Determination and Analysis of Ustiloxins A and B by LC-ESI-MS and HPLC in False Smut Balls of Rice
Abstract
:1. Introduction
2. Results and Discussion
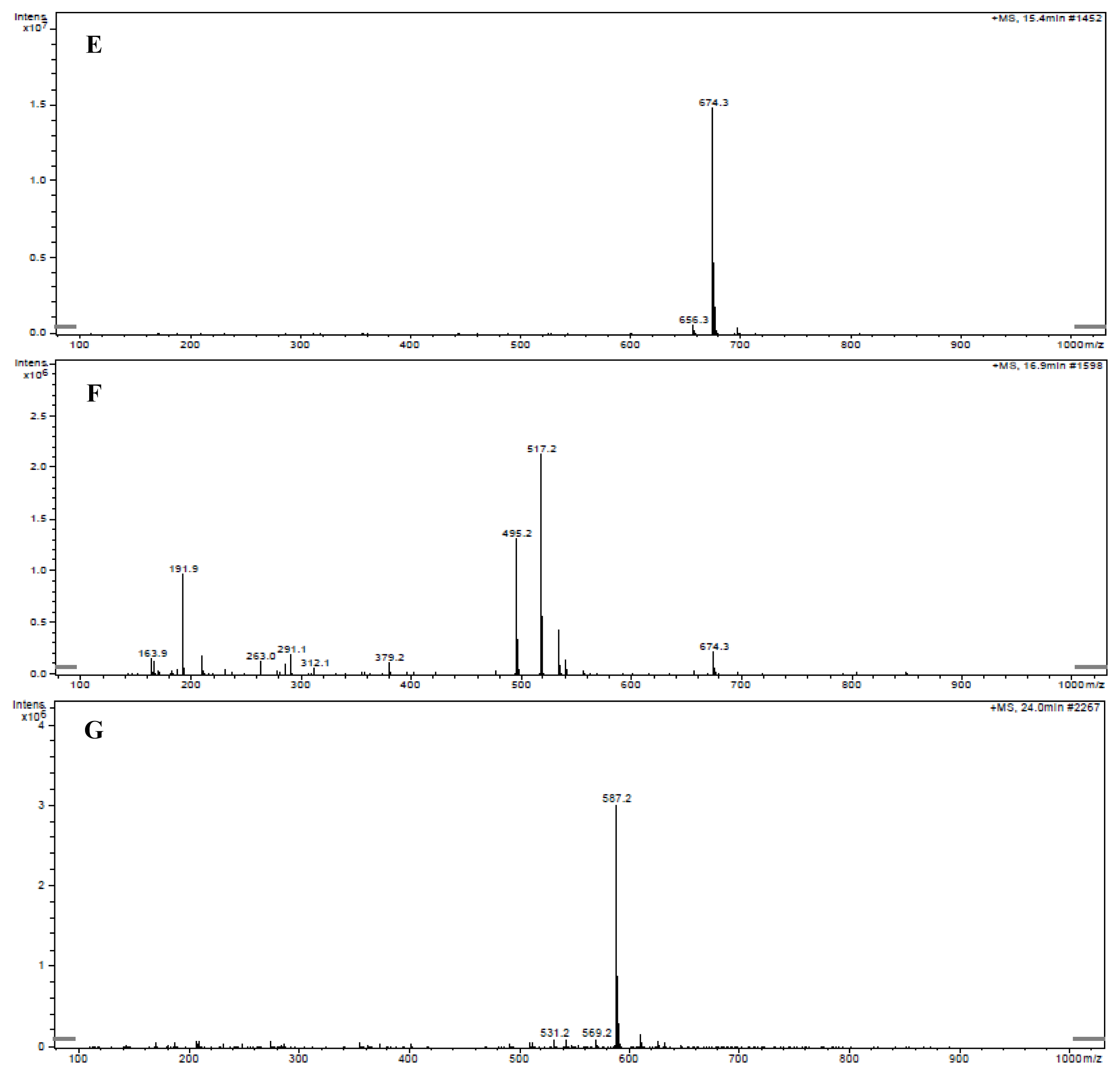
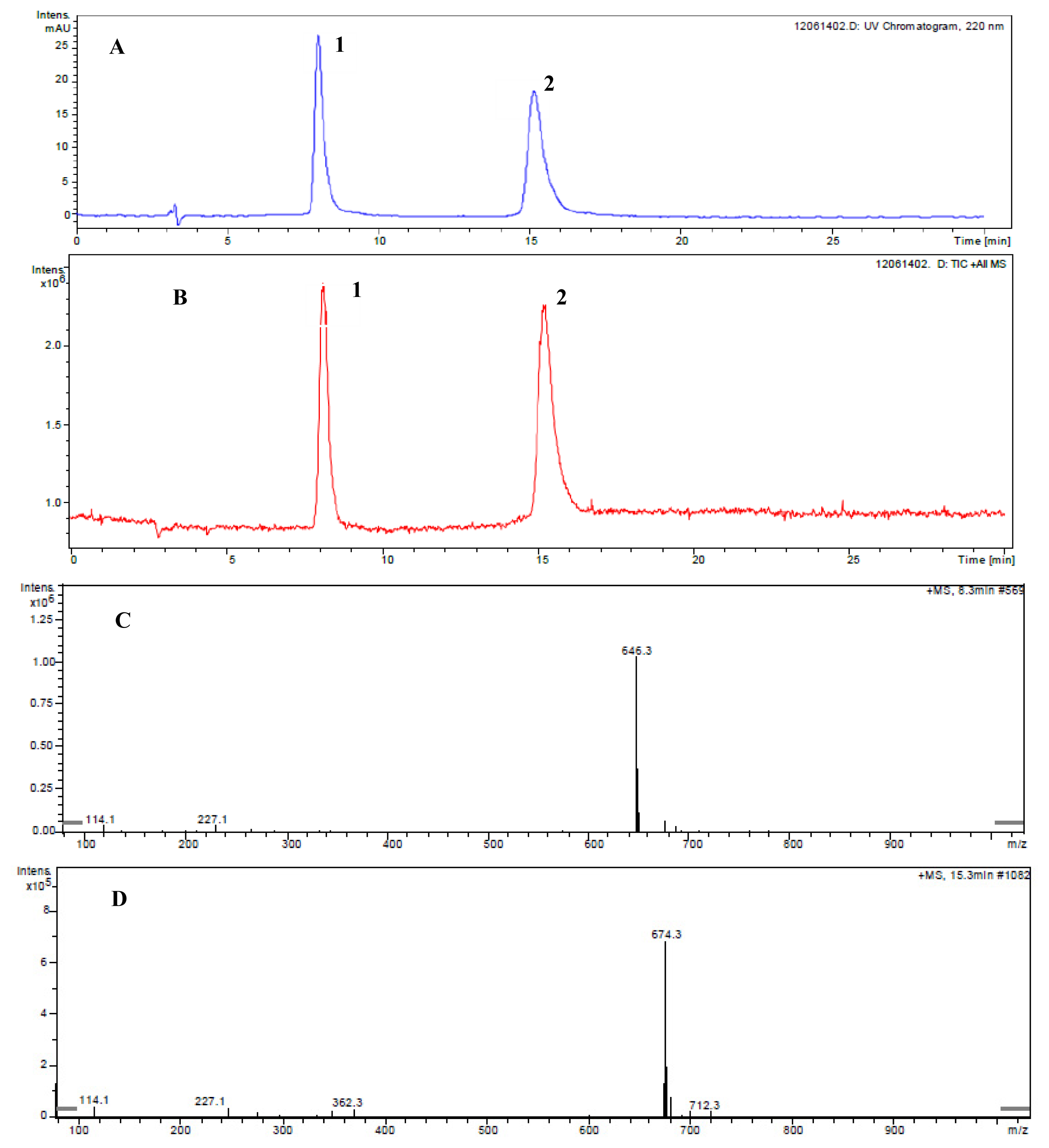
2.1. Primary Determination of the Ustiloxin-Like Compounds in Rice False Smut Balls by LC-ESI-MS
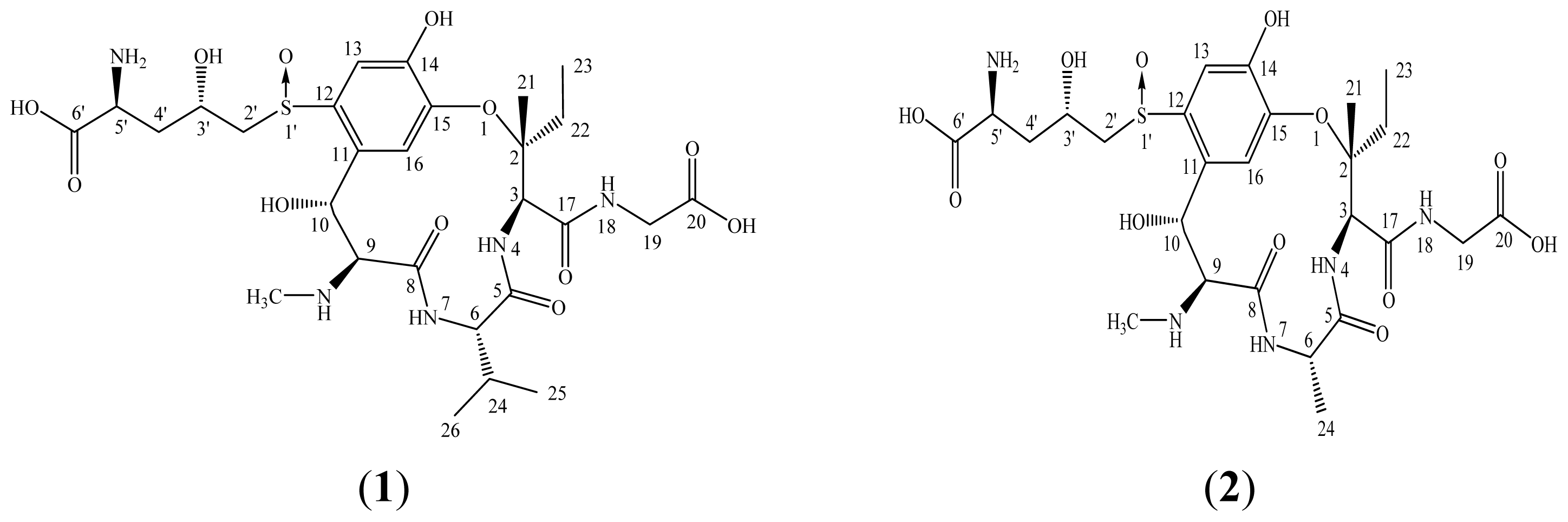
2.2. Elucidation of the Purified Ustiloxins A and B
2.3. Determination of Ustiloxins A and B by LC-ESI-MS
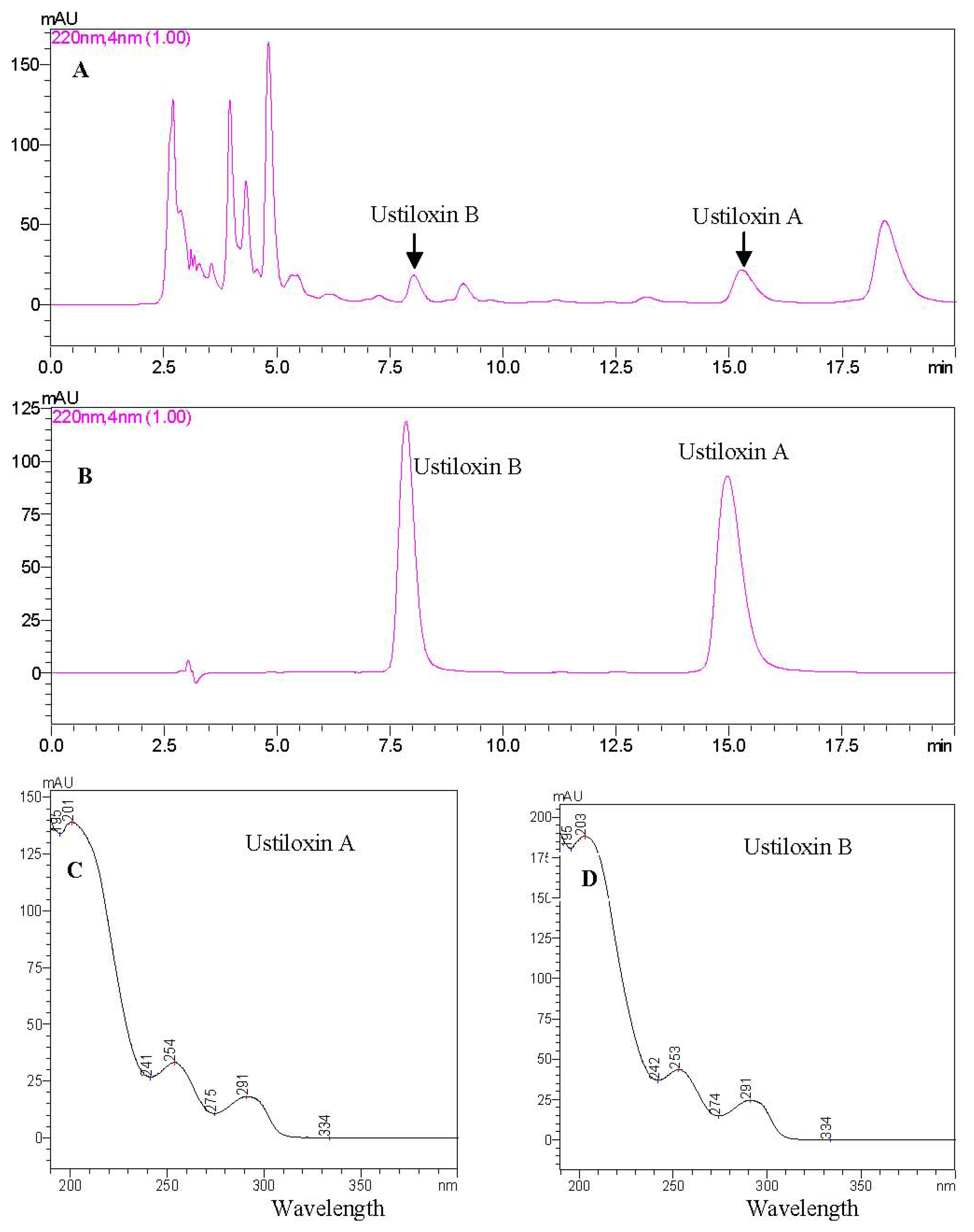
2.4. HPLC Analysis of Ustiloxins A and B in Rice False Smut Balls
2.4.1. Identification of Ustiloxins A and B by HPLC-UV
2.4.2. Calibration Curves
2.4.3. Precision
2.4.3. Accuracy
2.4.4. Limits of Detection and Quantification
2.4.5. Quantitative Determination of Ustiloxins A and B in Rice False Smut Balls
3. Experimental Section
3.1. General
3.2. Material
3.3. Extraction and Fractionation of the Ustiloxins
3.4. Physicochemical and Spectrometric Data of Ustiloxins A and B
3.5. HPLC Analysis of Ustiloxins A and B
3.5.1. Preparation of the Ustiloxin Standard Solutions
3.5.2. Preparation of Sample Solutions
3.5.3. Method Validation
3.5.4. Quantitative Determination of Ustiloxins A and B in Rice False Smut Balls
4. Conclusions
