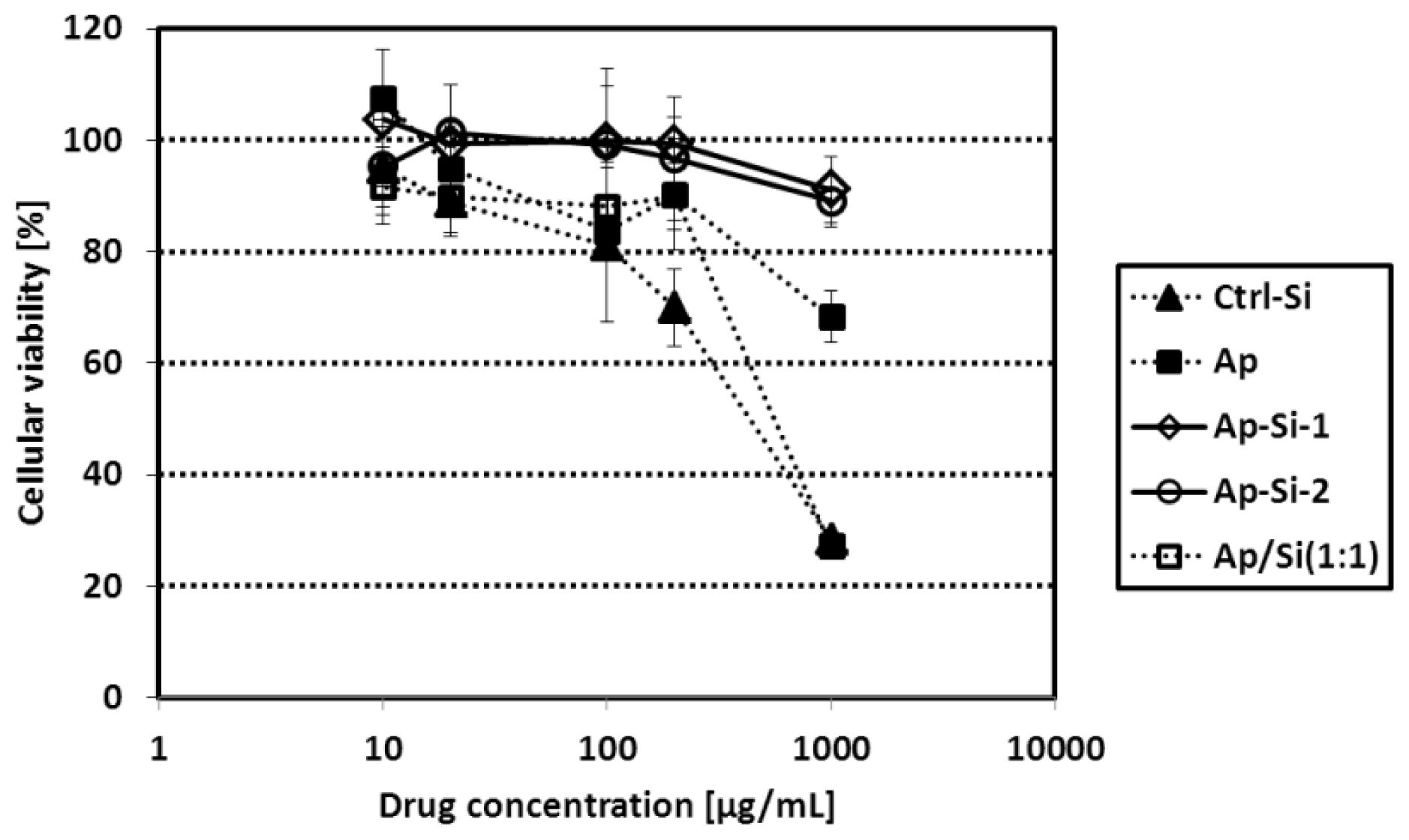
2.2. Cytotoxicity of Synthesized Si-QDs
2.3. In Vitro Effect of Ap-Si QDs
3. Experimental Section
3.1. Synthesis of Si-QDs
3.2. Measurement of Characteristics of Si-QDs
3.3. Cytotoxicity of Drug-Conjugated Si-QDs
3.4. Measurement of Cyclooxygenase-2 (COX-2) Inhibition
3.5. Michaelis-Menten Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar]
- Nirmal, M.; Brus, L.E. Luminescence photophysics in semiconductor nanocrystals. Acc. Chem. Res 1999, 32, 407–414. [Google Scholar]
- Haruta, M. Size- and support-dependency in the catalysis of gold. Catal. Today 1997, 36, 153–166. [Google Scholar]
- Kim, S.; Lim, Y.T.; Soltesz, E.G.; de Grand, A.M.; Lee, J.; Nakayama, A.; Parker, J.A.; Mihaljevic, T.; Laurence, R.G.; Dor, D.M. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat. Biotechnol 2004, 22, 93–97. [Google Scholar]
- Medintz, I.L.; Uyeda, H.T.; Goldman, E.R.; Mattoussi, H. Quantum dot bioconjugates for imaging, labelling and sensing. Nat. Mater 2005, 4, 435–446. [Google Scholar]
- Mattoussi, H.; Mauro, J.M.; Goldman, E.R.; Anderson, G.P.; Sundar, V.C.; Mikulec, F.V.; Bawendi, M.G. Self-assembly of CdSe-ZnS quantum dot bioconjugates using an engineered recombinant protein. J. Am. Chem. Soc 2000, 122, 12142–12150. [Google Scholar]
- Xiao, Y.; Gao, X.; Gannot, G.; Emmert-Buck, M.R.; Srivastava, S.; Wagner, P.D.; Amos, M.D.; Barker, P.E. Quantitation of HER2 and telomerase biomarkers in solid tumors with IgY antibodies and nanocrystal detection. Int. J. Cancer 2008, 122, 2178–2186. [Google Scholar]
- Ghazani, A.A.; Lee, J.A.; Klostranec, J.; Xiang, Q.; Dacosta, R.S.; Wilson, B.C.; Tsao, M.S.; Chan, W.C. High throughput quantification of protein expression of cancer antigens in tissue microarray using quantum dot nanocrystals. Nano Lett 2006, 6, 2881–2886. [Google Scholar]
- Eastman, P.S.; Ruan, W.; Doctolero, M.; Nuttall, R.; de Feo, G.; Park, J.S.; Chu, J.S.; Cooke, P.; Gray, J.W.; Li, S.; et al. Qdot nanobarcodes for multiplexed gene expression analysis. Nano Lett 2006, 6, 1059–1064. [Google Scholar]
- Liang, R.Q.; Li, W.; Li, Y.; Tan, C.Y.; Li, J.X.; Jin, Y.X.; Ruan, K.C. An oligonucleotide microarray for microRNA expression analysis based on labeling RNA with quantum dot and nanogold probe. Nucleic Acids Res 2005, 33, e17. [Google Scholar]
- Lucas, L.J.; Chesler, J.N.; Yoon, J.Y. Lab-on-a-chip immunoassay for multiple antibodies using microsphere light scattering and quantum dot emission. Biosens. Bioelectron 2007, 23, 675–681. [Google Scholar]
