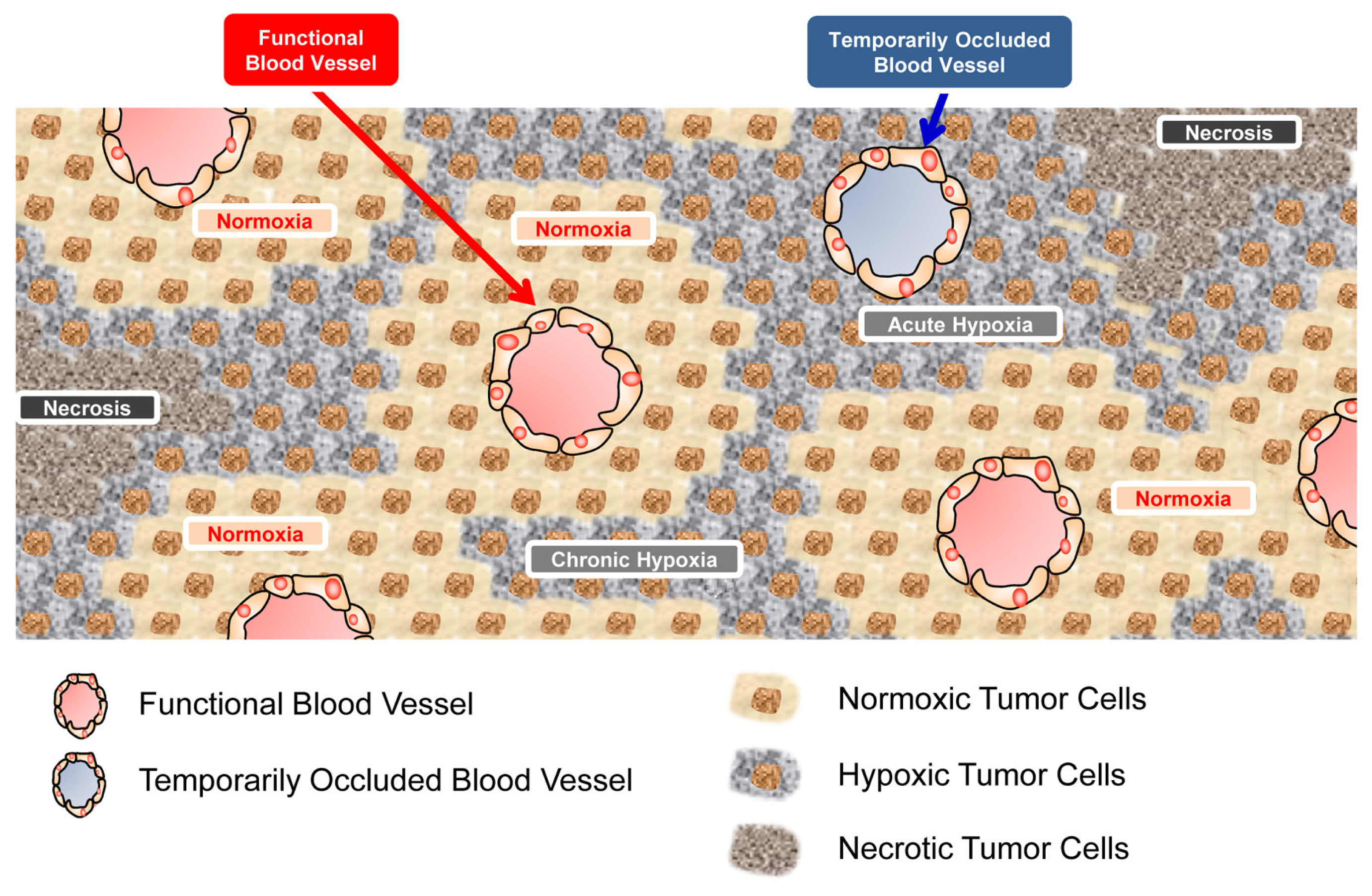
2.3. Glucose-Availability in a Micro Tumor Cord
Glucose is a simple sugar that primarily functions as an energy source providing adenosine triphosphate (ATP). The availability of glucose in a malignant solid tumor is one of the local regulators which affect the growth of individual cancer cells and the progression of whole tumors. Because of the limited distance that glucose as well as oxygen can diffuse from tumor blood vessels, cancer cells farther away from blood vessels cannot get enough glucose [
31]. We revealed that the decrease of glucose-availability results in the suppression of HIF-1α expression in pimonidazole-positive/HIF-1α-negative regions in a tumor cord, as described above (See Section 2.1) [
31]. So, how heterogeneous is glucose-availability/glucose-distribution in a malignant solid tumor? Walenta
et al. conducted metabolic mapping in both tumor xenografts and clinical biopsies with quantitative bioluminescence and single photon imaging techniques [
48]. They suggested a significant reduction in ATP and glucose especially in perinecrotic regions. Another group demonstrated the same possibility. When the glucose availability in perinecrotic regions of a micro tumor cord was increased through continuous administration of glucagon by using an osmotic pump, the expression of HIF-1α in these regions was significantly increased [
31]. In addition, we revealed, by performing an
in vitro experiment, that HIF-1α expression, HIF-1 activity, and the expression of HIF-1 downstream genes were suppressed when the glucose concentration in the culture medium was reduced to 0.45 g/L (2.49 mM) even under 1% and 0.02% oxygen conditions [
28,
31]. These results directly indicate that cancer cells in HIF-1α-positive/pimonidazole-negative regions should be supplied with enough glucose, at least higher than 0.45 g/L, for the expression of HIF-1α. On the other hand, cancer cells in perinecrotic/pimonidazole-positive regions are under low glucose conditions, less than 0.45 g/L (2.49 mM), resulting in a decreased level of HIF-1α even under hypoxic conditions (
Table 3).