Supplementary Materials
ijms-13-11275-s001.pdfAcknowledgements
References
- Koiso, Y.; Li, Y.; Iwasaki, S.; Hanaoka, K.; Kobayashi, T.; Sonoda, R.; Fujita, Y.; Yaegashi, H.; Sato, Z. Ustiloxins, antimitotic cyclic peptides from false smut balls on rice panicles caused by Ustilaginoidea virens. J. Antibiot 1994, 47, 765–773. [Google Scholar]
- Lu, S.; Liu, H.; Zhao, J.; Yin, C.; Zhou, L. Research progress of ustiloxins. Chin. Agric. Sci. Bull 2010, 26, 265–268. [Google Scholar]
- Zhou, L.; Lu, S.; Shan, T.; Wang, P.; Sun, W.; Chen, Z.; Wang, S. Chemistry and Biology of Mycotoxins from Rice False Smut Pathogen. In Mycotoxins: Properties, Applications and Hazards; Melborn, B.J., Greene, J.C., Eds.; Nova Science Publishers: New York, NY, USA, 2012; pp. 109–130. [Google Scholar]
- Tanaka, E.; Ashizawa, T.; Sonoda, R.; Tanaka, C. Villosiclava virens gen. nov., teleomorph of Ustilaginoidea virens, the causal agent of rice false smut. Mycotaxon 2008, 106, 491–501. [Google Scholar]
- Wang, S.; Li, M.; Dong, H.; Liu, X.; Bai, Y.; Yang, H. Sporulation, inoculation methods and pathogenicity of Ustilaginoidea albicans, the cause of white rice false smut in China. J. Phytopathol 2008, 156, 755–757. [Google Scholar]
