Molecular Cloning and Functional Analysis of Three FLOWERING LOCUS T (FT) Homologous Genes from Chinese Cymbidium
Abstract
:1. Introduction
2. Results
2.1. Isolation of three FT cDNAs from Cymbidium
2.2. Different Expression Patterns of Cymbidium FT genes under LD/SD
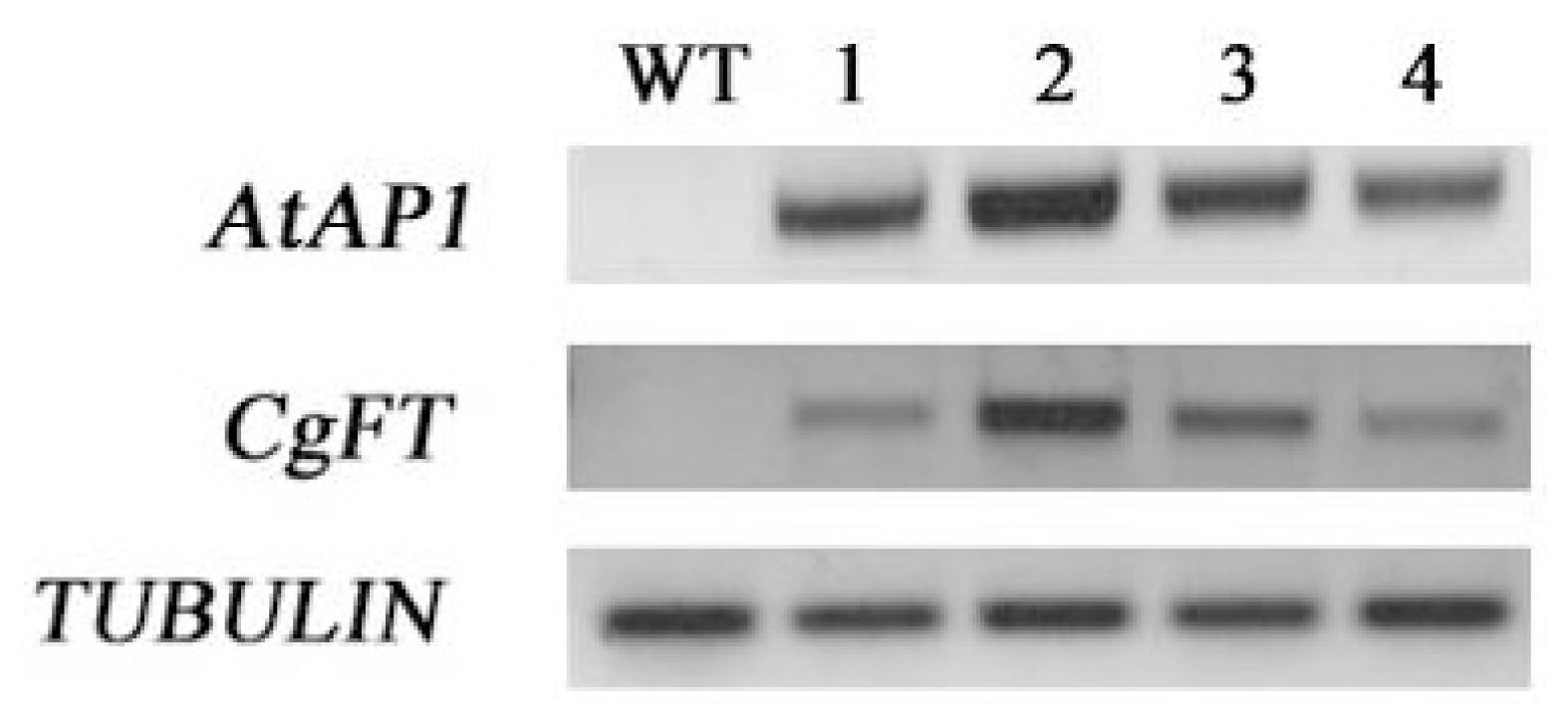
2.3. Ectopic Expression of CgFT Caused Early Flowering Phenotypes in Arabidopsis Thaliana
3. Discussion
3.1. Cloning and Characterization of three FT Homologous Genes from Chinese Cymbidium
3.2. Expression Patterns and Regulation
3.3. Constitutive Expression of CgFT Acts Similarly to Arabidopsis Thaliana FT in Regulating Flower Transition
4. Experimental Section
4.1. Plant Materials and Growth Conditions
4.2. Cloning CsFT, CgFT and CeFT cDNAs from Cymbidium
4.3. Real-Time PCR Analysis
4.4. Phylogenetic Analysis
4.5. Semi-Quantitative RT-PCR
4.6. Plant Transformation and Transgenic Plant Analysis
5. Conclusions
Acknowledgements
References
- Teixeira da Silva, J.A.; Chan, M.T.; Sanjaya Chai, M.L.; Tanaka, M. Priming abiotic factors for optimal hybrid Cymbidium (Orchidaceae) PLB and callus induction, plantlet formation, and their subsequent cytogenetic stability analysis. Sci. Hortic 2006, 109, 368–378. [Google Scholar]
- Teixeira da Silva, J.A.; Singh, N.; Tanaka, M. Priming biotic factors for optimal protocorm-like body and callus induction in hybrid Cymbidium (Orchidaceae), and assessment of cytogenetic stability in regenerated plants. Plant Cell Tissue Organ Cult 2006b, 84, 119–128. [Google Scholar]
- Teixeira da Silva, J.A.; Chin, D.P.; van, P.T.; Mii, M. Transgenic orchids. Sci. Hort 2011, 130, 673–680. [Google Scholar]
- Huan, L.V.T.; Takamura, T.; Tanaka, M. Callus formation and plant regeneration from callus through somatic embryo structures in Cymbidium orchid. Plant Sci 2004, 166, 1443–1449. [Google Scholar]
- Shimasaki, K.; Uemoto, S. Rhizome induction and plantlet regeneration of Cymbidium goeringii from flower bud cultures in vitro. Plant Cell Tissue Organ Cult 1991, 25, 49–52. [Google Scholar]
- Chang, C.; Chang, W.C. Plant regeneration from callus culture of Cymbidium ensifolium var. misericors. Plant Cell Rep 1998, 17, 251–255. [Google Scholar]
- Hsu, H.F.; Huang, C.H.; Chou, L.T.; Yang, C.H. Ectopic expression of an orchid (Oncidium Gower Ramsey) AGL6-like gene promotes flowering by activating flowering time genes in Arabidopsis thaliana. Plant Cell Physiol 2003, 44, 783–794. [Google Scholar]
- Hou, C.J.; Yang, C.H. Functional analysis of FT and TFL1 orthologs from orchid (Oncidium Gower Ramsey) that regulate the vegetative to reproductive transition. Plant Cell Physiol 2009, 50, 1544–1557. [Google Scholar]
