The Effects of G-CSF on Proliferation of Mouse Myocardial Microvascular Endothelial Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Mice Myocardical Microvascular Endothelial Cell Cultures
2.3. Proliferation Assay
2.4. Reverse Transcription and Polymerase Chain Reaction (RT-PCR)
2.5. Statistics
3. Results
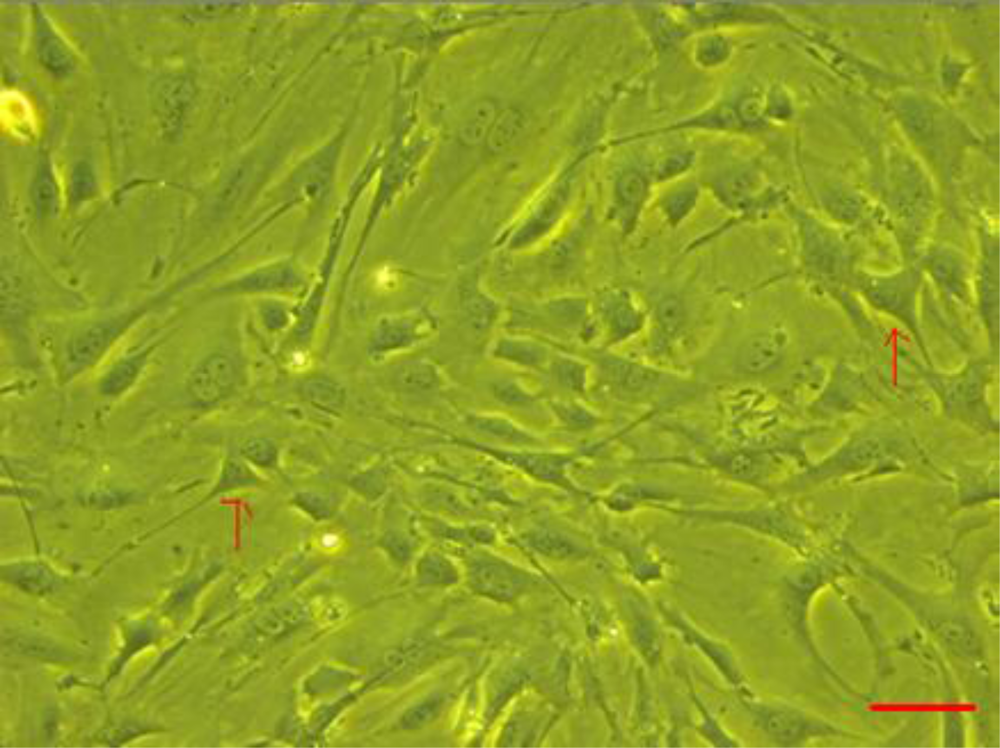
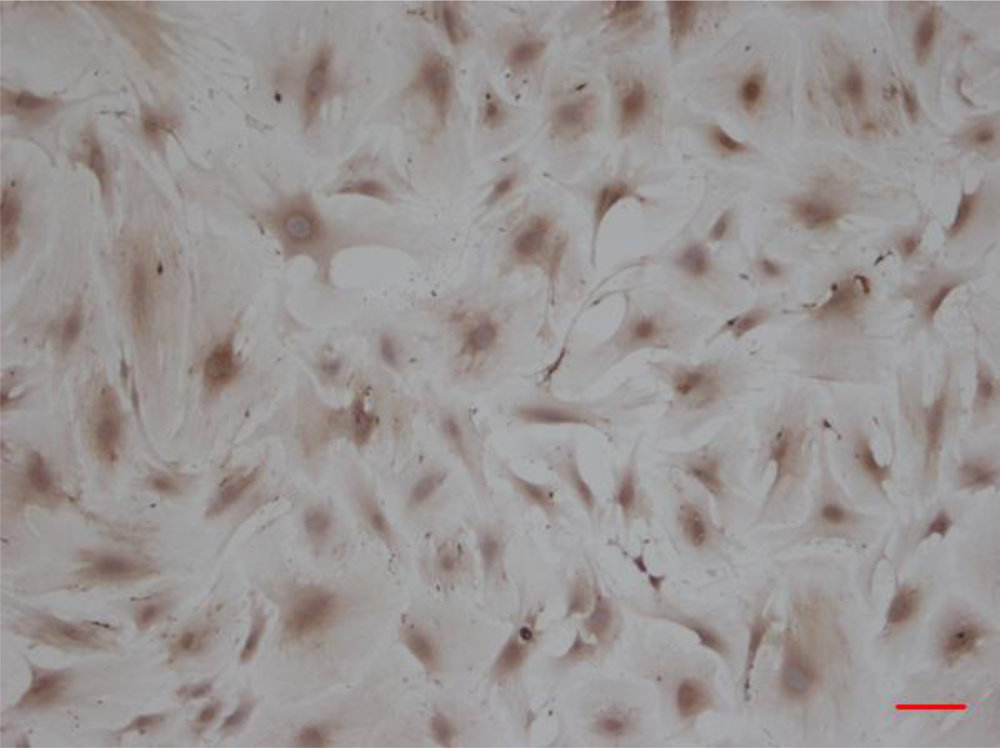
3.1. CMECs Morphology and Identification
3.2. CMEC Proliferative Capacity
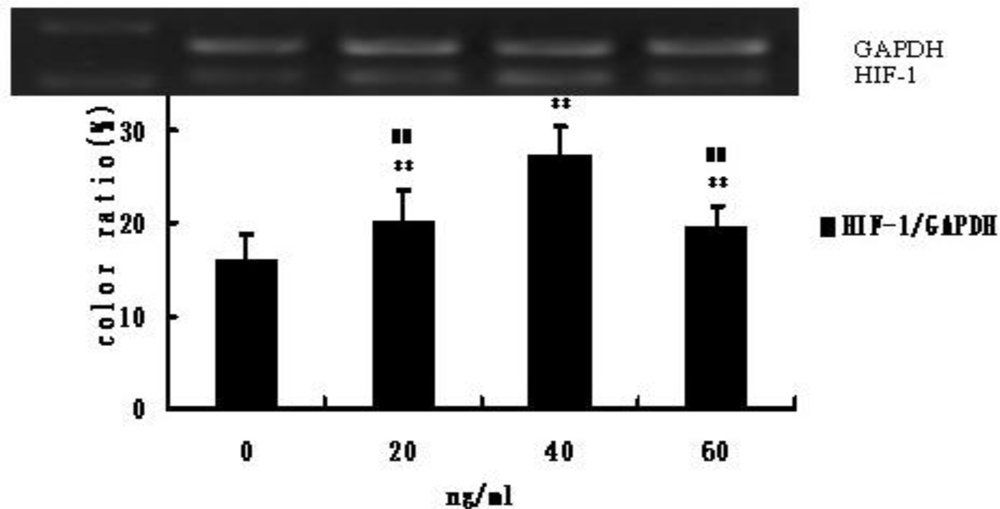
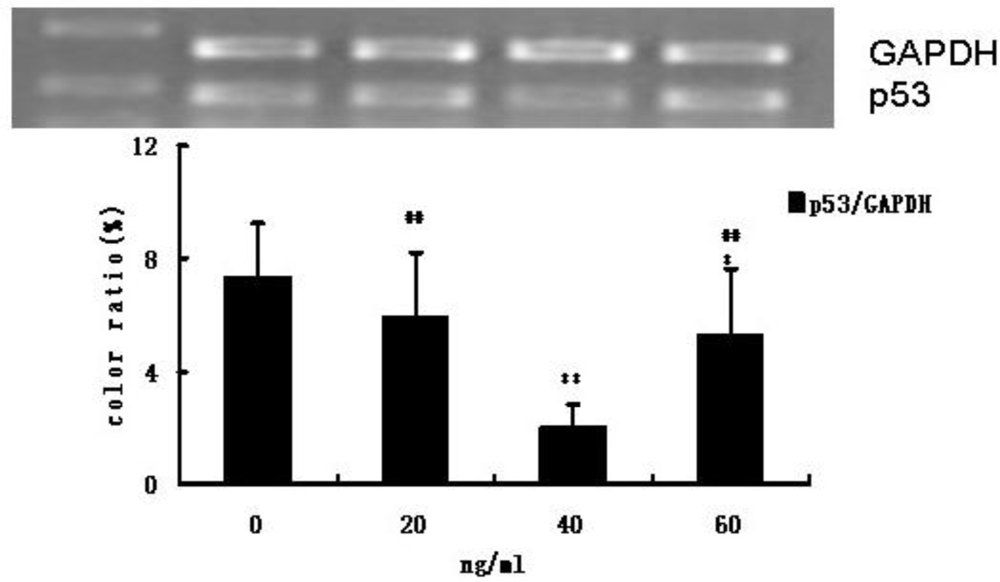
3.3. HIF-1 and p53 mRNA Expression
4. Discussion
5. Conclusion
Acknowledgments
References
- Hasegawa, H; Takano, H; Shiraishi, H; Ueda, K; Niitsuma, Y; Tadokoro, H; Komuro, I. Intracoronary injection of granulocyte colony-stimulating factor ameliorates the progression of left ventricular remodeling after myocardial ischemia/reperfusion in rabbits. Circ. J 2006, 70, 942–944. [Google Scholar]
- Jeon, O; Hwang, KC; Yoo, KJ; Kim, BS. Combined sustained delivery of basic fibroblast growth factor and administration of granulocyte colony-stimulating factor: Synergistic effect on angiogenesis in mouse ischemic limbs. J. Endovasc. Ther 2006, 13, 175–181. [Google Scholar]
- Lee, M; Aoki, M; Kondo, T; Kobayashi, K; Okumura, K; Komori, K; Murohara, T. Therapeutic angiogenesis with intramuscular injection of low-dose recombinant granulocyte-colony stimulating factor. Arterioscler. Thromb 2005, 25, 2535–2541. [Google Scholar]
- Ohki, Y; Heissig, B; Sato, Y; Akiyama, H; Zhu, Z; Hicklin, DJ; Shimada, K; Ogawa, H; Daida, H; Hattori, K; Ohsaka, A. Granulocyte colony-stimulating factor promotes neovascularization by releasing vascular endothelial growth factor from neutrophils. FASEB J 2005, 19, 2005–2007. [Google Scholar]
- Takagi, Y; Omura, T; Yoshiyama, M; Matsumoto, R; Enomoto, S; Kusuyama, T; Nishiya, D; Akioka, K; Iwao, H; Takeuchi, K; Yoshikawa, J. Granulocyte-colony stimulating factor augments neovascularization induced by bone marrow transplantation in rat hindlimb ischemia. J. Pharmacol. Sci 2005, 99, 45–51. [Google Scholar]
