Electric Field Induced Fluorescence Modulation of Single Molecules in PMMA Based on Electron Transfer
Abstract
:1. Introduction
2. Results
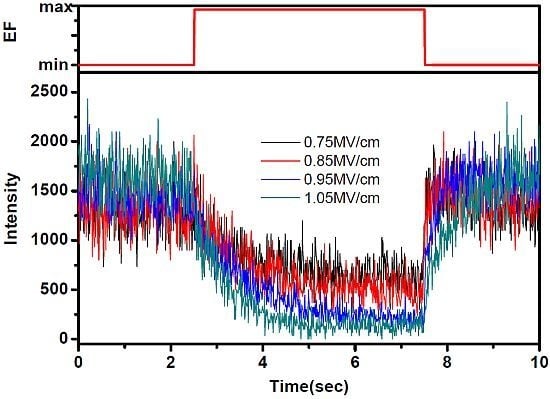
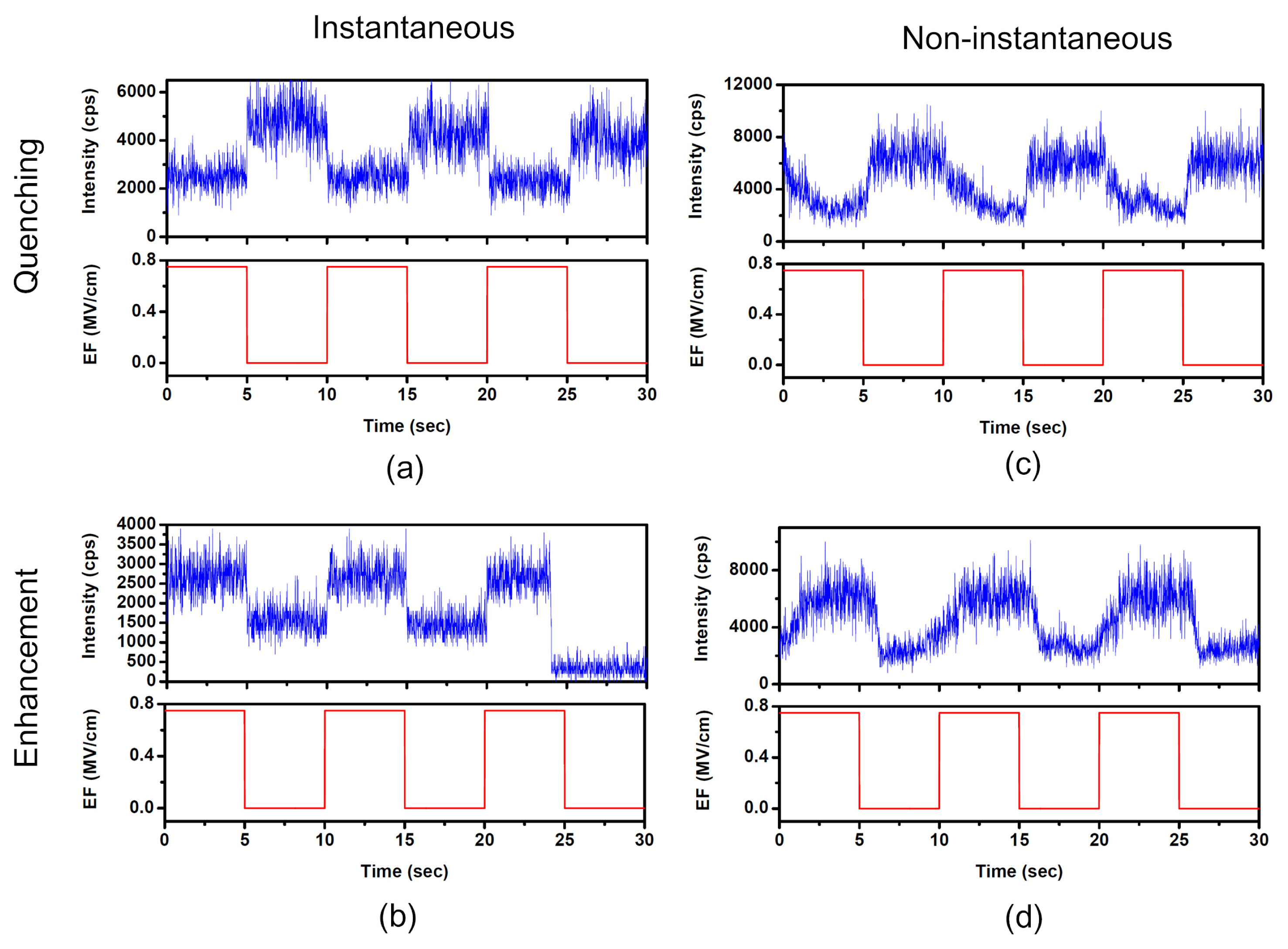
2.1. Diverse Responses of SMF in PMMA under EF
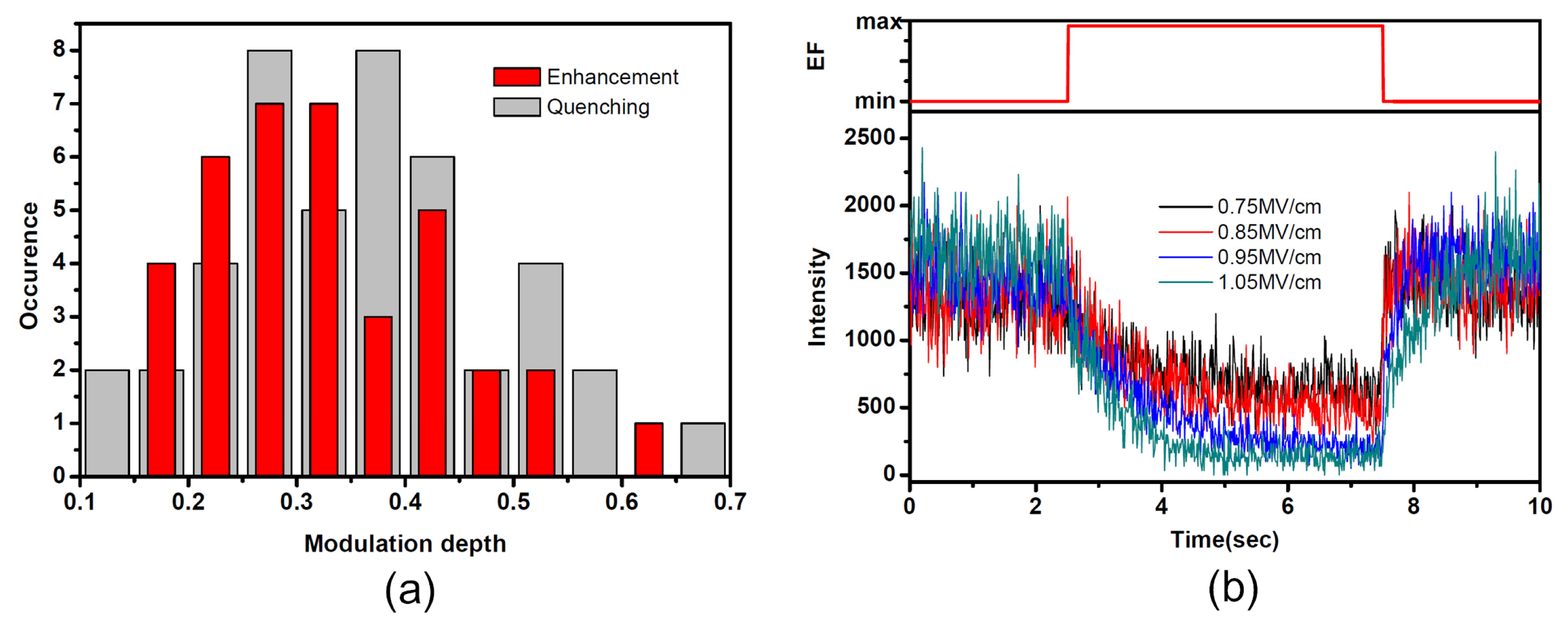
2.2. Diverse Modulation Depth and Response Time of SMF in PMMA under EF
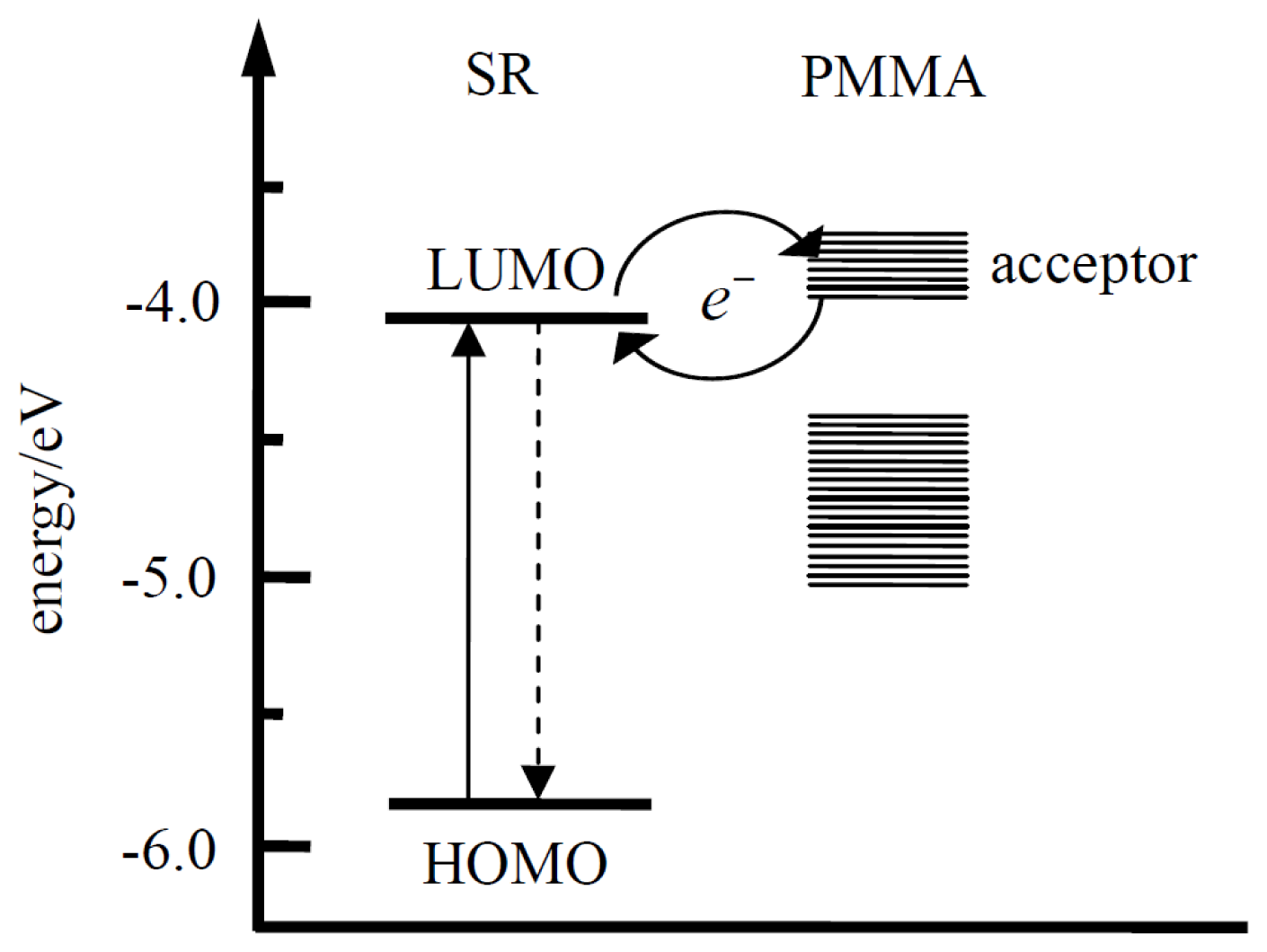
3. Discussion
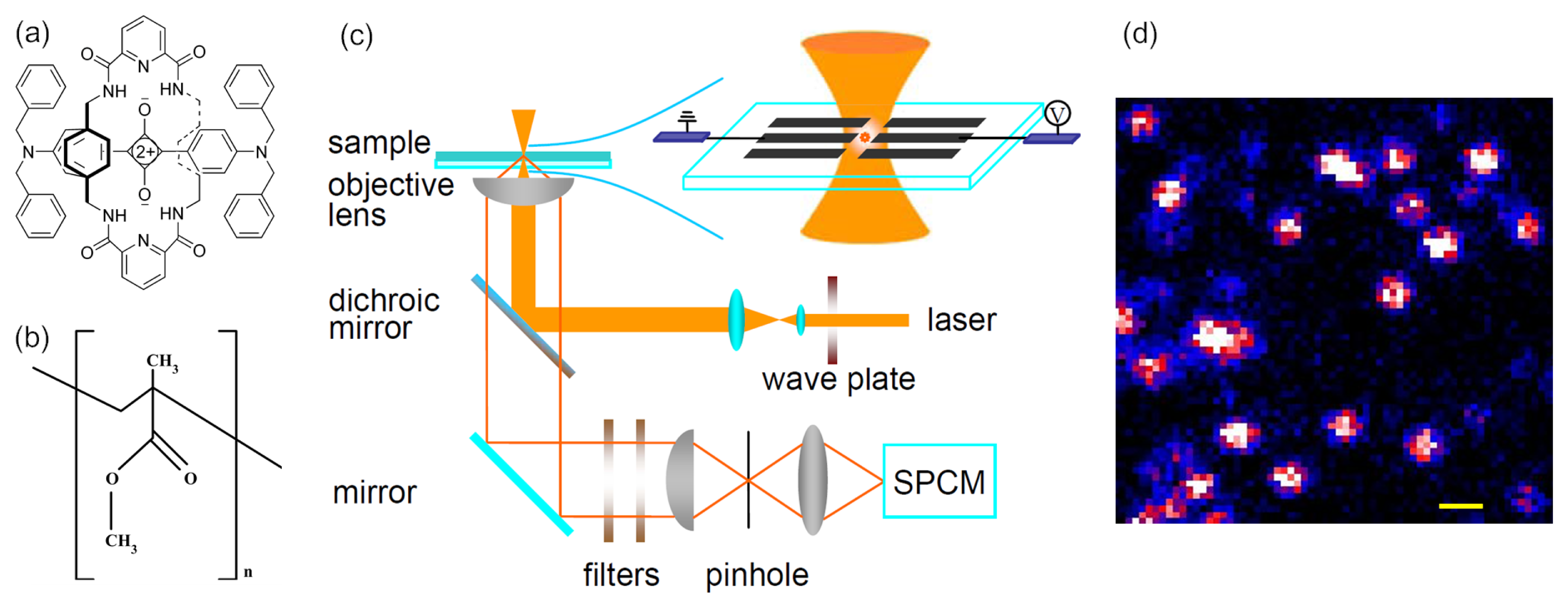
4. Experimental Section
4.1. Sample Preparation
4.2. Fluorescence Imaging of Single Dye Molecules in PMMA
5. Conclusions
Acknowledgements
References
- Moerner, W.; Orrit, M. Illuminating single molecules in condensed matter. Science 1999, 283, 1670–1676. [Google Scholar]
- Kulzer, F.; Orrit, M. Single-molecule optics. Annu. Rev. Phys. Chem 2004, 55, 585–611. [Google Scholar]
- Rezus, Y.; Walt, S.; Lettow, R.; Renn, A.; Zumofen, G.; Götzinger, S.; Sandoghdar, V. Single-photon spectroscopy of a single molecule. Phys. Rev. Lett. 2012, 108, 093601:1–093601:5. [Google Scholar]
- Lounis, B.; Orrit, M. Single-photon sources. Rep. Prog. Phys 2005, 68, 1129–1179. [Google Scholar]
