Effect of Maturity on Phenolics (Phenolic Acids and Flavonoids) Profile of Strawberry Cultivars and Mulberry Species from Pakistan
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Maturity on the Yield of Extract (%), Total Phenolics and Total Flavonoid Content in Strawberry and Mulberry Fruits
2.2. Effect of Maturity on Quantification of Flavonols and Phenolic Acid
3. Experimental Section
3.1. Collection of Samples
3.2. Reagents
3.3. Dry Matter Determination
3.4. Sample Extraction for Antioxidant Assay
3.5. Determination of Total Phenolics Content (TPC)
3.6. Determination of Total Flavonoids Content (TFC)
3.7. Extraction and Hydrolysis for Quantification
3.8. Instrumentation
3.9. Conditions Used for Phenolic Analysis
3.10. Statistical Analysis
4. Conclusions
References
- Iriti, M.; Faoro, F. Grape phytochemicals: A bouquet of old and new nutraceuticals for human health. Med. Hypotheses 2006, 67, 833–838. [Google Scholar]
- Zhang, Y.; Seeram, N.P.; Lee, R.; Feng, L.; Heber, D. Isolation and identification of strawberry phenolics with antioxidant and human cancer cell antiproliferative properties. J. Agric. Food Chem 2008, 6, 670–675. [Google Scholar]
- Cordenunsi, B.R.; Genovese, M.I.; Do-Nascimento, J.R.O.; Aymoto-Hassimotto, N.M.; Santos, R.J.D.; Lajolo, F.M. Effects of temperature on the chemical composition and antioxidant activity of three strawberry cultivars. Food Chem 2005, 91, 113–121. [Google Scholar]
- Reganold, J.P.; Andrews, P.K.; Reeve, J.R.; Carpenter-Boggs, L.; Schadt, C.W. Fruit and Soil Quality of Organic and Conventional Strawberry Agroecosystems. PLoS One 2010, 5, 1–14. [Google Scholar]
- Harborne, J.B.; Baxter, H.; Moss, G.P. Phytochemical dictionary. In Handbook of Bioactive Compounds from Plants, 2nd ed; Taylor and Francis: London, UK, 1999. [Google Scholar]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr 2000, 130, 2073–2085. [Google Scholar]
- Lodovici, M.; Guglielmi, F.; Meoni, M.; Dolara, P. Effect of natural phenolic acids on DNA oxidation in vitro. Food Chem. Toxicol 2001, 39, 1205–1210. [Google Scholar]
- Robbins, R. Phenolic acid in foods: An overview of analytical methodology. J. Agric. Food Chem 2003, 51, 2866–2887. [Google Scholar]
- Wang, S.Y.; Zheng, W. Effect of plant growth temperature on antioxidant capacity in strawberry. J. Agric. Food Chem 2001, 49, 4977–4982. [Google Scholar]
- Ayala-Zavala, J.F.; Wang, S.Y.; Wang, C.Y.; Gonzalez-Aguilar, G.A. Effect of storage temperatures on antioxidant capacity and aroma compounds in strawberry fruit. LWT Food Sci. Technol 2004, 37, 687–695. [Google Scholar]
- Serrano, M.; Guillen, F.; Martinez-Romero, D.; Castillo, S.; Valero, D. Chemical constituents and antioxidant activity of sweet cherry at different ripening stages. J. Agric. Food Chem 2005, 53, 2741–2745. [Google Scholar]
