Injurious Effects of Emodin on Maturation of Mouse Oocytes, Fertilization and Fetal Development via Apoptosis
Abstract
:1. Introduction
2. Results
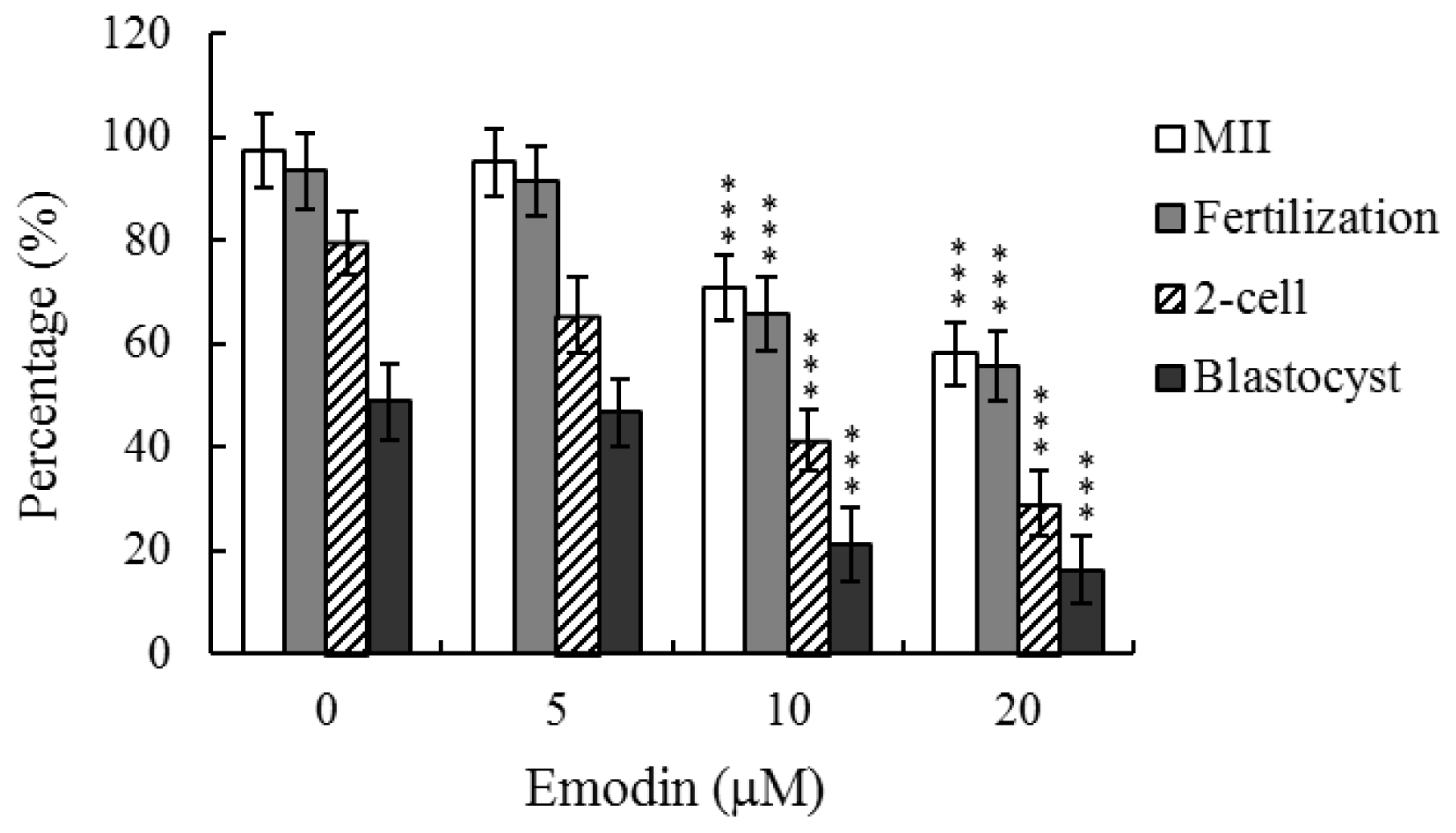
2.1. Effects of Emodin on Oocyte Maturation Status, Fertilization Rate, and in Vitro Embryo Development
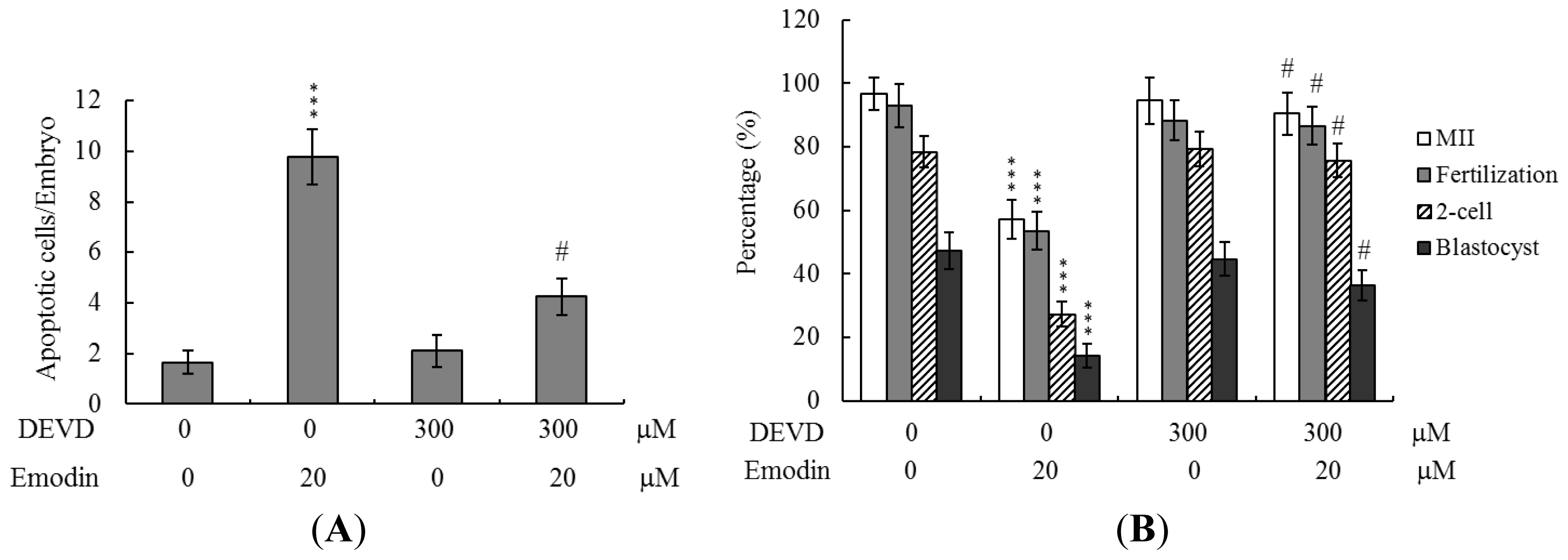
2.2. Effects of Emodin on Cell Proliferation and Apoptosis of Embryos during Oocyte Maturation in Vitro