- Pototschnig, M.; Chassagneux, Y.; Hwang, J.; Zumofen, G.; Renn, A.; Sandoghdar, V. Controlling the phase of a light beam with a single molecule. Phys. Rev. Lett. 2011, 107, 063001:1–063001:5. [Google Scholar]
- Azoulay, J.; Débarre, A.; Richard, A.; Tchénio, P. Quenching and enhancement of single-molecule fluorescence under metallic and dielectric tips. EPL (Europhys. Lett) 2000, 51, 374–380. [Google Scholar]
- Kühn, S.; Håkanson, U.; Rogobete, L.; Sandoghdar, V. Enhancement of single-molecule fluorescence using a gold nanoparticle as an optical nanoantenna. Phys. Rev. Lett. 2006, 97, 017402:1–017402:4. [Google Scholar]
- Zhang, J.; Fu, Y.; Chowdhury, M.H.; Lakowicz, J.R. Metal-enhanced single-molecule fluorescence on silver particle monomer and dimer: Coupling effect between metal particles. Nano Lett 2007, 7, 2101–2107. [Google Scholar]
- Anger, P.; Bharadwaj, P.; Novotny, L. Enhancement and quenching of single-molecule fluorescence. Phys. Rev. Lett. 2006, 96, 113002:1–113002:4. [Google Scholar]
- Hania, P.R.; Thomsson, D.; Scheblykin, I.G. Host matrix dependent fluorescence intensity modulation by an electric field in single conjugated polymer chains. J. Phys. Chem. B 2006, 110, 25895–25900. [Google Scholar]
