Peripheral Nerve Injuries and Transplantation of Olfactory Ensheathing Cells for Axonal Regeneration and Remyelination: Fact or Fiction?
Abstract
:1. Introduction
2. OEC Transplantation into Sciatic Nerve Supports Axonal Regeneration and Remyelination
3. Implantation of OEC-Seeded Scaffolds for Nerve Substance Defect Repair
3.1. OECs for Facial Nerve Repair
3.2. OECs in Dorsal Root Injury
4. Concluding Remarks
Supplementary Materials
ijms-13-12911-s001.pdfAcknowledgements
Abbreviations
| CHS | collagen-heparan sulphate |
| CNS | central nervous system |
| DREZ/DH | dorsal root entry zone/dorsal horn |
| ECM | extracellular matrix |
| EMG | electromyography |
| GFP | green fluorescent protein |
| CMAP | compound muscle action potential |
| Nav | voltage-gated TTX-sensitive sodium channels |
| Nav1.6 | voltage gated sodium channel subtype 1.6 |
| NGF | nerve growth factor |
| NCV | nerve conduction velocity; nerve growth factor |
| OB | olfactory bulb |
| OECs | olfactory ensheathing cells |
| OM | olfactory mucosa |
| PDLLA | poly D, L-lactic acid |
| PGA | polymer polyglycolic acid |
| PHB | poly-3-hydroxybutyrate |
| PLGL | poly [LA-co-(Glc-alt-Lys)] |
| PLLA | poly L-lactic acid |
| p75NGFR | p75 nerve growth factor receptor |
| PNS | peripheral nervous system |
| SFI | sciatic functional index |
| SpC | spinal cord |
References
- Furey, M.; Midha, R.; Xu, Q.B.; Belkas, J.; Gordon, T. Prolonged target deprivation reduces the capacity of injured motoneurons to regenerate. Neurosurgery 2007, 60, 723–733. [Google Scholar]
- Ramón-Cueto, A.; Nieto-Sampedro, M. Regeneration into the spinal cord of transected dorsal root axons is promoted by ensheathing glia transplants. Exp. Neurol 1994, 127, 232–244. [Google Scholar]
- Li, Y.; Field, P.M.; Raisman, G. Repair of adult rat corticospinal tract by transplants of olfactory ensheathing cells. Science 1997, 297, 2000–2002. [Google Scholar]
- Franklin, R.J.; Gilson, J.M.; Franceschini, I.A.; Barnett, S.C. Schwann cell-like myelination following transplantation of an olfactory bulb-ensheathing cell line into areas of demyelination in the adult CNS. Glia 1996, 17, 217–224. [Google Scholar]
- Imaizumi, T.; Lankford, K.L.; Waxman, S.G.; Greer, C.A.; Kocsis, J.D. Transplanted olfactory ensheathing cells remyelinate and enhance axonal conduction in the demyelinated dorsal columns of the rat spinal cord. J. Neurosci 1998, 18, 6176–6185. [Google Scholar]
- Kato, T.; Honmou, O.; Uede, T.; Hashi, K.; Kocsis, J.D. Transplantation of human olfactory ensheathing cells elicits remyelination of demyelinated rat spinal cord. Glia 2000, 39, 209–218. [Google Scholar]
- Sasaki, M.; Black, J.A.; Lankford, K.L.; Tokuno, H.A.; Waxman, S.G.; Kocsis, J.D. Molecular reconstruction of nodes of Ranvier after remyelination by transplanted olfactory ensheathing cells in the demyelinated spinal cord. J. Neurosci 2006, 28, 1803–1812. [Google Scholar]
- Feron, F.; Perry, C.; Cochrane, J.; Licina, P.; Nowitzke, A.; Urquhart, S.; Geraghty, T.; Mackay-Sim, A. Autologous olfactory ensheathing cell transplantation in human spinal cord injury. Brain 2005, 135, 3851–3860. [Google Scholar]
