Transcriptome Analysis Reveals Putative Genes Involved in Iridoid Biosynthesis in Rehmannia glutinosa
Abstract
:1. Introduction
2. Results and Discussion
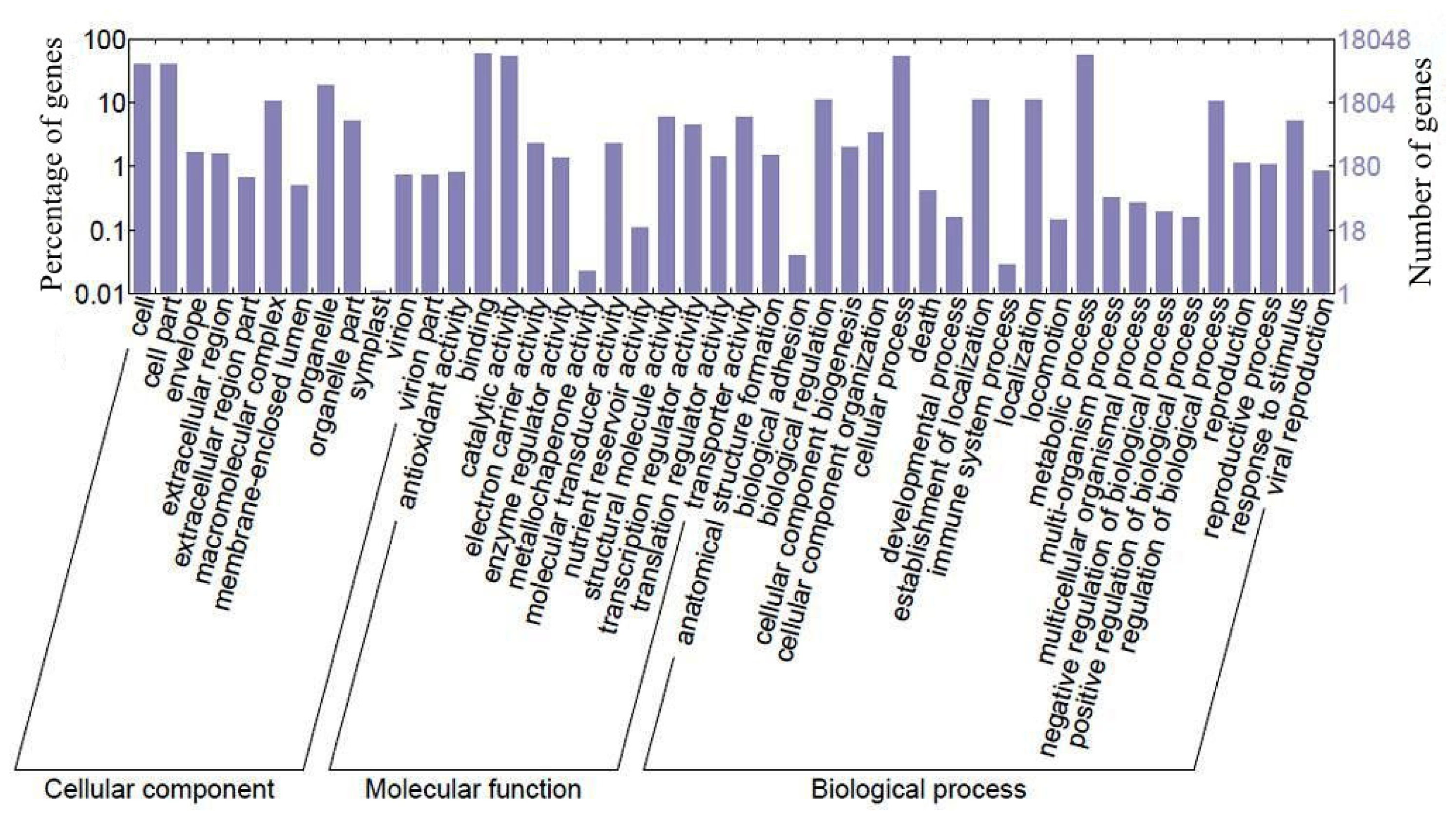
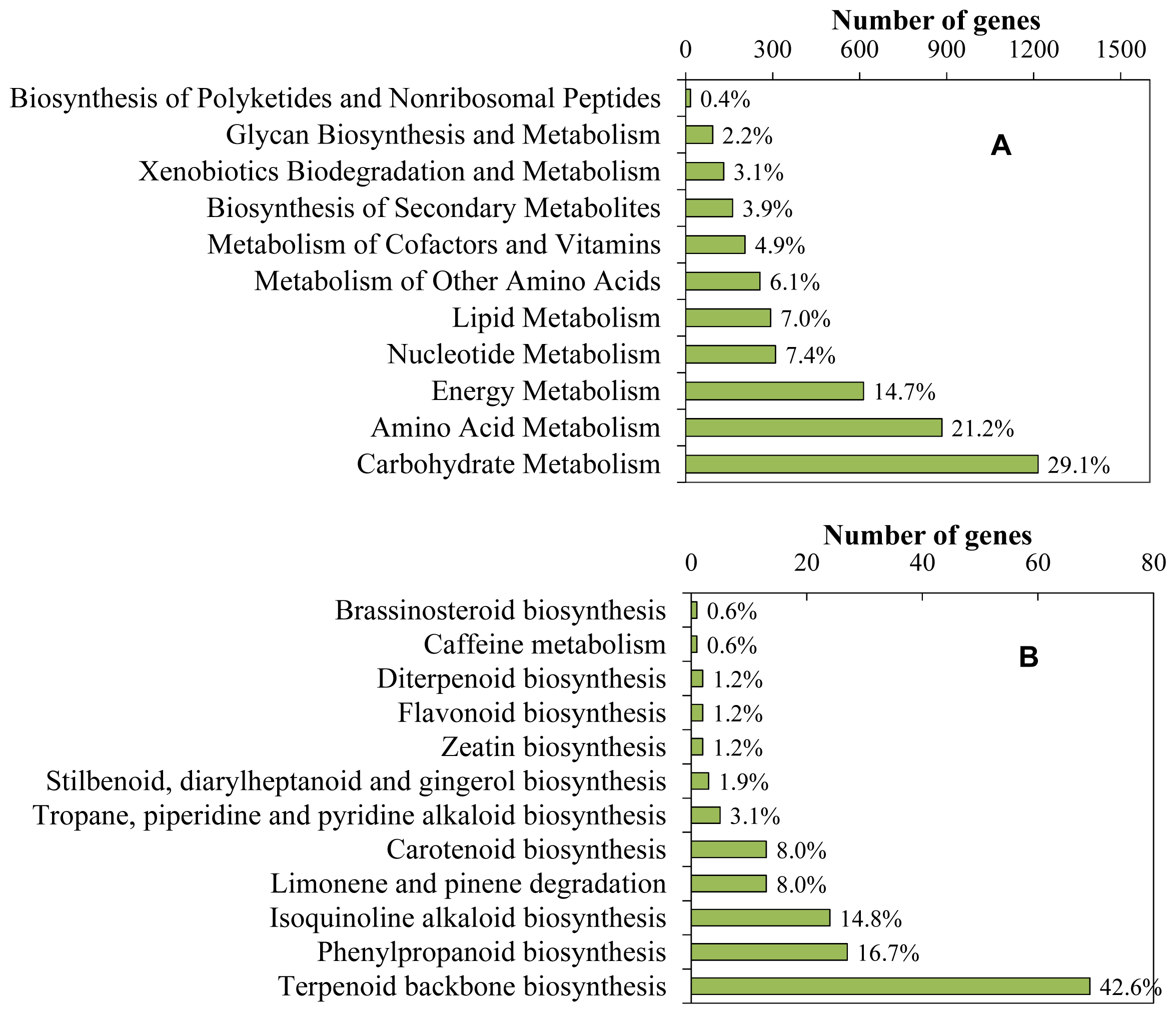
2.1. Sequence Generation and Function Analysis
