Binding of the Phage Display Derived Peptide CaIX-P1 on Human Colorectal Carcinoma Cells Correlates with the Expression of Carbonic Anhydrase IX
Abstract
:1. Introduction
2. Results and Discussion
2.1. CAIX Expression and Biding of CaIX-P1
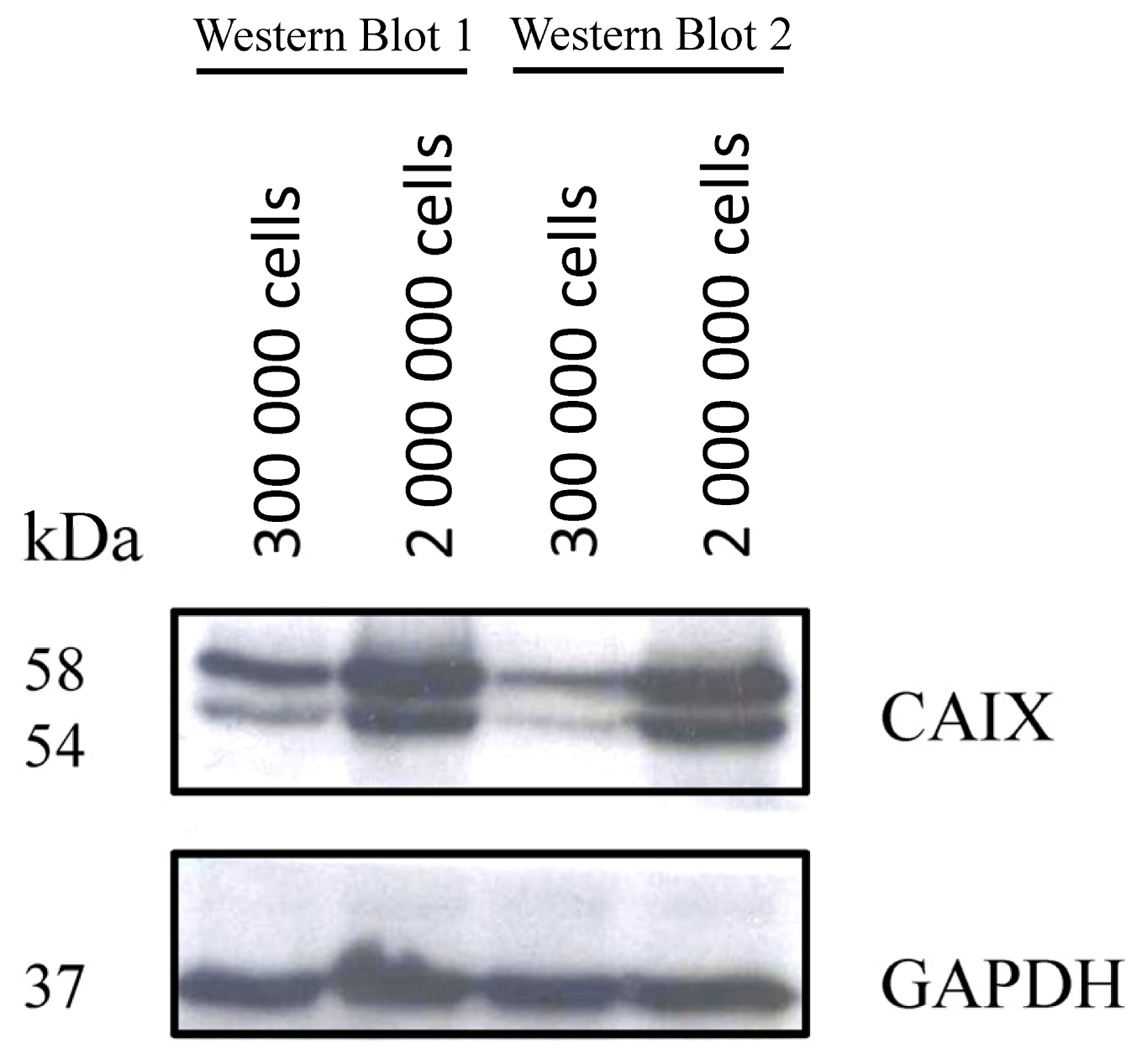
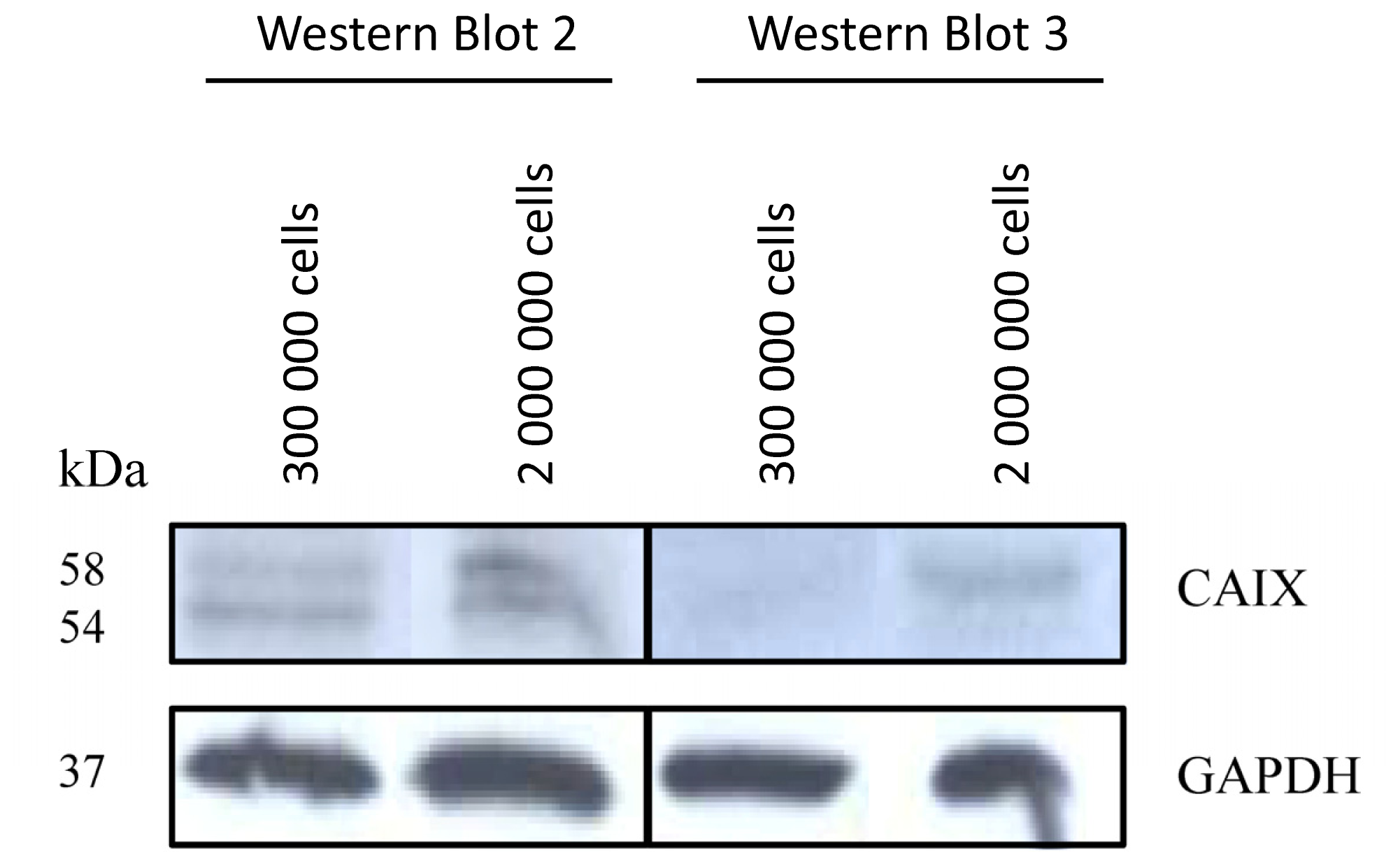
2.1.1. CAIX Expression in HCT116, HT29 and BxPC3 Cells
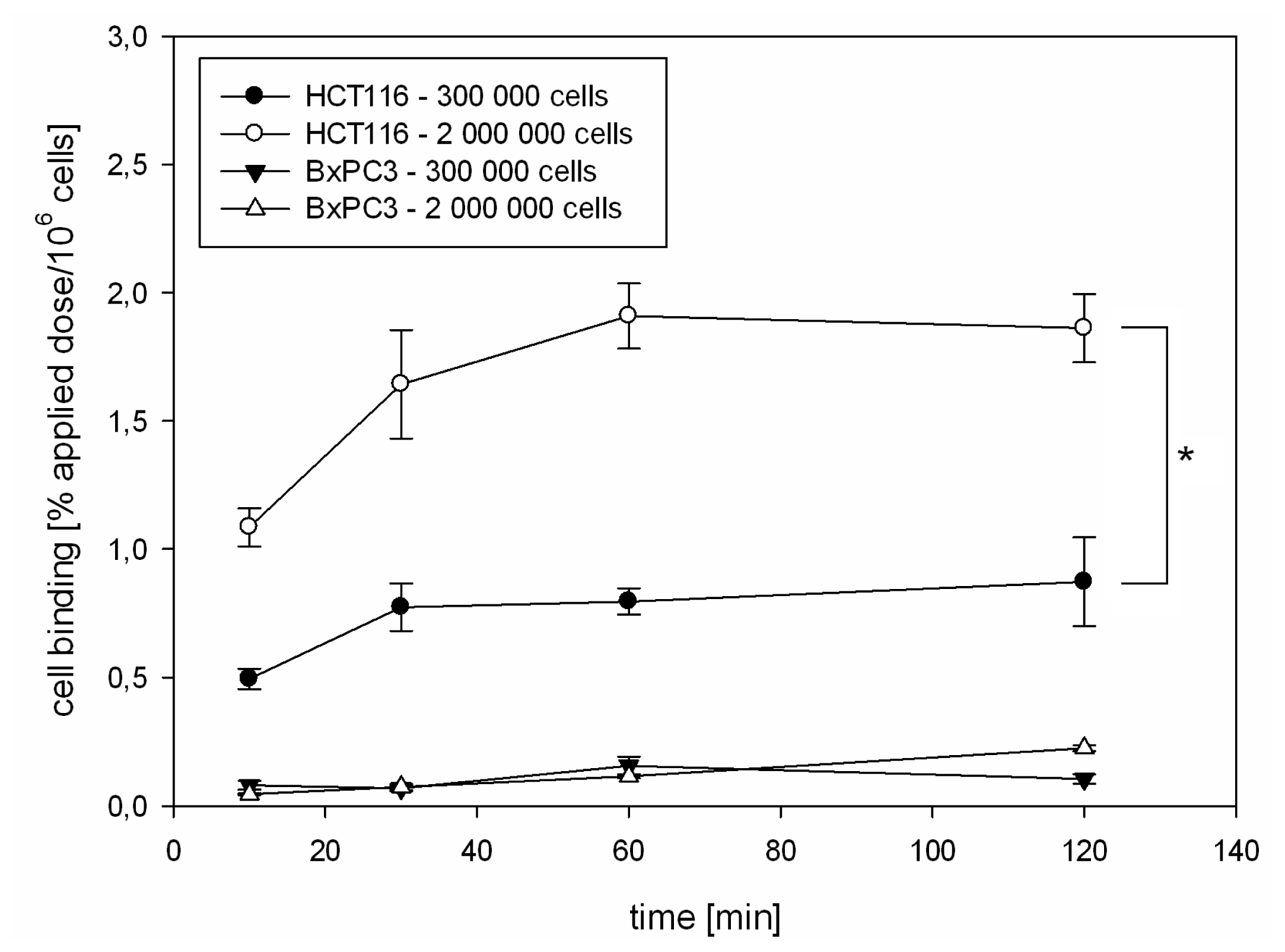
2.1.2. Kinetics of 125I-CaIX-P1 in HCT116 and BxPC3 Cells
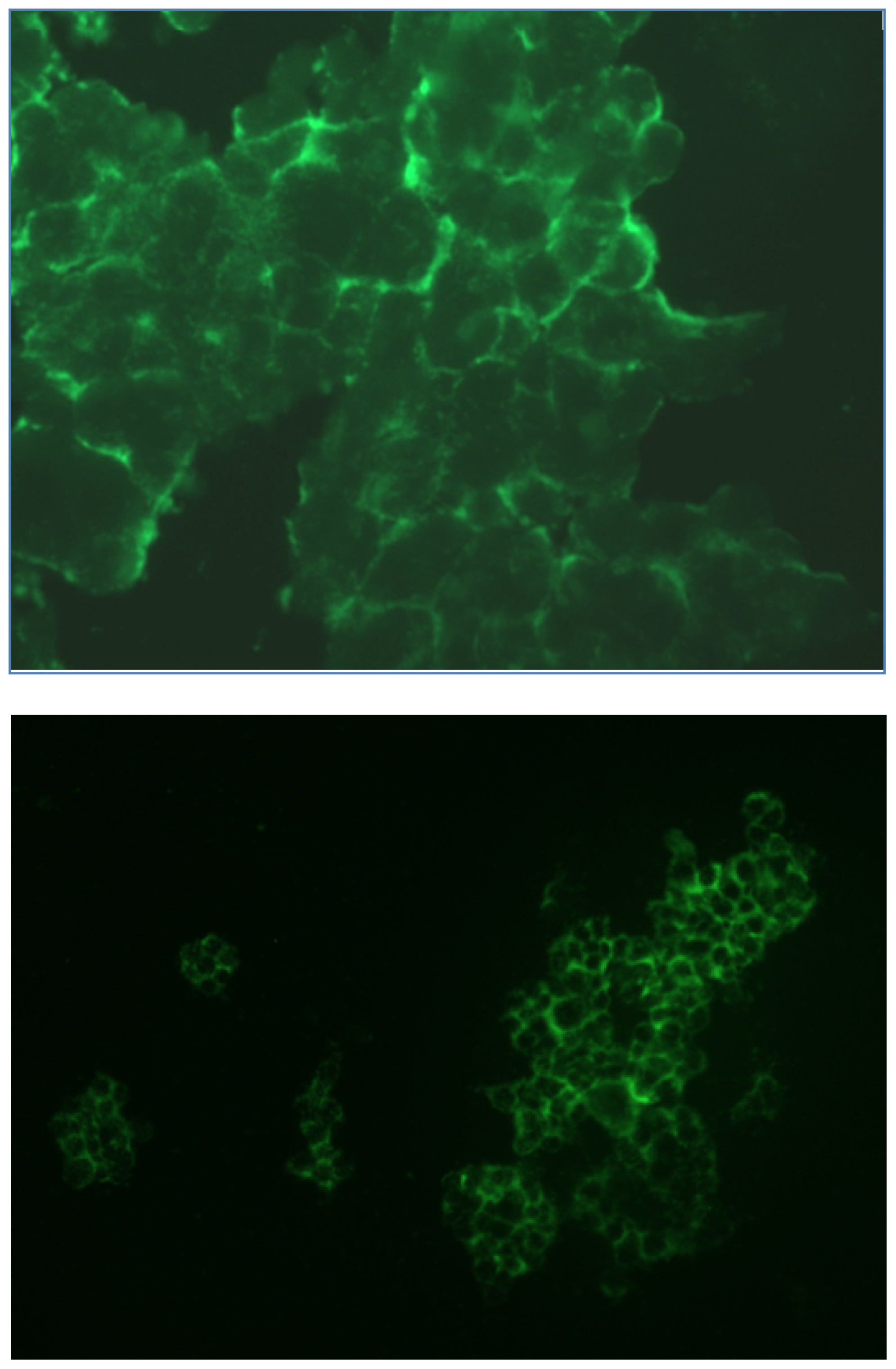
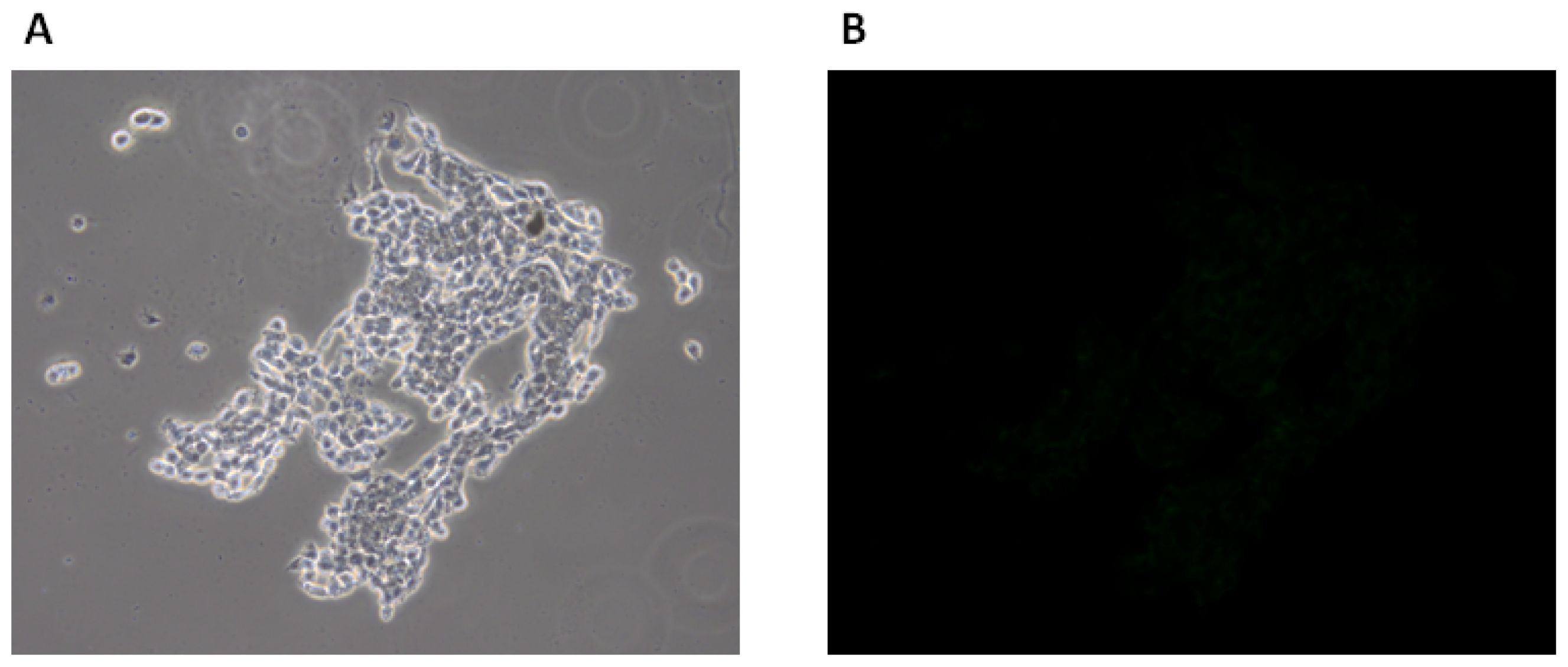
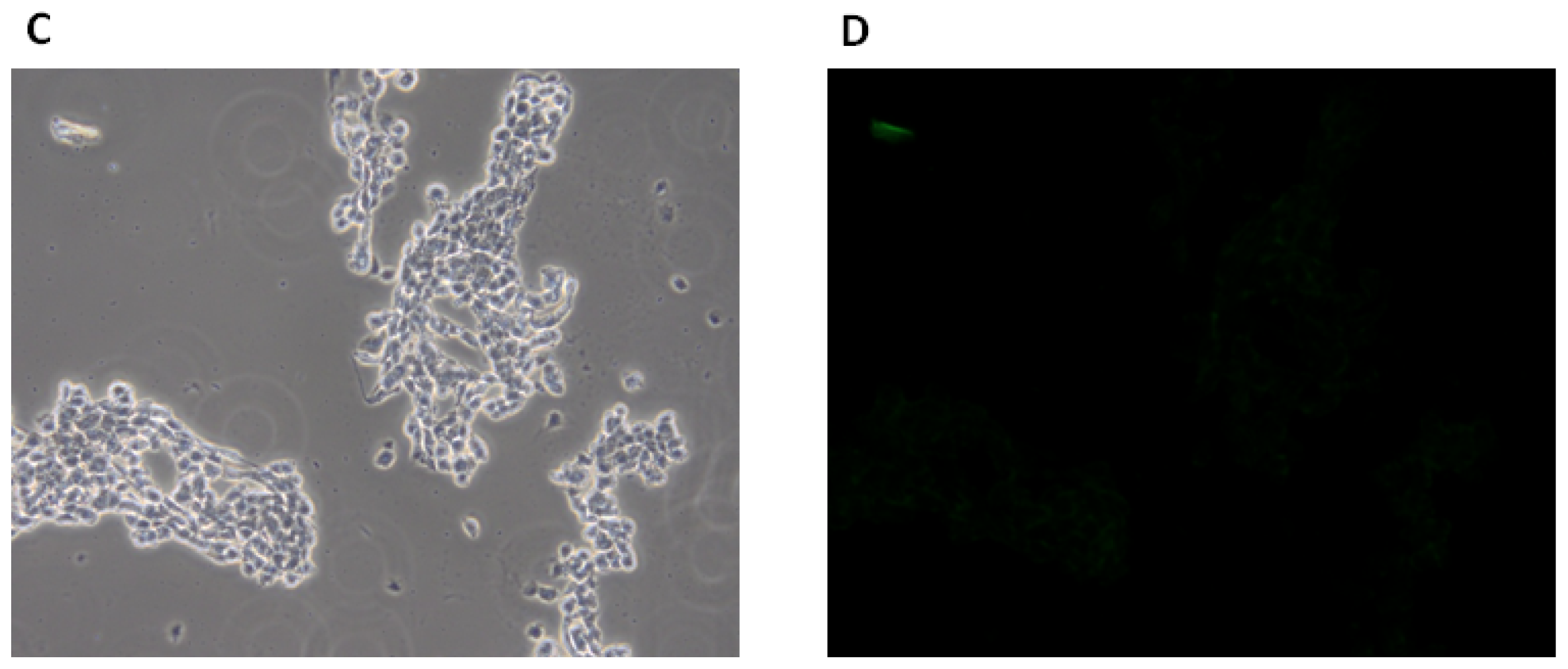
2.1.3. Fluorescence Microscopy Experiments
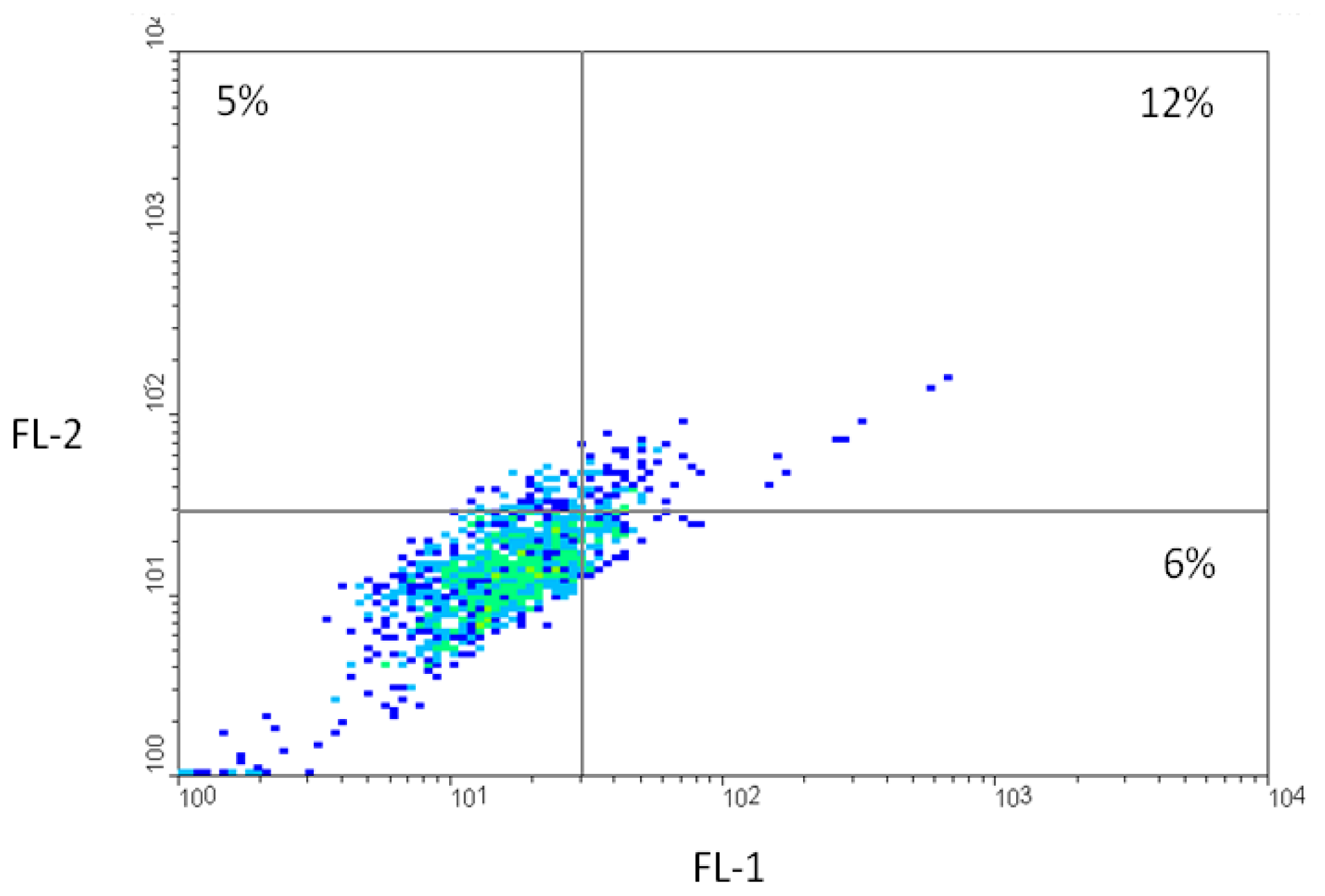
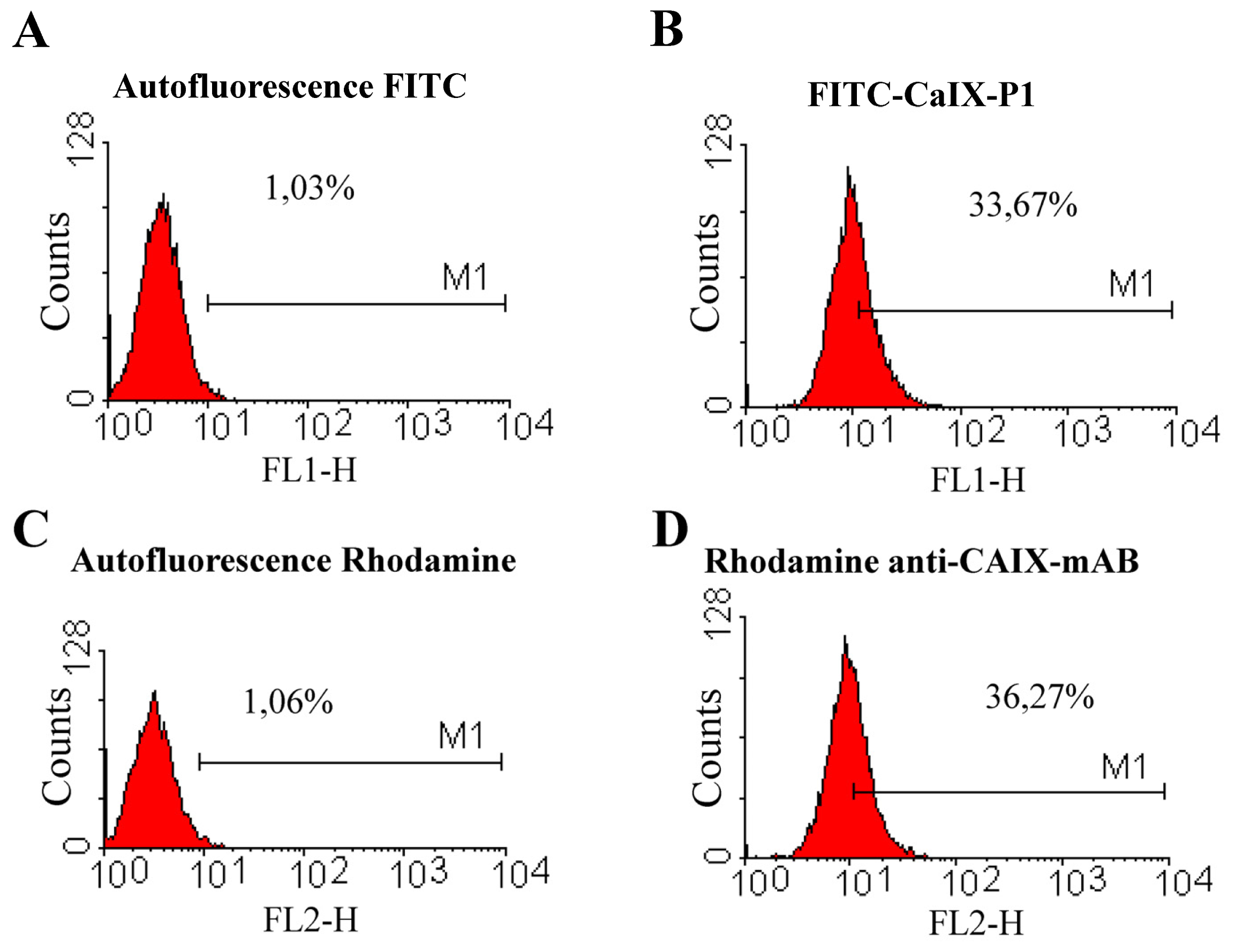
2.2. Fluorescence-Activated Cell Sorting (FACS)
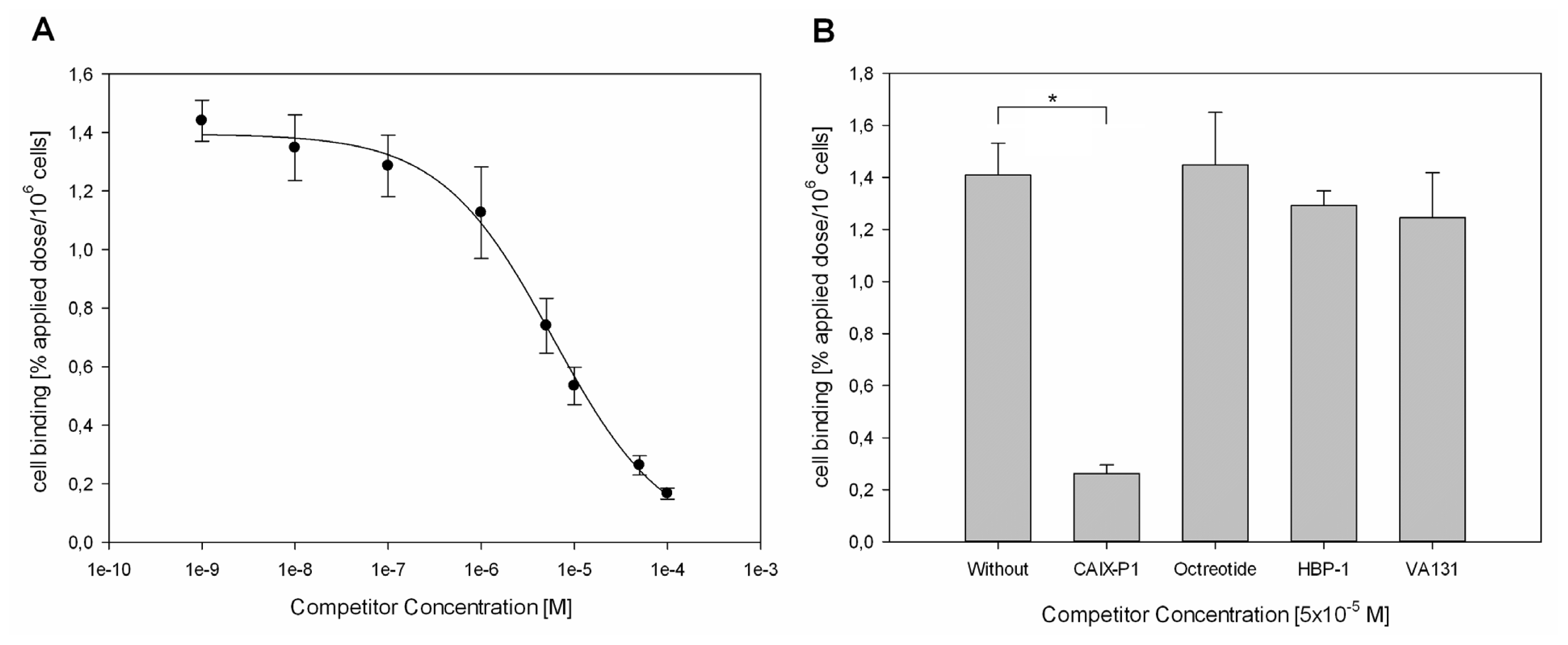
2.3. Competition Studies on HCT116 Cells
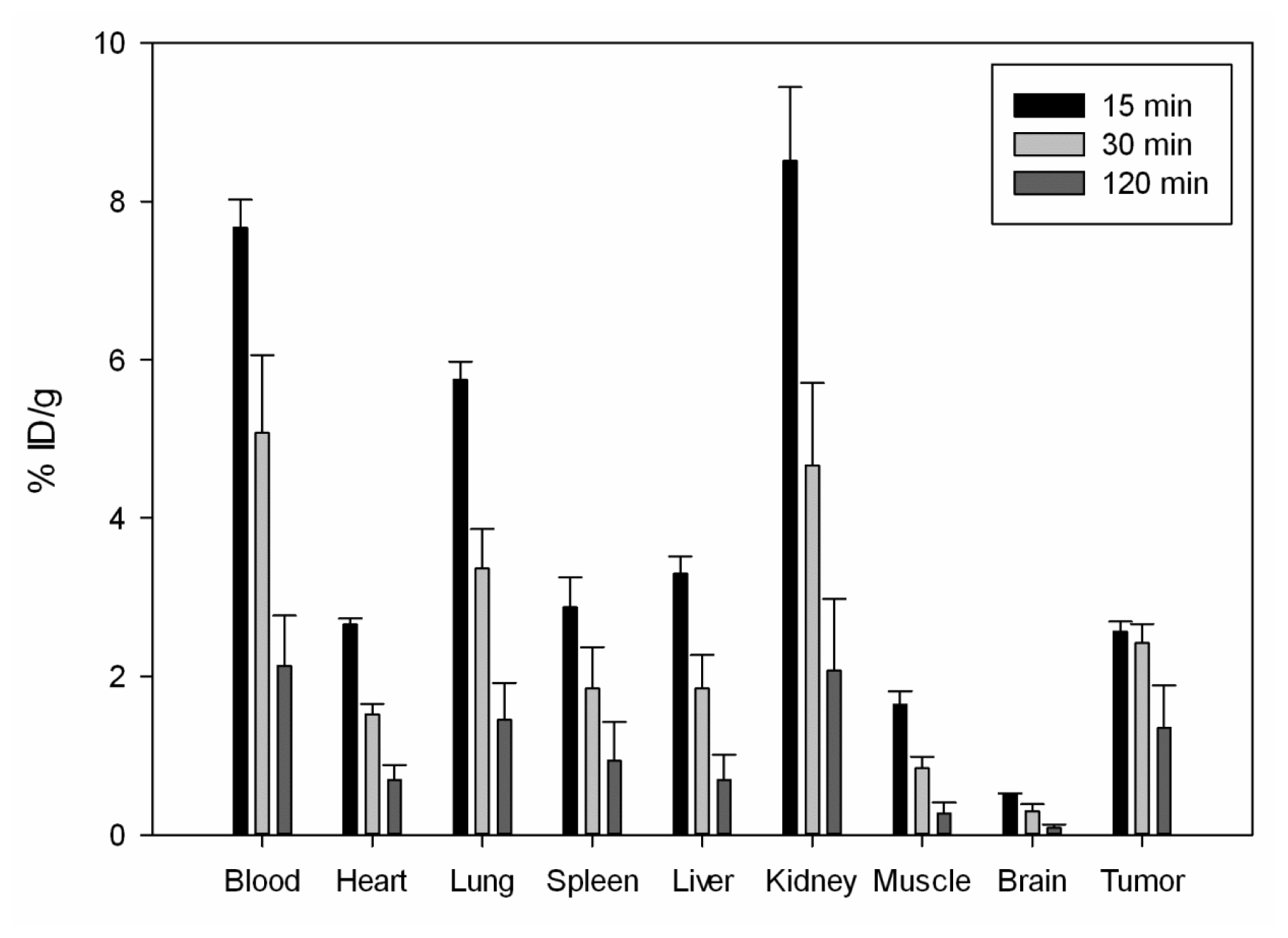
2.4. In Vivo Bio-Distribution
3. Experimental Section
3.1. Cell Lines
3.2. Peptides
3.3. In Vitro Binding Studies
3.4. Western-Blot Analysis
3.5. Fluorescence Microscopy Experiments
3.6. Fluorescence-Activated Cell Sorting (FACS)
3.7. In Vivo Organ Distribution
3.8. Data Analysis and Statistics
4. Conclusions
Acknowledgments
References
