Commercial Bee Pollen with Different Geographical Origins: A Comprehensive Approach
Abstract
:1. Introduction
2. Results and Discussion
2.1. Labeling and Information to Consumers
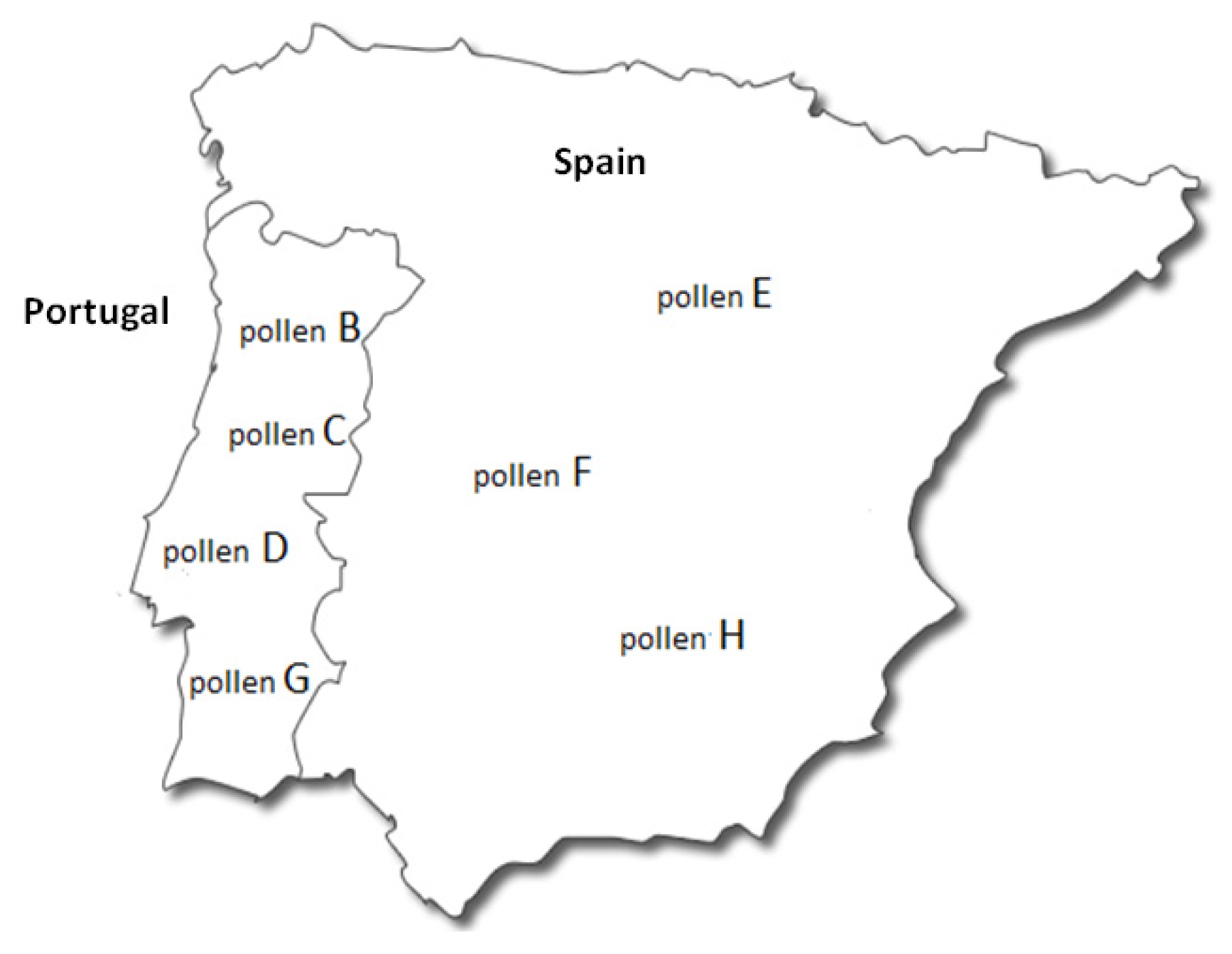
2.2. Pollinic Identification
2.3. Chemical Composition
2.4. Microbial Contamination
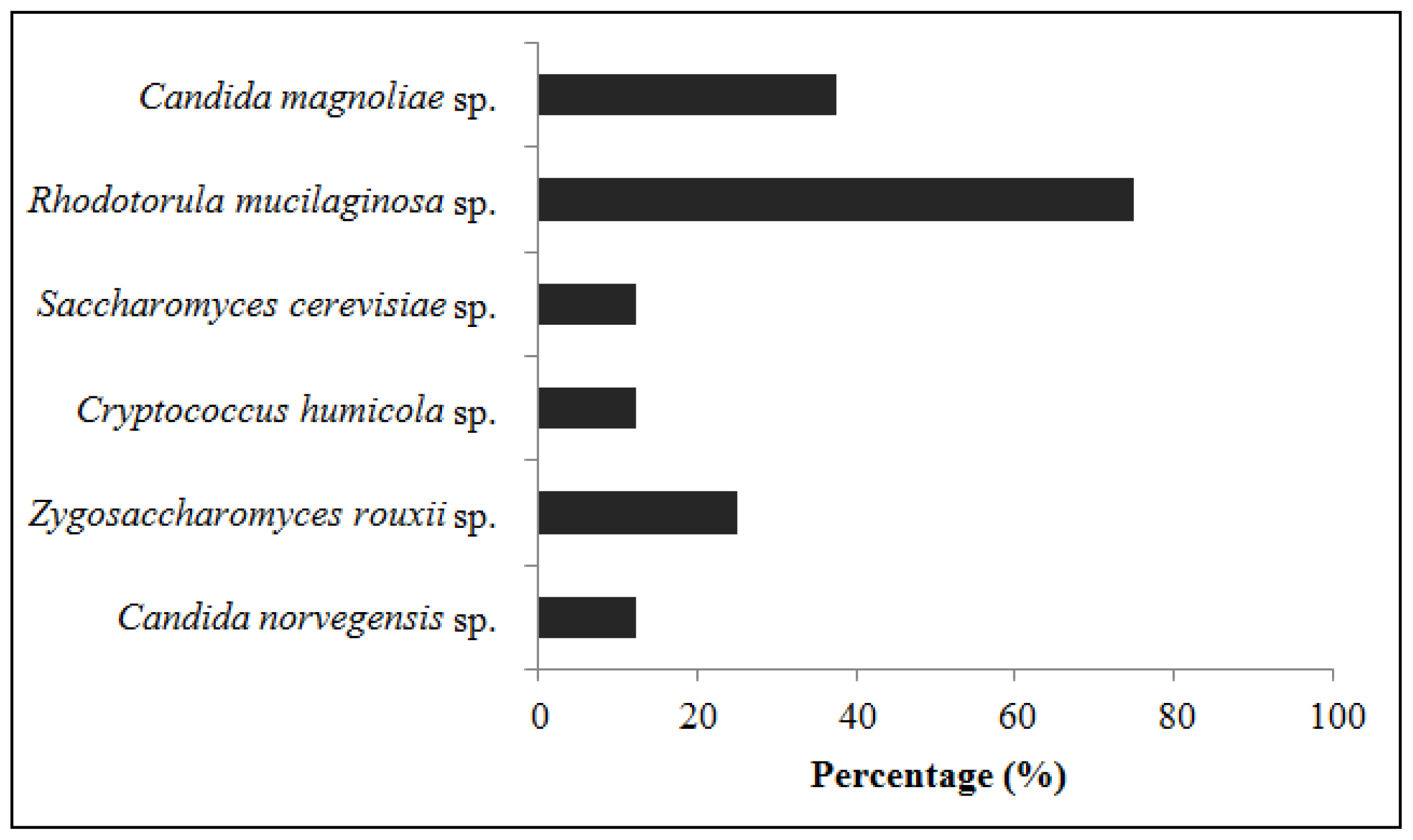
2.5. Isolation and Identification of Yeasts
3. Experimental Section
3.1. Samples
3.2. Chemicals and Materials
3.3. Labeling
3.4. Pollinic Identification
3.5. Physicochemical Analyses
3.5.1. Moisture
3.5.2. Ash
3.5.3. pH
3.5.4. Reducing Sugars
3.5.5. Water Activity
3.5.6. Lipids
3.5.7. Proteins
3.5.8. Carbohydrates
3.5.9. Energy
3.6. Microbiological Determinations
3.6.1. Sample Preparation
3.6.2. Enumeration of the Total Mesophilic Microorganisms
3.6.3. Enumeration of Yeast and Moulds
3.6.4. Sulfite Reducing Clostridium Spores
3.6.5. Fecal coliforms and Escherichia coli
3.6.6. Salmonella
3.7. Isolation and Identification of Yeasts
3.8. Statistical Analysis
4. Conclusions
References
- Almeida-Muradian, L.B.; Pamplona, L.C.; Coimbra, S.; Barth, O.M. Chemical composition and botanical evaluation of dried bee pollen pellets. J. Food Compos. Anal 2005, 18, 105–111. [Google Scholar]
