Fatty Acid Composition and Antioxidant Activity of Tea (Camellia sinensis L.) Seed Oil Extracted by Optimized Supercritical Carbon Dioxide
Abstract
:1. Introduction
2. Results and Discussion
2.1. Model Fitting
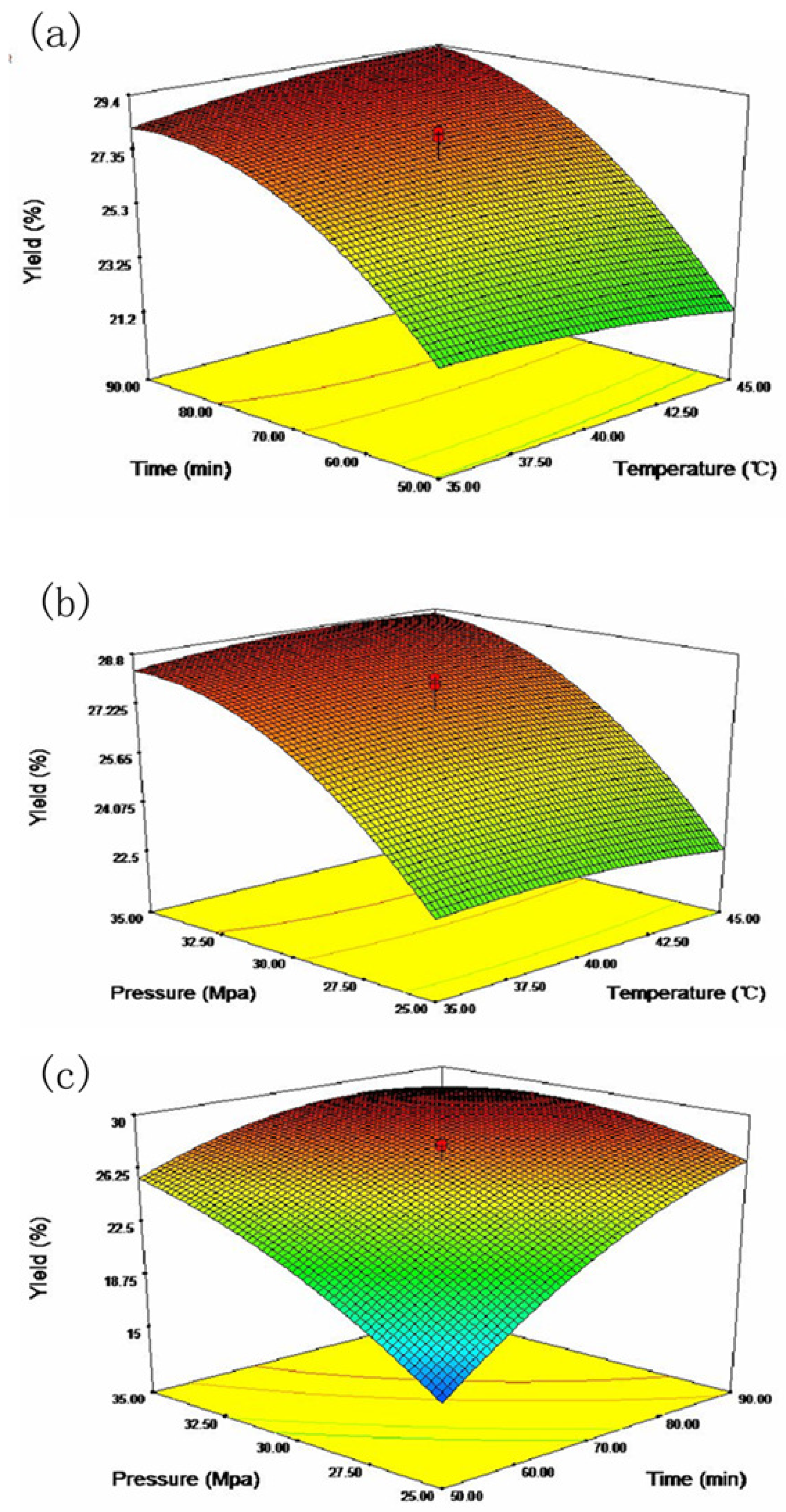
2.2. Response Surface Analysis
2.3. Comparison of SC-CO2 Extraction with SE
2.3.1. Oil Yields
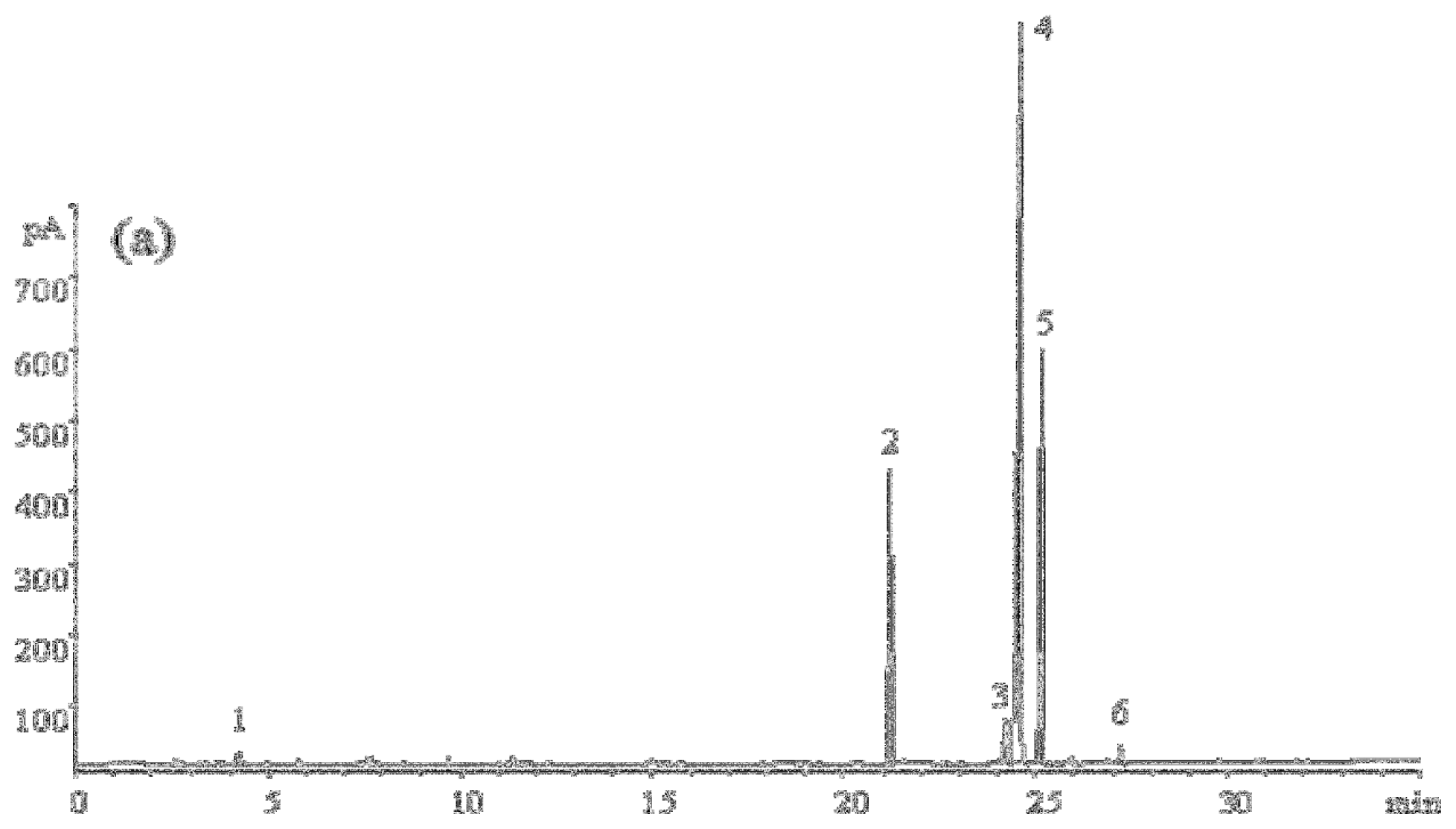
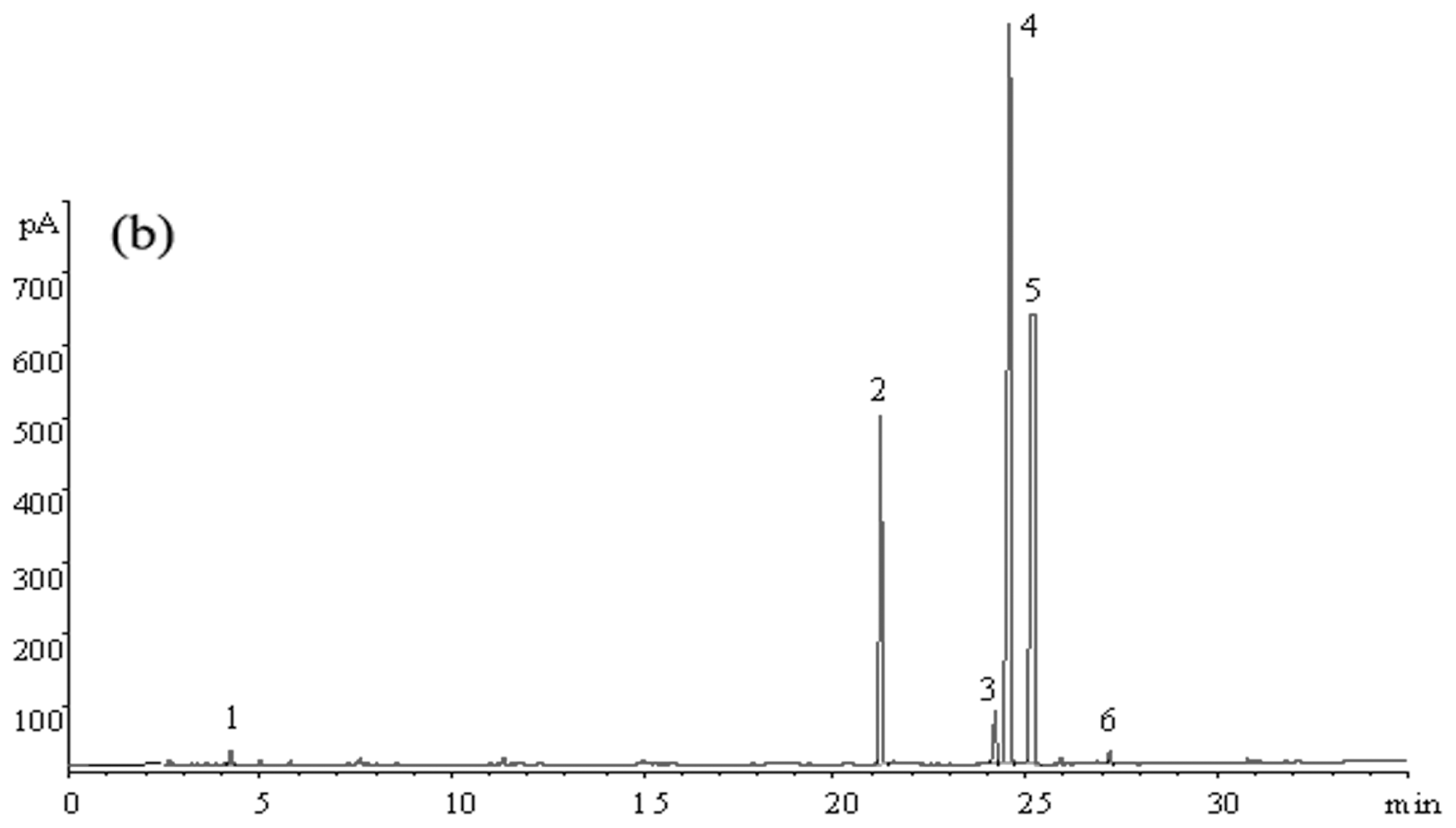
2.3.2. Fatty Acid Composition of Tea Seed Oils