- Gao, X.; Cui, Y.; Levenson, R.M.; Chung, L.W.K.; Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol 2004, 22, 969–976. [Google Scholar]
- Wu, X.; Liu, H.; Liu, J.; Haley, K.N.; Treadway, J.A.; Larson, J.P.; Ge, N.; Peale, F.; Bruchez, M.P. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat. Biotechnol. 2003, 21, 41–46. [Google Scholar]
- Sun, J.; Zhu, M.Q.; Fu, K.; Lewinski, N.; Drezek, R.A. Lead sulfide near-infrared quantum dot bioconjugates for targeted molecular imaging. Int. J. Nanomed 2007, 2, 235–240. [Google Scholar]
- Kobayashi, H.; Hama, Y.; Koyama, Y.; Barrett, T.; Regino, C.A.; Urano, Y.; Choyke, P.L. Simultaneous multicolor imaging of five different lymphatic basins using quantum dots. Nano Lett 2007, 7, 1711–1716. [Google Scholar]
- Hoshino, A.; Hanaki, K.; Suzuki, K.; Yamamoto, K. Applications of T-lymphoma labeled with fluorescent quantum dots to cell tracing markers in mouse body. Biochem. Biophys. Res. Commun 2004, 314, 46–53. [Google Scholar]
- Hoshino, A.; Manabe, N.; Fujioka, K.; Suzuki, K.; Yasuhara, M.; Yamamoto, K. Use of fluorescent quantum dot bioconjugates for cellular imaging of immune cells, cell organelle labeling, and nanomedicine: Surface modification regulates biological function, including cytotoxicity. J. Artif. Organs 2007, 10, 149–157. [Google Scholar]
- Manabe, N.; Hoshino, A.; Liang, Y.Q.; Goto, T.; Kato, N.; Yamamoto, K. Quantum dot conjugated with medicine as the drug tracer in vitro and in vivo. IEEE Trans. Nano Biosci 2006, 5, 263–267. [Google Scholar]
- Hoshino, A.; Nagao, T.; Nakasuga, A.; Ishida-Okawara, A.; Suzuki, K.; Yasuhara, M.; Yamamoto, K. Nanocrystal quantum dot-conjugated anti-myeloperoxidase antibody as the detector of activated neutrophils. IEEE Trans. Nano Biosci 2007, 6, 341–345. [Google Scholar]
- Hardman, R. A toxicologic review of quantum dots: Toxicity depends on physicochemical and environmental factors. Environ. Health Perspect 2006, 114, 165–172. [Google Scholar]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 2004, 4, 11–18. [Google Scholar]
- Mahendra, S.; Zhu, H.; Colvin, V.L.; Alvarez, P.J. Quantum dot weathering results in microbial toxicity. Environ. Sci. Technol 2008, 42, 9424–9430. [Google Scholar]
- Choi, H.S.; Liu, W.; Misra, P.; Tanaka, E.; Zimmer, J.P.; Ipe, B.I.; Bawendi, M.G.; Frangioni, J.V. Renal clearance of quantum dots. Nat. Biotechnol 2007, 25, 1165–1170. [Google Scholar]
- Heath, J.R.; Shiang, J.J.; Alivisatos, A.P. Germanium quantum dots: Optical properties and synthesis. J. Chem. Phys 1994, 101, 1607–1615. [Google Scholar]