- Patel, P.R.; Rao, T.V.R. Physiological changes in relation to growth and ripening of khirni [Manilkara hexandra (Roxb.) Dubard] fruit. Fruits 2009, 64, 139–146. [Google Scholar]
- Pineli, L.L.O.; Moretti, C.L.; Santos, M.S.; Campos, A.B.; Brasileiro, A.V.; Cordova, A.C; Chiarello, M.D. Antioxidants and other chemical and physical characteristics of two strawberry cultivars at different ripeness stages. J. Food Compos. Anal. 2011, 92, 831–838. [Google Scholar]
- Yang, J.; Gadi, R.; Thomson, T. Antioxidant capacity, total phenols, and ascorbic acid content of noni (Morinda citrifolia) fruits and leaves at various stages of maturity. Micronesica 2011, 41, 167–176. [Google Scholar]
- Shahidi, F.; Naczk, M. Phenolic compounds in fruits and vegetables. In Phenolics in Food and Nutraceutical; CRC Press: Tulsa, OK, USA, 2004; pp. 131–156. [Google Scholar]
- Dinelli, G.; Bonetti, A.; Minelli, M.; Marotti, I.; Catizone, P.; Mazzanti, A. Content of flavonols in Italian bean (Phaseolus vulgaris L.) ecotypes. Food Chem 2006, 90, 105–114. [Google Scholar]
- Justesen, U.; Knethsen, P. Composition of flavonoids in fresh herbs and calculation of flavonoids intake by use of herbs in traditional danish dishes. Food Chem 2001, 73, 245–250. [Google Scholar]
- Mabberley, D.J. The Plant-Book. A Portable Dictionary of the Higher Plants; Cambridge University Press: Cambridge, MA, USA, 1987. [Google Scholar]
- Scott, R.R.; Lei, Z.; Tong, W. Assessments of Commodity and Trade Issues Made by USDA Staff and not Necessarily Statements of Official U.S. Government Policy; GAIN Report Number: 10043; USDA Foreign Agricultural Service: Washington, D.C., USA, 2010. [Google Scholar]
- Azodanlou, R.; Darbellay, C.; Luisier, J.L.; Villettaz, J.C.; Amado, R. Quality assessment of strawberries (Fragaria species). J. Agric. Food Chem 2003, 51, 715–721. [Google Scholar]
- Mitcham, E.J. Strawberry. In The Commercial Storage of Fruits, Vegetables, and Florist and Nursery Crops; Gross, K.C., Wang, C.Y., Saltveit, M.E., Eds.; U.S. Department of Agriculture, Agricultural Research Service: Beltsville, MD, USA, 2004. [Google Scholar]
- Spayd, S.E.; Morris, J.R. Physical and chemical characteristics of puree from once-over harvested strawberries. J. Amer. Soc. Hort. Sci 1981, 106, 101–105. [Google Scholar]
- Terry, L.A.; Joyce, D.C.; Adikaram, N.K.B.; Khambay, B.P.S. Preformed antifungal compounds in strawberry fruit and flower tissues. Postharvest Biol. Technol 2004, 31, 201–212. [Google Scholar] [Green Version]
- Robards, K.; Antolovich, M. Analytical chemistry of fruit bioflavonoids. A review. Analyst 1997, 122, 11–34. [Google Scholar]