- Deindl, E; Zaruba, MM; Brunner, S; Huber, B; Mehl, U; Assmann, G; Hoefer, IE; Mueller-Hoecker, J; Franz, WM. G-CSF administration after myocardial infarction in mice attenuates late ischemic cardiomyopathy by enhanced arteriogenesis. FASEB J 2006, 20, 956–958. [Google Scholar]
- Kang, HJ; Kim, HS; Zhang, SY; Park, KW; Cho, HJ; Koo, BK; Kim, YJ; Soo Lee, D; Sohn, DW; Han, KS; Oh, BH; Lee, MM; Park, YB. Effects of intracoronary infusion of peripheral blood stem-cells mobilised with granulocyte-colony stimulating factor on left ventricular systolic function and restenosis after coronary stenting in myocardial infarction: The MAGIC cell randomised clinical trial. Lancet 2004, 363, 751–756. [Google Scholar]
- Ripa, RS; Jorgensen, E; Wang, Y; Thune, JJ; Nilsson, JC; Søndergaard, L; Johnsen, HE; Køber, L; Grande, P; Kastrup, J. Stem cell mobilization induced by subcutaneous granulocyte-colony stimulating factor to improve cardiac regeneration after acute ST-elevation myocardial infarction: Result of the double-blind, randomized, placebo-controlled stem cells in myocardial infarction (STEMMI) trial. Circulation 2006, 113, 1983–1992. [Google Scholar]
- Suzuki, K; Nagashima, K; Arai, M; Uno, Y; Misao, Y; Takemura, G; Nishigaki, K; Minatoguchi, S; Watanabe, S; Tei, C; Fujiwara, H. Effect of granulocyte colonystimulating factor treatment at a low dose but for a long duration in patients with coronary heart disease. Circ. J 2006, 70, 430–437. [Google Scholar]
- Wang, Y; Tagil, K; Ripa, RS; Nilsson, JC; Carstensen, S; Jørgensen, E; Sondergaard, L; Hesse, B; Johnsen, HE; Kastrup, J. Effect of mobilization of bone marrow stem cells by granulocyte colony stimulating factor on clinical symptoms, left ventricular perfusion and function in patients with severe chronic ischemic heart disease. Int. J. Cardiol 2005, 100, 477–483. [Google Scholar]
- Zohlnhofer, D; Ott, I; Mehilli, J; Schomig, K; Michalk, F; Ibrahim, T; Meisetschlager, G; von Wedel, J; Bollwein, H; Seyfarth, M; Dirschinger, J; Schmitt, C; Schwaiger, M; Kastrati, A; Schomig, A. Stem cell mobilization by granulocyte colony-stimulating factor in patients with acute myocardial infarction: a randomized controlled trial. JAMA 2006, 295, 1003–1010. [Google Scholar]