- Winum, J.Y.; Rami, M.; Scozzafava, A.; Montero, J.L.; Supuran, C. Carbonic anhydrase IX: A new druggable target for the design of antitumor agents. Med. Res. Rev 2008, 28, 445–463. [Google Scholar]
- Grabmaier, K.; A de Weijert, M.C.; Verhaegh, G.W.; Schalken, J.A.; Oosterwijk, E. Strict regulation of CAIX(G250/MN) by HIF-1alpha in clear cell renal cell carcinoma. Oncogene 2004, 23, 5624–5631. [Google Scholar]
- Pinheiro, C.; Sousa, B.; Albergaria, A.; Paredes, J.; Dufloth, R.; Vieira, D.; Schmitt, F.; Baltazar, F. GLUT1 and CAIX expression profiles in breast cancer correlate with adverse prognostic factors and MCT1 overexpression. Histol. Histopathol 2011, 26, 1279–1286. [Google Scholar]
- Vaupel, P.; Mayer, A. Hypoxia in cancer: Significance and impact on clinical outcome. Cancer Metastasis Rev 2007, 26, 225–239. [Google Scholar]
- Neri, D.; Supuran, C.T. Interfering with pH regulation in tumours as a therapeutic strategy. Nat. Rev. Drug Discov 2011, 10, 767–777. [Google Scholar]
- Supuran, C.T.; Winum, J.Y. Drug Design of Zinc-Enzyme Inhibitors–Functional, Structural, and Disease Applications; Wang, B., Ed.; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2009. [Google Scholar]
