Detection of Tumor Cell-Specific mRNA in the Peripheral Blood of Patients with Breast Cancer — Evaluation of Several Markers with Real-Time Reverse Transcription-PCR
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
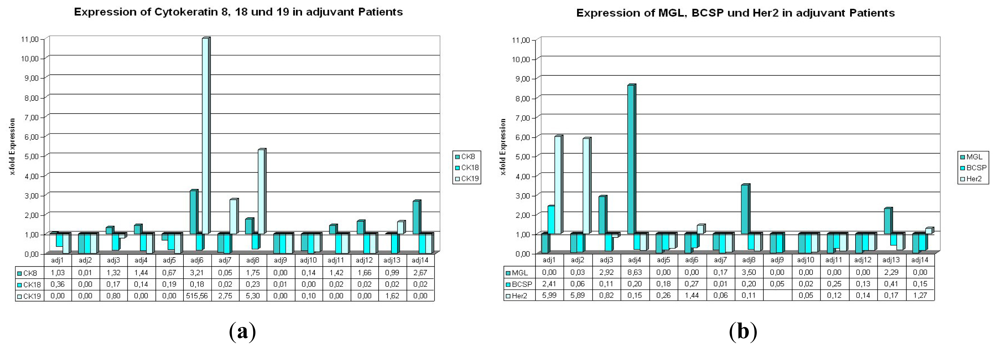
2.1.1. Expression of CK8, 18, and 19 in Patients with Primary Carcinoma Undergoing Adjuvant Therapy
2.1.2. Expression of BCSP, Her2, and MGL in Patients with Primary Carcinoma Undergoing Adjuvant Therapy
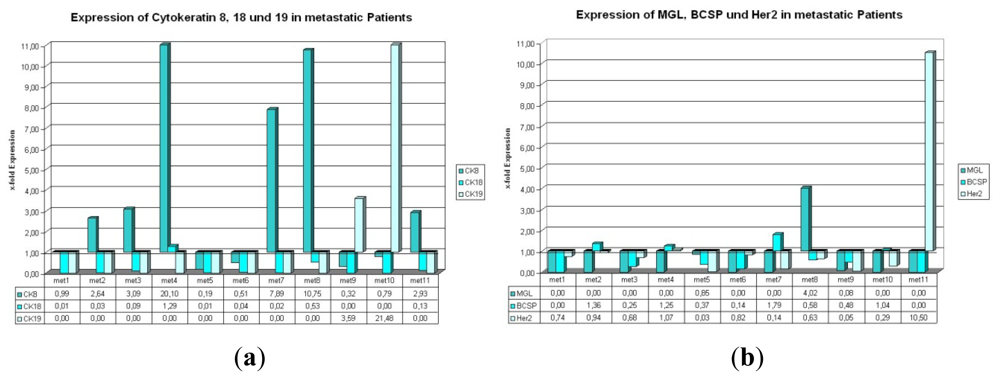
2.1.3. Expression of CK8, 18, and 19 in Metastatic Patients
2.1.4. Expression of BCSP, Her2, and MGL in Metastatic Patients
2.2. Discussion
3. Experimental Section
3.1. Blood Samples
3.2. Ethics Approval
3.3. RNA Isolation
3.4. Reverse Transcription
3.5. Nested PCR
3.6. Real-Time RT-PCR (TaqMan®)
3.7. Evaluation
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Zhang, L.; Zhou, W.; Velculescu, V.E.; Kern, S.E.; Hruban, R.H.; Hamilton, S.R.; Vogelstein, B.; Kinzler, K.W. Gene expression profiles in normal and cancer cells. Science 1997, 276, 1268–1272. [Google Scholar]
- Frasor, J.; Danes, J.M.; Komm, B.; Chang, K.C.; Lyttle, C.R.; Katzenellenbogen, B.S. Profiling of estrogen up- and down-regulated gene expression in human breast cancer cells: Insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinology 2003, 144, 4562–4574. [Google Scholar]
- Hedenfalk, I.; Duggan, D.; Chen, Y.; Radmacher, M.; Bittner, M.; Simon, R.; Meltzer, P.; Gusterson, B.; Esteller, M.; Kallioniemi, O.P.; et al. Gene-expression profiles in hereditary breast cancer. N. Engl. J. Med 2001, 344, 539–548. [Google Scholar]
- Pantel, K.; Brakenhoff, R.H. Dissecting the metastatic cascade. Nat. Rev. Cancer 2004, 4, 448–456. [Google Scholar]
- Ring, A.; Smith, I.E.; Dowsett, M. Circulating tumour cells in breast cancer. Lancet Oncol 2004, 5, 79–88. [Google Scholar]
- Smerage, J.B.; Hayes, D.F. The measurement and therapeutic implications of circulating tumour cells in breast cancer. Br. J. Cancer 2006, 94, 8–12. [Google Scholar]
- Braun, S.; Pantel, K.; Muller, P.; Janni, W.; Hepp, F.; Kentenich, C.R.; Gastroph, S.; Wischnik, A.; Dimpfl, T.; Kindermann, G.; et al. Cytokeratin-positive cells in the bone marrow and survival of patients with stage I, II, or III breast cancer. N. Engl. J. Med 2000, 342, 525–533. [Google Scholar]
- Braun, S.; Vogl, F.D.; Naume, B.; Janni, W.; Osborne, M.P.; Coombes, R.C.; Schlimok, G.; Diel, I.J.; Gerber, B.; Gebauer, G.; et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med 2005, 353, 793–802. [Google Scholar]