- Yamaguchi, M.; Hamamoto, R.; Uchiyama, S.; Ishiyama, K.; Hashimoto, K. Anabolic effects of bee pollen Cistus ladaniferus extract on bone components in the femoral diaphyseal and metaphyseal tissues of rats in vitro and in vivo. J. Health Sci 2006, 52, 43–49. [Google Scholar]
- Shoskes, D.A. Phytotherapy in chronic prostatitis. Urology 2002, 60, 35–37. [Google Scholar]
- Mizrahi, A.; Lensky, Y. Bee Products: Properties, Applications and Apitherapy; Springer-Verlag: New York, NY, USA; p. 1997.
- Wojcicki, J.; Samochowiec, L.; Bartlomowicz, B.; Hinek, A.; Jaworska, M.; Gawronska-Szklarz, B. Effect of pollen extract on the development of experimental atherosclerosis in rabbits. Atherosclerosis 1986, 62, 39–45. [Google Scholar]
- Zhang, X.; Habib, F.K.; Ross, M.; Burger, U.; Lewenstein, A.; Rose, K.; Jaton, J. Isolation and characterization of a cyclic hydroxamic acid from a pollen extract, which inhibits cancerous cell growth in vitro. J. Med. Chem 1995, 38, 735–738. [Google Scholar]
- Morais, M.; Moreira, L.; Feás, X.; Estevinho, L.M. Honeybee-collected pollen from five Portuguese Natural Parks: Palynological origin, phenolic content, antioxidant properties and antimicrobial activity. Food Chem. Toxicol 2011, 39, 1096–1101. [Google Scholar]
