Procyanidins from Wild Grape (Vitis amurensis) Seeds Regulate ARE-Mediated Enzyme Expression via Nrf2 Coupled with p38 and PI3K/Akt Pathway in HepG2 Cells
Abstract
:1. Introduction
2. Results and Discussion
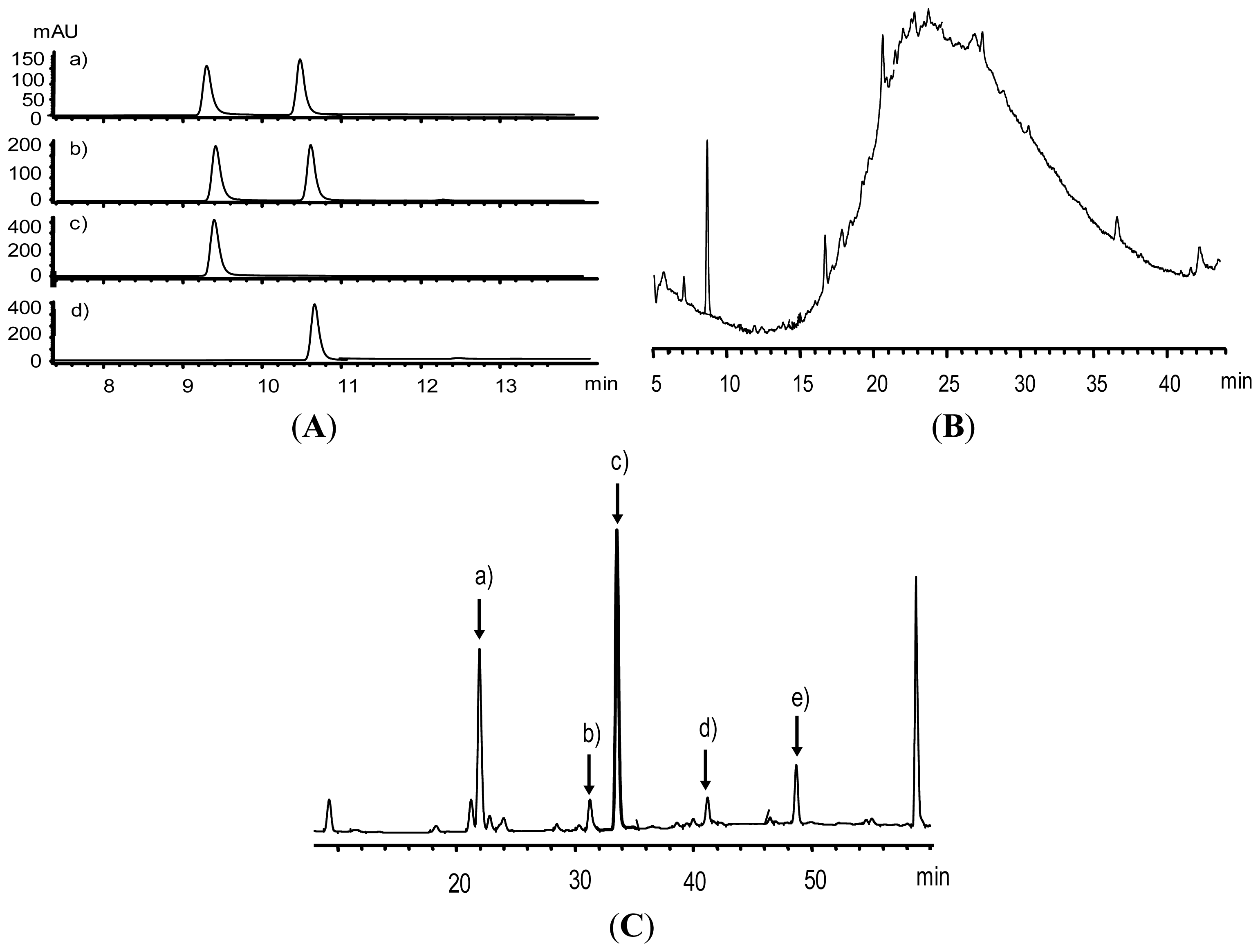
2.1. Flavan-3-ol Monomers in Fraction 4 (F4)
2.2. Analysis of Procyanidins Fraction 5 (F5)
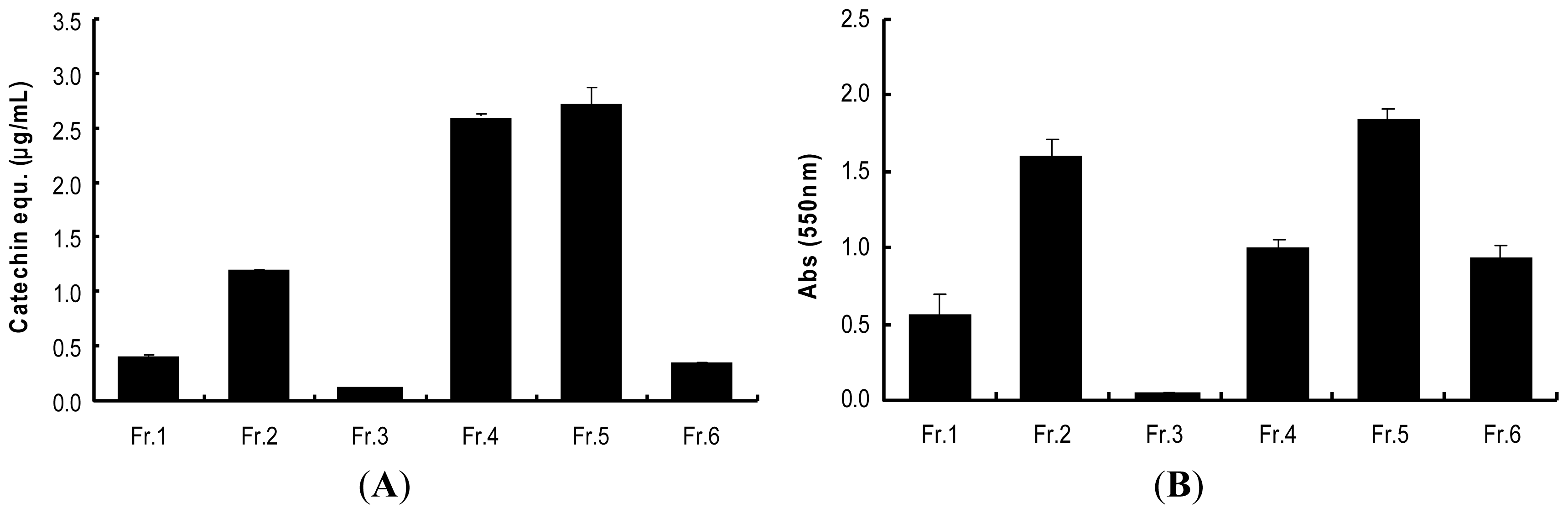
2.3. Determination of Procyanidins (F5) from Wild Grape Seed Column Fractions
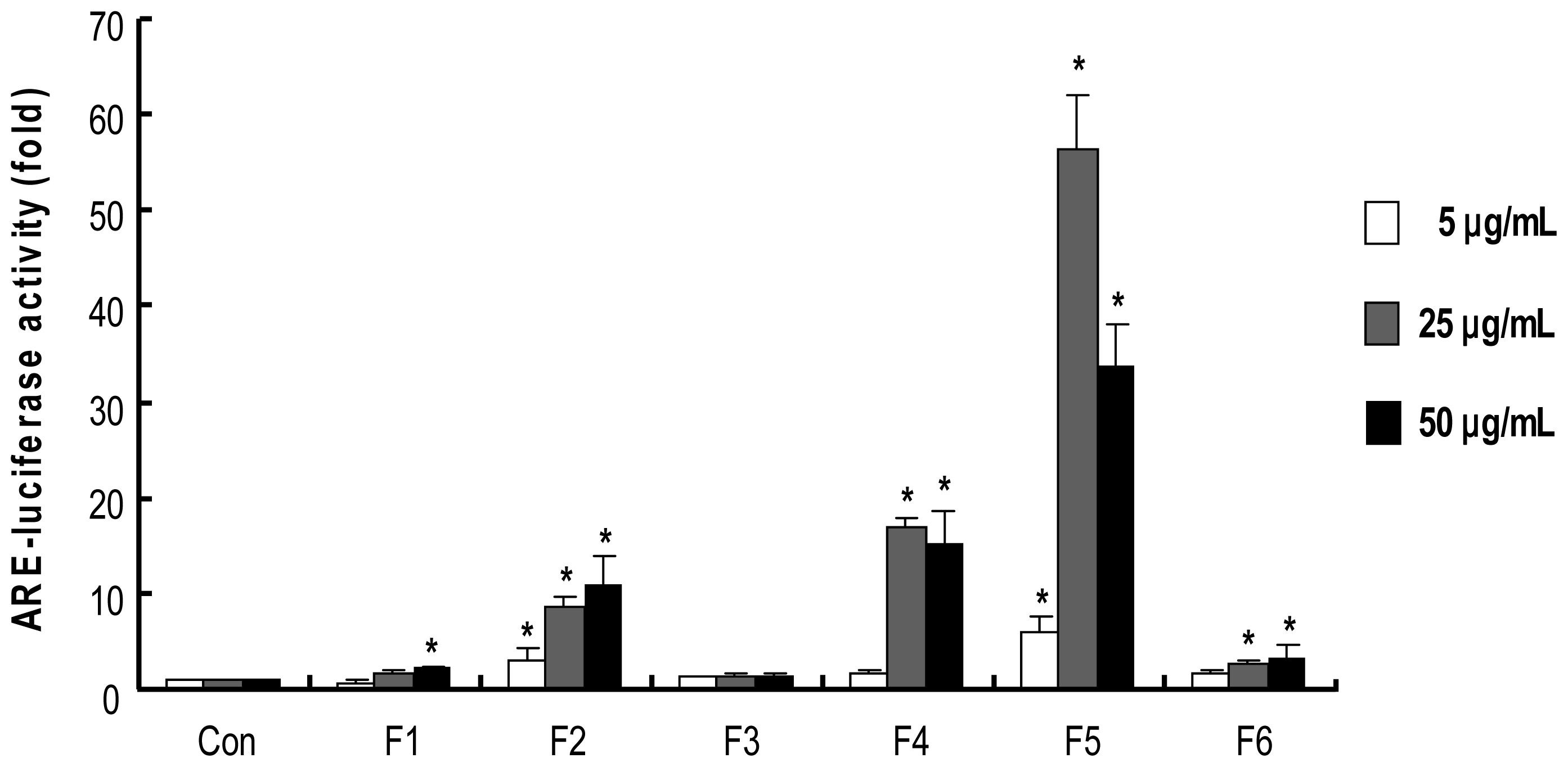
2.4. Procyanidins from Wild grape Seeds Induce ARE-Luciferase Reporter Gene Activity
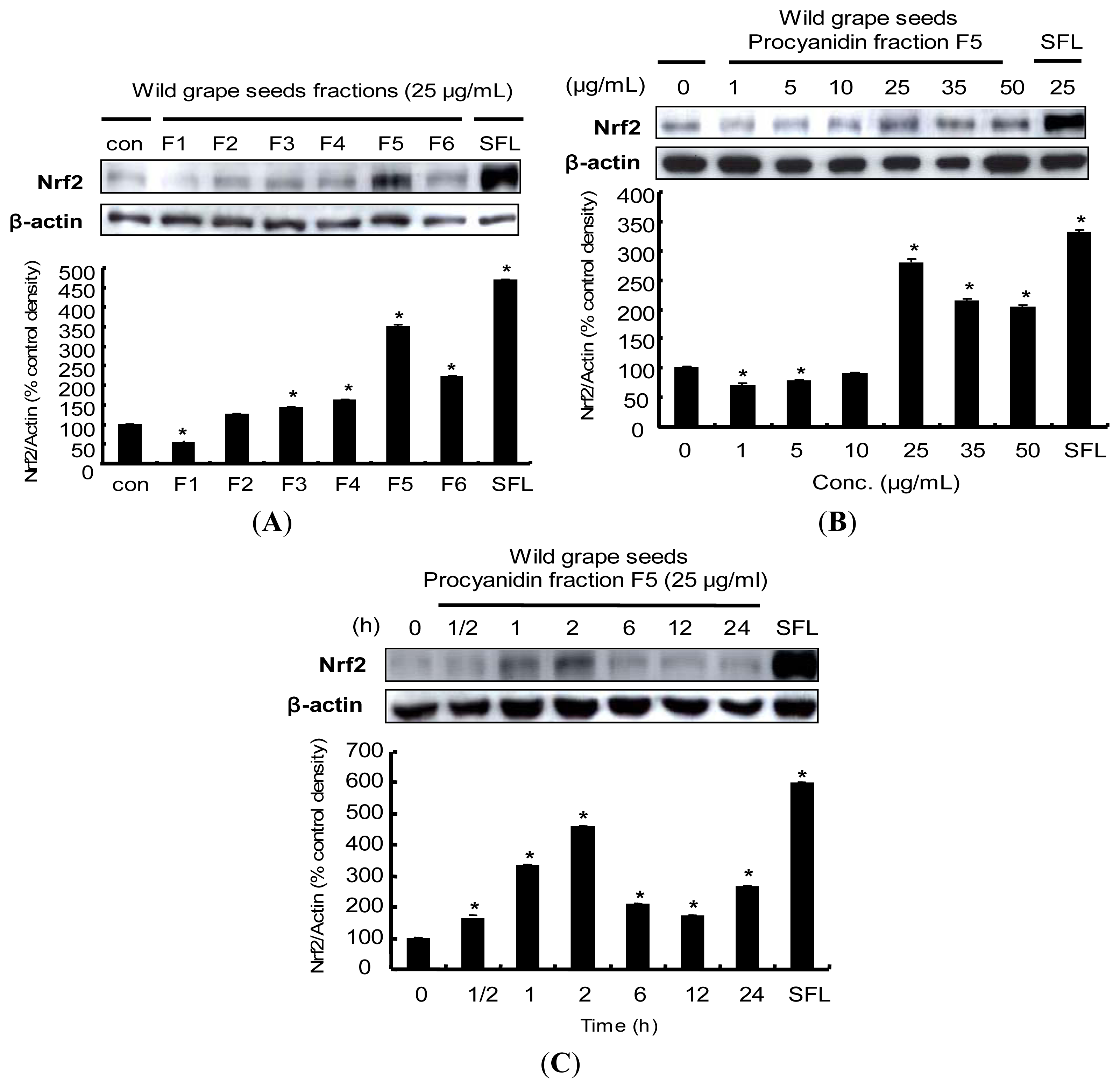
2.5. Induction of Nrf2 Protein Expression by Procyanidins from Wild Grapes Seeds in HepG2 Cells
2.6. Induction of the Nrf2-Driven Phase II Detoxifying/Antioxidant Enzyme Expression by Procyanidins from Wild Grape in HepG2 Cells
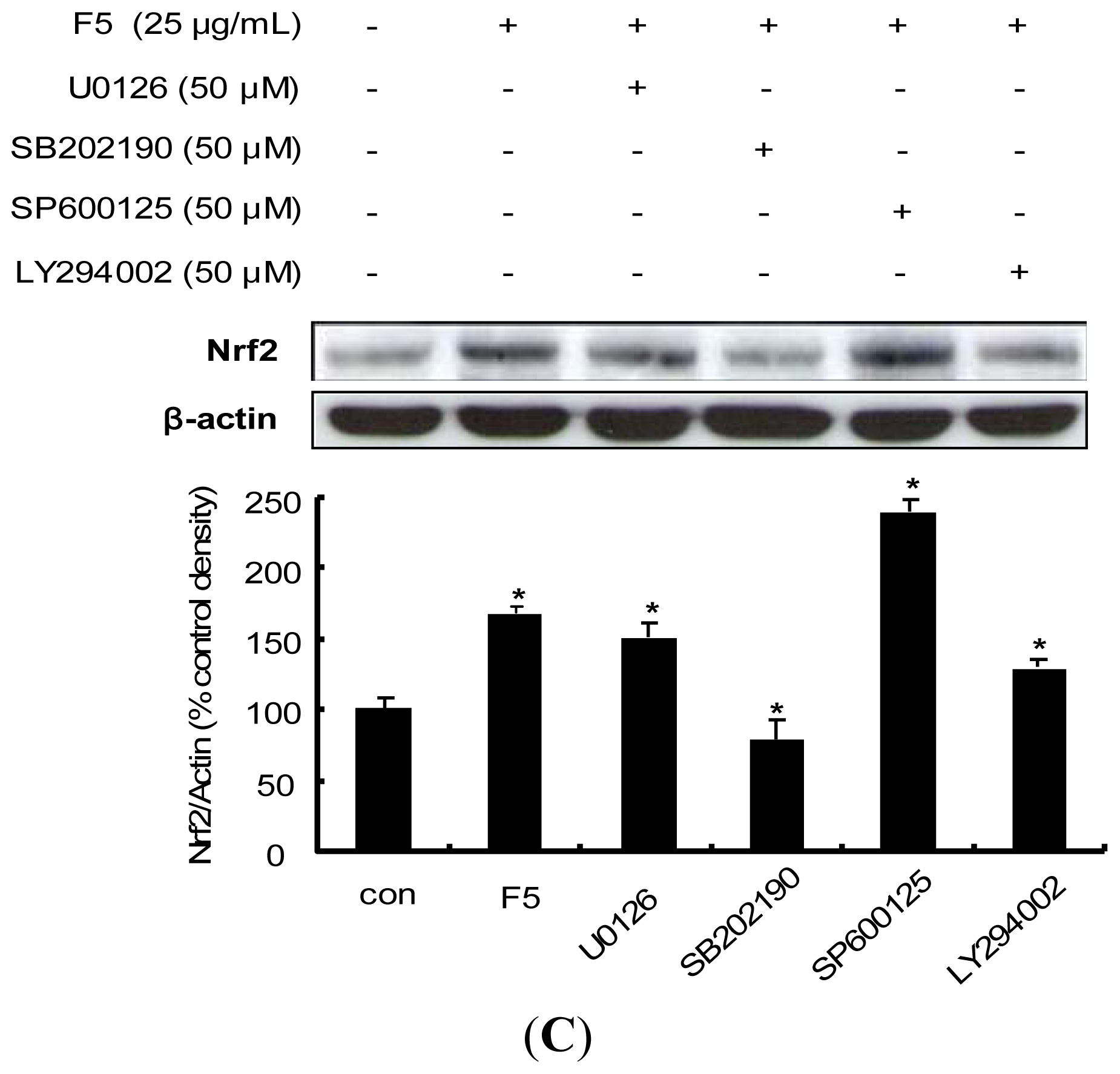
2.7. Procyanidin-Induced Nrf2 Expression Is Associated with MAPKs and PI3K/Akt Pathways
3. Experimental Section
3.1. Reagents
3.2. Extraction and Fractionation
3.3. LC-MS Analysis
3.4. Vanillin Assay
3.5. BuOH-HCl Hydrolysis
3.6. Depolymerization of Procyanidins with Phloroglucinol
3.7. Cell Culture and Treatments
3.8. ARE-Reporter Gene Activity
3.9. Western Blot Analysis
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Keum, Y.S.; Jeong, W.S.; Kong, A.N. Chemoprevention by isothiocyanates and their underlying molecular signaling mechanisms. Mutat. Res 2004, 555, 191–202. [Google Scholar]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar]
- Jeong, W.S.; Jun, M.; Kong, A.N. Nrf2: A potential molecular target for cancer chemoprevention by natural compounds. Antioxid. Redox. Signal 2006, 8, 99–106. [Google Scholar]
- Balogun, E.; Hoque, M.; Gong, P.; Killeen, E.; Green, C.J.; Foresti, R.; Alam, J.; Motterlini, R. Curcumin activates the haem oxygenase-1 gene via regulation of Nrf2 and the antioxidant-responsive element. Biochem. J 2003, 371, 887–895. [Google Scholar]
- Li, H.; Wang, Z.; Liu, Y. Review in the studies on tannins activity of cancer prevention and anticancer (in Chinese). Zhong Yao Cai 2003, 26, 444–448. [Google Scholar]