- Aherne, S.A.; OBbrien, N.M. Dietary flavonols: chemistry, food content, and metabolism. Nutrition 2002, 18, 75–81. [Google Scholar]
- Zadernowski, R.; Naczk, M.; Nesterowicz, J. Phenolic acid profiles in small berries. J. Agric. Food Chem 2005, 53, 2118–2124. [Google Scholar]
- Arabshahi-Delouee, S.; Urooj, A. Antioxidant properties of various solvent extracts of mulberry (Morus indica L.) leaves. Food Chem 2007, 102, 1233–1240. [Google Scholar]
- Ercisli, S.; Orhan, E. Chemical composition of white (Morus alba), red (Morus rubra) and black (Morus nigra) mulberry fruits. Food Chem 2007, 103, 1380–1384. [Google Scholar]
- Imran, M.; Talpur, F.N.; Jan, M.S.; Khan, A.; Khan, I. Analysis of nutritional components of some wild edible plants. J. Chem. Soc. Pak 2007, 29, 500–508. [Google Scholar]
- Sass-Kiss, A. Differences in anthocyanin and carotenoids content of fruits and vegetables. Food Res. Int 2005, 38, 1023–1029. [Google Scholar]
- Cieslik, E.; Greda, A.; Adamus, W. Contents of polyphenols in fruit and vegetables. Food Chem. 2006, 94, 135–142. [Google Scholar]
- Lin, J.Y.; Tang, C.Y. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem 2007, 101, 140–147. [Google Scholar]
- Kim, D.O.; Heo, H.J.; Kim, Y.J.; Yang, H.S.; Lee, C.Y. Sweet and sour cherry phenolics and their protective effects on neuronal cells. J. Agric. Food Chem 2005, 53, 9921–9927. [Google Scholar]
- Jakobek, L.; Seruga, M.; Medvidović-Kosanović, M.; Novak, I. Anthocyanin content and antioxidant activity of various red fruit juices. Dtsch. Lebensm. -Rundsch 2007, 103, 58–64. [Google Scholar]
- Jakobek, L.; Seruga, M.; Novak, I.; Medvidovic-Kosanovic, M. Flavonols, phenolic acids and antioxidant activity of some red fruits. Dtsch. Lebensm. -Rundsch 2007, 103, 369–378. [Google Scholar]
- Matilla, P.; Hellstrom, J.; Törrönen, R. Phenolic acids in berries, fruits and beverages. J. Agric. Food Chem 2006, 54, 7193–7199. [Google Scholar]
- Franke, A.A.; Custer, L.J.; Arakaki, C.; Murphy, S.P. Vitamin C and flavonoid levels of fruits and vegetables consumed in Hawaii. J. Food Compos. Anal. 2004, 17, 1–35. [Google Scholar]
- Olsson, M.E.; Gustavsson, K.; Andersson, S.; Nilsson, A.; Duan, R. Inhibition of cancer cell proliferation in vitro by fruit and berry extracts and correlations with antioxidant levels. J. Agric. Food Chem 2004, 52, 7264–7271. [Google Scholar]
- Sultana, B.; Anwar, F. Flavonols (kaempferol, quercetin, myricetin) contents of selected fruits, vegetables and medicinal plants. Food Chem 2008, 108, 879–884. [Google Scholar]
- Bohm, V.; Kuhnert, S.; Rohm, H.; Scholze, G. Improving the nutritional quality of microwave-vacuum dried strawberries: A preliminary study. Food Sci. Technol. Int 2006, 12, 67–75. [Google Scholar]
