Successful Treatment of Liver Aspergilloma by Caspofungin Acetate First-Line Therapy in a Non-Immunocompromised Patient
Abstract
:1. Introduction
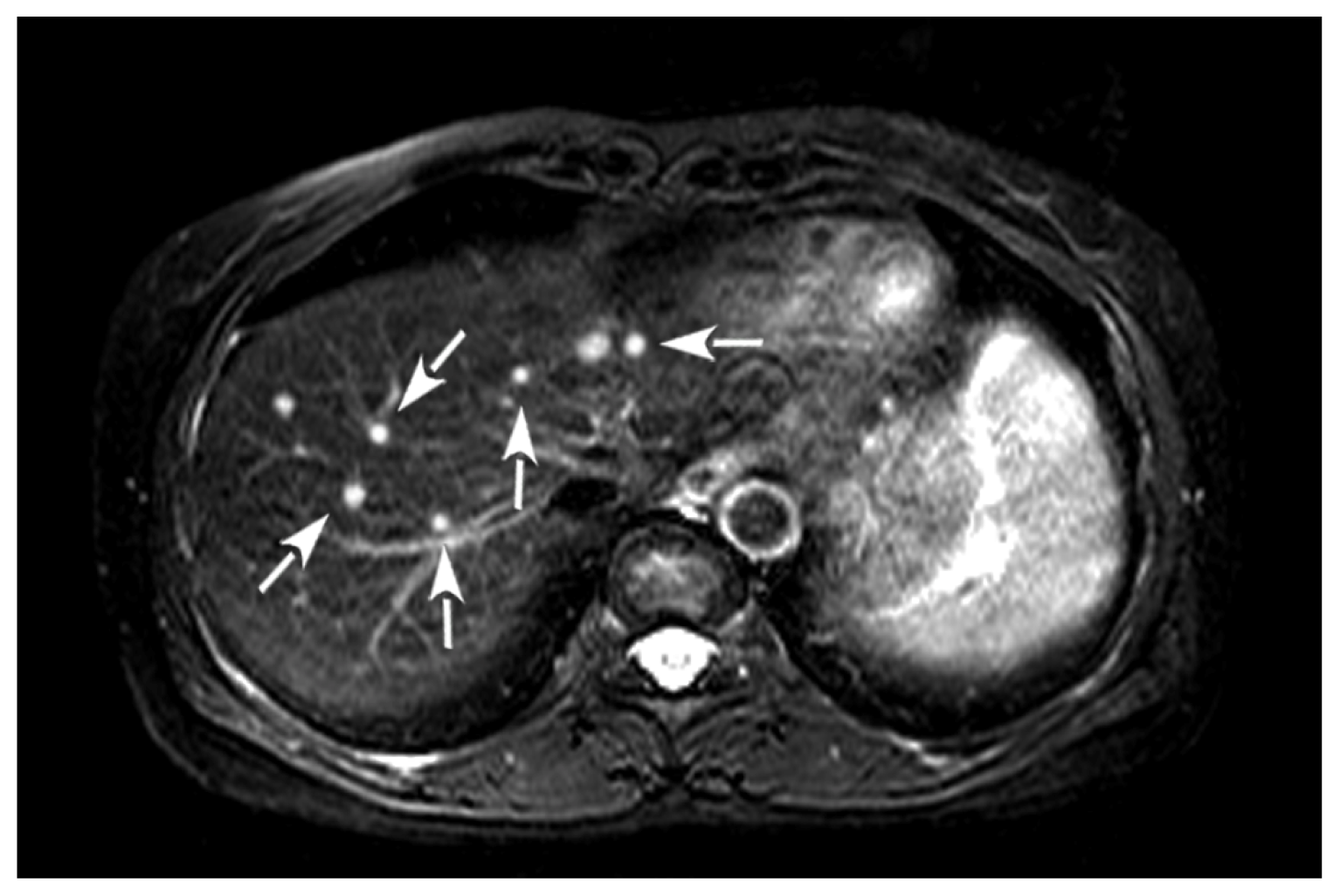
2. Case Presentation
3. Discussion
4. Experimental Methods
5. Conclusions
Acknowledgements
- Conflict of InterestThe authors declare no competing financial interests.