- Mackay-Sim, A.; Féron, F.; Cochrane, J.; Bassingthwaighte, L.; Bayliss, C.; Davies, W.; Fronek, P.; Gray, C.; Kerr, G.; Licina, P.; et al. Autologous olfactory ensheathing cell transplantation in human paraplegia: A 3-year clinical trial. Brain 2000, 132, 2376–2386. [Google Scholar]
- Huang, H.; Chen, L.; Wang, H.; Xiu, B.; Li, B.; Wang, R.; Zhang, J.; Zhang, F.; Gu, Z.; Li, Y.; et al. Influence of patients’ age on functional recovery after transplantation of olfactory ensheathing cells into injured spinal cord injury. Chin. Med. J 2003, 116, 1488–1491. [Google Scholar]
- Lima, C.; Pratas-Vital, J.; Escada, P.; Hasse-Ferreira, A.; Capucho, C.; Peduzzi, J.D. Olfactory mucosa autografts in human spinal cord injury: A pilot clinical study. J. Spinal Cord Med 2006, 38, 191–206. [Google Scholar]
- Deumens, R.; Koopmans, G.C.; Lemmens, M.; Möllers, S.; Honig, W.M.; Steinbusch, H.W.; Brook, G.; Joosten, E.A. Neurite outgrowth promoting effects of enriched and mixed OEC/ONF cultures. Neurosci. Lett 2006, 417, 20–26. [Google Scholar]
- Au, E.; Richter, M.W.; Vincent, A.J.; Tetzlaff, W.; Aebersold, R.; Sage, E.H.; Roskams, A.J. SPARC from olfactory ensheathing cells stimulates Schwann cells to promote neurite outgrowth and enhances spinal cord repair. J. Neurosci 2007, 29, 7208–7236. [Google Scholar]
- Cao, L.; Zhu, Y.L.; Su, Z.; Lv, B.; Huang, Z.; Mu, L.; He, C. Olfactory ensheathing cells promote migration of Schwann cells by secreted nerve growth factor. Glia 2007, 55, 897–904. [Google Scholar]
- Sasaki, M.; Lankford, K.L.; Zemedkun, M.; Kocsis, J.D. Identified olfactory ensheathing cells transplanted into the transected dorsal funiculus bridge the lesion and form myelin. J. Neurosci 2004, 26, 8485–8493. [Google Scholar]
- Dombrowski, M.A.; Sasaki, M.; Lankford, K.L.; Kocsis, J.D.; Radtke, C. Myelination and nodal formation of regenerated peripheral nerve fibers following transplantation of acutely prepared olfactory ensheathing cells. Brain Res 2006, 1127, 1–8. [Google Scholar]
- Cheng, S.Y.; Ruan, H.Z.; Wu, X.G. Olfactory ensheathing cells enhance functional recovery of injured sciatic nerve. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2003, 17, 18–36. [Google Scholar]
- Wang, C.; Shi, Z.; Wang, K. Effect of olfactory ensheathing cells transplantation on protecting spinal cord and neurons after peripheral nerve injury. Chin. J. Repar. Reconstr. Surg 2005, 19, 875–878. [Google Scholar]
- Delaviz, H.; Joghataie, M.T.; Mehdizadeh, M.; Bakhtiyari, M.; Nobakht, M.; Khoei, S. Transplantation of olfactory mucosa improve functional recovery and axonal regeneration following sciatic nerve repair in rats. Iran. Biomed. J 2008, 12, 197–202. [Google Scholar]
- Radtke, C.; Aizer, A.A.; Agulian, S.K.; Lankford, K.L.; Vogt, P.M.; Kocsis, J.D. Transplantation of olfactory ensheathing cells enhances peripheral nerve regeneration after microsurgical nerve repair. Brain Res 2009, 1274, 10–17. [Google Scholar]