- Cos, P.; de Bruyne, T.; Hermans, N.; Apers, S.; Berghe, D.V.; Vlietinck, A.J. Proanthocyanidins in health care: Current and new trends. Curr. Med. Chem 2004, 11, 1345–1359. [Google Scholar]
- Bagchi, D.; Bagchi, M.; Stohs, S.; Ray, S.D.; Sen, C.K.; Preuss, H.G. Cellular protection with proanthocyanidins derived from grape seeds. Ann. N. Y. Acad. Sci 2002, 957, 260–270. [Google Scholar]
- Aron, P.M.; Kennedy, J.A. Flavan-3-ols: Nature, occurrence and biological activity. Mol. Nutr. Food Res 2008, 52, 79–104. [Google Scholar]
- Schafer, A.; Hogger, P. Oligomeric procyanidins of French maritime pine bark extract (Pycnogenol) effectively inhibit alpha-glucosidase. Diabetes Res. Clin. Pract 2007, 77, 41–46. [Google Scholar]
- Kamimura, A.; Takahashi, T. Procyanidin B-3, isolated from barley and identified as a hair-growth stimulant, has the potential to counteract inhibitory regulation by TGF-beta1. Exp. Dermatol 2002, 11, 532–541. [Google Scholar]
- Verstraeten, S.V.; Hammerstone, J.F.; Keen, C.L.; Fraga, C.G.; Oteiza, P.I. Antioxidant and membrane effects of procyanidin dimers and trimers isolated from peanut and cocoa. J. Agric. Food Chem 2005, 53, 5041–5048. [Google Scholar]
- Busserolles, J.; Gueux, E.; Balasinska, B.; Piriou, Y.; Rock, E.; Rayssiguier, Y.; Mazur, A. In vivo antioxidant activity of procyanidin-rich extracts from grape seed and pine (Pinus maritima) bark in rats. Int. J. Vitam. Nutr. Res 2006, 76, 22–27. [Google Scholar]
- Faria, A.; Calhau, C.; de Freitas, V.; Mateus, N. Procyanidins as antioxidants and tumor cell growth modulators. J. Agric. Food Chem 2006, 54, 2392–2397. [Google Scholar]
- Selmi, C.; Mao, T.K.; Keen, C.L.; Schmitz, H.H.; Eric Gershwin, M. The anti-inflammatory properties of cocoa flavanols. J. Cardiovasc. Pharmacol 2006, 47, S163–S171. [Google Scholar]
- Karthikeyan, K.; Bai, B.R.; Devaraj, S.N. Cardioprotective effect of grape seed proanthocyanidins on isoproterenol-induced myocardial injury in rats. Int. J. Cardiol 2007, 115, 326–333. [Google Scholar]