- Baltrušaitytė, V.; Venskutonis, P.R.; Čkstarytė, V. Radical scavenging activity of different floral origin honey and beebread phenolic extracts. Food Chem 2007, 101, 502–514. [Google Scholar]
- Isidorov, V.A.; Isidorova, A.G.; Sczczepaniak, L.; Czyżewska, U. Gas chromatographic-mass spectrometric investigation of the chemical composition of beebread. Food Chem 2009, 115, 1056–1063. [Google Scholar]
- Villanueva, M.T.O.; Marquina, A.D.; Serrano, R.B.; Abellan, G.B. The importance of bee-collected pollen in the diet: A study of its composition. Int. J. Food Sci. Nutr 2002, 53, 217–224. [Google Scholar]
- Hani, B.; Dalila, B.; Saliha, D.; Harzallah, D.; Ghadbane, M.; Khennouf, S. Microbiological sanitary aspects of pollen. Adv. Environ. Biol 2012, 6, 1415–1420. [Google Scholar]
- Garcia-Villanova, R.J.; Cordón, C.; González, A.M.; Aparicio, P.; Garcia, M.E. Simultaneous immunoaffinity column cleanup and HPLC analysis of aflatoxins and ochratoxin A in Spanish bee pollen. J. Agric. Food Chem 2004, 52, 7235–7239. [Google Scholar]
- Decreto-Lei n.º 156/2008. Available online: http://www.nationalstemcentre.org.uk/dl/cb88b8db986af7cf354b280a1117755295a0d278/14762-Echium.pdf accessed on 18 July 2012.