References
- Oren, I.; Goldstein, N. Invasive pulmonary aspergillosis. Curr. Opin. Pulm. Med 2002, 8, 195–200. [Google Scholar]
- Chen, S.; Pu, J.L.; Yu, J.; Zhang, J.M. Multiple Aspergillus cerebellar abscesses in a middle-aged female: Case report and literature review. Int. J. Med. Sci 2011, 8, 635–639. [Google Scholar]
- Rossouw, I.; Goedhals, D.; van der Merwe, J.; Stallenberg, V.; Govender, N. A rare, fatal case of invasive spinal aspergillosis in an antiretroviral-naive, HIV-infected man with pre-existing lung colonization. J. Med. Microbiol 2011, 60, 1534–1538. [Google Scholar]
- Wightman, S.C.; Kim, A.W.; Proia, L.A.; Faber, L.P.; Gattuso, P.; Warren, W.H.; Liptay, M.J. An unusual case of Aspergillus fibrosing mediastinitis. Ann. Thorac. Surg 2009, 88, 1352–1354. [Google Scholar]
- Huang, C.W.; Lee, M.A.; Lu, R.H.; Peng, H.C.; Chao, H.S. A case of pulmonary aspergilloma and actinomycosis. J. Med. Microbiol 2011, 60, 543–546. [Google Scholar]
- Locasciulli, A.; Oneto, R.; Bacigalupo, A.; Socié, G.; Korthof, E.; Bekassy, A.; Schrezenmeier, H.; Passweg, J.; Führer, M. Severe Aplastic Anemia Working Party of the European Blood and Marrow Transplant Group. Outcome of patients with acquired aplastic anemia given first line bone marrow transplantation or immunosuppressive treatment in the last decade: A report from the European Group for Blood and Marrow Transplantation (EBMT). Haematologica 2007, 92, 11–18. [Google Scholar]
- Girmenia, C.; Pizzarelli, G.; Pozzi, E.; Cimino, G.; Gentile, G.; Martino, P. Improving outcomes of acute invasive Aspergillus rhinosinusitis in patients with hematologic malignancies or aplastic anemia: The role of voriconazole. Haematologica 2008, 93, 159–160. [Google Scholar]
- Bond, R.; Walter, A.; Trigg, M.E. Severe aplastic anemia associated with hepatitis and complicated by pulmonary aspergillosis: Response to immune suppression and antifungal therapy. Del. Med. J 2002, 74, 339–343. [Google Scholar]
- Hori, A.; Kami, M.; Kishi, Y.; Machida, U.; Matsumura, T.; Kashima, T. Clinical significance of extra-pulmonary involvement of invasive aspergillosis: A retrospective autopsy-based study of 107 patients. J. Hosp. Infect 2002, 50, 175–182. [Google Scholar]
- Potenza, L.; Barozzi, P.; Rossi, G.; Palazzi, G.; Vallerini, D.; Riva, G.; Cellini, M.; Morselli, M.; Volzone, F.; Venturelli, C.; et al. Assessment of Aspergillus-specific T cells for diagnosis of invasive aspergillosis in a leukemic child with liver lesions mimicking hepatosplenic candidiasis. Clin. Vaccine Immunol 2008, 15, 1625–1628. [Google Scholar]
- Van der Velden, W.J.; Blijlevens, N.M.; Klont, R.R.; Donnelly, J.P.; Verweij, P.E. Primary hepatic invasive aspergillosis with progression after rituximab therapy for a post transplantation lymphoproliferative disorder. Ann. Hematol 2006, 85, 621–623. [Google Scholar]
- Lee, T.Y.; Yang, S.S.; Chen, G.H.; Hwang, W.L.; Lin, Y.H.; Hwang, J.I. Hepatic abscess caused by Aspergillus fumigatus infection following splenectomy and immunosuppressive therapy. J. Formos. Med. Assoc 2003, 102, 501–505. [Google Scholar]
- Lin, S.J.; Schranz, J.; Teutsch, S.M. Aspergillosis case-fatality rate: Systematic review of the literature. Clin. Infect. Dis 2001, 32, 358–366. [Google Scholar]
- Vandecasteele, S.J.; Boelaert, J.R.; Verrelst, P.; Graulus, E.; Gordts, B.Z. Diagnosis and treatment of Aspergillus flavus sternal wound infections after cardiac surgery. Clin. Infect. Dis 2002, 35, 887–890. [Google Scholar]
- Schwartz, S.; Ruhnke, M.; Ribaud, P.; Corey, L.; Driscoll, T.; Cornely, O.A.; Schuler, U.; Lutsar, I.; Troke, P.; Thiel, E. Improved outcome in central nervous system aspergillosis, using voriconazole treat-ment. Blood 2005, 106, 2641–2645. [Google Scholar]