- Verdú, E.; Navarro, X.; Gudiño-Cabrera, G.; Rodríguez, F.J.; Ceballos, D.; Valero, A.; Nieto-Sampedro, M. Olfactory bulb ensheathing cells enhance peripheral nerve regeneration. Neuroreport 1999, 10, 1097–1101. [Google Scholar]
- Li, B.C.; Jiao, S.S.; Xu, C.; You, H.; Chen, J.M. PLGA conduit seeded with olfactory ensheathing cells for bridging sciatic nerve defect of rats. J. Biomed. Mater. Res. A 2010, 94, 769–780. [Google Scholar]
- You, H.; Wei, L.; Liu, Y.; Oudega, M.; Jiao, S.S.; Feng, S.N.; Chen, Y.; Chen, J.M.; Li, B.C. Olfactory ensheathing cells enhance Schwann cell-mediated anatomical and functional repair after sciatic nerve injury in adult rats. Exp. Neurol 2011, 238, 158–167. [Google Scholar]
- Guérout, N.; Duclos, C.; Drouot, L.; Abramovici, O.; Bon-Mardion, N.; Lacoume, Y.; Jean, L.; Boyer, O.; Marie, J.P. Transplantation of olfactory ensheathing cells promotes axonal regeneration and functional recovery of peripheral nerve lesion in rats. Muscle Nerve 2011, 34, 534–551. [Google Scholar]
- Penna, V.; Stark, G.B.; Wewetzer, K.; Radtke, C.; Lang, E.M. Comparison of Schwann Cells and Olfactory Ensheathing Cells for Peripheral Nerve Gap Bridging. Cells Tissues Organs 2012. [Google Scholar] [CrossRef]
- Guntinas-Lichius, O.; Angelov, D.N.; Tomov, T.L.; Dramiga, J.; Neiss, W.F.; Wewetzer, K. Transplantation of olfactory ensheathing cells stimulates the collateral sprouting from axotomized adult rat facial motoneurons. Exp. Neurol 2001, 172, 70–80. [Google Scholar]
- Guntinas-Lichius, O.; Wewetzer, K.; Tomov, T.L.; Azzolin, N.; Kazemi, S.; Streppel, M.; Neiss, W.F.; Angelov, D.N. Transplantation of olfactory mucosa minimizes axonal branching and promotes the recovery of vibrissae motor performance after facial nerve repair in rats. J. Neurosci 2002, 22, 7121–7131. [Google Scholar]
- Angelov, D.N.; Guntinas-Lichius, O.; Wewetzer, K.; Neiss, W.F.; Streppel, M. Axonal branching and recovery of coordinated muscle activity after transection of the facial nerve in adult rats. Adv. Anat. Embryol. Cell Biol 2005, 180, 1–130. [Google Scholar]
- Choi, D.; Raisman, G. Disorganization of the facial nucleus after nerve lesioning and regeneration in the rat: Effects of transplanting candidate reparative cells to the site of injury. Neurosurgery 2005, 56, 1093–1100. [Google Scholar]
- Guérout, N.; Paviot, A.; Bon-Mardion, N.; Duclos, C.; Genty, D.; Jean, L.; Boyer, O.; Marie, J.P. Co-transplantation of olfactory ensheathing cells from mucosa and bulb origin enhances functional recovery after peripheral nerve lesion. PLoS One 2011, 6, e23516. [Google Scholar]
- De Corgnol, A.C.; Guérout, N.; Duclos, C.; Vérin, E.; Marie, J.P. Olfactory ensheathing cells in a rat model of laryngeal reinnervation. Ann. Otol. Rhinol. Laryngol 2011, 120, 273–280. [Google Scholar]
- Paviot, A.; Guérout, N.; Bon-Mardion, N.; Duclos, C.; Jean, L.; Boyer, O.; Marie, J.P. Efficiency of laryngeal motor nerve repair is greater with bulbar than with mucosal olfactory ensheathing cells. Neurobiol. Dis 2011, 43, 688–694. [Google Scholar]