- National Non-Food Crops Centre. Echium. Available online: http://www.nationalstemcentre.org.uk/dl/cb88b8db986af7cf354b280a1117755295a0d278/14762-Echium.pdf accessed on 18 July 2012.
- Luz, C.; Bacha, G., Jr; Fonseca, R.L.E.; Sousa, P. Comparative pollen preferences by africanized honeybees Apis mellifera L. of two colonies in Para de Minas, Minas Gerais, Brazil. Ann. Braz. Acad. Sci. 2010, 82, 293–304. [Google Scholar]
- Herbert, J.R.; Shimanuki, H. Chemical composition and nutritive value of bee-collected and bee-stored pollen. Apidologie 1978, 9, 33–40. [Google Scholar]
- Coronel, B.B.; Grasso, S.C.; Pereira, G.; Fernández, A. Caracterización bromatológica del pólen apícola Argentino. Cienc. Docencia. Tecnol 2004, 15, 141–181. [Google Scholar]
- Morgano, M.A.; Milani, R.F.; Martins, M.C.T.; Rodriguez-Amaya, D.B. Determination of water content in Brazilian honeybee-collected pollen by Karl Fischer titration. Food Control 2011, 22, 1604–1608. [Google Scholar]
- Carpes, T. Estudo das Caracteristicas Fisico-Quimicas e Biológicas do Polén Apícola de Apis mellifera da região Sul do Brasil. Ph.D. Thesis, Universidade Federal do Paraná, Curitiba, Brazil, September 2008. [Google Scholar]
- Krell, R. Value Added Products from Beekeeping; FAO Agricultural Services: Rome, Italy, 1996. [Google Scholar]
- Bonvehi, J.S.; Jordà, R.E. Nutrient composition and microbiological quality of honeybee-collected pollen in Spain. J. Agric. Food Chem 1997, 45, 725–732. [Google Scholar]
- Estevinho, L.M.; Rodrigues, S.; Pereira, A.P.; Feás, X. Portuguese bee pollen: Palynological study, nutritional and microbiological evaluation. Int. J. Food Sci. Technol 2012, 47, 429–435. [Google Scholar]
- González, G.; Hinojo, M.J.; Mateo, R.; Medina, A.; Jiménez, M. Occurrence of mycotoxin producing fungi in bee pollen. Int. J. Food Microbiol 2005, 105, 1–9. [Google Scholar]
- González-Martin, I.; Hernàndez-Hierro, J.M.; Barros-Ferreiro, N.; Córdon-Marcos, C.; Garcìa-Villanova, R.J. Use of NIRS technology with a remote reflectance fibre-optic probe for predicting major components in bee pollen. Talanta 2007, 72, 998–1003. [Google Scholar]
- Marchini, L.C.; Reis, V.D.A.; Moreti, A.C.C.C. Composição físico-química de amostras de pólen coletado por abelhas africanizadas Apis mellifera (Hymenoptera: Apidae) em Piracicaba, Estado de São Paulo. Ciênc. Rural 2006, 36, 949–953. [Google Scholar]
- Bastos, D.H.M.; Rocha, C.I.; Cunha, I.B.S.; Carvalho, P.O.; Torres, E.A.S. Composição e qualidade de pólen apícola comercializado em algumas cidades nos estados de São Paulo e Minas Gerais. Revist. Inst. Adolfo Lutz 2003, 62, 239–244. [Google Scholar]
- Carpes, T.; Cabral, I.; Rosalen, P.L.; de Alencar, S.M.; Masson, M.L. Caracterização do potencial antimicrobiano dos extractos de pólen apícola da região Sul do Brasil. Alimentos Nutrição Araraquara 2009, 20, 271–277. [Google Scholar]
- Melo, I.L.P.; Freitas, A.S.; Barth, O.M.; Almeida-Muradian, L.B. Correlation between nutritional composition and floral origin of dried bee pollen. Revist. Inst. Adolfo Lutz 2009, 168, 1–9. [Google Scholar]
- Vit, P.; Santiago, B. Chemical composition of fresh bee pollen collected in the Misintá páramo from the Venezuelan Andes. Arch. Latinoam. Nutr 2008, 58, 411–415. [Google Scholar]