- Kurec, A.S.; Baltrucki, L.; Mason, D.Y.; Davey, F.R. Use of the APAAP method in the classification and diagnosis of hematologic disorders. Clin. Lab Med 1988, 8, 223–236. [Google Scholar]
- Noack, F.; Schmitt, M.; Bauer, J.; Helmecke, D.; Kruger, W.; Thorban, S.; Sandherr, M.; Kuhn, W.; Graeff, H.; Harbeck, N. A new approach to phenotyping disseminated tumor cells: Methodological advances and clinical implications. Int. J. Biol. Markers 2000, 15, 100–104. [Google Scholar]
- Fleming, T.P.; Watson, M.A. Mammaglobin, a breast-specific gene, and its utility as a marker for breast cancer. Ann. N. Y. Acad. Sci 2000, 923, 78–89. [Google Scholar]
- Inaba, S.; Li, C.; Shi, Y.E.; Song, D.Q.; Jiang, J.D.; Liu, J. Synuclein gamma inhibits the mitotic checkpoint function and promotes chromosomal instability of breast cancer cells. Breast Cancer Res. Treat 2005, 94, 25–35. [Google Scholar]
- Jiang, Y.; Liu, Y.E.; Goldberg, I.D.; Shi, Y.E. Gamma synuclein, a novel heat-shock protein-associated chaperone, stimulates ligand-dependent estrogen receptor alpha signaling and mammary tumorigenesis. Cancer Res 2004, 64, 4539–4546. [Google Scholar]
- Wu, K.; Quan, Z.; Weng, Z.; Li, F.; Zhang, Y.; Yao, X.; Chen, Y.; Budman, D.; Goldberg, I.D.; Shi, Y.E. Expression of neuronal protein synuclein gamma gene as a novel marker for breast cancer prognosis. Breast Cancer Res. Treat 2007, 101, 259–267. [Google Scholar]
- Wu, K.; Weng, Z.; Tao, Q.; Lin, G.; Wu, X.; Qian, H.; Zhang, Y.; Ding, X.; Jiang, Y.; Shi, Y.E. Stage-specific expression of breast cancer-specific gene gamma-synuclein. Cancer Epidemiol. Biomarkers Prev 2003, 12, 920–925. [Google Scholar]
- Guo, J.; Shou, C.; Meng, L.; Jiang, B.; Dong, B.; Yao, L.; Xie, Y.; Zhang, J.; Chen, Y.; Budman, D.R.; et al. Neuronal protein synuclein gamma predicts poor clinical outcome in breast cancer. Int. J. Cancer 2007, 121, 1296–1305. [Google Scholar]
- Pan, Z.Z.; Bruening, W.; Giasson, B.I.; Lee, V.M.; Godwin, A.K. Gamma-synuclein promotes cancer cell survival and inhibits stress- and chemotherapy drug-induced apoptosis by modulating MAPK pathways. J. Biol. Chem 2002, 277, 35050–35060. [Google Scholar]
- Giani, C.; Casalini, P.; Pupa, S.M.; De Vecchi, R.; Ardini, E.; Colnaghi, M.I.; Giordano, A.; Menard, S. Increased expression of c-erbB-2 in hormone-dependent breast cancer cells inhibits cell growth and induces differentiation. Oncogene 1998, 17, 425–432. [Google Scholar]
- Kim, Y.S.; Konoplev, S.N.; Montemurro, F.; Hoy, E.; Smith, T.L.; Rondon, G.; Champlin, R.E.; Sahin, A.A.; Ueno, N.T. HER-2/neu overexpression as a poor prognostic factor for patients with metastatic breast cancer undergoing high-dose chemotherapy with autologous stem cell transplantation. Clin. Cancer Res 2001, 7, 4008–4012. [Google Scholar]