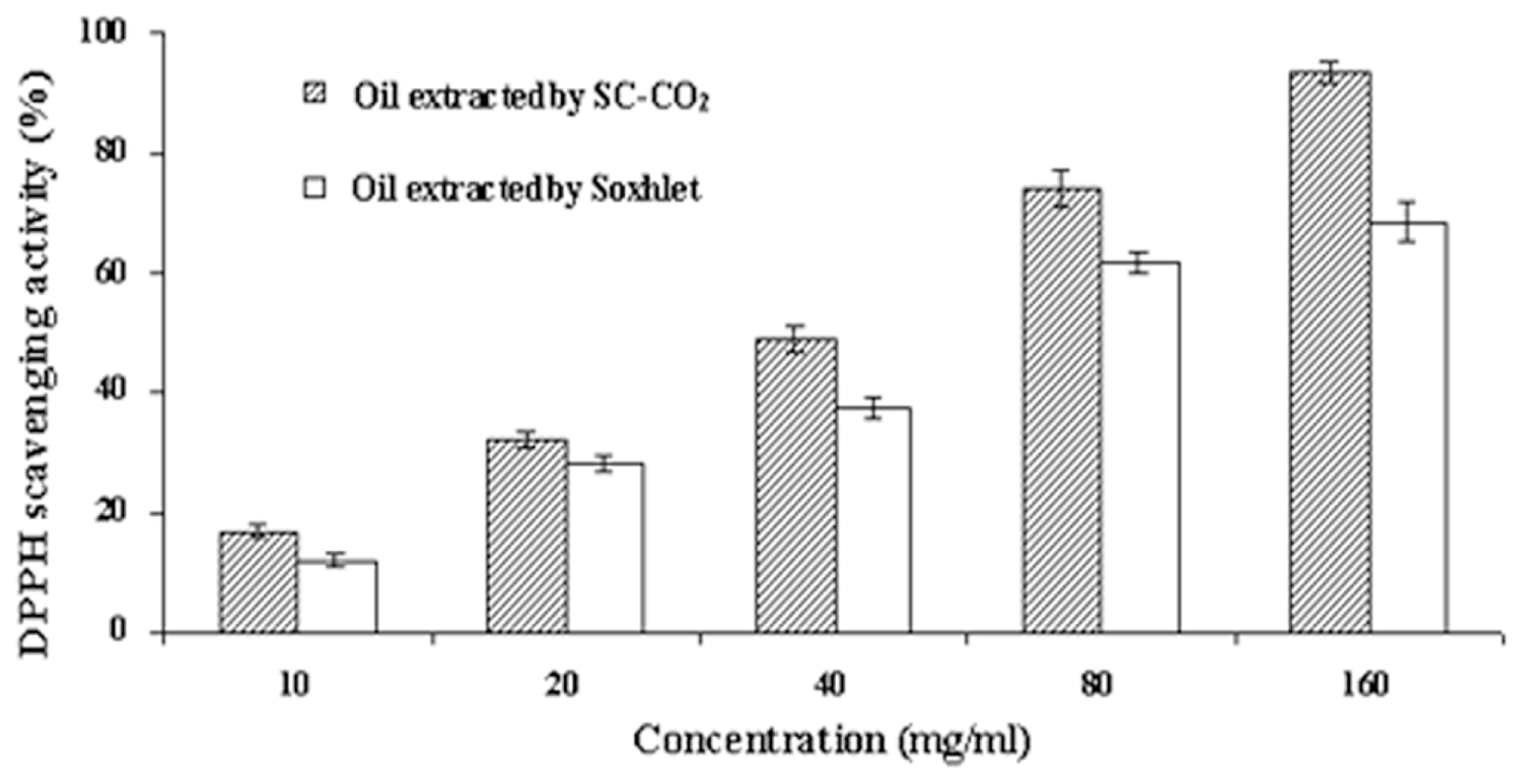
2.3.3. Antioxidant Activity of Tea Seed Oils
3. Experimental Section
3.1. Materials and Reagents
3.2. SC-CO2 Extraction
3.3. Soxhlet Extraction (SE)
3.4. Experimental Design and Statistical Analysis for Response Surface Methodology
3.5. Fatty Acid Composition
3.6. Antioxidant Activity
3.7. Statistical Analysis
4. Conclusions
- Conflict of InterestThe authors declare no conflict of interest.