- Ayaz, F.A.; Glew, R.H.; Millson, R.M.; Huang, H.S.; Chuang, L.H.; Sanz, C.; Hayırlıoglu-Ayaz, S. Nutrient contents of kale (Brassica oleraceae L. Var. acephala DC.). Food Chem 2006, 96, 572–579. [Google Scholar]
- Hervatin, H.L. Avaliação microbiológica e físico-quimica do pólen apícola in natura e desidratado sob diferentes temperaturas. M.Sc. Thesis, Universidade Estadual de Campinas, Campinas, Brazil, September 2009. [Google Scholar]
- Carvalho, C.M.; Rocha, A.; Estevinho, M.L.F. Identification of honey yeast based on RFLP analysis of the ITS region. Cienc. Tecnol. Aliment 2005, 5, 11–17. [Google Scholar]
- Snowdon, J.A.; Cliver, D.O. Microorganisms in honey. Int. J. Food Microbiol 1996, 31, 1–26. [Google Scholar]
- Stratford, M. Food and beverage yeasts. In Yeasts in Food and Beverages; Querol, A., Fleet, G.H., Eds.; Springer-Verlag: Berlin, Germany, 2006; pp. 335–379. [Google Scholar]
- Cascio, G.; Dalle Carbonare, L.; Maccacaro, L.; Caliari, F.; Ligozzi, M.; Cascio, V.; Fontana, R. First case of bloodstream infection due to Candida magnoliae in a Chinese oncological patient. J. Clin. Microbiol 2007, 10, 3470–3473. [Google Scholar]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of Melissopalinology. Bee World 1978, 59, 139–157. [Google Scholar]
- Zenebon, O.; Tiglea, N.S.P.P. Métodos Físico-Químicos Para Análise de Alimentos, 4th ed; Instituto Adolfo Lutz: São Paulo, Brazil, 2008; p. 1020. [Google Scholar]
- Silva, R.N.; Monteiro, V.N.; Alcanfor, J.A.X.; Assis, E.M.; Asquieri, E.R. Comparision methods for the determination of reducers sugars and total in honey. Ciênc. Tecnol. Aliment 2003, 23, 337–341. [Google Scholar]
- Barros, L.; Heleno, S.A.; Carvalho, A.M.; Ferreira, I.C.F.R. Lamiaceae often used in Portuguese folk medicine as a source of powerful antioxidants: Vitamins and phenolics. LWT-Food Sci. Technol 2010, 43, 544–550. [Google Scholar]
- Instituto Português da Qualidade, Géneros Alimentícios. Análise Microbiológica. Colheita de Amostras; NP-1828:1982; Instituto Português da Qualidade: Caparica, Portugal, 1982.
- Instituto Português da Qualidade, Microbiologia Alimentar—Regras Gerais Para a Contagem de Microrganismos a 30 °C; NP-3788:2002; Instituto Português da Qualidade: Caparica, Portugal, 2002.
- International Standards Organization, Microbiology of Food and Animal Feeding Stuffs— Horizontal Method for the Enumeration of Yeasts and Moulds—Part 2: Colony Count Technique in Products with Water Activity Less Than or Equal to 0.95; US ISO 21527-2:2008; International Standards Organization: Winterthur, Switzerland, 2008.
- International Standards Organization, Microbiology of Food and Animal Feeding Stuffs— Horizontal Method for the Enumeration of Sulfite-Reducing Bacteria Growing under Anaerobic Conditions; ISO 15213:2003; International Standards Organization: Winterthur, Switzerland, 2003.
- International Standards Organization, Microbiology of Food and Animal Feeding Stuffs— Horizontal Method for the Detection and Enumeration of Coliforms—Most Probable Number Technique; ISO 4831:2006; International Standards Organization: Winterthur, Switzerland, 2006.