- Navarro, X.; Valero, A.; Gudiño, G.; Forés, J.; Rodríguez, F.J.; Verdú, E.; Pascual, R.; Cuadras, J.; Nieto-Sampedro, M. Ensheathing glia transplants promote dorsal root regeneration and spinal reflex restitution after multiple lumbar rhizotomy. Ann. Neurol 1999, 45, 207–215. [Google Scholar]
- Goméz, V.M.; Averill, S.; King, V.; Yang, Q.; Doncel Pérez, E.; Chacón, S.C.; Ward, R.; Nieto-Sampedro, M.; Priestley, J.; Taylor, J. Transplantation of olfactory ensheathing cells fails to promote significant axonal regeneration from dorsal roots into the rat cervical cord. J. Neurocytol 2003, 32, 53–70. [Google Scholar]
- Riddell, J.S.; Enriquez-Denton, M.; Toft, A.; Fairless, R.; Barnett, S.C. Olfactory ensheathing cell grafts have minimal influence on regeneration at the dorsal root entry zone following rhizotomy. Glia 2004, 47, 150–167. [Google Scholar]
- Li, Y.; Carlstedt, T.; Berthold, C.H.; Raisman, G. Interaction of transplanted olfactory ensheathing cells and host astrocytic processes provides a bridge for axons to regenerate across the dorsal root entry zone. Exp. Neurol 2004, 188, 390–398. [Google Scholar]
- Ramer, L.M.; Richter, M.W.; Roskams, A.J.; Tetzlaff, W.; Ramer, M.S. Peripherally-derived olfactory ensheathing cells do not promote primary afferent regeneration following dorsal root injury. Glia 2004, 47, 189–206. [Google Scholar]
- Ibrahim, A.G.; Kirkwood, P.A.; Raisman, G.; Li, Y. Restoration of hand function in a rat model of repair of brachial plexus injury. Brain 2009, 132, 1288–1296. [Google Scholar]
- Wu, A.; Lauschke, J.L.; Gorrie, C.A.; Cameron, N.; Hayward, I.; Mackay-Sim, A.; Waite, P.M. Delayed olfactory ensheathing cell transplants reduce nociception after dorsal root injury. Exp. Neurol 2011, 238, 134–157. [Google Scholar]
- Tang, Z.P.; Liu, N.; Li, Z.W.; Xie, X.W.; Chen, Y.; Shi, Y.H.; Zeng, W.G.; Wang, S.X.; Chen, J.; Yang, J.; et al. In vitro evaluation of the compatibility of a novel collagen-heparan sulfate biological scaffold with olfactory ensheathing cells. Chin. Med. J 2010, 140, 1389–1394. [Google Scholar]
- Qian, L.M.; Zhang, Z.J.; Gong, A.H.; Qin, R.J.; Sun, X.L.; Cao, X.D.; Liu, J.B.; Jiang, P.; Chen, Y.C. A novel biosynthetic hybrid scaffold seeded with olfactory ensheathing cells for treatment of spinal cord injuries. Chin. Med. J. (Engl.) 2009, 122, 2032–2042. [Google Scholar]
- Shen, Y.; Qian, Y.; Zhang, H.; Zuo, B.; Lu, Z.; Fan, Z.; Zhang, P.; Zhang, F.; Zhou, C. Guidance of olfactory ensheathing cell growth and migration on electrospun silk fibroin scaffolds. Cell Transplant 2010, 19, 147–157. [Google Scholar]
- Wang, Y.; Wang, Y.; Yin, Y.; Li, S.; Yan, Q.; Wan, Z.; Han, Y. Interaction of olfactory ensheathing cells with nerve repairing scaffolds. J. Cent. South Univ. (Med. Sci.) 2009, 31, 382–387. [Google Scholar]
- Li, Y.; Yamamoto, M.; Raisman, G.; Choi, D.; Carlstedt, T. An experimental model of ventral root repair showing the beneficial effect of transplanting olfactory ensheathing cells. Neurosurgery 2007, 60, 731–743. [Google Scholar]