- Ashizawa, T.; Takahashi, M.; Moriwaki, J.; Hirayae, K. Quantification of the rice false smut pathogen Ustilaginoidea virens from soil in Japan using real-time PCR. Eur. J. Plant Pathol 2010, 128, 221–232. [Google Scholar]
- Luduena, R.F.; Roach, M.C.; Prasad, V.; Banerjee, M.; Koiso, Y.; Li, Y.; Iwasaki, S. Interaction of ustiloxin A with bovine brain tubulin. Biochem. Pharmacol 1994, 47, 1593–1599. [Google Scholar]
- Nakamura, K.; Izumiyama, N.; Ohtsubo, K.; Koiso, Y.; Iwasaki, S.; Sonoda, R.; Fujita, Y.; Yaegashi, H.; Sato, Z. “Lupinosis”-like lesions in mice caused by ustiloxin produced by Ustilaginoidea virens: A morphological study. Nat.”. Toxins 1994, 2, 22–28. [Google Scholar]
- Miyazaki, S.; Matsumoto, Y.; Uchihara, T.; Morimoto, K. High-performance liquid chromatographic determination of ustiloxin A in forage rice silage. J. Vet. Med. Sci 2009, 71, 239–241. [Google Scholar]
- Koiso, Y.; Natori, M.; Iwasaki, S.; Sato, S.; Sonoda, R.; Fujita, Y.; Yaegashi, H. Ustiloxin: A phytotoxin and a mycotoxin from false smut balls on rice panicles. Tetrahedron Lett 1992, 33, 4157–4160. [Google Scholar]
- Koiso, Y.; Morisaki, N.; Yamashita, Y.; Mitsui, Y.; Shirai, R.; Hashimoto, Y.; Iwasaki, S. Isolation and structure of an antimitotic cyclic peptide, ustiloxin F: Chemical interrelation with a homologous peptide, ustiloxin B. J. Antibiot 1998, 51, 418–422. [Google Scholar]