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human breast cancer: Correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987, 235, 177–182. [Google Scholar]
- Konecny, G.E.; Thomssen, C.; Luck, H.J.; Untch, M.; Wang, H.J.; Kuhn, W.; Eidtmann, H.; du Bois, A.; Olbricht, S.; Steinfeld, D.; et al. Her-2/neu gene amplification and response to paclitaxel in patients with metastatic breast cancer. J. Natl. Cancer Inst 2004, 96, 1141–1151. [Google Scholar]
- Moliterni, A.; Menard, S.; Valagussa, P.; Biganzoli, E.; Boracchi, P.; Balsari, A.; Casalini, P.; Tomasic, G.; Marubini, E.; Pilotti, S.; et al. HER2 overexpression and doxorubicin in adjuvant chemotherapy for resectable breast cancer. J. Clin. Oncol 2003, 21, 458–462. [Google Scholar]
- Tai, W.; Mahato, R.; Cheng, K. The role of HER2 in cancer therapy and targeted drug delivery. J. Controlled Release 2010, 146, 264–275. [Google Scholar]
- Bergqvist, J.; Ohd, J.F.; Smeds, J.; Klaar, S.; Isola, J.; Nordgren, H.; Elmberger, G.P.; Hellborg, H.; Bjohle, J.; Borg, A.L.; et al. Quantitative real-time PCR analysis and microarray-based RNA expression of HER2 in relation to outcome. Ann. Oncol 2007, 18, 845–850. [Google Scholar]
- Mendoza, G.; Portillo, A.; Olmos-Soto, J. Accurate breast cancer diagnosis through real-time PCR her-2 gene quantification usin immunohistochemically-identified biopsies. Oncol. Lett 2013, 5, 295–298. [Google Scholar]
- Bernard, P.S.; Wittwer, C.T. Real-time PCR technology for cancer diagnostics. Clin. Chem 2002, 48, 1178–1185. [Google Scholar]
- Krawczyk, N.; Banys, M.; Neubauer, H.; Solomayer, E.F.; Gall, C.; Hahn, M.; Becker, S.; Bachmann, R.; Wallwiener, D.; Fehm, T. HER2 status on persistent disseminated tumor cells after adjuvant therapy may differ from initial HER2 status on primary tumor. Anticancer Res 2009, 29, 4019–4024. [Google Scholar]
- Solomayer, E.F.; Becker, S.; Pergola-Becker, G.; Bachmann, R.; Kramer, B.; Vogel, U.; Neubauer, H.; Wallwiener, D.; Huober, J.; Fehm, T.N. Comparison of HER2 status between primary tumor and disseminated tumor cells in primary breast cancer patients. Breast Cancer Res. Treat 2006, 98, 179–184. [Google Scholar]
- Balbin, M.; Pendas, A.M.; Uria, J.A.; Jimenez, M.G.; Freije, J.P.; Lopez-Otin, C. Expression and regulation of collagenase-3 (MMP-13) in human malignant tumors. APMIS 1999, 107, 45–53. [Google Scholar]