- Supuran, C.T. Inhibition of carbonic anhydrase IX as a novel anticancer mechanism. World J. Clin. Oncol 2012, 3, 98–103. [Google Scholar]
- Dubois, L.; Lieuwes, N.G.; Maresca, A.; Thiry, A.; Supuran, C.T.; Scozzafava, A.; Wouters, B.G.; Lambin, P. Imaging of CA IX with fluorescent labelled sulfonamides distinguishes hypoxic and (re)-oxygenated cells in a xenograft tumour model. Radiother. Oncol 2009, 92, 423–428. [Google Scholar]
- Ahlskog, J.K.; Dumelin, C.E.; Trüssel, S.; Mårlind, J.; Neri, D. In vivo targeting of tumor-associated carbonic anhydrases using acetazolamide derivatives. Bioorg. Med. Chem. Lett 2009, 19, 4851–4856. [Google Scholar]
- Li, J.; Shi, L.; Wang, C.; Zhang, X.; Jia, L.; Li, X.; Zhou, W.; Qi, Y.; Zhang, L. Preliminary biological evaluation of 125I-labeled anti-carbonic anhydrase IX monoclonal antibody in the mice bearing HAT-29 tumors. Nucl. Med. Commun 2011, 32, 1190–1193. [Google Scholar]
- Wu, A.M.; Senter, P.D. Arming antibodies: Prospects and challenges for immunoconjugates. Nat. Biotechnol 2005, 23, 1137–1146. [Google Scholar]
- Haberkorn, U.; Eisenhut, M.; Altmann, A.; Mier, W. Endoradiotherapy with peptides-status and future development. Curr. Med. Chem 2008, 15, 219–234. [Google Scholar]