References
- Khan, N.; Mukhtar, H. Tea polyphenols for health promotion. Life Sci 2007, 81, 519–533. [Google Scholar]
- Tian, H.Z.; Qiu, A.Y.; Shi, X.H. Extraction of tea seed polysaccharide. China Oil (Chinese) 2004, 29, 27–29. [Google Scholar]
- Ravichandran, R.; Dhandapani, M. Composition characteristics and potential uses of south Indian tea seeds. J. Food Sci. Technol 1992, 29, 394–394. [Google Scholar]
- Sahari, M.A.; Ataii, D.; Hamedi, M. Characteristics of tea seed oil in comparison with sunflower and olive oils and its effect as a natural antioxidant. Am. Oil Chem. Soc 2004, 81, 585–588. [Google Scholar]
- Sprecher, H. Biochemistry of essential fatty acids. Prog. Lipid Res 1981, 20, 13–22. [Google Scholar]
- Bourre, J.M. Effects of nutrients (in food) on the structure and function of the nervous system: Update on dietary requirements for brain. Part 1: Micronutrients. J. Nutr. Health Aging 2006, 10, 386–399. [Google Scholar]
- Kim, N.H.; Choi, S.K.; Kim, S.J.; Moon, P.D.; Lim, H.S.; Choi, I.Y.; Na, H.J.; An, H.J.; Myung, N.Y.; Jeong, H.J. Green tea seed oil reduces weight gain in C57BL/6J mice and influences adipocyte differentiation by suppressing peroxisome proliferator-activated receptor-γ. Pflugers Arch.: Eur. J. Physiol 2008, 457, 293–302. [Google Scholar]
- Rajaei, A.; Barzegar, M.; Yamini, Y. Supercritical fluid extraction of tea seed oil and its comparison with solvent extraction. Eur. Food Res. Technol 2005, 220, 401–405. [Google Scholar]
- Machmudah, S.; Kawahito, Y.; Sasaki, M.; Goto, M. Supercritical CO2 extraction of rosehip seed oil: Fatty acids composition and process optimization. J. Supercrit. Fluids 2007, 41, 421–428. [Google Scholar]
- Jiao, S.; Li, D.; Huang, Z.; Zhang, Z.; Bhandari, B.; Chen, X.D.; Mao, Z. Optimization of supercritical carbon dioxide extraction of flaxseed oil using response surface methodology. Int. J. Food Eng 2008, 4. [Google Scholar] [CrossRef]
- Boutin, O.; Badens, E. Extraction from oleaginous seeds using supercritical CO2: Experimental design and products quality. J. Food Eng 2009, 92, 396–402. [Google Scholar]
- Mitra, P.; Ramaswamy, H.S.; Chang, K.S. Pumpkin (Cucurbita maxima) seed oil extraction using supercritical carbon dioxide and physicochemical properties of the oil. J. Food Eng 2009, 95, 208–213. [Google Scholar]
- Ixtaina, V.Y.; Vega, A.; Nolasco, S.M.; Tomás, M.C.; Gimeno, M.; Bárzana, E.; Tecante, A. Supercritical carbon dioxide extraction of oil from Mexican chia seed (Salvia hispanica L.): Characterization and process optimization. J. Supercrit. Fluids 2010, 55, 192–199. [Google Scholar]
- Ballard, T.S.; Mallikarjunan, P.; Zhou, K.; O’Keefe, S.F. Optimizing the extraction of phenolic antioxidants from peanut skins using response surface methodology. J. Agric. Food Chem 2009, 57, 3064–3072. [Google Scholar]
- Bhattacharjee, P.; Singhal, R.S.; Tiwari, S.R. Supercritical carbon dioxide extraction of cottonseed oil. J. Food Eng 2007, 79, 892–898. [Google Scholar]
