Comparative Study Using Different Infrared Zones of the Solventless Activation of Organic Reactions
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental
4. Conclusions
Acknowledgments
References and Notes
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Matlack, A.S. Introduction to Green Chemistry; Marcel Dekker: New York, NY, USA, 2001. [Google Scholar]
- Lancaster, M. Green Chemistry: An Introductory Text; Royal Society of Chemistry: Cambridge, UK, 2002. [Google Scholar]
- Hjeresen, D.L.; Schutt, D.L.; Boese, J.M. Green chemistry and education. J. Chem. Educ 2000, 77, 1543–1544. [Google Scholar]
- Collins, T.J. Introducing green chemistry in teaching and research. J. Chem. Educ 1995, 72, 965–966. [Google Scholar]
- Arroyo, G.; Hernández, E.; Martínez, J.; Miranda, R.; Noguez, O.; Penieres, J.; Rivero, C.; Velasco, B.; Vilchis, M.; Gómez, C. Prácticas de Laboratorio de Química Orgánica Verde; Editorial Tecnológica de Costa Rica: Cartago, Costa Rica, 2010. [Google Scholar]
- Miranda-Ruvalcaba, R.; Penieres-Carrillo, J.G.; Obaya-Valdivia, A.; Palma-Cruz, A.; Frontana-Uribe, B.; Nicolás-Vázquez, M.I.; Vargas-Rodríguez, Y.M.; Martínez, J.O.; Martín-Hernández, O.; Reyes-Sánchez, L.B.; et al. Química Verde Experimental 2011.
- Miranda, R.; Noguez, O.; Velasco, B.; Arroyo, G.; Penieres, G.; Martínez, O.; Delgado, F. Irradiación infrarroja una alternativa para la activación de reacciones y su contribución a la química verde. Educ. Quím 2009, 20, 421–425, and references therein. [Google Scholar]
- Noguez, M.O.; García, A.; Ibarra, C.; Cabrera, A.; Aceves, J.M.; Nicolás, M.I.; Miranda, R. Green synthesis of bis-biginelli esters, with vasodilatory effects, their mass spectrometric and physical studies. Trends Org. Chem 2009, 13, 75–82. [Google Scholar]
- Noguez, M.O.; Marcelino, V.; Rodríguez, H.; Martín, O.; Martínez, J.O.; Arroyo, G.A.; Pérez, F.J.; Suárez, M.; Miranda, R. Infrared assisted production of 3,4-Dihydro-2(1H)-pyridones in solvent-free conditions. Int. J. Mol. Sci 2011, 12, 2641–2649. [Google Scholar]
- Shortwave Electric Infrared the Facts. Copyright© ITW BGK Finishing Systems 05/03. Available online: http://www.bgk.com accessed on 11 August 2011.
- 3c 1H NMR (300 MHz, DMSO-d6, δ): 8.56 (s, 1H, HC=C), 7.96 (d, J = 7.9 Hz, 2H, H2,6), 7.7–755 (m, 3H, H3,4,5); 13C NMR (75 MHz, DMSO-d6, δ): 162.0 (HC=C), 134.8 (C4), 131.9 (C1), 131 (C3,5), 130 (C2,6), 114.4 (d, 3JH 13 CN = 8.6 Hz, CNtrans), 113.5 (d, 3JH 13 CN = 14.4 Hz, CNcis), 82 (HC=C). 3e 1H NMR (300 MHz, DMSO-d6, δ): 11.40 (s, 1H, NH), 11.22 (S, 1H, NH), 8.35 (S, 1H, HC=C), 8.13 (d, J2,3 = J6,5 7.1 Hz, 2H, H2,6), 7.44 (m, 3H, H3,4,5); 13C NMR (75 MHz, DMSO-d6, δ): 163.4 (C=Otrans), 162.0 (C=Ocis), 155.2 (HC=C), 150.6 (HNCONH), 133.6 (C2,6), 133.1 (C4) 132.5 (C1), 128.5 (C3,5), 119.3 (HC=C). 3g 1H NMR (300 MHz, CDCl3, δ): 7.22 (Ar), 9.93 (HN), 2.94 (3H), 4.28 (4H), 2.33 (7H), 3.54 (OCH3); 13C NMR (75 MHz, CDCl3, δ): 170. 4 (C2), 37.9 (C3), 31.3 (C4), 105. 1 (C5), 149.3 (C6), 18.9 (C7), 167.6 (C8), 51.8 (C9), 143.2 (C10), 127.2 (C11), 129.3 (C12), 126.0 (C13), 129.3 (C14), 127.2 (C15).