- Piljac-Zegarac, J.; Samec, D. Antioxidant stability of small fruits in postharvest storage at room and refrigerator temperatures. Food Res. Int 2011, 44, 345–350. [Google Scholar]
- Bae, S.H.; Suh, H.J. Antioxidant activities of five different mulberry cultivars in Korea. LWT Food Sci. Technol 2007, 40, 955–962. [Google Scholar]
- Imran, M.; Khan, H.; Shah, M.; Khan, R.; Khan, F. Chemical composition and antioxidant activity of certain Morus species. J. Zhejiang Univ. Sci. B 2010, 11, 973–980. [Google Scholar]
- Gungor, N.; Sengul, M. Antioxidant activity, total phenolic content and selected physicochemical properties of white mulberry (Morus alba. L.) fruits. Int. J. Food Prop 2008, 11, 44–52. [Google Scholar]
- Scalzo, J.; Politi, A.; Pellegrini, N.; Mezzetti, B.; Battino, M. Plant genotype affects total antioxidant capacity and phenolic contents in fruit. Nutrition 2005, 21, 207–213. [Google Scholar]
- Hakkinen, S.H.; Torronen, A.R. Content of flavonols and selected phenolic acids in strawberries and Vaccinium species: Influence of cultivar, cultivation site and technique. Food Res. Int 2000, 33, 517–524. [Google Scholar]
- Aminah, A.; Anna, P.K. Influence of ripening stages on physicochemical characteristics and antioxidant properties of bitter gourd (Momordica charantia). Int. Food Res. J 2011, 18, 895–900. [Google Scholar]
- Ferreira, I.C.F.R.; Baptista, P.; Vilas-Boas, M.; Barros, L. Free radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal. Food Chem 2007, 100, 1511–1516. [Google Scholar]
- Lugasi, A.; Hovari, J. Antioxidant properties of commercial alcoholic and nonalcoholic beverages. Nahrung 2003, 47, 79–86. [Google Scholar]
- Kevers, C.; Falkowski, M.; Tabart, J.; Defraigne, J.; Dommes, J.; Pincemail, J. Evolution of antioxidant capacity during storage of selected fruits and vegetables. J. Agric. Food Chem 2007, 55, 8596–8603. [Google Scholar]
- Hakkinen, S.; Heinonen, M.; Karenlampi, S.; Mykkanen, H.; Ruuskanen, J.; Torronnen, R. Screening of selected flavonoids and phenolic acids in 19 berries. Food Res. Int 1999, 32, 345–353. [Google Scholar]
- Kahkonen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem 1999, 47, 3954–3962. [Google Scholar]
- Maatta-Riihinen, M.; Kamal-Eldin, A.; Torronen, A.R. Identification and quantification of phenolic compounds in berries of Fragaria and Rubus species (Family Rosaceae). J. Agric. Food Chem 2004, 52, 6178–6187. [Google Scholar]
- Stohr, H.; Herrmann, K. The phenolics of fruits, the phenolics of strawberries and their changes during development and ripeness of the fruits. Z Lebensm-Unters Forsch 1975b, 159, 341–348. [Google Scholar]