- Chang, H.J.; Yang, M.J.; Yang, Y.H.; Hou, M.F.; Hsueh, E.J.; Lin, S.R. MMP13 is potentially a new tumor marker for breast cancer diagnosis. Oncol. Rep 2009, 22, 1119–1127. [Google Scholar]
- Lafleur, M.A.; Drew, A.F.; de Sousa, E.L.; Blick, T.; Bills, M.; Walker, E.C.; Williams, E.D.; Waltham, M.; Thompson, E.W. Upregulation of matrix metalloproteinases (MMPs) in breast cancer xenografts: A major induction of stromal MMP-13. Int. J. Cancer 2005, 114, 544–554. [Google Scholar]
- Selvamurugan, N.; Partridge, N.C. Constitutive expression and regulation of collagenase-3 in human breast cancer cells. Mol. Cell Biol. Res. Commun 2000, 3, 218–223. [Google Scholar]
- Nikseresht, M.; Seghatoleslam, A.; Monabati, A.; Talei, A.; Ghalati, F.B.; Owji, A.A. Overexpression of the novel human gene, UBE2Q2, in breast cancer. Cancer Genet. Cytogenet 2010, 197, 101–106. [Google Scholar]
- Fabre-Lafay, S.; Garrido-Urbani, S.; Reymond, N.; Goncalves, A.; Dubreuil, P.; Lopez, M. Nectin-4, a new serological breast cancer marker, is a substrate for tumor necrosis factor-alpha-converting enzyme (TACE)/ADAM-17. J. Biol. Chem 2005, 280, 19543–19550. [Google Scholar]
- Dontu, G. Breast cancer stem cell markers—The rocky road to clinical applications. Breast Cancer Res 2008, 10, 110. [Google Scholar]
- De Cremoux, P.; Tran-Perennou, C.; Brockdorff, B.L.; Boudou, E.; Brunner, N.; Magdelenat, H.; Lykkesfeldt, A.E. Validation of real-time RT-PCR for analysis of human breast cancer cell lines resistant or sensitive to treatment with antiestrogens. Endocr. Relat. Cancer 2003, 10, 409–418. [Google Scholar]
- Mitas, M.; Mikhitarian, K.; Walters, C.; Baron, P.L.; Elliott, B.M.; Brothers, T.E.; Robison, J.G.; Metcalf, J.S.; Palesch, Y.Y.; Zhang, Z.; et al. Quantitative real-time RT-PCR detection of breast cancer micrometastasis using a multigene marker panel. Int. J. Cancer 2001, 93, 162–171. [Google Scholar]
- Weissenstein, U.; Schumann, A.; Reif, M.; Link, S.; Toffol-Schmidt, U.D.; Heusser, P. Detection of circulating tumor cells in blood of metastatic breast cancer patients using a combination of cytokeratin and EpCAM antibodies. BMC Cancer 2012, 12, 206. [Google Scholar]
- Alix-Panabieres, C. EPISPOT assay: Detection of viable DTCs/CTCs in solid tumor patients. Recent Results Cancer Res 2012, 195, 69–76. [Google Scholar]
- Wang, N.; Shi, L.; Li, H.; Hu, Y.; Du, W.; Liu, W.; Zheng, J.; Huang, S.; Qu, X. Detection of circulating tumor cells and tumor stem cells in patients with breast cancer by using flow cytometry: A valuable tool for diagnosis and prognosis evaluation. Tumour Biol 2012, 33, 561–569. [Google Scholar]