| Substrate | Conditions | Time | ||
|---|---|---|---|---|
| Literature [8] | This work | |||
| Reagent | Product | Middle infrared/Temp | Near infrared/Temp | |
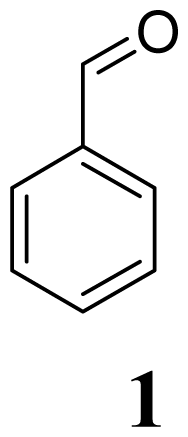 |  | 15 min/80 °C | 7 min/170 °C | |
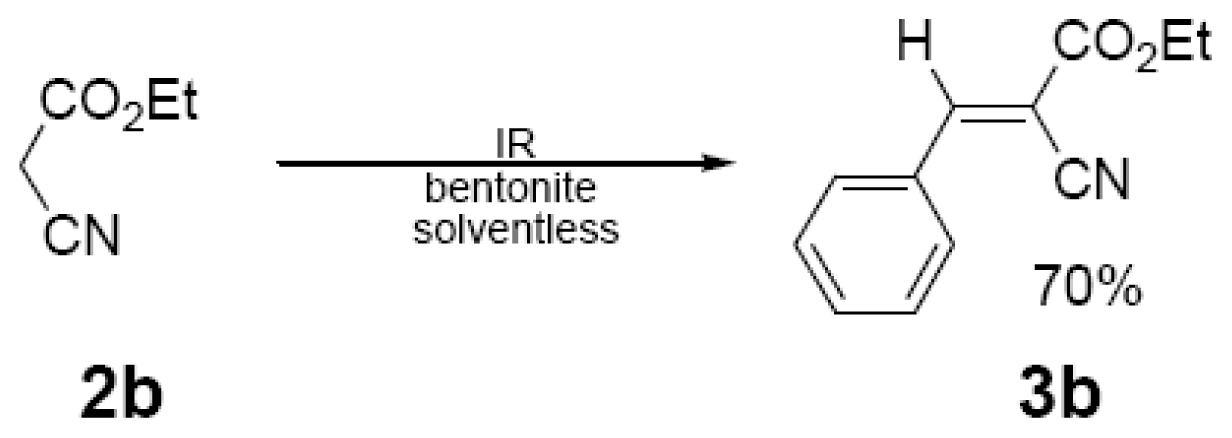 | 15 min/80 °C | 7 min/170 °C | ||
 | 15 min/80 °C | 7 min/170 °C | ||
 | 15 min/80 °C | 7 min/170 °C | ||
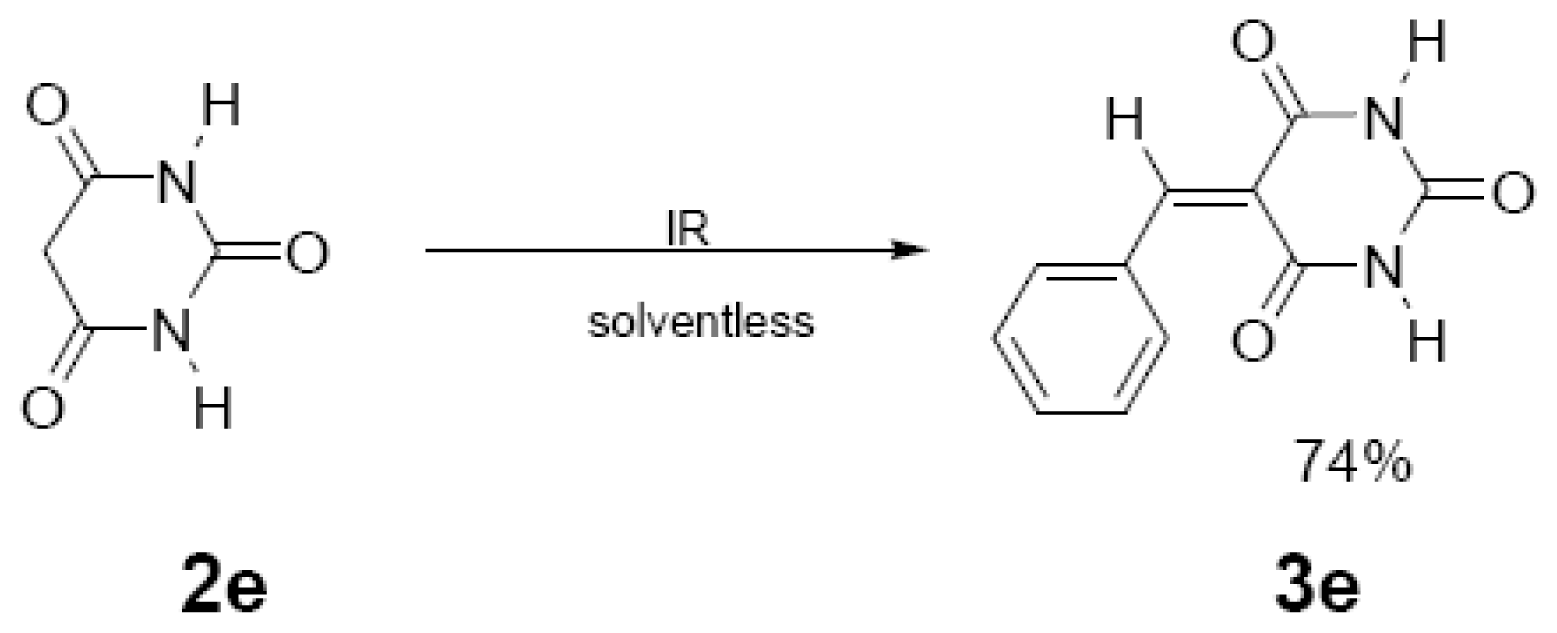 | 45 min/80 °C | 20 min/170 °C | ||
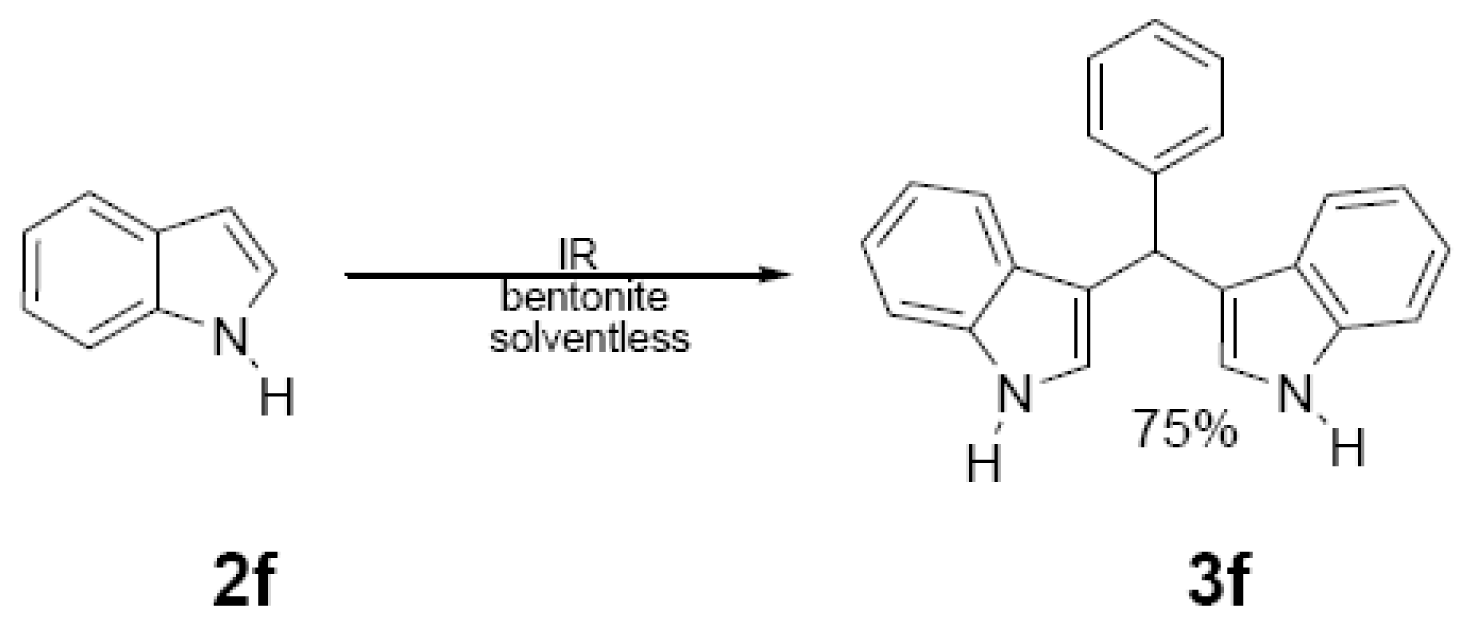 | 15 min/80 °C | 7 min/170 °C | ||
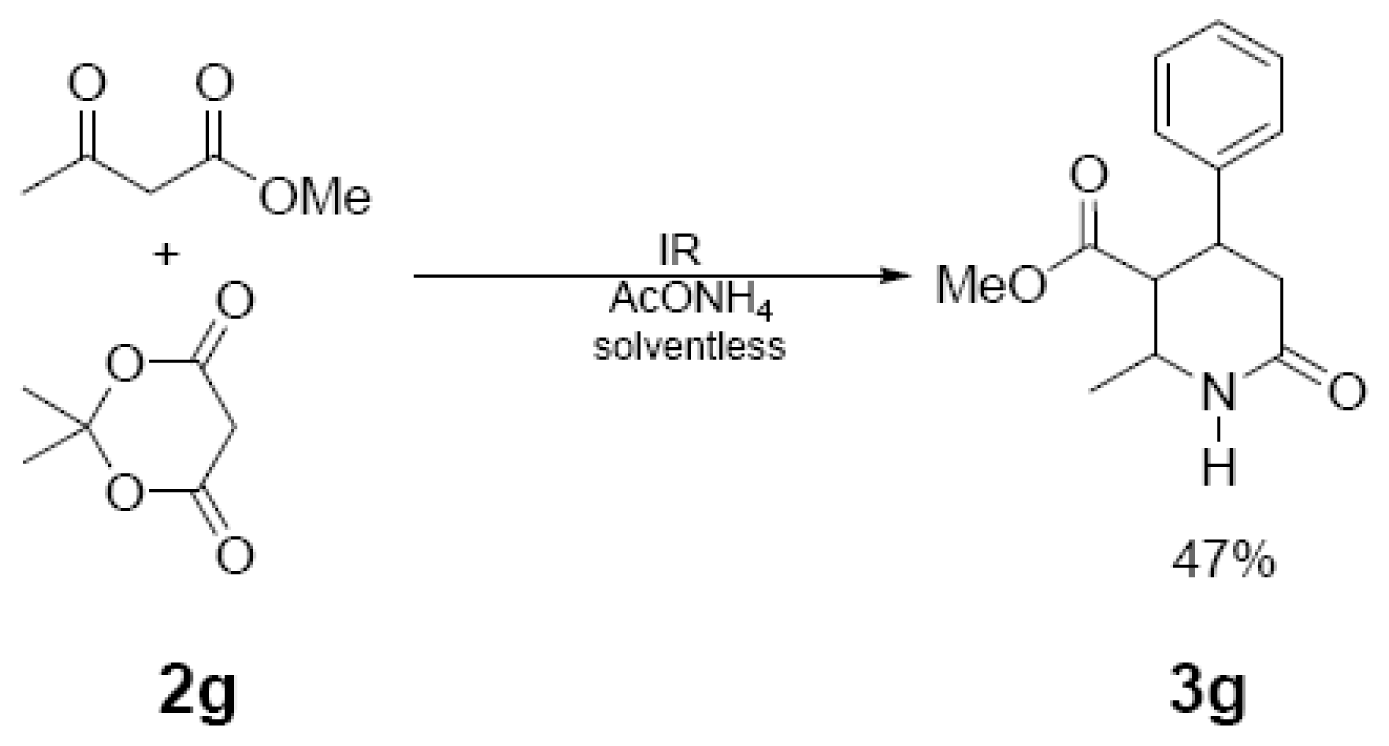 | 3 h/80 °C | 50 min/170 °C | ||