- Liu, S.; Yang, F.; Zhang, C.; Ji, H.; Hong, P.; Deng, C. Optimization of process parameters for supercritical carbon dioxide extraction of Passiflora seed oil by response surface methodology. J. Supercrit. Fluids 2009, 48, 9–14. [Google Scholar]
- Shao, P.; Sun, P.; Ying, Y. Response surface optimization of wheat germ oil yield by supercritical carbon dioxide extraction. Food Bioprod. Process 2008, 86, 227–231. [Google Scholar]
- Wei, Z.J.; Liao, A.M.; Zhang, H.X.; Liu, J.; Jiang, S.T. Optimization of supercritical carbon dioxide extraction of silkworm pupal oil applying the response surface methodology. Bioresour. Technol 2009, 100, 4214–4219. [Google Scholar]
- Celebi, N.; Yildiz, N.; Demir, A.S.; Calimli, A. Optimization of benzoin synthesis in supercritical carbon dioxide by response surface methodology (RSM). J. Supercrit. Fluids 2008, 47, 227–232. [Google Scholar]
- Wang, L.; Yang, B.; Du, X.; Yi, C. Optimisation of supercritical fluid extraction of flavonoids from Pueraria lobata. Food Chem 2008, 108, 737–741. [Google Scholar]
- Liu, J.; Lin, S.; Wang, Z.; Wang, C.; Wang, E.; Zhang, Y. Supercritical fluid extraction of flavonoids from Maydis stigma and its nitrite-scavenging ability. Food Bioprod. Process 2011, 89, 333–339. [Google Scholar]
- Thana, P.; Machmudah, S.; Goto, M.; Sasaki, M.; Pavasant, P.; Shotipruk, A. Response surface methodology to supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis. Bioresource Technol 2008, 99, 3110–3115. [Google Scholar]
- Tokue, C.; Kataoka, E.; Tanimura, W. Characterization of lipids in tea (Camellia sinensis) seeds cultivated in Taiwan and Japan. J. Jpn. Soc. Nutr. Food Sci. (Jpn.) 1989, 42, 71–77. [Google Scholar]
- Ravichandran, R. Fat stability and amino acids in south Indian tea seeds. Int. J. Food Sci. Technol 1993, 28, 639–646. [Google Scholar]
- Kris-Etherton, P.M. Monounsaturated fatty acids and risk of cardiovascular disease. Circulation 1999, 100, 1253–1258. [Google Scholar]
- Rajaei, A.; Barzegar, M.; Sahar, I.M. Comparison of antioxidative effect of tea and sesame seed oils extracted by different methods. J. Agric. Sci 2008, 10, 345–350. [Google Scholar]
- Li, B.; Xu, Y.; Jin, Y.X.; Wu, Y.Y.; Tu, Y.Y. Response surface optimization of supercritical fluid extraction of kaempferol glycosides from tea seed cake. Ind. Crop. Prod 2010, 32, 123–128. [Google Scholar]
- Wenli, Y.; Yaping, Z.; Jingjing, C.; Bo, S. Comparison of two kinds of pumpkin seed oils obtained by supercritical CO2 extraction. Eur. J. Lipid Sci. Technol 2004, 106, 355–358. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol 1995, 28, 25–30. [Google Scholar]
| Runs | Coded variables | Oil yield (%) | |||
|---|---|---|---|---|---|
| X1 (°C) | X2 (min) | X3 (MPa) | Exp. * | Pred. ** | |
| 1 | −1 | −1 | −1 | 16.9 | 16.4 |
| 2 | 1 | −1 | −1 | 14.6 | 14.8 |
| 3 | −1 | 1 | −1 | 26.4 | 26.2 |
| 4 | 1 | 1 | −1 | 27.1 | 27.0 |
| 5 | −1 | −1 | 1 | 26.1 | 25.8 |
| 6 | 1 | −1 | 1 | 25.2 | 25.0 |
| 7 | −1 | 1 | 1 | 28.0 | 27.4 |
| 8 | 1 | 1 | 1 | 28.8 | 29.0 |