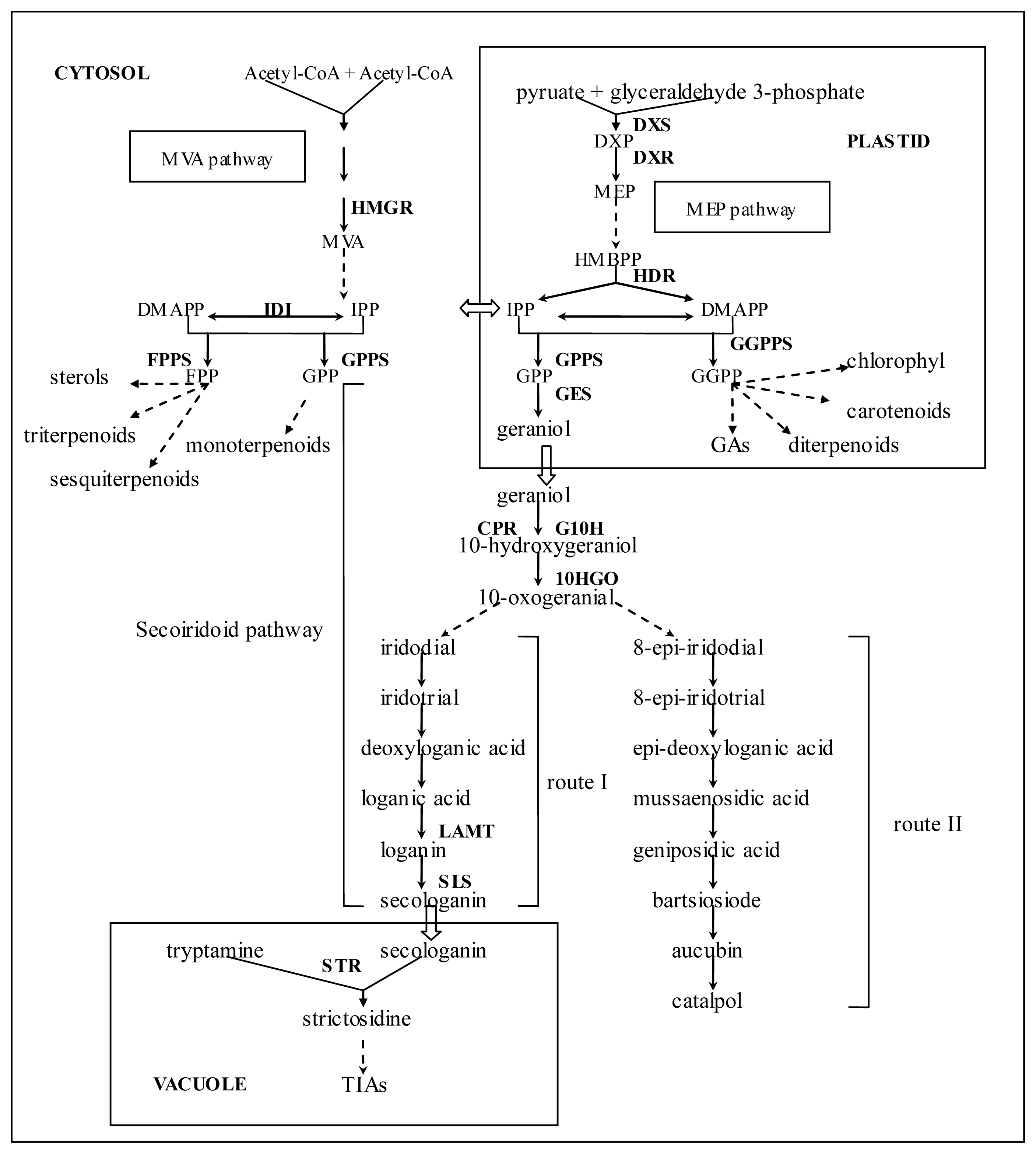
2.2. Identification of Putative Genes Related to Iridoid Biosynthesis
2.3. Identification of Genes Involved in Biosynthesis of Other Terpenoids
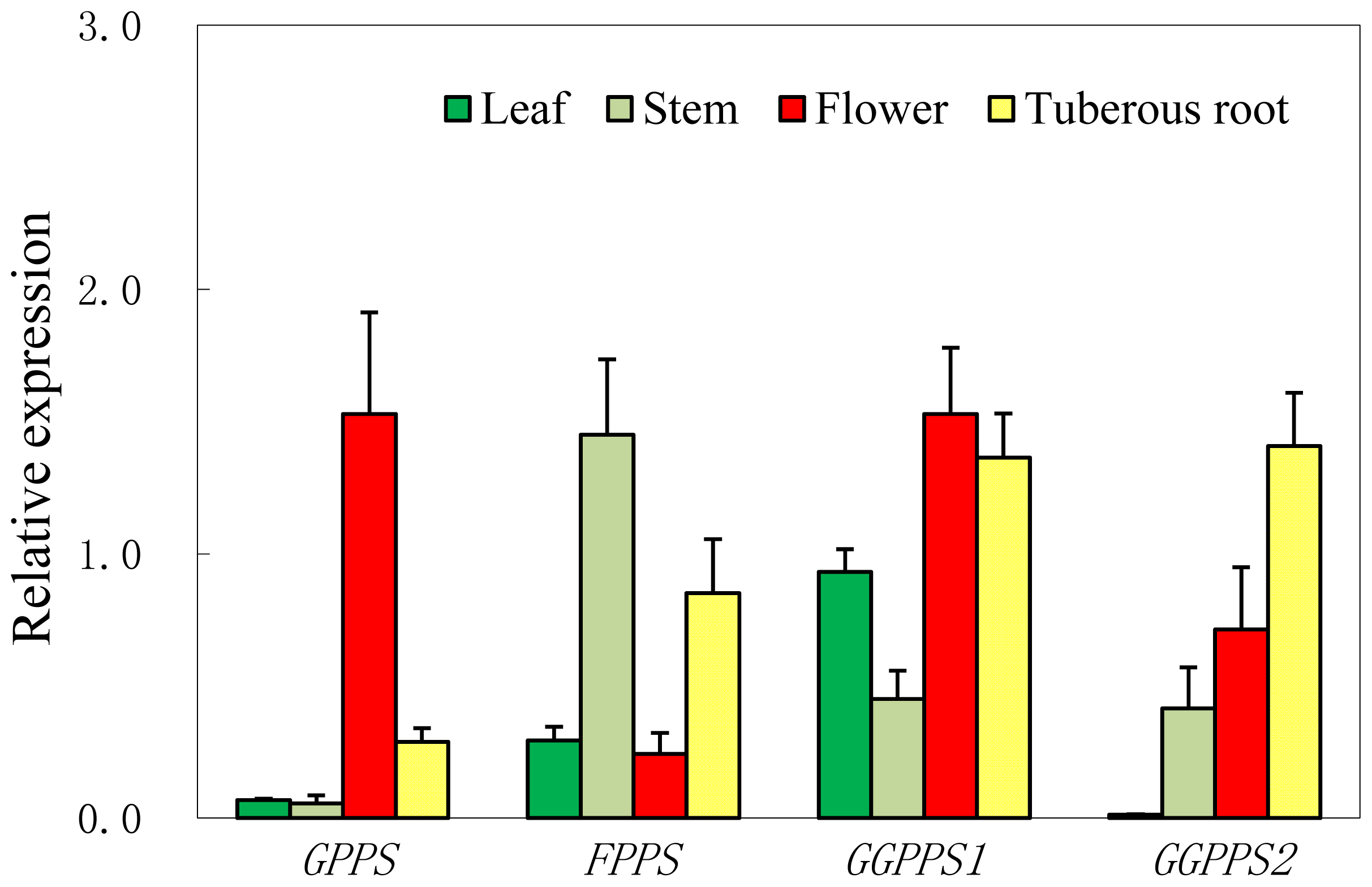
2.4. Expression Patterns of Four Short-Chain Prenyltransferase Genes
3. Experimental Section
3.1. Plant Materials
3.2. cDNA Library Construction and Sequencing
3.3. Sequence Assembly
3.4. Functional Annotation and Metabolic Pathway Analysis
3.5. Quantitative Real-Time PCR
4. Conclusions
Supplementary Materials
ijms-13-13748-s001.xls ijms-13-13748-s002.pdf ijms-13-13748-s003.xls ijms-13-13748-s004.jpgAcknowledgments
References
- World Preservation Society, Powerful and Unusual Herbs from the Amazon and China; World Preservation Society Inc: Greenacres, FL, USA, 2000; pp. 26–27.