- Markou, A.; Strati, A.; Malamos, N.; Georgoulias, V.; Lianidou, E.S. Molecular characterization of circulating tumor cells in breast cancer by a liquid bead array hybridization assay. Clin. Chem 2011, 57, 421–430. [Google Scholar]
- Dydensborg, A.B.; Herring, E.; Auclair, J.; Tremblay, E.; Beaulieu, J.F. Normalizing genes for quantitative RT-PCR in differentiating human intestinal epithelial cells and adenocarcinomas of the colon. Am. J. Physiol 2006, 290, G1067–G1074. [Google Scholar]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, RESEARCH0034. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar]
- Gervasoni, A.; Monasterio Munoz, R.M.; Wengler, G.S.; Rizzi, A.; Zaniboni, A.; Parolini, O. Molecular signature detection of circulating tumor cells using a panel of selected genes. Cancer Lett 2008, 263, 267–279. [Google Scholar]
- Bolke, E.; Orth, K.; Gerber, P.A.; Lammering, G.; Mota, R.; Peiper, M.; Matuschek, C.; Budach, W.; Rusnak, E.; Shaikh, S.; et al. Gene expression of circulating tumour cells in breast cancer patients. Eur. J. Med. Res 2009, 14, 426–432. [Google Scholar]
| Patient | Histology | T-stage | N-stage | Her2-status | Estrogen receptor (%) | Progesterone receptor (%) |
|---|---|---|---|---|---|---|
| Adj.1 | Inv. ductal | pT2 | pN3 | +++ | 0 | 0 |
| Adj.2 | Inv. ductal | pT1c | pN0 | ++ | 90 | 90 |
| Adj.3 | Inv. ductal | pT2 | pN0 | - | 90 | 90 |
| Adj.4 | Inv. lobular | pT1c | pN0 | - | 95 | 90 |
| Adj.5 | Inv. ductal | pT2 | pN0 | - | 30 | 50 |
| Adj.6 | Adeno-squamous | pT3 | pN0 | +++ | 0 | 0 |
| Adj.7 | Inv. ductal | pT1c | pN3a | + | 80 | 50 |
| Adj.8 | Inv. ductal | pT2 | pN3a | +++ | 30 | 10 |
| Adj.9 | Inv. ductal | pT2 | pN0 | + | 0 | 10 |
| Adj.10 | Inv. ductal | pT1c | pN0 | ++ | 90 | 60 |
| Adj.11 | Adenocarcinoma Lobular | pT2 | pN1 | + | 80 | 30 |
| Adj.12 | Inv. ductal | pT2 | pN0 | - | 90 | 30 |
| Adj.13 | Inv. ductal | pT2 | pN0 | + | 80 | 80 |
| Adj.14 | Inv. ductal pT1c | pN0 | - | 0 | 0 |
| Patient | Histology | T-stage | N-stage | Metastases | Her2- status | Estrogen receptor (%) | Progesterone receptor (%) |
|---|---|---|---|---|---|---|---|
| Met.1 | Inv. ductal | pT2 | pN0 | Liver, Bones | - | 0 | 0 |
| Met.2 | Inv. ductal | pT2 | pN0 | Brain | +++ | 0 | 0 |
| Met.3 | Inv. ductal | pT4 | pN0 | Bones | + | 0 | 0 |
| Met.4 | Inv. ductal | pT4 | pN1 | Lung | ++ | 0 | 0 |
| Met.5 | Inv. ductal | pT4 | pN0 | Lung | + | 60 | 40 |
| Met.6 | Inv. ductal | pT1c | pN0 | Bones, Liver | - | 0 | 0 |
| Met.7 | Inv. ductal | pT2 | pN0 | Liver | + | 0 | 0 |