- Marr, A.; Markert, A.; Altmann, A.; Askoxylakis, V.; Haberkorn, U. Biotechnology techniques for the development of new tumor specific peptides. Methods 2011, 55, 215–222. [Google Scholar]
- Askoxylakis, V.; Garcia-Boy, R.; Rana, S.; Krämer, S.; Hebling, U.; Mier, W.; Altmann, A.; Markert, A.; Debus, J.; Haberkorn, U. A new peptide ligand for targeting human carbonic anhydrase IX, identified through the phage display technology. PLoS One 2010, 5, e15962. [Google Scholar]
- Sansone, P.; Piazzi, G.; Paterini, P.; Strillacci, A.; Ceccarelli, C.; Minni, F.; Biasco, G.; Chieco, P.; Bonafé, M. Cyclooxygenase-2/carbonic anhydrase-IX up-regulation promotes invasive potential and hypoxia survival in colorectal cancer cells. J. Cell. Mol. Med 2009, 13, 3876–3887. [Google Scholar]
- Hunakova, L.; Bodo, J.; Chovancova, J.; Sulikova, G.; Pastorekova, S.; Sedlak, J. Expression of new prognostic markers, peripheral-type benzodiazepine receptor and carbonic anhydrase IX, in human breast and ovarian carcinoma cell lines. Neoplasma 2007, 54, 541–548. [Google Scholar]
- Askoxylakis, V.; Millonig, G.; Wirkner, U.; Schwager, C.; Rana, S.; Altmann, A.; Haberkorn, U.; Debus, J.; Mueller, S.; Huber, P.E. Investigation of tumor hypoxia using a two-enzyme system for in vitro generation of oxygen deficiency. Radiat. Oncol 2011, 6, 35. [Google Scholar]