- Kosar, M.; Kafkas, E.; Paydas, S.; Baser, K.H.C. Phenolic composition of strawberry genotypes at different maturation stages. J. Agric. Food Chem 2004, 52, 1586–1589. [Google Scholar]
- Ndri, D.; Calani, L.; Mazzeo, T.; Scazzina, F.; Rinaldi, M.; Rio, D.D.; Pellegrini, N.; Brighenti, F. Effects of different maturity stages on antioxidant content of Ivorian Gnagnan (Solanum indicum L.) berries. Molecules 2010, 15, 7125–7138. [Google Scholar]
- Vasco, C.; Riihinen, K.; Ruales, J.; Kamal-Eldin, A. Chemical composition and phenoliccompound profile of mortiño (Vaccinium floribundum Kunth). J. Agric. Food Chem 2009, 57, 8274–8281. [Google Scholar]
- Hernanz, D.; Recamales, A.F.; Melendez-Martinez, A.J.; Gonzalez-Miret, M.L.; Heredia, F.J. Assessment of the differences in the phenolic composition of five strawberry cultivars (Fragaria ×ananassa Duch.) grown in two different soilless systems. J. Agric. Food Chem 2007, 55, 1846–1852. [Google Scholar]
- Häkkinen, S.H.; Kärenlampi, S.O.; Mykkänen, H.M.; Törrönen, A.R. Influence of domestic processing and storage on flavonol contents in berries. J. Agric. Food Chem 2000, 48, 2960–2965. [Google Scholar]
- McDonald, M.; Hughes, M.; Burns, J. Survey of the free and conjugated myricetin and quercetin content of red wines of different geographical origins. J. Agric. Food Chem 1998, 46, 368–375. [Google Scholar]
- Lakenbrink, C.; Lapczynski, S.; Maiwald, B.; Engelhardt, U.H. Flavonoids and other polyphenols in consumer brews of tea and other caffeinated beverages. J. Agric. Food Chem 2000, 48, 2848–2852. [Google Scholar]
- Trichopoulou, A.; Vasilpoulou, E.; Hollman, P. Nutritional composition and flavonoid content of edible wild greens and green pies: A potential rich source of antioxidant nutrients in the Mediterranean diet. Food Chem 2000, 70, 319–323. [Google Scholar]
- Vuorinen, H.; Maata, K.; Torronen, R. Content of the flavonols myricetin, quercetin, and kaempferol in Finnish berry wines. J. Agric. Food Chem 2000, 48, 2675–2680. [Google Scholar]
- Soleas, G.J.; Diamandis, E.P.; Goldberg, D.M. Resveratrol: a molecule whose time has come? And gone? Clin. Biochem 1997, 30, 91–113. [Google Scholar]
- Bilyk, A.; Sapers, G.M. Varietal differences in the quercetin, kaempferol, and myricetin contents of highbush blueberry, cranberry, and thornless blackberry. J. Agric. Food Chem 1986, 34, 585–588. [Google Scholar]
- Memon, A.A.; Najma, M.; Luthria, D.L.; Bhanger, M.I.; Pitafi, A.A. Phenolic acids profiling and antioxidant potential of mulberry (Morus laevigata W., Morus nigra L., Morus alba L.) leaves and fruits grown in Pakistan. Pol. J. Food Nutr. Sci 2010, 60, 25–32. [Google Scholar]