- Hubert, P.; Nguen-Huu, J.-J.; Boulanger, B.; Chapuzet, E.; Chiap, P.; Cohen, N.; Compagnon, P.-A.; Dewe, W.; Feinberg, M.; Lallier, M.; et al. Harmonization of strategies for the validation of quantitative analytical procedures. A SFSTP proposal—Part I. J. Pharm. Biomed. Anal 2004, 36, 579–586. [Google Scholar]
- Hubert, P.; Nguen-Huu, J.-J.; Boulanger, B.; Chapuzet, E.; Chiap, P.; Cohen, N.; Compagnon, P.-A.; Dewe, W.; Feinberg, M.; Lallier, M.; et al. Harmonization of strategies for the validation of quantitative analytical procedures. A SFSTP proposal—Part II. J. Pharm. Biomed. Anal 2007, 45, 70–81. [Google Scholar]
- Nagaraju, P.T.; Sreekanh, R.K.P.; Chinna, G.P.; Venugopal, N.; Ravindranath, S.; Bhanu, P.B.; Sade, S. Reverse phase HPLC method for determination of Ritonavir in pharmaceutical preparations. Int. J. Pharm. Sci. Rev. Res 2012, 13, 55–57. [Google Scholar]
- Lv, Z.; Dong, J.; Zhang, B. Rapid identification and detection of flavonoid compounds from bamboo leaves by LC-(ESI)-IT-TOF/MS. BioResources 2011, 7, 1405–1418. [Google Scholar]