- Zhang, R.X.; Li, M.X.; Jia, Z.P. Rehmannia glutinosa: Review of botany, chemistry and pharmacology. J. Ethnopharmacol 2008, 117, 199–214. [Google Scholar]
- Luo, Y.Y.; Zhang, S.Q.; Suo, J.Z.; Sun, D.Y.; Cui, X.C. Determination of catalpol in rehmannia root by high performance liquid chromatography. Chin. Pharm. J 1994, 29, 38–40. [Google Scholar]
- Tundis, R.; Loizzo, M.R.; Menichini, F.; Statti, G.A.; Menichini, F. Biological and pharmacological activities of iridoids: recent developments. Mini Rev. Med. Chem 2008, 8, 399–420. [Google Scholar]
- Zhang, X.; Zhang, A.; Jiang, B.; Bao, Y.; Wang, J.; An, L. Further pharmacological evidence of he neuroprotective effect of catalpol from Rehmannia glutinosa. Phytomedicine 2008, 15, 484–490. [Google Scholar]
- Bi, J.; Wang, X.B.; Chen, L.; Hao, S.; An, L.J.; Jiang, B.; Guo, L. Catalpol protects mesencephalic neurons against MPTP induced neurotoxicity via attenuation of mitochondrial dysfunction and MAO-B activity. Toxicol. In Vitro 2008, 22, 1883–1889. [Google Scholar]
- Cai, Q.Y.; Chen, X.S.; Zhan, X.L.; Yao, Z.X. Protective effects of catalpol on oligodendrocyte death and myelin breakdown in a rat model of chronic cerebral hypoperfusion. Neurosci. Lett 2011, 497, 22–26. [Google Scholar]
- Jensen, S.R. Plant iridoids, Their Biosynthesis and Distribution in Angiosperms. In Ecological Chemistry and Biochemistry of Plant Terpenoids; Harborne, J.B., Tomas-Barberan, F.A., Eds.; Clarendon Press: Oxford, UK, 1991; pp. 133–158. [Google Scholar]
- Damtoft, S.; Jensen, S.R.; Jessen, C.U.; Knudsen, T.B. Late stages in the biosynthesis of aucubin in Scrophularia. Phytochemistry 1993, 33, 1089–1093. [Google Scholar]
- Damtoft, S.; Jensen, S.R.; Weiergang, I. Early stages in the biosynthesis of aucubin and harpagide. Phytochemistry 1994, 35, 621–622. [Google Scholar]
- Damtoft, S. Biosynthesis of catalpol. Phytochemstry 1994, 35, 1187–1189. [Google Scholar]
- Jensen, S.R.; Franzyk, H.; Wallander, E. Chemotaxonomy of the Oleaceae: Iridoids as taxonomic markers. Phytochemry 2002, 60, 213–231. [Google Scholar]
- Hedhili, S.; Courdavault, V.; Giglioli-Guivarc’h, N.; Gantet, P. Regulation of the terpene moiety biosynthesis of Catharanthus roseus terpene indole alkaloids. Phytochem. Rev 2007, 6, 341–351. [Google Scholar]
- El-Sayed, M.; Verpoorte, R. Catharanthus terpenoid indole alkaloids: Biosynthesis and regulation. Phytochem. Rev 2007, 6, 277–305. [Google Scholar]
- Oudin, A.; Courtois, M.; Rideau, M.; Clastre, M. The iridoid pathway in Catharanthus roseus alkaloid biosynthesis. Phytochem. Rev 2007, 6, 259–276. [Google Scholar]
- Chahed, K.; Oudin, A.; Guivarc’h, N.; Hamdi, S.; Chenieux, J.C.; Rideau, M.; Clastre, M. 1-Deoxy-d-xylulose-5-phosphate synthase from periwinkle: cDNA identification and induced gene expression in terpenoidindole alkaloid-producing cells. Plant Physiol. Biochem 2000, 38, 559–566. [Google Scholar]
- Veau, B.; Courtois, M.; Oudin, A.; Chenieux, J.C.; Rideau, M.; Clastre, M. Cloning and expression of cDNAs encoding two enzymes of the MEP pathway in Catharanthus roseus. Biochem. Biophys. Acta 2000, 1517, 159–163. [Google Scholar]
- Meijer, A.H.; Lopes Cardoso, M.I.; Voskuilen, J.T.; de Waal, A.; Verpoorte, R.; Hoge, J.H. Isolation and characterization of a cDNA clone from Catharanthus roseus encoding NADPH: Cytochrome P-450 reductase, an enzyme essential for reactions catalysed by cytochrome P-450 monooxygenases in plants. Plant J 1993, 4, 47–60. [Google Scholar]