- Voca, S.; Dobricevic, N.; Dragovic-Uzelac, V.; Duralija, B.; Druzic, J. Fruit quality of new early ripening strawberry cultivars in Croatia. Food Technol. Biotechnol 2008, 46, 292–298. [Google Scholar]
- Jamil, A.; Anwar, F.; Ashraf, M. Plant Tolerance to Biotic and Abiotic Stresses through Modern Genetic Engineering. In Crops, Growth, Quality and Biotechnology; Dris, R., Ed.; WFL Publisher: Helsinki, Finland, 2005; pp. 1276–1299. [Google Scholar]
- Ali, Q.; Ashraf, M. Exogenously applied glycine, betaine enhances seed and seed oil quality of maize (Zea mays L.) under water deficit conditions. Environ. Exp. Bot 2011, 71, 249–259. [Google Scholar]
- Karjalainen, R.; Lehtinen, A.; Hietaniemi, V.; Pihlava, J.M.; Jokinen, K.; Keinänen, M.; Julkunen-Tiito, R. Benzothiadiazole and glycine betaine treatments enhance phenolic compound production in strawberry. Acta Hortic 2002, 567, 353–356. [Google Scholar]
- Chaovanalikit, A.; Wrolstad, R.E. Total anthocyanins and total phenolics of fresh and processed cherries and their antioxidant properties. J. Food Sci 2004, 69, 67–72. [Google Scholar]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem 2002, 50, 3010–3014. [Google Scholar]
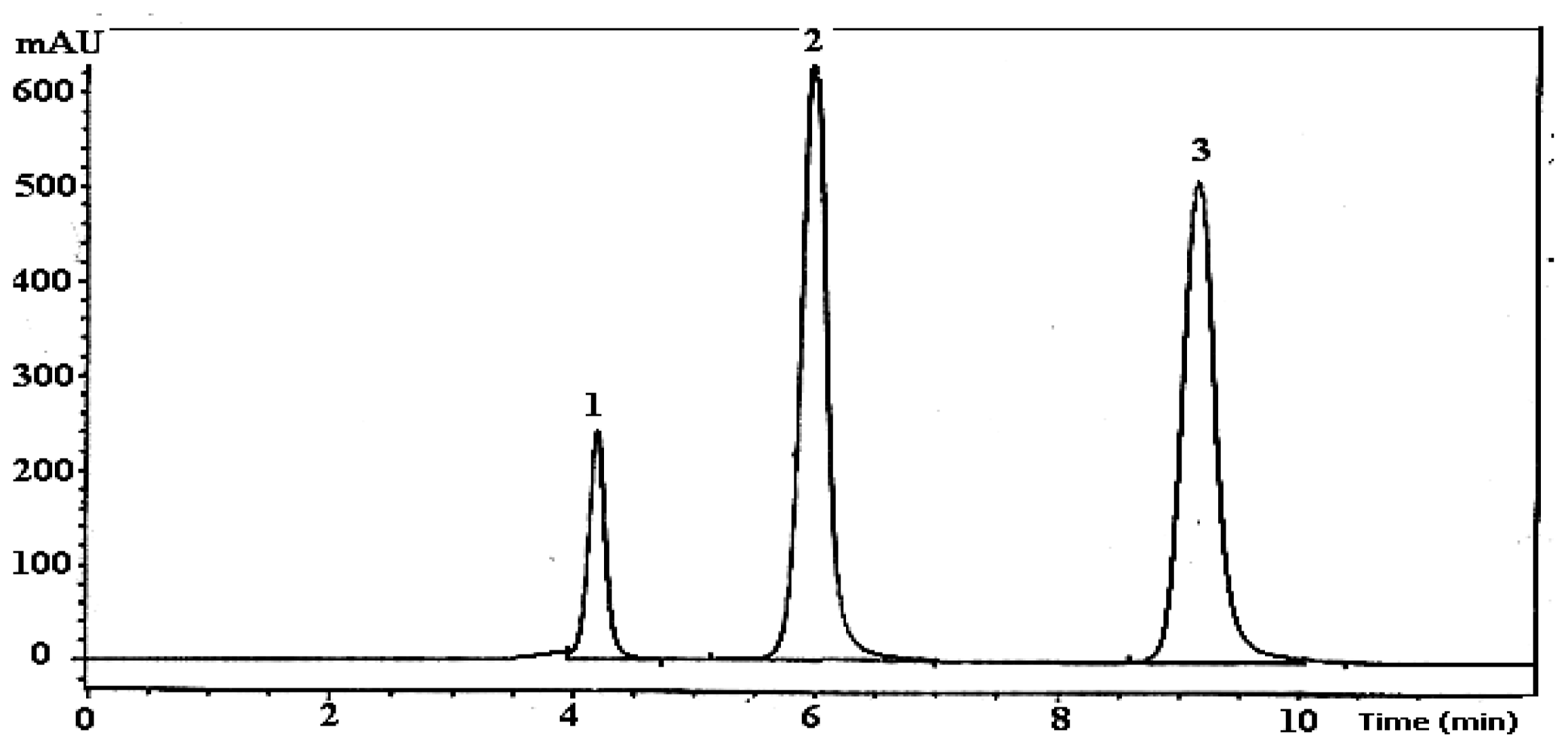

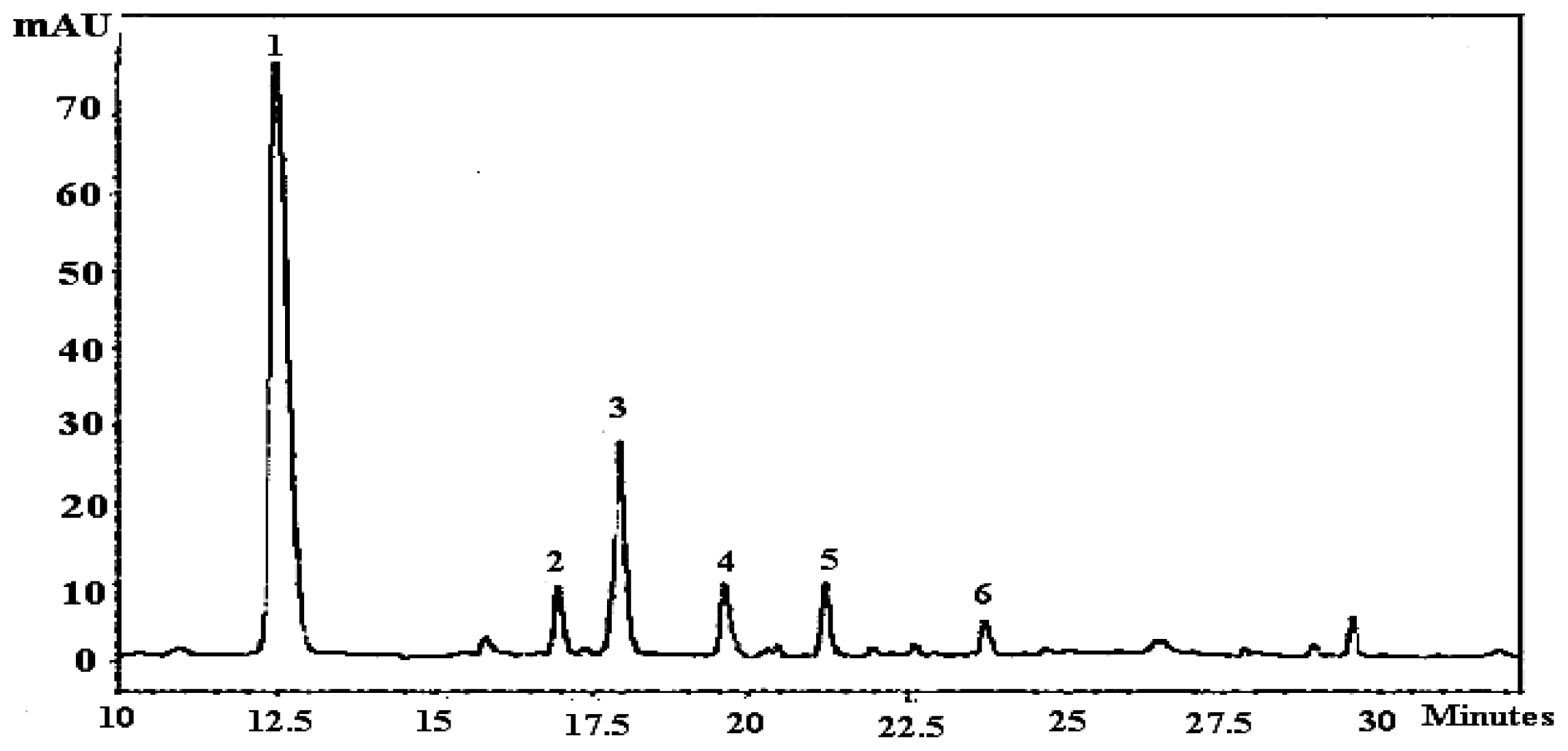

| Fruits | Species | Maturity stages | % extraction yield | Total Phenolics A | Total flavonoids B |
|---|---|---|---|---|---|
| Strawberry | Korona | Un-ripened | 08.9 ± 1.1 c | 581 ± 18 c | 123 ± 10 c |
| Semi-ripened | 31.6 ± 3.2 b | 938 ± 42 b | 174 ± 15 b | ||
| Fully-ripened | 53.3 ± 4.8 a | 1884 ± 69 a | 327 ± 17 a | ||
| Tufts | Un-ripened | 08.5 ± 1.6 c | 491 ± 22 c | 83 ± 06 b | |
| Semi-ripened | 24.4 ± 1.7 b | 794 ± 44 b | 98 ± 07 b | ||
| Fully-ripened | 43.4 ± 2.5 a | 1662 ± 88 a | 197 ± 07 a | ||
| Mulberry | M. laevigata | Un-ripened | 14.6 ± 1.0 c | 201 ± 07 c | 304 ± 16 c |
| Semi-ripened | 33.3 ± 2.7 b | 466 ± 17 b | 559 ± 34 b | ||
| Fully-ripened | 52.3 ± 2.3 a | 1803 ± 67 a | 615 ± 26 a | ||
| M. macroura | Un-ripened | 12.3 ± 1.2 c | 219 ± 06 c | 145 ± 09 c | |
| Semi-ripened | 35.4 ± 3.6 b | 508 ± 14 b | 282 ± 07 a | ||
| Fully-ripened | 54.0 ± 4.2 a | 2067 ± 67 a | 249 ± 08 b | ||
| M. nigra | Un-ripened | 11.4 ± 0.8 b | 395 ± 17 c | 245 ± 06 c | |
| Semi-ripened | 24.0 ± 2.6 a | 1722 ± 37 b | 706 ± 32 b | ||
| Fully-ripened | 28.3 ± 2.1 a | 2287 ± 41 a | 1021 ± 75 a | ||
| M. alba | Un-ripened | 06.9 ± 0.7 c | 575 ± 12 c | 110 ± 07 c | |
| Semi-ripened | 30.8 ± 2.8 b | 1071 ± 31 b | 392 ± 06 b | ||
| Fully-ripened | 40.2 ± 2.0 a | 1872 ± 57 a | 625 ± 17 a |
| Strawberry (Korona) | Strawberry (Tufts) | |||||
|---|---|---|---|---|---|---|
| Flavonols | Un-ripened | Semi-ripened | Fully-ripened | Un-ripened | Semi-ripened | Fully-ripened |