- Brase, S.; Encinas, A.; Keck, J.; Nising, C.F. Chemistry and biology of mycotoxins and related fungal metabolites. Chem. Rev 2009, 109, 3903–3990. [Google Scholar]
- Li, Y.; Koiso, Y.; Kobayashi, H.; Hashimoto, Y.; Iwasaki, S. Ustiloxins, new antimitotic cyclic peptides: Interaction with procine brain tubulin. Biochem. Pharmacol 1995, 49, 1367–1372. [Google Scholar]
- Li, P.; Evans, C.D.; Forbeck, E.M.; Park, H.; Bai, R.; Hamel, E.; Joullie, M.M. Total synthesis and biological evaluation of ustiloxin natural products and two analogs. Bioorg. Med. Chem. Lett 2006, 16, 4804–4807. [Google Scholar]
- Li, P.; Evans, C.D.; Wu, Y.; Cao, B.; Hamel, E.; Joullie, M.M. Evolution of the total syntheses of ustiloxin natural products and their analogues. J. Am. Chem. Soc 2008, 130, 2351–2364. [Google Scholar]

| Compound | Intra-day (n = 5) | Inter-day (n = 9) | ||
|---|---|---|---|---|
| Concentration (μg/mL) | RSD (%) | Concentration (μg/mL) | RSD (%) | |
| Ustiloxin A | 12.5 | 0.47 | 12.5 | 0.24 |
| 50.0 | 0.14 | 50.0 | 0.27 | |
| 150.0 | 0.07 | 150.0 | 0.31 | |
| Ustiloxin B | 12.5 | 1.82 | 12.5 | 1.63 |
| 50.0 | 0.29 | 50.0 | 0.13 | |
| 150.0 | 1.43 | 150.0 | 1.45 | |
| Compound | Added concentration (μg/mL) | Recovery yield (%) | RSD (%) |
|---|---|---|---|
| Ustiloxin A | 16.0 | 97.4 | 0.49 |