 | 3 h/80 °C | 50 min/170 °C | ||
 | 3 h/80 °C | 1.5 h/170 °C | ||
 | 40 min/80 °C | 20 min/170 °C | ||
| Compound | m/z (% relative abundance) M+• | Others Peaks m/z (% relative abundance) |
|---|---|---|
| 3a | 248 (23) | 176 (100), 172 (13), 77 (20) |
| 3b | 201 (39) | 129 (99), 125 (22), 77 (18) |
| 3c | 173 (95) | 172 (100), 156 (8),146 (5), 128 (23), 101 (8) |
| 3d | 154 (100) | 127 (75), 100 (11) |
| 3e | 216 (63) | 215 (100), 172 (63), 145 (7), 127 (17), 102 (26) |
| 3f | 322 (100) | 321 (34), 245 (52) |
| 3g | 229 (88) | 214 (35), 186 (66), 131 (100), 91 (42), 77 (39) |
| 3h | 329 (44) | 300 (21), 284 (25), 256 (30), 252 (100) |
| 3i | 260 (27) | 231 (66), 187 (39), 183 (100) |
| 3j | 181 (63) | 105 (100), 77 (34) |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Córdova, M.O.N.; Ramírez, C.I.F.; Bejarano, B.V.; Razo, G.A.A.; Flores, F.J.P.; Tellez, V.C.; Ruvalcaba, R.M. Comparative Study Using Different Infrared Zones of the Solventless Activation of Organic Reactions. Int. J. Mol. Sci. 2011, 12, 8575-8580. https://doi.org/10.3390/ijms12128575
Córdova MON, Ramírez CIF, Bejarano BV, Razo GAA, Flores FJP, Tellez VC, Ruvalcaba RM. Comparative Study Using Different Infrared Zones of the Solventless Activation of Organic Reactions. International Journal of Molecular Sciences. 2011; 12(12):8575-8580. https://doi.org/10.3390/ijms12128575
Chicago/Turabian StyleCórdova, María Olivia Noguez, Carlos I. Flores Ramírez, Benjamín Velasco Bejarano, Gabriel A. Arroyo Razo, Francisco J. Pérez Flores, Vladimir Carranza Tellez, and René Miranda Ruvalcaba. 2011. "Comparative Study Using Different Infrared Zones of the Solventless Activation of Organic Reactions" International Journal of Molecular Sciences 12, no. 12: 8575-8580. https://doi.org/10.3390/ijms12128575