- Schindler, F.; Lupton, J.M.; Müller, J.; Feldmann, J.; Scherf, U. How single conjugated polymer molecules respond to electric fields. Nat. Mater 2006, 5, 141–146. [Google Scholar]
- Hania, P.R.; Scheblykin, I.G. Electric field induced quenching of the fluorescence of a conjugated polymer probed at the single molecule level. Chem. Phys. Lett 2005, 414, 127–131. [Google Scholar]
- Holman, M.W.; Liu, R.; Adams, D.M. Single-molecule spectroscopy of interfacial electron transfer. J. Am. Chem. Soc 2003, 125, 12649–12654. [Google Scholar]
- Biju, V.; Micic, M.; Hu, D.; Lu, H.P. Intermittent single-molecule interfacial electron transfer dynamics. J. Am. Chem. Soc 2004, 126, 9374–9381. [Google Scholar]
- Heinlein, T.; Knemeyer, J.P.; Piestert, O.; Sauer, M. Photoinduced electron transfer between fluorescent dyes and guanosine residues in DNA-hairpins. J. Phys. Chem. B 2003, 107, 7957–7964. [Google Scholar]
- Asbury, J.B.; Hao, E.; Wang, Y.; Ghosh, H.N.; Lian, T. Ultrafast electron transfer dynamics from molecular adsorbates to semiconductor nanocrystalline thin films. J. Phys. Chem. B 2001, 105, 4545–4557. [Google Scholar]
- Zhang, G.; Xiao, L.; Chen, R.; Gao, Y.; Wang, X.; Jia, S. Single-molecule interfacial electron transfer dynamics manipulated by an external electric current. Phys. Chem. Chem. Phys 2011, 13, 13815–13820. [Google Scholar]