| Nerve lesion model | OEC condition | OEC application | Outcome | Limits | Reference |
|---|---|---|---|---|---|
| Sciatic nerve crush lesion (rat) | GFP-OECs purified 30,000 cells/μL and 10 μL used | OEC injection proximal and distal to lesion | myelin formation and axonal regeneration high density of Na(v)1.6 newly formed nodes of Ranvier | no functional testing performed | Dombrowski et al., 2006 [16] |
| Sciatic nerve transection and silicone entubulation (rat) | OB OECs | OECs injected in silicone tube | improvement of CMAP increased nerve fiber regeneration and thickness of myelination | no limits or side effects reported | Cheng et al., 2003 [17] |
| Sciatic nerve transaction (rat) | OB OECs | OEC injection in lesion side | enhancement of axonal regeneration reduction of motoneuron apoptosis | no significant difference in neuronal survival in experimental and control groups | Wang et al., 2005 [18] |
| Sciatic nerve transaction (rat) | olfactory mucosa transplantation | olfactory mucosa transplantation | SFI increased | Control group only nontransected animals | Delaviz et al.. 2008 [19] |
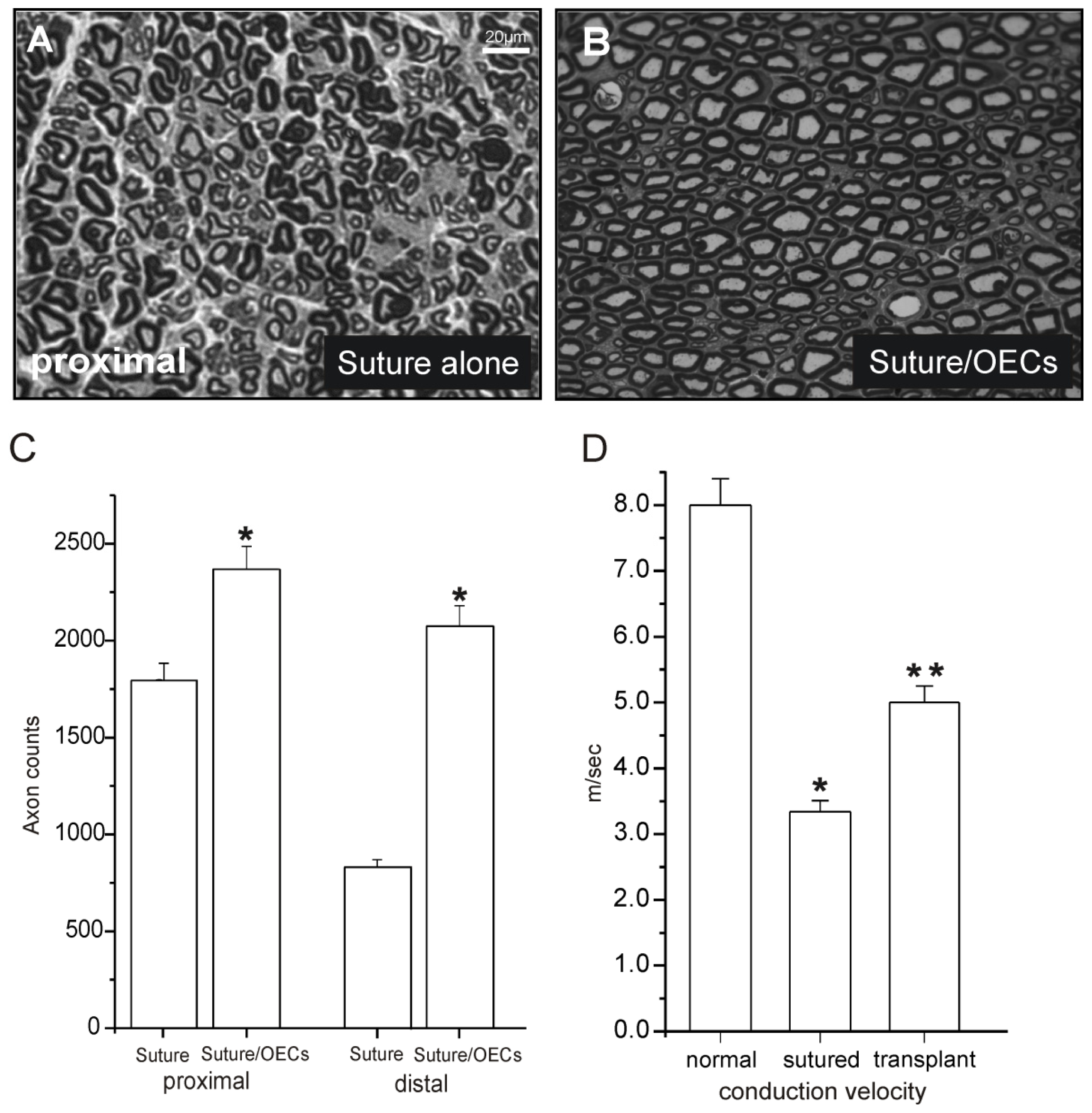
| Sciatic nerve transaction and microsurgical repair by suture (rat) | GFP-OECs purified/PKH labeled 30,000 cells/μL and 10 μL used | OECs injection proximal and distal to lesion | Axonal regeneration and remyelination newly formed nodes of Ranvier functional improvement | Observation interval limited to 3 weeks | Radtke et al., 2009 [20] |
| Sciatic nerve lesion 12–15mm gap and tube implantation (rat) | Purified PKH-labelled OB OECs 120,000 cells/tube | Silicone tubel prefilled with OECs in laminin gel | Enhancement axonal regeneration increased CMAP functional improvement | Regeneration limit at 15 mm Regeneration in 50% of animals | Verdu et al., 1999 [21] |
| Sciatic nerve lesion 10 mm PLGA conduit implantation (rat) | CM-Dil labeled OECs in 1 × 10,000 μL and 50 μL used | PLGA filled with OECs OECs in EMC | Nerve fiber regenation motor function recovery NCV and CMAP recovery | No recovery SFI after 12 weeks | Li et al., 2010 [22] |
| Sciatic nerve lesion 20 mm and PLGA conduit implantation (rat) | Purified OECs Hoechst-labelled 3 × 100,000 μL and 20 μL used | PLGA filled with OECs OECs in EMC | Enhancement axonal regeneration increased myelinated fibers recovery sensory and motor function | 20% of rats showed autophagia and heel ulcers | You et al., 2010 [23] |
| Sciatic nerve lesion and 20 mm resection, no surgical repair (rat) | Cultured OECs from olfactory bulb GFP-labelled cells, purity was determined by p75NGFR | Cell suspension was laid into transaction site immediately after resection | Muscle strength and morphometric axon counting with complete restoration, increase of neurotrophic factors | OECs did not directly on axonal regrowth, but seem to create favorable microenviroment | Guerout et al., 2011a [24] |
| Sciatic nerve lesion 15 mm and biogenic conduit implantation (rat) | Purified neonatal OECs or purified neonatal SCs | Conduits filled with OECs or SCs | Improvement in motor function | Recovery better after SCs compared to OECs with conduit implantation nerve transplant best results | Penna et al., 2012 [25] |