- Kaluz, S.; Kaluzová, M.; Chrastina, A.; Olive, P.L.; Pastoreková, S.; Pastorek, J.; Lerman, M.I.; Stanbridge, E.J. Lowered oxygen tension induces expression of the hypoxia marker MN/carbonic anhydrase IX in the absence of hypoxia-inducible factor 1 alpha stabilization: A role for phosphatidylinositol 3′-kinase. Cancer Res 2002, 62, 4469–4477. [Google Scholar]
- Nothelfer, E.M.; Zitzmann-Kolbe, S.; Garcia-Boy, R.; Krämer, S.; Herold-Mende, C.; Altmann, A.; Eisenhut, M.; Mier, W.; Haberkorn, U. Identification and characterization of a peptide with affinity to head and neck cancer. J. Nucl. Med 2009, 50, 426–434. [Google Scholar]
- Zhang, J.; Spring, H.; Schwab, M. Neuroblastoma tumor cell-binding peptides identified through random peptide phage display. Cancer Lett 2001, 171, 153–164. [Google Scholar]
- Di Cianni, A.; Carotenuto, A.; Brancaccio, D.; Novellino, E.; Reubi, J.C.; Beetschen, K.; Papini, A.M.; Ginanneschi, M. Novel octreotide dicarba-analogues with high affinity and different selectivity for somatostatin receptors. J. Med. Chem 2010, 53, 6188–6197. [Google Scholar]