- da Silva Porto, P.A.; Laranjinha, J.A.; de Freitas, V.A. Antioxidant protection of low density lipoprotein by procyanidins: Structure/activity relationships. Biochem. Pharmacol 2003, 66, 947–954. [Google Scholar]
- Chang, W.C.; Hsu, F.L. Inhibition of platelet aggregation and arachidonate metabolism in platelets by procyanidins. Prostaglandins Leukot. Essent. Fatty Acids 1989, 38, 181–188. [Google Scholar]
- Sharma, S.D.; Meeran, S.M.; Katiyar, S.K. Dietary grape seed proanthocyanidins inhibit UVB-induced oxidative stress and activation of mitogen-activated protein kinases and nuclear factor-kappaB signaling in in vivo SKH-1 hairless mice. Mol. Cancer Ther 2007, 6, 995–1005. [Google Scholar]
- Matito, C.; Agell, N.; Sanchez-Tena, S.; Torres, J.L.; Cascante, M. Protective effect of structurally diverse grape procyanidin fractions against UV-induced cell damage and death. J. Agric. Food Chem 2011, 59, 4489–4495. [Google Scholar]
- Gali, H.U.; Perchellet, E.M.; Gao, X.M.; Laks, P.E.; Perchellet, J.P. Inhibitory effects of semisynthetic flavonoid derivatives on the biochemical markers of tumor promotion in mouse epidermis in vivo. Cancer Lett 1993, 72, 149–156. [Google Scholar]
- Gerhauser, C. Cancer chemopreventive potential of apples, apple juice, and apple components. Planta Med 2008, 74, 1608–1624. [Google Scholar]
- Nomoto, H.; Iigo, M.; Hamada, H.; Kojima, S.; Tsuda, H. Chemoprevention of colorectal cancer by grape seed proanthocyanidin is accompanied by a decrease in proliferation and increase in apoptosis. Nutr. Cancer 2004, 49, 81–88. [Google Scholar]
- Gosse, F.; Guyot, S.; Roussi, S.; Lobstein, A.; Fischer, B.; Seiler, N.; Raul, F. Chemopreventive properties of apple procyanidins on human colon cancer-derived metastatic SW620 cells and in a rat model of colon carcinogenesis. Carcinogenesis 2005, 26, 1291–1295. [Google Scholar]
- Eng, E.T.; Ye, J.; Williams, D.; Phung, S.; Moore, R.E.; Young, M.K.; Gruntmanis, U.; Braunstein, G.; Chen, S. Suppression of estrogen biosynthesis by procyanidin dimers in red wine and grape seeds. Cancer Res 2003, 63, 8516–8522. [Google Scholar]