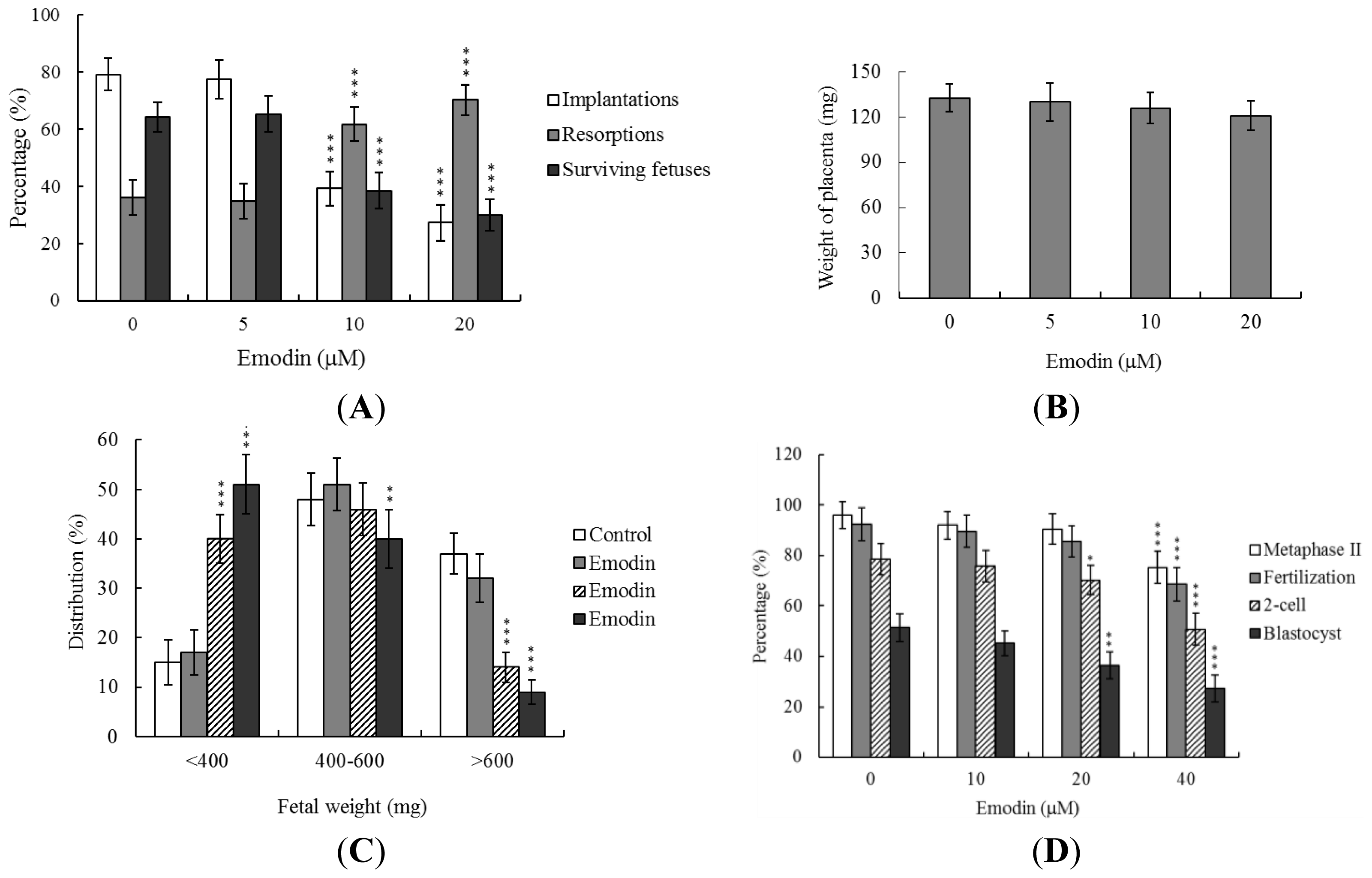
2.3. Developmental Potential of Blastocysts from Oocytes Treated with Emodin and in Vivo Effects of Emodin Intake on Oocyte Development
2.4. Effects of Emodin Intake on the Developmental Potential of Blastocyst-Stage Embryos
2.5. Apoptotic Effects of Emodin on Oocyte Maturation Status, Fertilization Rate, and Embryo Development during IVM
3. Discussion
4. Experimental Section
4.1. Chemicals and Reagents
4.2. COC Collection and in Vitro Maturation (IVM)
4.3. Maturation Status Assessment
4.4. In Vivo Maturation
4.5. Effects of Emodin Intake on Oocyte Maturation in an Animal Model
4.6. In Vitro Fertilization
4.7. Fertilization Assessment
4.8. Cell Proliferation
4.9. TUNEL Assay of Blastocysts
4.10. Blastocyst Development Following Embryo Transfer
4.11. Statistical Analysis
5. Conclusions
Acknowledgements
- Conflicts of InterestThe authors declare no conflicts of interest.