| Facial nerve lesion (rats) 5 mm interstump distance silicone tube | OB OECs deplated of fibroblasts 200,000 OECs | Collagen gel containing OECs in silicone tube | Increased motoneurons 10 fold increase in motoneurons increased sproutuing and pathfinding | No functional alterations | Guntinas-Lichius et al., 2001 [26] |
| Facial nerve lesion (rat) end-to end anastomosis | OM freshly prepared detection by y-chromsome | OM laid over sutured epineurium | Reduction of collateral branching promatio of functional recovery sustained expression trophic factors | No improvement of accuracy of reinnervation | Guntinas-Lichius et al., 2002 [27] |
| Facial nerve lesion (rat) | OB OECs and OM transplantation | OM pieces were applied OEC suspension injected | Moderate nerve regeneration | Only OM yielded in major improvement | Angelov et al., 2005 [28] |
| Fiacial nerve lesion (rat) and immediate repair by suture | Mixed OECs and S-type OECs | Bolus of cultured cells was applied to the cut ends before suture | Increased rate of eye closure recovery | Disorganization of the facial nucleus and aberrant nerve branching unchanged | Choi and Raisman, 2005 [29] |
| recurrent laryngeal nerve section/anastomosis (rat) | OECs from mucosa (OM-OECs), or olfactory bulb (OB-OECs) or co-transplantation of both | Cells were laid over section/anastomosis site immediately at the time of surgery (6 ×10,000 cells) | Co-transplantation of OM-OECs and OB-OECs supported major functional recovery with reduction of synkinesis | OM-OECs or OB-OECs displayed opposite abilities to improve functional recovery | Guerout et al., 2011b [30] |
| Vagus nerve transaction and immediate repair by suture | Cultivated olfactory bulb or cultivated olfactory mucosa of non-cultivated olfact. mucosa | best vocal fold angular movement with cultivated olfactory mucosa in all cell groups less synkinesis | de Corgnol et al., 2011 [31] | ||
| Complete vagus nerve lesion and anastomosis in rat | GPF OM and OB OECs 5 × 1,000,000 cells/animal | OB or OM OECs in matrigel per micropipette in anatomosis side | Improvement of reinnervation (EMG) increased myelinated fibers functional improvement | OM OECs improves muscular activity but no increases in number of myelinated fibers | Pavoit et al., 2011 [32] |
| Transection of dorsal roots L3-L6 in rats | OECs from olfactory nerve and glomerular layer, immunopurified marked with PKH28 | Impantation into DREZ | promotion of central regeneration and functional reconnection of regenerating sensory afferents, reflex recovery | immunoreactive fibers entering DH with lower density than contalateral side | Navarro et al., 1999 [33] |
| Dorsal root rhizotomy at C3-T3 in rats | purified OB-OECs | direct OEC transplantation dorsal horn OEC transplants or into the DREZ | axons regenerated at the rhizotomy site | no regeneration across DREZ no regeneration into dorsal horn | Gomez et al., 2003 [34] |