- Vetter, H.P.; Mangold, U.; Schröder, G.; Marner, F.J.; Werck-Reichhart, D.; Schröder, J. Molecular analysis and heterologous expression of an inducible cytochrome P450 protein from periwinkle (Catharanthus roseus L.). Plant Physiol 1992, 100, 998–1007. [Google Scholar]
- Collu, G.; Unver, N.; Peltenburg-Looman, A.M.; van der Heijden, R.; Verpoorte, R.; Memelink, J. Geraniol 10-hydroxylase, a cytochrome P450 enzyme involved in terpenoid indole alkaloid biosynthesis. FEBS Lett 2001, 508, 215–220. [Google Scholar]
- Collu, G.; Garcia, A.A.; van der Heijden, R.; Verpoorte, R. Activity of the cytochrome P450 enzyme geraniol 10-hydroxylase and alkaloid production in plant cell cultures. Plant Sci 2002, 162, 165–172. [Google Scholar]
- Burlat, V.; Oudin, A.; Courtois, M.; Rideau, M.; St-Pierre, B. Co-expression of three MEP pathway genes and geraniol 10-hydroxylase in internal phloem parenchyma of Catharanthus roseus implicates multicellular translocation of intermediates during the biosynthesis of monoterpene indole alkaloids and isoprenoid-derived primary metabolites. Plant J 2004, 38, 131–141. [Google Scholar]
- Oudin, A.; Mahroug, S.; Courdavault, V.; Hervouet, N.; Zelwer, C.; Rodríguez-Concepción, M.; St-Pierre, B.; Burlat, V. Spatial distribution and hormonal regulation of gene products from methyl erythritol phosphate and monoterpene-secoiridoid pathways inCatharanthus roseus. Plant Mol. Biol 2007, 65, 13–30. [Google Scholar]
- Murata, J.; Roepke, J.; Gordon, H.; de Luca, V. The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell 2008, 20, 524–542. [Google Scholar]
- Broun, P. Transcription factors as tools for metabolic engineering in plants. Curr. Opin. Plant Biol 2004, 7, 202–209. [Google Scholar]
- Van der Fits, L.; Memelink, J. ORCA3, a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 2000, 289, 295–297. [Google Scholar]
- Wang, Z.; Fang, B.; Chen, J.; Zhang, X.; Luo, Z.; Huang, L.; Chen, X.; Li, Y. De novo assembly and characterization of root transcriptome using Illumina paired-end sequencing and development of cSSR markers in sweet potato (Ipomoea batatas). BMC Genomics 2010, 11, 726–739. [Google Scholar]
- Wu, S.; Schalk, M.; Clark, A.; Miles, R.B.; Coates, R.; Chappell, J. Redirection of cytosolic or plastidic isoprenoid precursors elevates terpene production in plants. Nature Biotech 2006, 24, 1441–1447. [Google Scholar]
- Contin, A.; van der Heijden, R.; Lefeber, A.W.; Verpoorte, R. The iridoid glucoside secologanin is derived from the novel triose phosphate/pyruvate pathway in a Catharanthus roseus cell culture. FEBS Lett 1998, 434, 413–416. [Google Scholar]
- Yamazaki, Y.; Kitajima, M.; Arita, M.; Takayama, H.; Sudo, H.; Yamazaki, M.; Aimi, N.; Saito, K. Biosynthesis of camptothecin. In silico and in vivo tracer study from [1–13C]glucose. Plant Physiol 2004, 134, 161–170. [Google Scholar]
- Li, H.; Yang, S.Q.; Wang, H.; Tian, J.; Gao, W.Y. Biosynthesis of the iridoid glucoside, lamalbid, in Lamium barbatum. Phytochemstry 2010, 71, 1690–1694. [Google Scholar]
- Courdavault, V.; Thiersault, M.; Courtois, M.; Gantet, P.; Oudin, A.; Doireau, P.; St-Pierre, B.; Giglioli-Guivarc’h, N. CaaX-prenyltransferases are essential for expression of genes involved in the early stages of monoterpenoid biosynthetic pathway in Catharanthus roseus cells. Plant Mol. Biol 2005, 57, 855–870. [Google Scholar]
- Iijima, Y.; Gang, D.R.; Fridman, E.; Lewinsohn, E.; Pichersky, E. Characterization of geraniol synthase from the peltate glands of sweet basil. Plant Physiol 2004, 134, 370–379. [Google Scholar]