| 32.0 | 96.9 | 0.26 | |
| 64.0 | 93.4 | 1.73 | |
| Ustiloxin B | 16.0 | 90.0 | 0.65 |
| 32.0 | 91.4 | 1.85 | |
| 64.0 | 96.5 | 1.34 | |
| Compound | Average (mg/g, n = 6) | RSD (%) |
|---|---|---|
| Ustiloxin A | 0.57 ± 0.0078 | 1.38 |
| Ustiloxin B | 0.38 ± 0.0042 | 1.11 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shan, T.; Sun, W.; Liu, H.; Gao, S.; Lu, S.; Wang, M.; Sun, W.; Chen, Z.; Wang, S.; Zhou, L. Determination and Analysis of Ustiloxins A and B by LC-ESI-MS and HPLC in False Smut Balls of Rice. Int. J. Mol. Sci. 2012, 13, 11275-11287. https://doi.org/10.3390/ijms130911275
Shan T, Sun W, Liu H, Gao S, Lu S, Wang M, Sun W, Chen Z, Wang S, Zhou L. Determination and Analysis of Ustiloxins A and B by LC-ESI-MS and HPLC in False Smut Balls of Rice. International Journal of Molecular Sciences. 2012; 13(9):11275-11287. https://doi.org/10.3390/ijms130911275
Chicago/Turabian StyleShan, Tijiang, Weibo Sun, Hao Liu, Shan Gao, Shiqiong Lu, Mingan Wang, Wenxian Sun, Zhiyi Chen, Shu Wang, and Ligang Zhou. 2012. "Determination and Analysis of Ustiloxins A and B by LC-ESI-MS and HPLC in False Smut Balls of Rice" International Journal of Molecular Sciences 13, no. 9: 11275-11287. https://doi.org/10.3390/ijms130911275