- Warner, J.H. Solution-phase synthesis of germanium nanoclusters using sulfur. Nanotechnology 2006, 17, 5613–5619. [Google Scholar]
- Prabakar, S.; Shiohara, A.; Hanada, S.; Fujioka, K.; Yamamoto, K.; Tilley, R.D. Size controlled synthesis of germanium nanocrystals with hydride reducing agents and their biological applications. Chem. Mater 2010, 22, 482–486. [Google Scholar]
- Sato, K.; Hirakuri, K. Improved luminescence intensity and stability of nanocrystalline silicon due to the passivation of non-luminescent states. J. Appl. Phys 2005, 97, 104326. [Google Scholar]
- Wang, L.; Reipa, V.; Blasic, J. Silicon nanoparticles as a luminescent label to DNA. Bioconjugate Chem 2004, 15, 409–412. [Google Scholar]
- Warner, J.H.; Hoshino, A.; Yamamoto, K.; Tilley, R.D. Water-soluble photoluminescent silicon quantum dots. Angew. Chem. Int. Ed 2005, 44, 4550–4554. [Google Scholar]
- Shiohara, A.; Hanada, S.; Prabakar, S.; Fujioka, K.; Lim, T.; Yamamoto, K.; Northcote, P.; Tilley, R.D. Chemical reactions on surface molecules attached to silicon quantum dots. J. Am. Chem. Soc. 2010, 132, 248–253. [Google Scholar]
- Neugart, F.; Zappe, A.; Jelezko, F.; Tietz, C.; Boudou, J.P.; Krueger, A.; Wrachtrup, J. Dynamics of diamond nanoparticles in solution and cells. Nano Lett 2007, 7, 3588–3591. [Google Scholar]
- Takagahara, T.; Takeda, K. Theory of the quantum confinement effect on excitons in quantum dots of indirect-gap materials. Phys. Rev. B 1992, 46, 15578–15581. [Google Scholar]
- Nishiguchi, K.; Zhao, X.; Oda, S. Nanocrystalline silicon electron emitter with a high efficiency enhanced by a planarization technique. J. Appl. Phys 2002, 92, 2748–2757. [Google Scholar]
- Allen, T.M.; Cullis, P.R. Drug delivery systems: Entering the mainstream. Science 2004, 303, 1818–1822. [Google Scholar]
- Harada, A.; Kataoka, K. Chain length recognition: Core-shell supramolecular assembly from oppositely charged block copolymers. Science 1999, 283, 65–67. [Google Scholar]
- Kakizawa, Y.; Kataoka, K. Block copolymer micelles for delivery of gene and related compounds. Adv. Drug Delivery Rev 2002, 54, 203–222. [Google Scholar]
- Medina, O.P.; Zhu, Y.; Kairemo, K. Targeted liposomal drug delivery in cancer. Curr. Pharm. Des 2004, 10, 2981–2989. [Google Scholar]
- Pillai, O.; Panchagnula, R. Polymers in drug delivery. Curr. Opin. Chem. Biol 2001, 5, 447–451. [Google Scholar]