| 9 | −1.68 | 0 | 0 | 25.8 | 26.6 |
| 10 | 1.68 | 0 | 0 | 26.7 | 26.5 |
| 11 | 0 | −1.68 | 0 | 16.4 | 16.7 |
| 12 | 0 | 1.68 | 0 | 28.1 | 28.3 |
| 13 | 0 | 0 | −1.68 | 18.3 | 18.5 |
| 14 | 0 | 0 | 1.68 | 27.8 | 28.1 |
| 15 | 0 | 0 | 0 | 26.6 | 27.2 |
| 16 | 0 | 0 | 0 | 26.5 | 27.2 |
| 17 | 0 | 0 | 0 | 27.9 | 27.2 |
| 18 | 0 | 0 | 0 | 26.6 | 27.2 |
| 19 | 0 | 0 | 0 | 27.8 | 27.2 |
| 20 | 0 | 0 | 0 | 28.1 | 27.2 |
| Source | Sum of squares | df | Mean square | F-value | p-value |
|---|---|---|---|---|---|
| Model | 373.26 | 9 | 41.47 | 89.63 | <0.0001 |
| X1 | 2.54 × 10−3 | 1 | 2.54 × 10−3 | 5.50E-03 | 0.9424 |
| X2 | 162.97 | 1 | 162.97 | 352.19 | <0.0001 |
| X3 | 111.81 | 1 | 111.81 | 241.64 | <0.0001 |
| X1X2 | 2.76 | 1 | 2.76 | 5.97 | 0.0347 |
| X1X3 | 0.28 | 1 | 0.28 | 0.61 | 0.4537 |
| X2X3 | 34.03 | 1 | 34.03 | 73.54 | <0.0001 |
| X12 | 0.90 | 1 | 0.90 | 1.94 | 0.1942 |
| X22 | 39.88 | 1 | 39.88 | 86.19 | <0.0001 |
| X32 | 27.47 | 1 | 27.47 | 59.37 | <0.0001 |
| Residual | 4.63 | 10 | 0.46 | ||
| Lack of fit | 1.77 | 5 | 0.35 | 0.62 | 0.6932 |
| Pure error | 2.86 | 5 | 0.57 | ||
| Corrected total | 377.89 | 19 |
| Methods | C7:0 * | C16:0 * | C18:0 * | C18:1 * | C18:2 * | C20:1 * | SFA * | MUFA * | PUFA * | Others |
|---|---|---|---|---|---|---|---|---|---|---|
| SC-CO2 | 0.7 | 15.3 | 3.3 | 57.5 | 22.3 | 0.9 | 18.6 | 58.4 | 22.3 | 0.7 |
| Soxhlet | 0.7 | 17.7 | 3.8 | 52.9 | 24.2 | 0.7 | 21.5 | 53.6 | 24.2 | 0.7 |
| Independent variables | Coded symbols | Levels | ||||
|---|---|---|---|---|---|---|
| −1.68 | −1 | 0 | 1 | 1.68 | ||
| Extraction temperature (°C) | X1 | 31.59 | 35 | 40 | 45 | 48.41 |
| Extraction time (min) | X2 | 36.36 | 50 | 70 | 90 | 103.64 |
| Extraction pressure (Mpa) | X3 | 21.59 | 25 | 30 | 35 | 38.41 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Y.; Sun, D.; Chen, H.; Qian, L.; Xu, P. Fatty Acid Composition and Antioxidant Activity of Tea (Camellia sinensis L.) Seed Oil Extracted by Optimized Supercritical Carbon Dioxide. Int. J. Mol. Sci. 2011, 12, 7708-7719. https://doi.org/10.3390/ijms12117708
Wang Y, Sun D, Chen H, Qian L, Xu P. Fatty Acid Composition and Antioxidant Activity of Tea (Camellia sinensis L.) Seed Oil Extracted by Optimized Supercritical Carbon Dioxide. International Journal of Molecular Sciences. 2011; 12(11):7708-7719. https://doi.org/10.3390/ijms12117708
Chicago/Turabian StyleWang, Yuefei, Da Sun, Hao Chen, Lisheng Qian, and Ping Xu. 2011. "Fatty Acid Composition and Antioxidant Activity of Tea (Camellia sinensis L.) Seed Oil Extracted by Optimized Supercritical Carbon Dioxide" International Journal of Molecular Sciences 12, no. 11: 7708-7719. https://doi.org/10.3390/ijms12117708
APA StyleWang, Y., Sun, D., Chen, H., Qian, L., & Xu, P. (2011). Fatty Acid Composition and Antioxidant Activity of Tea (Camellia sinensis L.) Seed Oil Extracted by Optimized Supercritical Carbon Dioxide. International Journal of Molecular Sciences, 12(11), 7708-7719. https://doi.org/10.3390/ijms12117708