- Liu, S.; Liu, Z.; Chen, K.; Yan, Y.; Watzlowik, P.; Wester, H.J.; Chin, F.T.; Chen, X. 18F-labeled galacto and PEGylated RGD dimers for PET imaging of αvβ3 integrin expression. Mol. Imaging Biol 2010, 12, 530–538. [Google Scholar]
- Hruby, V.J. Designing peptide receptor agonists and antagonists. Nat. Rev. Drug. Discov 2002, 1, 847–858. [Google Scholar]
- Rana, S.; Nissen, F.; Marr, A.; Markert, A.; Altmann, A.; Mier, W.; Debus, J.; Haberkorn, U.; Askoxylakis, V. Optimization of a novel peptide ligand targeting human carbonic anhydrase IX. PLoS One 2012, 7, e38279. [Google Scholar]
- Hilpert, K.; Winkler, D.F.; Hancock, R.E. Peptide arrays on cellulose support: SPOT synthesis, a time and cost efficient method for synthesis of large numbers of peptides in a parallel and addressable fashion. Nat. Protoc 2007, 2, 1333–1349. [Google Scholar]
- Winkler, D.F.; Hilpert, K.; Brandt, O.; Hancock, R.E. Synthesis of peptide arrays using SPOT technology and the CelluSpots-method. Methods Mol. Biol 2009, 570, 157–174. [Google Scholar]
- Ahmed, S.; Mathews, A.S.; Byeon, N.; Lavasanifar, A.; Kaur, K. Peptide arrays for screening cancer specific peptides. Anal. Chem 2010, 82, 7533–7541. [Google Scholar]
- Zoller, F.; Haberkorn, U.; Mier, W. Miniproteins as phage display-scaffolds for clinical applications. Molecules 2011, 16, 2467–2485. [Google Scholar]