- Hoshino, A.; Manabe, N.; Fujioka, K.; Hanada, S.; Yasuhara, M.; Kondo, A.; Yamamoto, K. GFP expression by intracellular gene delivery of GFP-coding fragments using nanocrystal quantum dots. Nanotechnology 2008, 19, 495102. [Google Scholar]
- Willoughby, D.A.; Moore, A.R.; Colville-Nash, P.R. COX-1, COX-2, and COX-3 and the future treatment of chronic inflammatory disease. Lancet 2000, 355, 646–648. [Google Scholar]
- Fujioka, K.; Hiruoka, M.; Sato, K.; Manabe, N.; Miyasaka, R.; Hanada, S.; Hoshino, A.; Tilley, R.D.; Manome, Y.; Hirakuri, K.; et al. Luminescent passive-oxidized silicon quantum dots as biological staining labels and their cytotoxicity effects at high concentration. Nanotechnology 2008, 19, 415102. [Google Scholar]
- Maeda, H.; Wu, J.; Sawa, T.; Matsumura, Y.; Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. J. Control. Release 2000, 65, 271–284. [Google Scholar]
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Adv. Enzyme Reg 2001, 41, 189–207. [Google Scholar]
- Yamada, T.; Iwasaki, Y.; Tada, H.; Iwabuki, H.; Chuah, M.; Vanden Driessche, T.; Fukuda, H.; Kondo, A.; Ueda, M.; Seno, M.; et al. Nanoparticles for the delivery of genes and drugs to human hepatocytes. Nat. Biotechnol 2003, 21, 885–890. [Google Scholar]
- Shiohara, A.; Hoshino, A.; Hanaki, K.; Suzuki, K.; Yamamoto, K. On the cyto-toxicity caused by quantum dots. Microbiol. Immunol 2004, 48, 669–675. [Google Scholar]



| Sample | Particle size (nm) | Weight ratio of Ap | Estimated IC50 (mg/mL) | Calculated Ki |
|---|---|---|---|---|
| Ap-Si-1 | 6.5 | 0.23 | 0.16 | 0.013 |
| Ap-Si-2 | 8.5 | 0.30 | 0.19 | 0.005 |
| Ap | N.D. | N.C. | 0.23 | 22 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hanada, S.; Fujioka, K.; Futamura, Y.; Manabe, N.; Hoshino, A.; Yamamoto, K. Evaluation of Anti-Inflammatory Drug-Conjugated Silicon Quantum Dots: Their Cytotoxicity and Biological Effect. Int. J. Mol. Sci. 2013, 14, 1323-1334. https://doi.org/10.3390/ijms14011323
Hanada S, Fujioka K, Futamura Y, Manabe N, Hoshino A, Yamamoto K. Evaluation of Anti-Inflammatory Drug-Conjugated Silicon Quantum Dots: Their Cytotoxicity and Biological Effect. International Journal of Molecular Sciences. 2013; 14(1):1323-1334. https://doi.org/10.3390/ijms14011323
Chicago/Turabian StyleHanada, Sanshiro, Kouki Fujioka, Yasuhiro Futamura, Noriyoshi Manabe, Akiyoshi Hoshino, and Kenji Yamamoto. 2013. "Evaluation of Anti-Inflammatory Drug-Conjugated Silicon Quantum Dots: Their Cytotoxicity and Biological Effect" International Journal of Molecular Sciences 14, no. 1: 1323-1334. https://doi.org/10.3390/ijms14011323