- Kardailsky, I.; Shukla, V.K.; Ahn, J.H.; Dagenais, N.; Christensen, S.K.; Nguyen, J.T.; Chory, J.; Harrison, M.J.; Weigel, D. Activation tagging of the floral inducer FT. Science 1999, 286, 1962–1965. [Google Scholar]
- Kobayashi, Y.; Kaya, H.; Goto, K.; Iwabuchi, M.; Araki, T. A pair of related genes with antagonistic roles in mediating flowering signals. Science 1999, 286, 1960–1962. [Google Scholar]
- Komeda, Y. Genetic regulation of time to flower in Arabidopsis thaliana. Ann. Rev. Plant Biol 2004, 55, 521–535. [Google Scholar]
- Samach, A.; Wigge, P.A. Ambient temperature perception in plants. Curr. Opin. Plant Biol 2005, 8, 483–486. [Google Scholar]
- Koornneef, M.; Hanhart, C.J.; van der Veen, J.H. A genetic and physiological analysis of late flowering mutants in Arabidopsis thaliana. Mol. Gen. Genet 1991, 229, 57–66. [Google Scholar]
- Ahn, J.H.; Miller, D.; Winter, V.J.; Banfield, M.J.; Lee, J.H.; Yoo, S.Y.; Henz, S.R.; Brady, R.L.; Weigel, D. A divergent external loop confers antagonistic activity on floral regulators FT and TFL1. EMBO J 2006, 25, 605–614. [Google Scholar]
- Kojima, S.; Takahashi, Y.; Kobayashi, Y.; Monna, L.; Sasaki, T.; Araki, T.; Yano, M. Hd3a, a rice ortholog of the Arabidopsis FT gene, promotes transition to flowering downstream of Hd1 under short-day conditions. Plant Cell Physiol 2002, 43, 1096–1105. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 1962, 15, 473–497. [Google Scholar]
- Cheng, C.L.; Acedo, G.N.; Cristinsin, M.; Conkling, M.A. Sucrose mimics the light induction of Arabidopsis nitrate reductase gene transcription. Proc. Natl. Acad. Sci. USA 1992, 89, 1861–1864. [Google Scholar]
- Corbesier, L.; Vincent, C.; Jang, S.; Fornara, F.; Fan, Q.; Searle, I.; Giakountis, A.; Farrona, S.; Gissot, L.; Turnbull, C.; et al. FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 2007, 316, 1030–1033. [Google Scholar]
- Fujiwara, S.; Oda, A.; Yoshida, R.; Niinuma, K.; Miyata, K.; Tomozoe, Y.; Tajima, T.; Nakagawa, M.; Hayashi, K.; Coupland, G.; et al. Circadian clock proteins LHY and CCA1 regulate SVP protein accumulation to control flowering in Arabidopsis. Plant Cell 2008, 20, 2960–2971. [Google Scholar]
- Shalit, A.; Rozmana, A.; Goldshmidt, A.; Alvarez, J.B.; Browman, J.L.; Eshed, Y.; Lifschitz, E. The flowering hormone florigen functions as a general systemic regulator of growth and termination. Proc. Natl. Acad. Sci. USA 2009, 106, 8392–8397. [Google Scholar]
- Wellmer, F.; Alves-Ferreira, M.; Dubois, A.; Riechmann, J.L.; Meyerowitz, E.M. Genome-wide analysis of gene expression during early Arabidopsis flower development. PLoS Genet 2006, 2, 1012–1024. [Google Scholar]
- Carmona, M.J.; Calonje, M.; Martinez-Zapater, J.M. The FT/TFL1 gene family in grapevine. Plant Mol. Biol 2007, 63, 637–650. [Google Scholar]