A relationship between the intra-tumor heterogeneity of glucose availability and tumor metastasis was also demonstrated by using clinical data on head and neck carcinomas [
48]. There was a significant difference between the metastatic group and non-metastatic group with regard to the glucose content of primary lesions. Interestingly, the glucose level of the metastatic group was significantly lower. This study implies that glucose-availability in primary tumors might be associated with a poor prognosis. In addition to glucose-availability, the expression of a glucose transporter gene, which functions in glucose-uptake into cells, is recognized as an important prognostic factor.
2.4. pH in a Micro Tumor Cord
Several studies have shown that the extracellular pH in human tumors as well as experimental xenografts is low, around 6.0. Such a microenvironment is, at least in part, caused by CO
2 and lactate produced by and secreted from cancer cells through accelerated glycolysis and lactic acid fermentation [
6]. Because hypoxia is one of the most remarkable triggers for the acceleration [
49], there is a possibility that intratumoral pH mapping is affected by the distance from functional tumor blood vessels. Cancer cells shift their metabolic pathway from mitochondrial oxidative phosphorylation to glycolysis and lactic acid fermentation so as to adjust their oxygen-demand to meet the limited oxygen-supply under hypoxic conditions [
50,
51]. HIF-1 has been reported to play important roles in the metabolic reprogramming through the following functions. First, HIF-1 upregulates glucose metabolism. HIF-1 induces the expression of genes involved in glucose uptake (such as GLUT1 and GLUT3) [
52] and enzymatic breakdown of glucose to pyruvate (such as HK2) [
53]. Furthermore, due to the increase of lactate dehydrogenase A (LDH-A) expression, HIF-1 facilitates lactic acid fermentation (further exchange of pyruvate to lactate) [
6]. Second, HIF-1, by inducing the expression of pyruvate dehydrogenase kinase 1 (PDK1) and inactivating the pyruvate dehydrogenase complex (PDH), inhibits the conversion of pyruvate to acetyl-CoA and decreases the supply of a substrate to the tricarboxylic acid (TCA) cycle and the electron transport chain [
54,
55]. Third, HIF-1 is reported to act in the downregulation of mitochondrial function. Experiments using functional VHL-deficient renal cell carcinoma cell lines revealed that HIF-1 can reduce mitochondrial biogenesis by inducing the expression of MAX interactor 1 (MXI-1), which represses c-Myc transcriptional activity, and promoting MXI-1-independent but proteasome-dependent degradation of c-Myc. MXI-1 helps to inhibit the expression of genes associated with mitochondrial DNA replication [
56–
58]. On the other hand, HIF-1 is also known to induce the autophagy of mitochondria by inducing the expression of BNIP3 [
59]. Another mechanism by which HIF-1 controls mitochondrial function is the regulation of the expression of the COX4 subunits by activating transcription of the genes encoding COX4-2 and LON, a mitochondrial protease that is required for degradation of COX4-1 [
60].
In addition to the HIF-1-mediated metabolic reprogramming, HIF-1-dependent expression of carbonic anhydrase 9 (CA9) on the tumor cell surface also contributes to extracellular acidification by hydrating CO
2 to HCO
3− and H
+[
61]. The accumulated lactate within tumor cells can be co-transported out of the cell with H
+ via monocarboxylate transporters (MCTs), resulting in a decrease of extracellular pH (
Table 4) [
62].