| Doral root entry zone/dorsal horn rhizotomy in rats | purified by p75NGFR OECs identification by β-gal 30,000–200,000 cells | injection of OEC suspension at DREZ/DH | no advantage in promoting ingrowth of afferent fibers in DREZ | no evidence of functional recovery of afferent fibers, minimal ingrowth of afferent fibers in SpC | Riddell et al., 2004 [35] |
| Dorsal root transection at L4 in rats | endogenous matrix containing GFP-OECs | direct application to surfaces of rootlet and SpC combined with fibrin glue | regenerated dorsal root axons crossed repaired DREZ | transplanted cells did not enter the spinal cord itself | Li et al., 2004 [36] |
| Cervical or lumbar dorsal root lesion in rats | GFP-OECs from lamina propria | OECs transplanted into DRG, intact or injured dorsal roots or the dorsal columns via DREZ | OECs migration into the DRG/dorsal root | OECs migrated within the PNS but did not cross the DREZ no primary afferent regeneration | Ramer et al., 2004 [37] |
| Dorsal roots transection C5-T2 acute and chronic lesion (rats) | GFP-OECs from OB | OECs injection in roots C4-T1 | restoration fore-paw function recovery sensory input axonal regeneration | none of chronically rhizotomized rats showed electrophysiological responses | Ibrahim et al., 2009 [38] |
| Dorsal root injury at C7 and C8 in rats | GFP-cultures enriched for OECs 6 × 10,000 cells | stereotactic injection into dorsal horn | attenuation of neuropathic pain | no improvement sensory function increasement of selfmutilation no functional improvement | Wu et al., 2010 [39] |
| Avulsion of ventral root at S1 and reimplantation (rat) | GFP-OECs and fibroblasts 1:1 | OECs transplanted at SpC interface OECs matrix cut into pieces | increase of fibers crossing lesion side migration of OECs | 20% of fibers enter roots without OEC transplantation | Li et al., 2007 [17] |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Radtke, C.; Kocsis, J.D. Peripheral Nerve Injuries and Transplantation of Olfactory Ensheathing Cells for Axonal Regeneration and Remyelination: Fact or Fiction? Int. J. Mol. Sci. 2012, 13, 12911-12924. https://doi.org/10.3390/ijms131012911
Radtke C, Kocsis JD. Peripheral Nerve Injuries and Transplantation of Olfactory Ensheathing Cells for Axonal Regeneration and Remyelination: Fact or Fiction? International Journal of Molecular Sciences. 2012; 13(10):12911-12924. https://doi.org/10.3390/ijms131012911
Chicago/Turabian StyleRadtke, Christine, and Jeffery D. Kocsis. 2012. "Peripheral Nerve Injuries and Transplantation of Olfactory Ensheathing Cells for Axonal Regeneration and Remyelination: Fact or Fiction?" International Journal of Molecular Sciences 13, no. 10: 12911-12924. https://doi.org/10.3390/ijms131012911
APA StyleRadtke, C., & Kocsis, J. D. (2012). Peripheral Nerve Injuries and Transplantation of Olfactory Ensheathing Cells for Axonal Regeneration and Remyelination: Fact or Fiction? International Journal of Molecular Sciences, 13(10), 12911-12924. https://doi.org/10.3390/ijms131012911