- Raina, K.; Singh, R.P.; Agarwal, R.; Agarwal, C. Oral grape seed extract inhibits prostate tumor growth and progression in TRAMP mice. Cancer Res 2007, 67, 5976–5982. [Google Scholar]
- College, J.N.M. Dictionary of Chinese Traditional Medicines; Shanghai Science and Technology Press: Shanghai, China, 1984. [Google Scholar]
- Lee, H.R.; Bak, M.J.; Jeong, W.S.; Kim, Y.C.; Chung, S.K. Antioxidant properties of proanthocyanidin fraction isolated from wild grape (Vitis amurensis) seed. J. Korean Soc. Appl. Biol. Chem 2009, 52, 539–544. [Google Scholar]
- Wang, J.N.; Hano, Y.; Nomura, T.; Chen, Y.J. Procyanidins from the seeds of Vitis amurensis. Phytochemistry 2000, 53, 1097–1102. [Google Scholar]
- Lee, E.O.; Lee, H.J.; Hwang, H.S.; Ahn, K.S.; Chae, C.; Kang, K.S.; Lu, J.; Kim, S.H. Potent inhibition of Lewis lung cancer growth by heyneanol A from the roots of Vitis amurensis through apoptotic and anti-angiogenic activities. Carcinogenesis 2006, 27, 2059–2069. [Google Scholar]
- Huang, K.S.; Lin, M.; Cheng, G.F. Anti-inflammatory tetramers of resveratrol from the roots of Vitis amurensis and the conformations of the seven-membered ring in some oligostilbenes. Phytochemistry 2001, 58, 357–362. [Google Scholar]
- Jang, M.H.; Piao, X.L.; Kim, H.Y.; Cho, E.J.; Baek, S.H.; Kwon, S.W.; Park, J.H. Resveratrol oligomers from Vitis amurensis attenuate beta-amyloid-induced oxidative stress in PC12 cells. Biol. Pharm. Bull 2007, 30, 1130–1134. [Google Scholar]
- Kim, J.Y.; Jeong, H.Y.; Lee, H.K.; Kim, S.; Hwang, B.Y.; Bae, K.; Seong, Y.H. Neuroprotection of the leaf and stem of Vitis amurensis and their active compounds against ischemic brain damage in rats and excitotoxicity in cultured neurons. Phytomedicine 2011, 19, 150–159. [Google Scholar]
- Park, M.J.; Kang, H.S.; Jin, J.S.; Kim, Y.H.; OK, S.; Jeong, J.H.; Lee, H.R.; Lim, H.J.; Jo, W.K.; Kim, Y.C.; Chung, S.K.; Jeong, W.S. Chemopreventive properties of Wild grape (Vitis amurensis) extracts. J. Clin. Biochem. Nutr 2008, 43, 59–61. [Google Scholar]