| Myricetin | 13.2 ± 0.5 c | 28.5 ± 1.2 a | 20.8 ± 0.5 b | 12.8 ± 0.3 c | 20.1 ± 0.8 b | 23.1 ± 1.4 a |
| Quercetin | 1.4 ± 0.1 c | 5.5 ± 0.1 b | 5.5 ± 0.1 b | 5.9 ± 0.9 b | 11.2 ± 0.4 a | 10.9 ± 0.8 a |
| Kaempferol | 19.9 ± 1.0 b | 18.4 ± 0.9 b | 98.1 ± 3.2 a | 23.2 ± 0.8 c | 31.2 ± 1.7 b | 78.6 ± 2.8 a |
| Total flavonols | 34.5 | 52.4 | 128.5 | 41.9 | 62.5 | 112.6 |
| Phenolic acid | ||||||
| p-coumaric | 22.2± 1.5 c | 31.4 ± 2.0 b | 47.5 ± 2.9 a | 17.3 ± 1.0 b | 18.5 ± 1.4 b | 34.9 ± 2.7 a |
| p-hydroxy-benzoic | 33.6 ± 2.4 c | 52.1 ± 3.5 b | 65.4 ± 4.1 a | 21.4 ± 1.8 c | 38.9 ± 2.5 b | 47.9 ± 3.4 a |
| Chlorogenic | 11.5 ± 0.4 b | 12.7 ± 0.8 b | 18.2 ± 1.4 a | 14.2 ± 0.6 b | 16.9 ± 0.8 a | 12.2 ± 0.7 c |
| Ferulic | 8.4 ± 0.5 c | 12.5 ± 1.1 b | 17.3 ± 1.3 a | 7.6 ± 0.8 c | 12.5 ± 0.9 b | 24.1 ± 1.5 a |
| Gallic | 8.6 ± 0.5 c | 15.3 ± 1.4 b | 22.8 ±1.9 a | 6.8 ± 0.5 c | 10.2 ± 0.8 b | 24.6 ± 1.9 a |
| Vanillic | ND | 3.8 ± 0.1 b | 16.1 ± 1.0 a | ND | 2.8 ± 0.4 b | 11.8 ± 0.9 a |
| ∑ HBA | 42.12 | 71.23 | 104.29 | 35.79 | 51.96 | 84.39 |
| ∑ HCA | 42.07 | 56.65 | 82.93 | 38.98 | 47.88 | 71.11 |
| ∑ PHA | 84.19 | 127.88 | 187.22 | 74.77 | 99.84 | 155.50 |
| M. laevigata | M. macroura | |||||
|---|---|---|---|---|---|---|
| Flavonols | Un-ripened | Semi-ripened | Fully-ripened | Un-ripened | Semi-ripened | Fully-ripened |
| Myricetin | 11.5 ± 0.8 b | 22.3 ± 0.5 a | 20.0 ± 1.7 a | 11.5 ± 0.9 c | 19.2 ± 1.3 b | 22.5 ± 1.3 a |
| Quercetin | 7.0 ± 0.7 c | 88.8 ± 4.8 b | 145.7 ± 8.4 a | 7.0 ± 0.2 c | 13.9 ± 0.8 b | 21.7 ± 0.6 a |
| Kaempferol | 9.8 ± 0.6 c | 32.2 ± 2.5 b | 56.1 ± 3.9 a | 9.8 ± 0.6 a | 9.8 ± 0.3 a | 8.1 ± 0.4 b |
| Total flavonols | 28.3 | 143.3 | 221.8 | 28.3 | 42.9 | 52.3 |
| Phenolic acid | ||||||
| p-coumaric | ND | 15.9 ± 1.1 b | 27.3 ± 2.5 a | 5.1 ± 0.7 b | 3.2 ± 0.4 c | 13.2 ± 1.3 a |
| p-hydroxy-benzoic | 1.1 ± 0.1 c | 4.1 ± 0.2 b | 7.3 ± 0.7 a | 5.1 ± 0.5 c | 17.5± 1.3 b | 24.1 ± 1.8 a |
| Chlorogenic | 3.4 ± 0.4 c | 7.1 ± 0.8 b | 12.9 ± 1.1 a | 4.2 ± 0.2 c | 13.2 ± 0.7 b | 23.2 ± 1.7 a |
| Ferulic | 12.4 ± 0.9 b | ND | 17.2 ± 0.9 a | 6.3 ± 0.3 b | 7.7 ± 0.5 b | 13.4 ± 1.1 a |
| Gallic | 5.2 ± 0.6 c | 8.8 ± 0.9 b | 14.2 ± 1.4 a | 4.2 ± 0.5 b | 3.5 ± 0.9 b | 9.8 ± 0.7 a |
| Vanillic | 8.5 ± 0.2 c | 13.3 ± 0.8 b | 21.1 ± 0.9 a | 3.2 ± 0.5 b | 1.9 ± 0.6 c | 16.1 ± 1.8 a |
| ∑ HBA | 14.8 | 22.0 | 42.6 | 12.5 | 22.9 | 50.0 |
| ∑ HCA | 15.8 | 26.2 | 57.4 | 15.6 | 24.1 | 49.8 |
| ∑ PHA | 30.6 | 48.2 | 100.0 | 28.1 | 47.0 | 99.8 |
| M. nigra | M. alba | |||||
| Flavonols | Un-ripened | Semi-ripened | Fully-ripened | Un-ripened | Semi-ripened | Fully-ripened |
| Myricetin | 52.6 ± 3.1 c | 63.3 ± 4.7 a | 56.1 ± 4.8 b | 45.0± 2.2 c | 78.1 ± 3.7 b | 88.4 ± 4.8 a |