- International Standards Organization, Microbiology of Food and Animal Feeding Stuffs— Horizontal Method for the Detection of Salmonella spp.; ISO 6579:2002; International Standards Organization: Winterthur, Switzerland, 2002.
| In the packaging (%) | |
|---|---|
| Trade name | 100.0 |
| Net quantity (g or kg) | 100.0 |
| Date of minimum durability | 100.0 |
| Name, company or business name | 87.5 |
| Social grower/packer/seller | 87.5 |
| established in the EU | 87.5 |
| Lot | 100.0 |
| Address | 87.5 |
| Place of origin | 87.5 |
| * Storage conditions | 75.0 |
| * Information to consumers | 75.0 |
| Family | Samples | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | H | ||
| Asteraceae | Leontodon spp. (%) | - | 5.3 ± 1.9 3 | - | - | 1.2 ± 0.1 3 | - | 18.2 ± 3.4 2 | - |
| Boraginaceae | Echium spp. (%) | 2.0 ± 0.4 3 | 4.6 ± 1.4 3 | 7.5 ± 1.6 3 | - | 4.2 ± 0.8 3 | 1.2 ± 0.2 3 | 6.2 ± 2.3 3 | 49.6 ± 5.2 1 |
| Cistaceae | Cistus spp. (%) | 68.2 ± 8.9 1 | - | 83.6 ± 9.2 1 | 2.4 ± 0.5 3 | 90.6 ± 9.3 1 | - | 5.2 ± 1.5 3 | - |
| Ericaceae | Erica spp. (%) | - | - | - | 58.0 ± 7.7 1 | 1.7 ± 0.2 3 | - | - | - |
| Fabaceae | Cytisus spp. (%) | - | 57.0 ± 9.2 1 | 2.9 ± 0.3 3 | 3 ± 0.8 3 | - | 69.8 ± 6.7 1 | - | 8 ± 1.0 3 |
| Acacia spp. (%) | - | 2.5 ± 0.6 3 | - | - | 1.5 ± 0.3 3 | - | - | - | |
| Trifolium spp. (%) | 4.4 ± 1.2 3 | - | 4.8 ± 1.0 3 | 16.3 ± 2.4 2 | - | - | 45.4 ± 7.1 1 | 2.0 ± 1.0 3 | |
| Fagaceae | Quercus spp. (%) | - | - | - | - | - | - | 16.0 ± 0.8 2 | - |
| Castanea spp. (%) | 15.8 ± 3.2 2 | 13.8 ± 2.3 3 | 1.2 ± 0.3 3 | - | - | 24.8 ± 5.1 2 | - | 5.2 ± 1.3 3 | |
| Lamiaceae | Thymus spp. (%) | - | - | - | 10.3 ± 3.9 3 | - | 1.4 ± 0.4 3 | - | 9.0 ± 2.1 3 |
| Mimosaceae | Mimosa spp. (%) | - | - | - | 10.0 ± 3.6 3 | - | - | - | - |
| Myrtaceae | Eucalyptus spp. (%) | - | - | - | - | - | - | 9.0 ± 1.9 3 | - |
| Pinaceae | Pinus spp. (%) | - | 12.4 ± 2.7 3 | - | - | - | - | - | - |
| Rosaceae | Prunus spp. (%) | 5.0 ± 1.6 3 | 4.4 ± 0.9 3 | - | - | 0.8 ± 0.2 3 | 2.8 ± 0.4 3 | - | - |
| - | Rubus spp. (%) | 4.6 ± 1.3 3 | - | 26.2 ± 4.3 2 | |||||
| A | B | C | D | E | F | G | H | |
|---|---|---|---|---|---|---|---|---|
| Water content | 7.63 ± 0.27 ab | 7.91 ± 0.04 ab | 6.02 ± 0.07 c | 6.02 ± 0.18 c | 7.97 ± 0.07 a | 6.95 ± 0.39 bc | 8.40 ± 0.82 a | 7.82 ± 0.25 ab |
| Water activity | 0.43 ± 0.03 a | 0.35 ± 0.02 cd | 0.33 ± 0.01 d | 0.26 ± 0.01 e | 0.41 ± 0.004 ab | 0.33 ± 0.02 d | 0.40 ± 0.03 ab | 0.38 ± 0.017 bc |
| pH | 4.92 ± 0.02 ab | 4.62 ± 0.04 c | 4.71 ± 0.03 bc | 5.17 ± 0.09 a | 4.49 ± 0.14 cd | 4.99 ± 0.17 ab | 4.23 ± 0.14 d | 5.03 ± 0.10 a |