- Chardon, F.; Damerval, C. Phylogenomic analysis of the PEBP gene family in cereals. J. Mol. Evol 2005, 61, 579–590. [Google Scholar]
- Faure, S.; Higgins, J.; Turner, A.; Laurie, D.A. The FLOWERING LOCUS T like gene family in barley (Hordeum vulgare). Genetics 2007, 176, 599–609. [Google Scholar]
- Igasaki, T.; Watanabe, Y.; Nishiguchi, M.; Kotoda, N. The FLOWERING LOCUS T/TERMINAL FLOWER1 family in Lombardy poplar. Plant Cell Physiol 2008, 49, 291–300. [Google Scholar]
- Danilevskaya, O.N.; Meng, X.; Hou, Z.; Ananiev, E.V.; Simmons, C.R. A genomic and expression compendium of the expanded PEBP gene family from maize. Plant Physiol 2008, 146, 250–264. [Google Scholar]
- Yan, L.; Fu, D.; Li, C.; Blechl, A.; Tranquilli, G.; Bonafede, M.; Sanchez, A.; Valarik, M.; Yasuda, S.; Dubcovsky, J. The wheat and barley vernalization gene VRN3 is an orthologue of FT. Proc. Natl. Acad. Sci. USA 2006, 103, 1640–1644. [Google Scholar]
- Jeffares, D.C.; Mourier, T.; Penny, D. The biology of intron gain and loss. Trends Genet 2006, 22, 16–22. [Google Scholar]
- Roy, S.W.; Gilbert, W. Rates of intron loss and gain: Implications for early eukaryotic evolution. Proc. Natl. Acad. Sci. USA 2005, 102, 7158–7162. [Google Scholar]
- Searle, I.; Coupland, G. Induction of flowering by seasonal changes in photoperiod. Eur. Mol. Biol. Organ J 2004, 23, 1217–1222. [Google Scholar]
- Hayama, R.; Coupland, G. The molecular basis of diversity in the photoperiodic flowering responses of Arabidopsis and rice. Plant Physiol 2004, 135, 677–684. [Google Scholar]
- Lifschitz, E.; Eshed, Y. Universal florigenic signals triggered by FT homologues regulate growth and flowering cycles in perennial day-neutral tomato. J. Exp. Bot 2006, 57, 3405–3414. [Google Scholar]
- Hayama, R.; Agashe, B.; Luley, E.; King, R.; Coupland, G. A circadian rhythm set by dusk determines the expression of FT homologs and the short-day photoperiodic flowering response in Pharbitis. Plant Cell 2007, 19, 2988–3000. [Google Scholar]
- Lin, M.K.; Belanger, H.; Lee, Y.J.; Varkonyi-Gasic, E.; Taoka, K.I.; Miura, E.; Xoconostle-Cazares, B.; Gendler, K.; Jorgensen, R.A.; Phinney, B.; et al. FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 2007, 19, 1488–1506. [Google Scholar]
- Komiya, R.; Ikegami, A.; Tamaki, S.; Yokoi, S.; Shimamoto, K. Hd3a and RFT1 are essential for flowering in rice. Development 2008, 135, 767–774. [Google Scholar]
- Takahashi, Y.; Teshima, K.M.; Yokoi, S.; Innan, H.; Shimamoto, K. Variations in Hd1 proteins, Hd3a promoters, and Ehd1 expression levels contribute to diversity of flowering time in cultivated rice. Proc. Natl. Acad. Sci. USA 2009, 106, 4555–4560. [Google Scholar]