References
- Yim, H.; Lee, Y.H.; Lee, C.H.; Lee, S.K. Emodin, an anthraquinone derivative isolated from the rhizomes of Rheum palmatum, selectively inhibits the activity of casein kinase II as a competitive inhibitor. Planta Med 1999, 65, 9–13. [Google Scholar]
- Yang, F.; Zhang, T.; Tian, G.; Cao, H.; Liu, Q.; Ito, Y. Preparative isolation and purification of hydroxyanthraquinones from Rheum officinale Baill by high-speed counter-current chromatography using pH-modulated stepwise elution. J. Chromatogr. A 1999, 858, 103–107. [Google Scholar]
- Huang, H.C.; Chu, S.H.; Chao, P.D. Vasorelaxants from Chinese herbs, emodin and scoparone, possess immunosuppressive properties. Eur. J. Pharmacol 1991, 198, 211–213. [Google Scholar]
- Zhou, X.M.; Chen, Q.H. Biochemical study of Chinese rhubarb. XXII. Inhibitory effect of anthraquinone derivatives on Na+-K+-ATPase of the rabbit renal medulla and their diuretic action. Yaoxue Xuebao 1988, 23, 17–20. [Google Scholar]
- Koyama, M.; Kelly, T.R.; Watanabe, K.A. Novel type of potential anticancer agents derived from chrysophanol and emodin. Some structure-activity relationship studies. J. Med. Chem 1988, 31, 283–284. [Google Scholar]
- Zhang, L.; Lau, Y.K.; Xia, W.; Hortobagyi, G.N.; Hung, M.C. Tyrosine kinase inhibitor emodin suppresses growth of HER-2/neu-overexpressing breast cancer cells in athymic mice and sensitizes these cells to the inhibitory effect of paclitaxel. Clin. Cancer Res 1999, 5, 343–353. [Google Scholar]
- Shieh, D.E.; Chen, Y.Y.; Yen, M.H.; Chiang, L.C.; Lin, C.C. Emodin-induced apoptosis through p53-dependent pathway in human hepatoma cells. Life Sci 2004, 74, 2279–2290. [Google Scholar]
- Wang, C.-G.; Yang, J.-Q.; Liu, B.-Z.; Jin, D.-T.; Wang, C.; Zhong, L.; Zhu, D.; Wu, Y. Anti-tumor activity of emodin against human chronic myelocytic leukemia K562 cell lines in vitro and in vivo. Eur. J. Pharmacol 2010, 627, 33–41. [Google Scholar]
- Su, Y.T.; Chang, H.L.; Shyue, S.K.; Hsu, S.L. Emodin induces apoptosis in human lung adenocarcinoma cells through a reactive oxygen species-dependent mitochondrial signaling pathway. Biochem. Pharmacol 2005, 70, 229–241. [Google Scholar]
- Lin, S.Y.; Lai, W.W.; Ho, C.C.; Yu, F.S.; Chen, G.W.; Yang, J.S.; Liu, K.C.; Lin, M.L.; Wu, P.P.; Fan, M.J.; et al. Emodin induces apoptosis of human tongue squamous cancer SCC-4 cells through reactive oxygen species and mitochondria-dependent pathways. Anticancer Res 2009, 29, 327–335. [Google Scholar]
- Kuo, T.C.; Yang, J.S.; Lin, M.W.; Hsu, S.C.; Lin, J.J.; Lin, H.J.; Hsia, T.C.; Liao, C.L.; Yang, M.D.; Fan, M.J.; et al. Emodin has cytotoxic and protective effects in rat C6 glioma cells: Roles of Mdr1a and nuclear factor κB in cell survival. J. Pharmacol. Exp. Ther 2009, 330, 736–744. [Google Scholar]
- Chang, M.H.; Huang, F.J.; Chan, W.H. Emodin induces embryonic toxicity in mouse blastocysts through apoptosis. Toxicology 2012, 299, 25–32. [Google Scholar]
- Sartori, R.; Sartor-Bergfelt, R.; Mertens, S.A.; Guenther, J.N.; Parrish, J.J.; Wiltbank, M.C. Fertilization and early embryonic development in heifers and lactating cows in summer and lactating and dry cows in winter. J. Dairy Sci 2002, 85, 2803–2812. [Google Scholar]
- Banwell, K.M.; Lane, M.; Russell, D.L.; Kind, K.L.; Thompson, J.G. Oxygen concentration during mouse oocyte in vitro maturation affects embryo and fetal development. Hum. Reprod 2007, 22, 2768–2775. [Google Scholar]
- De Castro, E.P.L.A.; Hansen, P.J. Interactions between oxygen tension and glucose concentration that modulate actions of heat shock on bovine oocytes during in vitro maturation. Theriogenology 2007, 68, 763–770. [Google Scholar]
- Hardy, K. Cell death in the mammalian blastocyst. Mol. Hum. Reprod 1997, 3, 919–925. [Google Scholar]
- Hardy, K.; Stark, J.; Winston, R.M. Maintenance of the inner cell mass in human blastocysts from fragmented embryos. Biol. Reprod 2003, 68, 1165–1169. [Google Scholar]