- Surh, Y.J.; Kundu, J.K.; Na, H.K. Nrf2 as a master redox switch in turning on the cellular signaling involved in the induction of cytoprotective genes by some chemopreventive phytochemicals. Planta Med 2008, 74, 1526–1539. [Google Scholar]
- Eggler, A.L.; Gay, K.A.; Mesecar, A.D. Molecular mechanisms of natural products in chemoprevention: Induction of cytoprotective enzymes by Nrf2. Mol. Nutr. Food Res 2008, 52, S84–S94. [Google Scholar]
- Ramos-Gomez, M.; Kwak, M.K.; Dolan, P.M.; Itoh, K.; Yamamoto, M.; Talalay, P.; Kensler, T.W. Sensitivity to carcinogenesis is increased and chemoprotective efficacy of enzyme inducers is lost in nrf2 transcription factor-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 3410–3415. [Google Scholar]
- McCarty, M.F. Polyphenol-mediated inhibition of AP-1 transactivating activity may slow cancer growth by impeding angiogenesis and tumor invasiveness. Med. Hypotheses 1998, 50, 511–514. [Google Scholar]
- Sun, Z.; Huang, Z.; Zhang, D.D. Phosphorylation of Nrf2 at multiple sites by MAP kinases has a limited contribution in modulating the Nrf2-dependent antioxidant response. PLoS One 2009, 4, e6588:1–e6588:9. [Google Scholar]
- Yu, R.; Chen, C.; Mo, Y.Y.; Hebbar, V.; Owuor, E.D.; Tan, T.H.; Kong, A.N. Activation of mitogen-activated protein kinase pathways induces antioxidant response element-mediated gene expression via a Nrf2-dependent mechanism. J. Biol. Chem 2000, 275, 39907–39913. [Google Scholar]
- Nandakumar, V.; Singh, T.; Katiyar, S.K. Multi-targeted prevention and therapy of cancer by proanthocyanidins. Cancer Lett 2008, 269, 378–387. [Google Scholar]
- Shih, P.H.; Yen, G.C. Differential expressions of antioxidant status in aging rats: The role of transcriptional factor Nrf2 and MAPK signaling pathway. Biogerontology 2007, 8, 71–80. [Google Scholar]
- Lim, J.H.; Park, H.S.; Choi, J.K.; Lee, I.S.; Choi, H.J. Isoorientin induces Nrf2 pathway-driven antioxidant response through phosphatidylinositol 3-kinase signaling. Arch. Pharm. Res 2007, 30, 1590–1598. [Google Scholar]