- Ikeda, H.; Esaki, N.; Nakai, S.; Hashimoto, K.; Uesato, S.; Soda, K.; Fujita, T. Acyclic monoterpene primary alcohol: NADP+ oxidoreductase of Rauwolfia serpentine cells: the key enzyme in biosynthesis of monoterpene alcohols. J. Biochem 1991, 109, 341–347. [Google Scholar]
- Sun, Y.; Luo, H.; Li, Y.; Sun, C.; Song, J.; Niu, Y.; Zhu, Y.; Dong, L.; Lv, A.; Tramontano, E.; et al. Pyrosequencing of the Camptotheca acuminata transcriptome reveals putative genes involved in camptothecin biosynthesis and transport. BMC Genomics 2011, 12, 533–543. [Google Scholar]
- Welsch, R.; Wüst, F.; Bär, C.; Al-Babili, S.; Beyer, P. A third phytoene synthase is devoted to abiotic stress-induced abscisic acid formation in rice and defines functional diversification of phytoene synthase genes. Plant Physiol 2008, 147, 367–380. [Google Scholar]
- Keller, Y.; Bouvier, F.; d’Harlingue, A.; Camara, B. Metabolic compartmentation of plastid prenyllipid biosynthesis-evidence for the involvement of a multifunctional geranylgeranyl reductase. Eur. J. Biochem 1998, 251, 413–417. [Google Scholar]
- Tanaka, R.; Oster, U.; Kruse, E.; Rudiger, W.; Grimm, B. Reduced activity of geranylgeranyl reductase leads to loss of chlorophyll and tocopherol and to partially geranylgeranylated chlorophyll in transgenic tobacco plants expressing antisense RNA for geranylgeranyl reductase. Plant Physiol 1999, 120, 695–704. [Google Scholar]
- Devarenne, T.P.; Ghosh, A.; Chappell, J. Regulation of squalene synthase, a key enzyme of sterol biosynthesis, in tobacco. Plant Physiol 2002, 129, 1095–1106. [Google Scholar]
- McGarbey, D.; Croteau, R. Terpenoid metabolism. Plant Cell 1995, 7, 1015–1026. [Google Scholar]
- Bouvier, F.; Rahier, A.; Camara, B. Biogenesis, molecular regulation and function of plant isoprenoids. Prog. Lipid Res 2005, 44, 357–429. [Google Scholar]
- Tholl, D.; Lee, S. Terpene specialized metabolism in. Arabidopsis thaliana. Arabidopsis Book 2011. [Google Scholar] [CrossRef]
- Burke, C.C.; Croteau, R. Interactions with the small subunit of geranyl diphosphate synthase modifies the chain length specificity of geranylgeranyl diphosphate synthase to produce geranyl diphosphate. J. Biol. Chem 2002, 277, 3141–3149. [Google Scholar]
- Tholl, D.; Kish, C.M.; Orlova, I.; Sherman, D.; Gershenzon, J.; Pichersky, E.; Dudareva, N. Formation of monoterpenes in Antirrhinum majus and Clarkia breweri flowers involves heterodimeric geranyl diphosphate synthases. Plant Cell 2004, 16, 977–992. [Google Scholar]
- Wang, G.; Dixon, R.A. Heterodimeric geranyl(geranyl)diphosphate synthase from hop (Humulus lupulus) and the evolution of monoterpene biosynthesis. Proc. Natl. Acad. Sci. USA 2009, 106, 9914–9919. [Google Scholar]
- Orlova, I.; Nagegowda, D.A.; Kish, C.M.; Gutensohn, M.; Maeda, H.; Varbanova, M.; Fridman, E.; Yamaguchi, S.; Hanada, A.; Kamiya, Y.; et al. The small subunit of snapdragon geranyl diphosphate synthase modifies the chain length specificity of tobacco geranylgeranyl diphosphate synthase in planta. Plant Cell 2009, 21, 4002–4017. [Google Scholar]
- Vandermoten, S.; Haubruge, E.; Cusson, M. New insights into short-chain prenyltransferases: structural features, evolutionary history and potential for selective inhibition. Cell. Mol. Life Sci 2009, 66, 3685–3695. [Google Scholar]
- Schmidt, A.; Wächtler, B.; Temp, U.; Krekling, T.; Séguin, A.; Gershenzon, J. A bifunctional geranyl and geranylgeranyl diphosphate synthase is involved in terpene oleoresin formation in Picea abies. Plant Physiol 2010, 152, 639–655. [Google Scholar]
- Knudsen, J.T.; Gershenzon, J. The Chemical Diversity of Floral Scent. In Biology of Floral Scent; Dudareva, N., Pichensky, E., Eds.; CRC Press: Oxon, UK, 2006; pp. 27–52. [Google Scholar]