Based on the notion of HIF-1-mediated metabolic reprogramming described above, only HIF-1α-positive hypoxic regions would be under acidic conditions in a micro tumor cord and normoxic regions would be under neutral pH. However, it does not seem to be that simple. It is now evident that HIF-1α expression and HIF-1 activity are regulated by various factors even under normoxic conditions; e.g., through the activation of oncogenes or loss of tumor suppressor genes [
63], by increased levels of metabolites such, as succinate and furmarate, by reactive oxygen species (ROS) [
64], and the products of glycolysis, such as lactate and pyruvate
etc.[
65]. Moreover, a number of oncogenes and tumor suppressor genes, such as AMP-activated protein kinase (AMPK), NF-kB, myc, epidermal growth factor (EGF), insulin-like growth factor I, phosphoinositol 3 kinase (PI3K), mTOR, and Kirsten rat sarcoma viral oncogene homolog (KRAS), have been directly associated with the metabolic reprogramming [
66,
67]. Also, c-Myc has been shown to be linked to the regulation of glycolysis in aerobic cells through the direct activation of LDHA and almost all glycolytic genes [
68,
69], and mutated Ras is known to enhance glycolysis, partly by increasing the stability of c-Myc [
70]. From these points of view, metabolic reprogramming and the resultant acidic conditions should be generated not only in hypoxic regions in a HIF-1-dependent manner but also in normoxic regions. In fact, Warburg
et al. reported in their pioneering studies in the 1920s that tumor tissues metabolize more glucose to lactate than normal tissues even in the presence of enough O
2, a phenomenon known as the Warburg effect [
71]. If so, pH in normoxic regions may be dependent on the extent of the Warburg effect (
Table 4). In order to spatio-temporally understand the pH map in a conglomerate of micro tumor cords, it is critical to elucidate how tumor-specific microenvironments and mutations of oncogenes and/or tumor-suppressor genes influence the Warburg effect.
Intratumoral pH has been indirectly measured by using fluorescence ratio imaging microscopy. Dellian
et al. reported that steep interstitial pH gradients exist between tumor blood vessels; the pH decreased by an average of 0.10 pH units over a distance of 40 microns away from the blood vessel wall, and by 0.33 pH units over a 70 microns distance [
72]. They additionally reported that the maximum pH drop, defined as the pH difference between the inter-vessel midpoint and the vessel wall, was positively correlated with the inter-vessel distance [
72]. Moreover, Helmlinger
et al. found a plateau phase of the mean pH, the pH = 6.91 100–170 μm from blood vessels. Furthermore, the pH decreased further to a second plateau at 6.70 in anoxic/necrotic regions [
41].
2.5. Cell Cycle Status and Proliferative Activity of Cells in a Micro Tumor Cord
A fundamental biological process that is dramatically influenced by oxygen-availability is cell proliferation. In many cell types, hypoxia suppresses cell proliferation. Because cancer cells away from functional blood vessels receive minimum amounts of nutrients and oxygen from the circulation, they show the lowest proliferative index. In 1997, a quantitative comparison of immunostaining for hypoxia and proliferation indicated an inverse relationship between pimonidazole binding and proliferation markers in human squamous cell carcinomas [
73]. Koshiji
et al. demonstrated that acute stabilization of HIF-1α and subsequent activation of HIF-1 induce cell cycle arrest under hypoxia (1% O
2). HIF-1 inhibits the function of c-Myc through physical binding to its
N-terminal region [
74]. Over 40% of human cancers exhibit overexpressed c-Myc, regulating the aberrant tumor cell proliferation and metabolism. c-Myc controls the G
1/S transition by associating with MAX. This heterodimer promotes the expression of genes such as cyclin D2 (CCND2), E2F1 and ornithine decarboxylase 1 (ODC1). Simultaneously, c-Myc inhibits the expression of CDKN1A and CDKN1B encoding the cyclin-dependent kinase inhibitors (CDKIs) p21 and p27, respectively[
75]. Through multiple mechanisms, HIF-1 effectively inhibits c-Myc-dependent cell proliferation, and then seems to induce cell cycle arrest at G
1. So, G
1 arrest would be induced in the HIF-1α-positive/pimonidazole-negative layer of a micro tumor cord (
Table 5) [
56,
76], however, it is also true that a proliferation-dependent enzymatic reaction could be induced in this layer, meaning that HIF-1α-positive cells are still proliferative [
28]. From this point of view, the proliferative potential of HIF-1α-positive/pimonidazole-negative cells still remains controversial (
Table 5).
In contrast, the proliferative activity of cancer cells seems to be relatively attenuated in pimonidazole-positive/HIF-1α-negative layers. We recently revealed that a low glucose-mediated decrease in HIF-1α expression results in the suppression of p27 expression, leading to release from cell cycle arrest and subsequent transition from the G
1 to S phase [
31]. As a result, the proportion of cancer cells in S-phase significantly increases in pimonidazole-positive/HIF-1α-negative layers (although a substantial number of cells seem to remain in G
1/G
0). We additionally found that, after the progression of a certain percentage of cells into S-phase, the cell cycle remains at S-phase without efficient DNA synthesis probably because of the depletion of glucose and oxygen [
31].
Liu
et al. investigated biological functions such as the cell cycle and proliferation in lung cancer cells treated with cycling hypoxia
in vitro. They found that repeated exposure of A549 and H446 lung cancer cells to 0.1% O
2 hypoxia and 20% O
2 normoxia for 20 cycles resulted in higher proliferation rates than in the parental cell lines based on their MTT assay. They also showed that the S-phase population of A549 and H446 cells exposed to cycling hypoxia increased. These results suggest that cycling hypoxia could promote cellular proliferation [
77].