- Zmeili, O.S.; Soubani, A.O. Pulmonary aspergillosis: A clinical update. QJM 2007, 100, 317–334. [Google Scholar]
- Jain, L.R.; Denning, D.W. The efficacy and tolerability of voriconazole in the treatment of chronic cavitary pulmonary aspergillosis. J. Infect 2006, 52, e133–e137. [Google Scholar]
- Izumikawa, K.; Ohtsu, Y.; Kawabata, M.; Takaya, H.; Miyamoto, A.; Sakamoto, S.; Kishi, K.; Tsuboi, E.; Homma, S.; Yoshimura, K. Clinical efficacy of micafungin for chronic pulmonary aspergillosis. Med. Mycol 2007, 45, 273–278. [Google Scholar]
- Denning, D.W. Echinocandins: A new class of antifungal. J. Antimicrob. Chemother 2002, 49, 889–891. [Google Scholar]
- Kirkpatrick, W.R.; Perea, S.; Coco, B.J.; Patterson, T.F. Efficacy of caspofungin alone and in combination with voriconazole in a Guinea pig model of invasive aspergillosis. Antimicrob. Agents Chemother 2002, 46, 2564–2568. [Google Scholar]
- Steinbach, W.J.; Perfect, J.R.; Schell, W.A.; Walsh, T.J.; Benjamin, D.K., Jr. In vitro analyses, animal models, and 60 clinical cases of invasive Aspergillus terreus infection. Antimicrob. Agents Chemother. 2004, 48, 3217–3225. [Google Scholar]
- Steinbach, W.J.; Benjamin, D.K., Jr; Kontoyiannis, D.P.; Perfect, J.R.; Lutsar, I.; Marr, K.A.; Lionakis, M.S.; Torres, H.A.; Jafri, H.; Walsh, T.J. Infections due to Aspergillus terreus: A multicenter retrospective analysis of 83 cases. Clin. Infect. Dis. 2004, 39, 192–198. [Google Scholar]
- Castagnola, E.; Machetti, M.; Cappelli, B.; Molinari, A.C.; Morreale, G.; Dodero, P.; Tomà, P.; Faraci, M. Caspofungin associated with liposomal amphotericin B or voriconazole for treatment of refractory fungal pneumonia in children with acute leukaemia or undergoing allogeneic bone marrow transplant. Clin. Microbiol. Infect 2004, 10, 255–257. [Google Scholar]
- Candoni, A.; Mestroni, R.; Damiani, D.; Tiribelli, M.; Michelutti, A.; Silvestri, F.; Castelli, M.; Viale, P.; Fanin, R. Caspofungin as first line therapy of pulmonary invasive fungal infections in 32 immunocompromised patients with hematologic malignancies. Eur. J. Haematol 2005, 75, 227–233. [Google Scholar]


| Age | Gender | Underlying disease | Immune status | Involvement | Diagnosis | Treatment | Course | Reference |
|---|---|---|---|---|---|---|---|---|
| 11 | Male | AML | Compromised | Liver | Biopsy | Caspofungin Amphotericin B | Cured | 10 |
| 53 | Female | NHL | Compromised | Liver | Biopsy | Voriconazole | Died | 11 |
| 66 | Male | Aplastic anemia | Compromised | Liver | Biopsy | Amphotericin B | Died | 12* |
| 55 | Female | Aplastic anemia | Non-compromised | Liver | Biopsy | Caspofungin | Cured | Our case |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bai, Q.-X.; Huan, Y.; Wang, J.-H.; Yang, L.-J.; Dong, H.-J. Successful Treatment of Liver Aspergilloma by Caspofungin Acetate First-Line Therapy in a Non-Immunocompromised Patient. Int. J. Mol. Sci. 2012, 13, 11063-11070. https://doi.org/10.3390/ijms130911063
Bai Q-X, Huan Y, Wang J-H, Yang L-J, Dong H-J. Successful Treatment of Liver Aspergilloma by Caspofungin Acetate First-Line Therapy in a Non-Immunocompromised Patient. International Journal of Molecular Sciences. 2012; 13(9):11063-11070. https://doi.org/10.3390/ijms130911063
Chicago/Turabian StyleBai, Qing-Xian, Yi Huan, Jian-Hong Wang, Li-Jie Yang, and Hong-Juan Dong. 2012. "Successful Treatment of Liver Aspergilloma by Caspofungin Acetate First-Line Therapy in a Non-Immunocompromised Patient" International Journal of Molecular Sciences 13, no. 9: 11063-11070. https://doi.org/10.3390/ijms130911063