- Byrne, A.T.; Southgate, J.; Brison, D.R.; Leese, H.J. Analysis of apoptosis in the preimplantation bovine embryo using TUNEL. J. Reprod. Fertil 1999, 117, 97–105. [Google Scholar]
- Chan, W.H. Ginkgolide B induces apoptosis and developmental injury in mouse embryonic stem cells and blastocysts. Hum. Reprod 2006, 21, 2985–2995. [Google Scholar]
- Hsuuw, Y.D.; Chang, C.K.; Chan, W.H.; Yu, J.S. Curcumin prevents methylglyoxal-induced oxidative stress and apoptosis in mouse embryonic stem cells and blastocysts. J. Cell. Physiol 2005, 205, 379–386. [Google Scholar]
- Shiao, N.H.; Chan, W.H. Injury effects of ginkgolide B on maturation of mouse oocytes, fertilization, and fetal development in vitro and in vivo. Toxicol. Lett 2009, 188, 63–69. [Google Scholar]
- Chan, W.H. Impact of genistein on maturation of mouse oocytes, fertilization, and fetal development. Reprod. Toxicol 2009, 28, 52–58. [Google Scholar]
- Wei, W.T.; Chen, H.; Ni, Z.L.; Liu, H.B.; Tong, H.F.; Fan, L.; Liu, A.; Qiu, M.X.; Liu, D.L.; Guo, H.C.; et al. Antitumor and apoptosis-promoting properties of emodin, an anthraquinone derivative from Rheum officinale Baill, against pancreatic cancer in mice via inhibition of Akt activation. Int. J. Oncol 2011, 39, 1381–1390. [Google Scholar]
- Cross, J.C.; Werb, Z.; Fisher, S.J. Implantation and the placenta: Key pieces of the development puzzle. Science 1994, 266, 1508–1518. [Google Scholar]
- Pampfer, S.; de Hertogh, R.; Vanderheyden, I.; Michiels, B.; Vercheval, M. Decreased inner cell mass proportion in blastocysts from diabetic rats. Diabetes 1990, 39, 471–476. [Google Scholar]
- Kelly, S.M.; Robaire, B.; Hales, B.F. Paternal cyclophosphamide treatment causes postimplantation loss via inner cell mass-specific cell death. Teratology 1992, 45, 313–318. [Google Scholar]
- Lane, M.; Gardner, D.K. Differential regulation of mouse embryo development and viability by amino acids. J. Reprod. Fertil 1997, 109, 153–164. [Google Scholar]
- Wei, T.N.; Hu, J.D.; Chen, Y.Y.; Chen, X.J.; Liu, T.B.; Lu, L.H. Effect of emodin on induction of apoptosis in jurkat cells and its possible mechanisms. Zhongguo Shiyan Xueyexue Zazhi 2009, 17, 1203–1206. [Google Scholar]
- Institute of Laboratory Animal Resources (Commission on Life Sciences NRC), Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA; p. 1996.
- Chan, W.H. Citrinin induces apoptosis via a mitochondria-dependent pathway and inhibition of survival signals in embryonic stem cells, and causes developmental injury in blastocysts. Biochem. J 2007, 404, 317–326. [Google Scholar]
- Huang, L.H.; Shiao, N.H.; Hsuuw, Y.D.; Chan, W.H. Protective effects of resveratrol on ethanol-induced apoptosis in embryonic stem cells and disruption of embryonic development in mouse blastocysts. Toxicology 2007, 242, 109–122. [Google Scholar]
- Hardy, K.; Handyside, A.H.; Winston, R.M. The human blastocyst: Cell number, death and allocation during late preimplantation development in vitro. Development 1989, 107, 597–604. [Google Scholar]
- Gardner, R.L.; Davies, T.J. Lack of coupling between onset of giant transformation and genome endoreduplication in the mural trophectoderm of the mouse blastocyst. J. Exp. Zool 1993, 265, 54–60. [Google Scholar]



© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, M.-H.; Chang, S.-C.; Chan, W.-H. Injurious Effects of Emodin on Maturation of Mouse Oocytes, Fertilization and Fetal Development via Apoptosis. Int. J. Mol. Sci. 2012, 13, 13911-13925. https://doi.org/10.3390/ijms131113911
Chang M-H, Chang S-C, Chan W-H. Injurious Effects of Emodin on Maturation of Mouse Oocytes, Fertilization and Fetal Development via Apoptosis. International Journal of Molecular Sciences. 2012; 13(11):13911-13925. https://doi.org/10.3390/ijms131113911
Chicago/Turabian StyleChang, Mei-Hui, Shao-Chung Chang, and Wen-Hsiung Chan. 2012. "Injurious Effects of Emodin on Maturation of Mouse Oocytes, Fertilization and Fetal Development via Apoptosis" International Journal of Molecular Sciences 13, no. 11: 13911-13925. https://doi.org/10.3390/ijms131113911