- Ohki, Y; Heissig, B; Sato, Y; Akiyama, H; Zhu, Z; Hicklin, DJ; Shimada, K; Ogawa, H; Daida, H; Hattori, K; Ohsaka, A. Granulocyte colony-stimulating factor promotes neovascularization by releasing vascular endothelial growth factor from neutrophils. FASEB J 2005, 19, 2005–2007. [Google Scholar]
- Sano, M; Minamino, T; Toko, H; Miyauchi, H; Orimo, M; Qin, YJ; Akazawa, H; Tateno, K; Kayama, Y; Harada, M; Shimizu, I; Asahara, T; Hamada, H; Tomita, S; Molkentin, JD; Zou, YZ; Komuro, I. p53-induced inhibition of Hif-1 causes cardiac dysfunction during pressure overload. Nature 2007, 446, 444–448. [Google Scholar]
- Alaiti, MA; Ishikawa, M; Costa, MA. Bone marrow and circulating stem/progenitor cells for regenerative cardiovascular therapy. Transl. Res 2010, 156, 112–129. [Google Scholar]
- Kim, Y-J; Shin, J-I; Park, K-W; Lee, H-Y; Kang, H-J; Koo, B-K; Park, B-J; Sohn, D-W; Oh, B-H; Park, Y-B. The effect of granulocyte-colony stimulating factor on endothelial function in patients with myocardial infarction. Heart 2009, 95, 1320–1325. [Google Scholar]
- Toblli, JE; Cao, G; DeRosa, G; Di Gennaro, F; Forcada, P. Angiotensin-Converting enzyme inhibition and angiogenesis in myocardium of obese Zucker rats. Am. J. Hypertens 2004, 17, 172–180. [Google Scholar]
- Hull, MA; Hewett, PW; Brough, JL; Hawkey, CJ. Isolation and culture of human gastric endothelial cells. Gastroenterology 1996, 111, 1230–1240. [Google Scholar]
- Schaeffer, P; Laplace, M-C; Prabonnaud, V; Bernat, A; Gully, D; Lespy, L; Herbert, J-M. Neurotensin induces the release of prostacyclin from human umbilical vein endothelial cells in vitro and increases plasma prostacyclin levels in the rat. Eur. J. Pharmacol 1997, 323, 215–221. [Google Scholar]
- Marin, V; Kaplanski, G; Grès, S; Farnarier, C; Bongrand, P. Endothelial cell culture: protocol to obtain and cultivate human umbilical endothelial cells. J. Immunol. Methods 2001, 254, 183–190. [Google Scholar]
- Mountain, DJH; Singh, M; Singh, K. Interleukin-1β-mediated inhibition of the processes of angiogenesis in cardiac microvascular endothelial cells. Life Sci 2008, 82, 1224–1230. [Google Scholar]