- Kong, F.; Liu, B.; Xia, Z.; Sato, S.; Kim, B.M.; Watanabe, S.; Yamada, T.; Tabata, S.; Kanazawa, A.; Harada, K.; et al. Two coordinately regulated homologs of FLOWERING LOCUS T are involved in the control of photoperiodic flowering in soybean. Plant Physiol 2010, 154, 1220–1231. [Google Scholar]
- Liu, Y.G.; Chen, Y.L. High-efficiency thermal asymmetric interlaced PCR for amplification of unknown flanking sequences. BioTechniques 2007, 43, 649–656. [Google Scholar]
- National Center for Biotechnology Information. Available online: http://www.ncbi.nlm.nih.gov/ accessed on 1 September 2010.
- European Bioinformatics Institute. Available online: http://www.ebi.ac.uk/Tools/clustalw2/ accessed on 1 September 2010.
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evol 2007, 24, 1596–1599. [Google Scholar]
- Wu, P.Z.; Li, J.; Wei, Q.; Zeng, L.; Chen, Y.P.; Li, M.R.; Jiang, H.W.; Wu, G.J. Cloning and functional characterization of an acyl-acyl carrier protein thioesterase (JcFATB1) from Jatropha curcas. Tree Physiol 2009, 29, 1299–1305. [Google Scholar]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium mediated transformation of Arabidopsis thaliana. Plant J 1998, 16, 735–743. [Google Scholar]





| Genotype | No. of plants | Days to bolting | Rosette leaf number at bolting | Days to anthesis | Rosette leaf number at anthesis |
|---|---|---|---|---|---|
| WT | 20 | 22.7 ± 3.2 | 8.1 ± 1.9 | 30.4 ± 5.0 | 10.4 ± 2.1 |
| 35S::CgFT (line 7) | 22 | 13.0 ± 1.5 ** | 4.0 ± 0.0 ** | 22.7 ± 3.3 ** | 4.2 ± 0.6 ** |
| 35S::CgFT (line 30) | 13 | 14.1 ± 2.0 ** | 4.0 ± 0.0 ** | 23.8 ± 3.9 ** | 4.2 ± 0.6 ** |
| 35S::CgFT (line 9) | 12 | 16.5 ± 3.7 ** | 4.3 ± 0.8 ** | 26.6 ± 3.1 * | 4.8 ± 1.0 ** |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, W.; Fang, Z.; Zeng, S.; Zhang, J.; Wu, K.; Chen, Z.; Teixeira da Silva, J.A.; Duan, J. Molecular Cloning and Functional Analysis of Three FLOWERING LOCUS T (FT) Homologous Genes from Chinese Cymbidium. Int. J. Mol. Sci. 2012, 13, 11385-11398. https://doi.org/10.3390/ijms130911385
Huang W, Fang Z, Zeng S, Zhang J, Wu K, Chen Z, Teixeira da Silva JA, Duan J. Molecular Cloning and Functional Analysis of Three FLOWERING LOCUS T (FT) Homologous Genes from Chinese Cymbidium. International Journal of Molecular Sciences. 2012; 13(9):11385-11398. https://doi.org/10.3390/ijms130911385
Chicago/Turabian StyleHuang, Weiting, Zhongming Fang, Songjun Zeng, Jianxia Zhang, Kunlin Wu, Zhilin Chen, Jaime A. Teixeira da Silva, and Jun Duan. 2012. "Molecular Cloning and Functional Analysis of Three FLOWERING LOCUS T (FT) Homologous Genes from Chinese Cymbidium" International Journal of Molecular Sciences 13, no. 9: 11385-11398. https://doi.org/10.3390/ijms130911385