| Ash | 3.16 ± 0.03 a | 1.85 ± 0.05 cd | 2.38 ± 0.39 bc | 2.23 ± 0.03 bcd | 2.16 ± 0.29 cd | 2.83 ± 0.29 ab | 1.67 ± 0.29 d | 0.5 ± 0.01 e |
| Proteins | 20.84 ± 1.16 ab | 20.03 ± 1.49 b | 25.15 ± 1.64 a | 22.01 ± 1.43 ab | 22.45 ± 0.89 ab | 20.69 ± 2.64 b | 18.79 ± 1.62 b | 12.50 ± 0.5 c |
| Lipids | 2.66 ± 0.06 c | 2.35 ± 0.11 d | 2.79 ± 0.10 bc | 2.71 ± 0.03 c | 2.86 ± 0.09 bc | 3.33 ± 0.16 a | 3.06 ± 0.08 ab | 2.75 ± 0.13 c |
| Carbohydrates | 73.35 ± 1.21 bc | 75.76 ± 1.48 b | 69.68 ± 2.08 c | 73.06 ± 1.39 bc | 72.53 ± 1.04 bc | 73.15 ± 2.88 bc | 76.49 ± 1.95 b | 84.25 ± 0.58 a |
| Reducing sugars | 26.09 ± 3.60 c | 33.19 ± 2.36 abc | 35.68 ± 5.07 abc | 38.62 ± 2.19 ab | 41.79 ± 3.21 a | 34.52 ± 1.05 abc | 26.90 ± 3.30 c | 29.39 ± 4.71 bc |
| Energy kcal | 400.70 ± 0.36 d | 404.37 ± 0.59 c | 404.43 ± 1.10 c | 404.66 ± 0.03 c | 405.66 ± 1.51 c | 405.30 ± 1.73 c | 408.62 ± 1.06 b | 411.75 ± 0.63 a |
| A | B | C | D | E | F | G | H | p-Value | |
|---|---|---|---|---|---|---|---|---|---|
| Aerobic mesophiles (cfu/g) | <10 a | <10 a | <10 a | <10 a | <10 a | 8.7 × 103 ± 7.8 × 10 b | <10 a | <10 a | 0.032 |
| Moulds and yeasts (cfu/g) | <10 a | <10 a | <10 a | 8.8 × 102 ± 6.9 × 10 b | < 10 a | 9.4 × 102 ± 7.1 × 10 b | 2.6 × 102 ± 5.2 × 10 ab | 6.9 × 102 ± 4.4 × 101 b | 0.001 |
| Fecal coliforms (MPN/g) | <1 | <1 | <1 | <1 | <1 | <1 | <1 | <1 | n.a. |
| Escherichia coli (MPN/g) | Absent | Absent | Absent | Absent | Absent | Absent | Absent | Absent | n.a. |
| Sulphite reducing clostridium (MPN/g) | Absent | Absent | Absent | Absent | Absent | Absent | Absent | Absent | n.a. |
| Salmonella sp. (in 10 g) | Absent | Absent | Absent | Absent | Absent | Absent | Absent | Absent | n.a. |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nogueira, C.; Iglesias, A.; Feás, X.; Estevinho, L.M. Commercial Bee Pollen with Different Geographical Origins: A Comprehensive Approach. Int. J. Mol. Sci. 2012, 13, 11173-11187. https://doi.org/10.3390/ijms130911173
Nogueira C, Iglesias A, Feás X, Estevinho LM. Commercial Bee Pollen with Different Geographical Origins: A Comprehensive Approach. International Journal of Molecular Sciences. 2012; 13(9):11173-11187. https://doi.org/10.3390/ijms130911173
Chicago/Turabian StyleNogueira, Carla, Antonio Iglesias, Xesus Feás, and Leticia M. Estevinho. 2012. "Commercial Bee Pollen with Different Geographical Origins: A Comprehensive Approach" International Journal of Molecular Sciences 13, no. 9: 11173-11187. https://doi.org/10.3390/ijms130911173