- Kennedy, J.A.; Jones, G.P. Analysis of proanthocyanidin cleavage products following acid-catalysis in the presence of excess phloroglucinol. J. Agric. Food Chem 2001, 49, 1740–1746. [Google Scholar]
- Souguet, J.M.; Cheynier, V.; Brossand, F.; Moutounet, M. Polymeric proanthocyanidins from grape skins. Phytochemistry 1996, 43, 509–512. [Google Scholar]
- Yu, R.; Lei, W.; Mandlekar, S.; Weber, M.J.; Der, C.J.; Wu, J.; Kong, A.N. Role of a mitogen-activated protein kinase pathway in the induction of phase II detoxifying enzymes by chemicals. J. Biol. Chem 1999, 274, 27545–27552. [Google Scholar]
- Martin, D.; Rojo, A.I.; Salinas, M.; Diaz, R.; Gallardo, G.; Alam, J.; de Galarreta, C.M.; Cuadrado, A. Regulation of heme oxygenase-1 expression through the phosphatidylinositol 3-kinase/Akt pathway and the Nrf2 transcription factor in response to the antioxidant phytochemical carnosol. J. Biol. Chem 2004, 279, 8919–8929. [Google Scholar]
- Keum, Y.S.; Yu, S.; Chang, P.P.; Yuan, X.; Kim, J.H.; Xu, C.; Han, J.; Agarwal, A.; Kong, A.N. Mechanism of action of sulforaphane: Inhibition of p38 mitogen-activated protein kinase isoforms contributing to the induction of antioxidant response element-mediated heme oxygenase-1 in human hepatoma HepG2 cells. Cancer Res 2006, 66, 8804–8813. [Google Scholar]
- McNally, S.J.; Harrison, E.M.; Ross, J.A.; Garden, O.J.; Wigmore, S.J. Curcumin induces heme oxygenase 1 through generation of reactive oxygen species, p38 activation and phosphatase inhibition. Int. J. Mol. Med 2007, 19, 165–172. [Google Scholar]
- Gong, P.; Hu, B.; Cederbaum, A.I. Diallyl sulfide induces heme oxygenase-1 through MAPK pathway. Arch. Biochem. Biophys 2004, 432, 252–260. [Google Scholar]
- Tanigawa, S.; Fujii, M.; Hou, D.X. Action of Nrf2 and Keap1 in ARE-mediated NQO1 expression by quercetin. Free Radic. Biol. Med 2007, 42, 1690–1703. [Google Scholar]
- Hanneken, A.; Lin, F.F.; Johnson, J.; Maher, P. Flavonoids protect human retinal pigment epithelial cells from oxidative-stress-induced death. Invest. Ophthalmol. Vis. Sci 2006, 47, 3164–3177. [Google Scholar]