- John, H.; Maronde, E.; Forssmann, W.G.; Meyer, M.; Adermann, K. N-terminal acetylation protects glucagon-like peptide GLP-1-(7-34)-amide from DPP-IV-mediated degradation retaining cAMP- and insulin-releasing capacity. Eur. J. Med. Res 2008, 13, 73–78. [Google Scholar]
- Gentilucci, L.; de Marco, R.; Cerisoli, L. Chemical modifications designed to improve peptide stability: incorporation of non-natural amino acids, pseudo-peptide bonds, and cyclization. Curr. Pharm. Des 2010, 16, 3185–3203. [Google Scholar]
- Askoxylakis, V.; Zitzmann-Kolbe, S.; Zoller, F.; Altmann, A.; Markert, A.; Rana, S.; Marr, A.; Mier, W.; Debus, J.; Haberkorn, U. Challenges in optimizing a prostate carcinoma binding peptide, identified through the phage display technology. Molecules 2011, 16, 1559–1578. [Google Scholar]
- Aloj, L.; Morelli, G. Design, synthesis and preclinical evaluation of radiolabeled peptides for diagnosis and therapy. Curr. Pharm. Des 2004, 10, 3009–3031. [Google Scholar]
- Pnwar, P.; Iznaga-Escobar, N.; Mishra, P.; Srivastava, V.; Sharma, R.K.; Chandra, R.; Mishra, A.K. Radiolabeling and biological evaluation of DOTA-Ph-Al derivative conjugated to anti-EGFR antibody ior egf/r3 for targeted tumor imaging and therapy. Cancer Biol. Ther 2005, 4, 854–860. [Google Scholar]




© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Askoxylakis, V.; Ehemann, V.; Rana, S.; Krämer, S.; Rahbari, N.N.; Debus, J.; Haberkorn, U. Binding of the Phage Display Derived Peptide CaIX-P1 on Human Colorectal Carcinoma Cells Correlates with the Expression of Carbonic Anhydrase IX. Int. J. Mol. Sci. 2012, 13, 13030-13048. https://doi.org/10.3390/ijms131013030
Askoxylakis V, Ehemann V, Rana S, Krämer S, Rahbari NN, Debus J, Haberkorn U. Binding of the Phage Display Derived Peptide CaIX-P1 on Human Colorectal Carcinoma Cells Correlates with the Expression of Carbonic Anhydrase IX. International Journal of Molecular Sciences. 2012; 13(10):13030-13048. https://doi.org/10.3390/ijms131013030
Chicago/Turabian StyleAskoxylakis, Vasileios, Volker Ehemann, Shoaib Rana, Susanne Krämer, Nuh N. Rahbari, Jürgen Debus, and Uwe Haberkorn. 2012. "Binding of the Phage Display Derived Peptide CaIX-P1 on Human Colorectal Carcinoma Cells Correlates with the Expression of Carbonic Anhydrase IX" International Journal of Molecular Sciences 13, no. 10: 13030-13048. https://doi.org/10.3390/ijms131013030