- Verberk, R.; van Oijen, A.M.; Orrit, M. Simple model for the power-law blinking of single semiconductor nanocrystals. Phys. Rev. B 2002, 66, 233202:1–233202:4. [Google Scholar]
- Kuno, M.; Fromm, D.; Johnson, S.; Gallagher, A.; Nesbitt, D. Modeling distributed kinetics in isolated semiconductor quantum dots. Phys. Rev. B 2003, 67, 125304:1–125304:15. [Google Scholar]
- Issac, A.; von Borczyskowski, C.; Cichos, F. Correlation between photoluminescence intermittency of CdSe quantum dots and self-trapped states in dielectric media. Phys. Rev. B 2005, 71, 161302:1–161302:4. [Google Scholar]
- Haase, M.; Hübner, C.G.; Reuther, E.; Herrmann, A.; Müllen, K.; Basché, T. Exponential and power-law kinetics in single-molecule fluorescence intermittency. J. Phys. Chem. B 2004, 108, 10445–10450. [Google Scholar]
- Haase, M.; Hübner, C.G.; Nolde, F.; Müllen, K.; Basché, T. Photoblinking and photobleaching of rylene diimide dyes. Phys. Chem. Chem. Phys 2011, 13, 1776–1785. [Google Scholar]
- Cichos, F.; von Borczyskowski, C.; Orrit, M. Power-law intermittency of single emitters. Curr. Opin. Colloid Interface Sci 2007, 12, 272–284. [Google Scholar]