- Hsieh, T.C.; Lu, X.; Wang, Z.; Wu, J.M. Induction of quinone reductase NQO1 by resveratrol in human K562 cells involves the antioxidant response element ARE and is accompanied by nuclear translocation of transcription factor Nrf2. Med. Chem 2006, 2, 275–285. [Google Scholar]
- Kode, A.; Rajendrasozhan, S.; Caito, S.; Yang, S.R.; Megson, I.L.; Rahman, I. Resveratrol induces glutathione synthesis by activation of Nrf2 and protects against cigarette smoke-mediated oxidative stress in human lung epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol 2008, 294, L478–L488. [Google Scholar]
- Chen, C.Y.; Jang, J.H.; Li, M.H.; Surh, Y.J. Resveratrol upregulates heme oxygenase-1 expression via activation of NF-E2-related factor 2 in PC12 cells. Biochem. Biophys. Res. Commun 2005, 331, 993–1000. [Google Scholar]
- Seo, J.Y.; Lee, Y.S.; Kim, H.J.; Lim, S.S.; Lim, J.S.; Lee, I.A.; Lee, C.H.; Yoon Park, J.H.; Kim, J.S. Dehydroglyasperin C isolated from licorice caused Nrf2-mediated induction of detoxifying enzymes. J. Agric. Food Chem 2011, 58, 1603–1608. [Google Scholar]
- Ross, D. Quinone reductases multitasking in the metabolic world. Drug Metab. Rev 2004, 36, 639–654. [Google Scholar]
- Jana, S.; Mandlekar, S. Role of phase II drug metabolizing enzymes in cancer chemoprevention. Curr. Drug Metab 2009, 10, 595–616. [Google Scholar]
- Joung, E.J.; Li, M.H.; Lee, H.G.; Somparn, N.; Jung, Y.S.; Na, H.K.; Kim, S.H.; Cha, Y.N.; Surh, Y.J. Capsaicin induces heme oxygenase-1 expression in HepG2 cells via activation of PI3K-Nrf2 signaling: NAD(P)H:quinone oxidoreductase as a potential target. Antioxid. Redox. Signal 2007, 9, 2087–2098. [Google Scholar]
- Zhang, Y.; Guan, L.; Wang, X.; Wen, T.; Xing, J.; Zhao, J. Protection of chlorophyllin against oxidative damage by inducing HO-1 and NQO1 expression mediated by PI3K/Akt and Nrf2. Free Radic. Res 2008, 42, 362–371. [Google Scholar]