- Seo, H-W; Kim, E-J; Na, H; Lee, M-O. Transcriptional activation of hypoxia-inducible factor-1α by HDAC4 and HDAC5 involves differential recruitment of p300 and FIH-1. FEBS Lett 2009, 583, 55–60. [Google Scholar]
- Ma, JW; Qiao, ZY; Xiang, X. Optimisation of extraction procedure for black fungus polysaccharides and effect of the polysaccharides on blood lipid and myocardium antioxidant enzymes activities. Carbohydr. Polym 2011, 84, 1061–1068. [Google Scholar]
- Wang, GL; Semenza, GL. Desferrioxamine induces erythropoietin gene expression and hypoxia-inducible factor 1 DNA-binding activity: implications for models of hypoxia signal transduction. Blood 1993, 82, 3610–3615. [Google Scholar]
- Wenger, RH. Cellular adaptation to hypoxia: O2-sensing protein hydroxylases, hypoxia-inducible transcription factors, and O2-regulated gene expression. FASEB J 2002, 16, 1151–1162. [Google Scholar]
- Narasimhan, M; Rose, R; Karthikeyan, M; Rathinavelu, A. Detection of HDM2 and VEGF co-expression in cancer cell lines: novel effect of HDM2 antisense treatment on VEGF expression. Life Sci 2007, 81, 1362–1372. [Google Scholar]
- Cadwell, C; Zambetti, GP. The effects of wild-type p53 tumor suppressor activity and mutant p53 gain-of-function on cell growth. Gene 2001, 277, 15–30. [Google Scholar]

© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Zou, Y.; Ge, J.; Zhang, D.; Guan, A.; Wu, J.; Li, L. The Effects of G-CSF on Proliferation of Mouse Myocardial Microvascular Endothelial Cells. Int. J. Mol. Sci. 2011, 12, 1306-1315. https://doi.org/10.3390/ijms12021306
Li J, Zou Y, Ge J, Zhang D, Guan A, Wu J, Li L. The Effects of G-CSF on Proliferation of Mouse Myocardial Microvascular Endothelial Cells. International Journal of Molecular Sciences. 2011; 12(2):1306-1315. https://doi.org/10.3390/ijms12021306
Chicago/Turabian StyleLi, Jiming, Yunzeng Zou, Junbo Ge, Daifu Zhang, Aili Guan, Jian Wu, and Lei Li. 2011. "The Effects of G-CSF on Proliferation of Mouse Myocardial Microvascular Endothelial Cells" International Journal of Molecular Sciences 12, no. 2: 1306-1315. https://doi.org/10.3390/ijms12021306