- Treussart, F.; Alléaume, R.; Le Floc’h, V.; Xiao, L.; Courty, J.M.; Roch, J.F. Direct measurement of the photon statistics of a triggered single photon source. Phys. Rev. Lett. 2002, 89, 093601:1–093601:4. [Google Scholar]
- Song, H.; Reed, M.A.; Lee, T. Single molecule electronic devices. Adv. Mater 2011, 23, 1583–1608. [Google Scholar]
- Krevelen, D.W.; te Nijenhuis, K. Properties of Polymers: Their Correlation with Chemical Structure; Their Numerical Estimation and Prediction from Additive Group Contributions; Elsevier Science: Oxford, UK, 2009; p. 331. [Google Scholar]
- Chowdry, A.; Westgate, C. The role of bulk traps in metal-insulator contact charging. J. Phys. D Appl. Phys 1974, 7, 713–725. [Google Scholar]
- Murata, Y. Photoelectric emission and contact charging of some synthetic high polymers. Jpn. J. Appl. Phys 1979, 18, 1–8. [Google Scholar]
- Schönenberger, C. Charge flow during metal-insulator contact. Phys. Rev. B 1992, 45, 3861–3864. [Google Scholar]
- Holmlin, R.E.; Haag, R.; Chabinyc, M.L.; Ismagilov, R.F.; Cohen, A.E.; Terfort, A.; Rampi, M.A.; Whitesides, G.M. Electron transport through thin organic films in metal-insulator-metal junctions based on self-assembled monolayers. J. Am. Chem. Soc 2001, 123, 5075–5085. [Google Scholar]