| Met.8 | Inv. ductal | pT3c | pN1 | Liver, Bones, Brain | - | 0 | 0 |
| Met.9 | Inv. ductal | pT3 | pN0 | Bones | +++ | 20 | 10 |
| Met.10 | Inv. ductal | pT2 | pN1 | Lung, Bones | - | 30 | 10 |
| Met.11 | Inv. ductal | pT1b | pN0 | Bones | - | 90 | 70 |
| Her2 Status Tumor/CTCs | Adjuvant | Metastatic | ||
|---|---|---|---|---|
| Number | % | Number | % | |
| +/+ | 3/14 | 22% | 1/11 | 9% |
| +/− | 1/14 | 7% | 6/11 | 55% |
| −/+ | 6/14 | 43% | 1/11 | 9% |
| −/− | 4/14 | 28% | 3/11 | 27% |
| Gene | Forward primer | Reverse primer |
|---|---|---|
| CK8 | 5′-cgtcaagctgctggac-3′ | 5′-aggctgtagcggccgg-3′ |
| CK19 | 5′-gcctggttcaagccgg-3′ | 5′-ctcctgattcccgctc-3′ |
| BCSP | 5′-acactgtgtggccaagac-3′ | 5′-ccactctgggtctgcc-3′ |
| Gene | Forward primer | Reverse primer | Hydrolysis probe |
|---|---|---|---|
| B2M | 5′-ggccgagatggctccg-3′ | 5′-gatgaaaccccatagc-3′ | 5′-aggctatccagattca-3′ |
| CK8 | 5′-cctacaggaagctgga-3′ | 5′-gctcagaccaatagcc-3′ | 5′-ggagagccggggagtc-3′ |
| CK19 | 5′-ccgaggttacacctgc-3′ | 5′-gatcagcgccgatatg-3′ | 5′-ctgagcatgagctgcc-3′ |
| BCSP | 5′-ggagaacatcgtcacc-3′ | 5′-ggatgcctcactcctg-3′ | 5′-tgcgcaaggagaggcc-3′ |
| Gene | R2-value | PCR efficiency |
|---|---|---|
| B2M | 0,9845 | 98,45% |
| CK8 | 0,9923 | 99,23% |
| CK19 | 0,9066 | 90,66% |
| BCSP | 0,9894 | 98,94% |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Andergassen, U.; Hofmann, S.; Kölbl, A.C.; Schindlbeck, C.; Neugebauer, J.; Hutter, S.; Engelstädter, V.; Ilmer, M.; Friese, K.; Jeschke, U. Detection of Tumor Cell-Specific mRNA in the Peripheral Blood of Patients with Breast Cancer — Evaluation of Several Markers with Real-Time Reverse Transcription-PCR. Int. J. Mol. Sci. 2013, 14, 1093-1104. https://doi.org/10.3390/ijms14011093
Andergassen U, Hofmann S, Kölbl AC, Schindlbeck C, Neugebauer J, Hutter S, Engelstädter V, Ilmer M, Friese K, Jeschke U. Detection of Tumor Cell-Specific mRNA in the Peripheral Blood of Patients with Breast Cancer — Evaluation of Several Markers with Real-Time Reverse Transcription-PCR. International Journal of Molecular Sciences. 2013; 14(1):1093-1104. https://doi.org/10.3390/ijms14011093
Chicago/Turabian StyleAndergassen, Ulrich, Simone Hofmann, Alexandra C. Kölbl, Christian Schindlbeck, Julia Neugebauer, Stefan Hutter, Verena Engelstädter, Matthias Ilmer, Klaus Friese, and Udo Jeschke. 2013. "Detection of Tumor Cell-Specific mRNA in the Peripheral Blood of Patients with Breast Cancer — Evaluation of Several Markers with Real-Time Reverse Transcription-PCR" International Journal of Molecular Sciences 14, no. 1: 1093-1104. https://doi.org/10.3390/ijms14011093