- Sun, P.; Guo, Y.H.; Qi, J.J.; Zhou, L.L.; Li, X.E. Isolation and expression analysis of tuberous root development related genes in Rehmannia glutinosa. Mol. Biol. Rep 2010, 37, 1069–1079. [Google Scholar]
| Database | No. of matches | Percentage |
|---|---|---|
| Nr | 25,700 | 43.7% |
| KEGG | 25,259 | 42.9% |
| UniProt | 25,468 | 43.3% |
| COG | 5,701 | 9.7% |
| Nt | 22,427 | 38.1% |
| Total | 29,053 | 49.0% |
| Enzyme name | Enzyme code | Number of unigenes in 454 library | Number of reads in 454 library |
|---|---|---|---|
| acetoacetyl-coenzyme A thiolase (AACT) | 2.3.1.9 | 5 | 9 |
| 3-hydroxy-3-methylglutaryl-coenzyme A synthase (HMGS) | 2.3.3.10 | 5 | 32 |
| 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGR) | 1.1.1.34 | 10 | 185 |
| mevalonate kinase (MK) | 2.7.1.36 | 3 | 14 |
| phosphomevelonate kinase (PMK) | 2.7.4.2 | 2 | 2 |
| diphosphomevalonate decarboxylase (DPMDC) | 4.1.1.33 | 4 | 19 |
| 1-deoxy-d-xylulose-5-phosphate synthase (DXS) | 2.2.1.7 | 6 | 13 |
| 1-deoxy-d-xylulose-5-phosphate reductoisomerase (DXR) | 1.1.1.267 | 4 | 36 |
| 4-diphosphocytidyl-2-C-methyl-d-erythritol synthase (CMS) | 2.7.7.60 | 1 | 1 |
| 4-diphosphocytidyl-2C-methyl-d-erythritol kinase (CMK) | 2.7.1.148 | 1 | 2 |
| 2-C-methyl-d-erythritol 2,4-cyclodiphosphate synthase (MECS) | 4.6.1.12 | 4 | 4 |
| 1-hydroxy-2-methyl-butenyl 4-diphosphate synthase (HDS) | 1.17.7.1 | 4 | 7 |
| 1-hydroxy-2-methyl-butenyl 4-diphosphate reductase (HDR) | 1.17.1.2 | 3 | 7 |
| isopentenyl diphosphate isomerase (IDI) | 5.3.3.2 | 6 | 17 |
| farnesyl diphosphate synthase (FPP) | 2.2.1.10 | 4 | 19 |
| geranyl diphosphate synthase (GPPS) | 2.5.1.1 | 1 | 3 |
| geranylgeranyl pyrophosphate synthase (GGPPS) | 1.3.1.74 | 9 | 21 |
| geraniol synthase (GES) | 3.1.7.3 | 1 | 2 |
| geraniol 10-hydroxylase (G10H) | 1.14.14.1 | 1 | 2 |
| cytochrome P450 reductase (CPR) | 1.6.2.4 | 5 | 58 |
| 10-hydroxygeraniol oxidoreductase (10HGO) | 1.1.1.255 | 6 | 97 |
| Total number | 85 | 550 |
| Gene | Primer sequences (forward and reverse) |
|---|---|
| GPPS | 5′-GGGAAAACTGCTGGGAAGG-3′ |
| 5′-TTAGCCAGAGCAACCAAAGGA-3′ | |
| FPPS | 5′-CAGAGGAAAGCCTTACTATGTGGAT-3′ |
| 5′-TGAACAATGCGGCGGTGA-3′ | |
| GGPPS1 | 5′-TTGCCCTGTATGGACAACGA-3′ |
| 5′-CGCCGATAACACGAACGATT-3′ | |
| GGPPS2 | 5′-AGGGCACAGGCTAAGAAAGAAT-3′ |
| 5′-CCTCTATCAACAGCATAATCAACAAAGT-3′ | |
| TIP41 | 5′-ACGCCTCGGATTTCTCATTC-3′ |
| 5′-TCCAGCCGCATAGAGCATC-3′ |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, P.; Song, S.; Zhou, L.; Zhang, B.; Qi, J.; Li, X. Transcriptome Analysis Reveals Putative Genes Involved in Iridoid Biosynthesis in Rehmannia glutinosa. Int. J. Mol. Sci. 2012, 13, 13748-13763. https://doi.org/10.3390/ijms131013748
Sun P, Song S, Zhou L, Zhang B, Qi J, Li X. Transcriptome Analysis Reveals Putative Genes Involved in Iridoid Biosynthesis in Rehmannia glutinosa. International Journal of Molecular Sciences. 2012; 13(10):13748-13763. https://doi.org/10.3390/ijms131013748
Chicago/Turabian StyleSun, Peng, Shuhui Song, Lili Zhou, Bing Zhang, Jianjun Qi, and Xianen Li. 2012. "Transcriptome Analysis Reveals Putative Genes Involved in Iridoid Biosynthesis in Rehmannia glutinosa" International Journal of Molecular Sciences 13, no. 10: 13748-13763. https://doi.org/10.3390/ijms131013748
APA StyleSun, P., Song, S., Zhou, L., Zhang, B., Qi, J., & Li, X. (2012). Transcriptome Analysis Reveals Putative Genes Involved in Iridoid Biosynthesis in Rehmannia glutinosa. International Journal of Molecular Sciences, 13(10), 13748-13763. https://doi.org/10.3390/ijms131013748