- Kang, E.S.; Woo, I.S.; Kim, H.J.; Eun, S.Y.; Paek, K.S.; Kim, H.J.; Chang, K.C.; Lee, J.H.; Lee, H.T.; Kim, J.H.; et al. Up-regulation of aldose reductase expression mediated by phosphatidylinositol 3-kinase/Akt and Nrf2 is involved in the protective effect of curcumin against oxidative damage. Free Radic. Biol. Med 2007, 43, 535–545. [Google Scholar]
- Chen, C.; Pung, D.; Leong, V.; Hebbar, V.; Shen, G.; Nair, S.; Li, W.; Kong, A.N. Induction of detoxifying enzymes by garlic organosulfur compounds through transcription factor Nrf2: Effect of chemical structure and stress signals. Free Radic. Biol. Med 2004, 37, 1578–1590. [Google Scholar]
- Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1985, 25, 223–230. [Google Scholar]
- Creaser, C.S.; Koupai-Abyazani, M.R.; Stephenson, G.R. Gas chromatographic-mass spectrometric characterization of flavanones in citrus and grape juices. Analyst 1992, 117, 1105–1109. [Google Scholar]
- Jeong, W.S.; Keum, Y.S.; Chen, C.; Jain, M.R.; Shen, G.; Kim, J.H.; Li, W.; Kong, A.N. Differential expression and stability of endogenous nuclear factor E2-related factor 2 (Nrf2) by natural chemopreventive compounds in HepG2 human hepatoma cells. J. Biochem. Mol. Biol 2005, 38, 167–176. [Google Scholar]


© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bak, M.-J.; Jun, M.; Jeong, W.-S. Procyanidins from Wild Grape (Vitis amurensis) Seeds Regulate ARE-Mediated Enzyme Expression via Nrf2 Coupled with p38 and PI3K/Akt Pathway in HepG2 Cells. Int. J. Mol. Sci. 2012, 13, 801-818. https://doi.org/10.3390/ijms13010801
Bak M-J, Jun M, Jeong W-S. Procyanidins from Wild Grape (Vitis amurensis) Seeds Regulate ARE-Mediated Enzyme Expression via Nrf2 Coupled with p38 and PI3K/Akt Pathway in HepG2 Cells. International Journal of Molecular Sciences. 2012; 13(1):801-818. https://doi.org/10.3390/ijms13010801
Chicago/Turabian StyleBak, Min-Ji, Mira Jun, and Woo-Sik Jeong. 2012. "Procyanidins from Wild Grape (Vitis amurensis) Seeds Regulate ARE-Mediated Enzyme Expression via Nrf2 Coupled with p38 and PI3K/Akt Pathway in HepG2 Cells" International Journal of Molecular Sciences 13, no. 1: 801-818. https://doi.org/10.3390/ijms13010801