| Quercetin | 8.1 ± 0.6 c | 43.5 ± 2.4 a | 11.7 ± 0.8 b | 1.3 ± 0.1 a | 1.3 ± 0.1 a | 0.7 ± 0.1 b |
| Kaempferol | 8.6 ± 0.7 c | 56.6 ± 3.5 a | 31.7 ± 1.9 b | 15.0± 0.1 a | 6.3 ± 0.1 b | 5.2 ± 0.2 b |
| Total flavonols | 69.3 | 163.4 | 99.5 | 61.3 | 85.7 | 94.3 |
| Phenolic acid | ||||||
| p-coumaric | 4.2 ± 0.5 c | 7.6 ± 0.6b | 21.2 ± 1.1 a | 1.1 ± 0.1 b | 2.0 ± 0.2 b | 4.0 ± 0.3 a |
| p-hydroxy-benzoic | ND | ND | 5.3 ± 0.5 a | 3.2 ± 0.5 b | 12.1 ± 1.2 a | 13.3 ± 1.2 a |
| Chlorogenic | 2.6 ± 0.5 b | 3.4 ± 0.2 b | 6.8 ± 0.1 a | 5.3 ± 0.4 c | 9.0 ± 0.7 b | 17.3 ± 1.8 a |
| Ferulic | ND | 2.4 ± 0.4 b | 7.5 ± 0.8 a | ND | ND | ND |
| Gallic | 2.5 ± 0.4 b | 3.9 ± 0.4 b | 8.3 ± 0.9 a | 3.6 ± 0.5 c | 6.2 ± 0.8 b | 8.1 ± 0.5 a |
| Vanillic | 6.1 ± 0.7 c | 10.2 ± 0.9 b | 18.3 ± 1.5 a | 1.7± 0.0 b | 2.2 ± 0.1 b | 5.7 ± 0.5 a |
| ∑ HBA | 8.6 | 14.1 | 31.9 | 8.5 | 20.5 | 27.1 |
| ∑ HCA | 6.8 | 13.4 | 35.5 | 6.4 | 11.0 | 21.3 |
| ∑ PHA | 15.4 | 27.5 | 67.4 | 14.9 | 31.5 | 48.4 |
| Fruits | Cultivar/Species | Un-ripened | Semi-ripened | Fully-ripened |
|---|---|---|---|---|
| Strawberry | Korona | Green/hard | Reddish green/semi-soft | Red/soft |
| Tufts | Green/hard | Reddish green/semi-soft | Red/soft | |
| Long mulberry | M. laevigata | Light Green/hard | Red/semi-soft | Black/soft |
| M. macroura | Light Green/hard | Light yellow/semi-soft | Off-white/soft | |
| Small mulberry | M. nigra | Light Green/hard | Red/semi-soft | Black/soft |
| M. alba | Light Green/hard | Light yellow/semi-soft | Off-white/soft |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mahmood, T.; Anwar, F.; Abbas, M.; Saari, N. Effect of Maturity on Phenolics (Phenolic Acids and Flavonoids) Profile of Strawberry Cultivars and Mulberry Species from Pakistan. Int. J. Mol. Sci. 2012, 13, 4591-4607. https://doi.org/10.3390/ijms13044591
Mahmood T, Anwar F, Abbas M, Saari N. Effect of Maturity on Phenolics (Phenolic Acids and Flavonoids) Profile of Strawberry Cultivars and Mulberry Species from Pakistan. International Journal of Molecular Sciences. 2012; 13(4):4591-4607. https://doi.org/10.3390/ijms13044591
Chicago/Turabian StyleMahmood, Tahir, Farooq Anwar, Mateen Abbas, and Nazamid Saari. 2012. "Effect of Maturity on Phenolics (Phenolic Acids and Flavonoids) Profile of Strawberry Cultivars and Mulberry Species from Pakistan" International Journal of Molecular Sciences 13, no. 4: 4591-4607. https://doi.org/10.3390/ijms13044591
APA StyleMahmood, T., Anwar, F., Abbas, M., & Saari, N. (2012). Effect of Maturity on Phenolics (Phenolic Acids and Flavonoids) Profile of Strawberry Cultivars and Mulberry Species from Pakistan. International Journal of Molecular Sciences, 13(4), 4591-4607. https://doi.org/10.3390/ijms13044591