- Arunkumar, E.; Forbes, C.C.; Noll, B.C.; Smith, B.D. Squaraine-derived rotaxanes: Sterically protected fluorescent near-IR dyes. J. Am. Chem. Soc 2005, 127, 3288–3289. [Google Scholar]
- Duke, C.; Fabish, T. Charge-induced relaxation in polymers. Phys. Rev. Lett 1976, 37, 1075–1078. [Google Scholar]
- Raju, G.G. Dielectrics in Electric Fields; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, R.; Gao, Y.; Zhang, G.; Wu, R.; Xiao, L.; Jia, S. Electric Field Induced Fluorescence Modulation of Single Molecules in PMMA Based on Electron Transfer. Int. J. Mol. Sci. 2012, 13, 11130-11140. https://doi.org/10.3390/ijms130911130
Chen R, Gao Y, Zhang G, Wu R, Xiao L, Jia S. Electric Field Induced Fluorescence Modulation of Single Molecules in PMMA Based on Electron Transfer. International Journal of Molecular Sciences. 2012; 13(9):11130-11140. https://doi.org/10.3390/ijms130911130
Chicago/Turabian StyleChen, Ruiyun, Yan Gao, Guofeng Zhang, Ruixiang Wu, Liantuan Xiao, and Suotang Jia. 2012. "Electric Field Induced Fluorescence Modulation of Single Molecules in PMMA Based on Electron Transfer" International Journal of Molecular Sciences 13, no. 9: 11130-11140. https://doi.org/10.3390/ijms130911130