Antinociceptive and Anti-Inflammatory Activities of the Ethanol Extract of Annona muricata L. Leaves in Animal Models
Abstract
:1. Introduction
2. Results and Discussion
2.1. Acute Toxicity
2.2. Writhing Response Induced by Acetic Acid in Mice
2.3. Effects on Formalin-Induced Nociception in Mice
2.4. Effects on Hot-Plate Latency Assay in Mice
2.5. Effects on Carrageenan-induced Edema in Rats
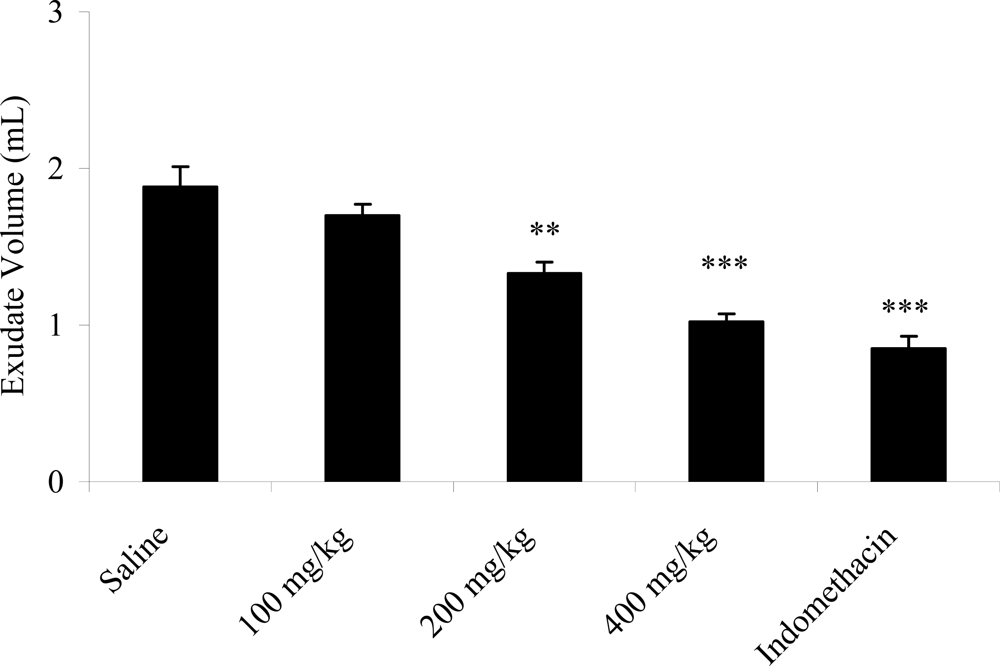
2.6. Effects on Carrageenan-Induced Pleurisy in Rats
3. Experimental Section
3.1. Plant Material and Extraction
3.2. Chemicals
3.3. Animals
3.4. Acute Toxicity
3.5. Acetic Acid-Induced Writhing Response in Mice
3.6. Formalin-Induced Nociception in Mice
3.7. Hot-Plate Latency Assay in Mice
3.8. Carrageenan-Induced Edema in Rats
3.9. Carrageenan-Induced Pleurisy in Rats
3.10. Calculations and Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Di Stasi, LC; Hiruma-Lima, CA. Plantas Medicinais na Amazônia e na Mata Atlântica, 2nd ed; Editora UNESP: São Paulo, Brazil, 2002; pp. 87–112. [Google Scholar]
- Sousa, MP; Matos, MEO; Matos, FJA; Machados, MIL; Craveiro, AA. Constituintes Químicos Ativos e Propriedades Biológicas de Plantas Medicinais Brasileiras, 2nd ed; Editora UFC: Fortaleza, Brazil, 2004; pp. 281–283. [Google Scholar]
- Lorenzi, H; Matos, FJA. Plantas Medicinais No Brasil: Nativas e Exóticas, 2nd ed; Instituto Plantarum: Nova Odessa, Brazil, 2008; pp. 62–63. [Google Scholar]
- Leboeuf, M; Legueut, C; Cavé, A; Desconclois, JF; Forgacs, P; Jacquemin, H. Alkaloids of Annonaceae. XXIX. Alkaloids of Annona muricata. Planta Med 1981, 42, 37–44. [Google Scholar]
- Leboeuf, M; Cavé, A; Bhaumik, PK; Mukherjee, B; Mukherjee, R. The phytochemistry of the annonaceae. Phytochemistry 1982, 21, 2783–2813. [Google Scholar]
- Pélissler, Y; Marion, C; Kone, D; Lamaty, G; Menut, C; Besslere, JM. Volatile components of Annona muricata L. J. Essent. Oil Res 1994, 6, 411–414. [Google Scholar]
- Kossouoh, C; Moudachirou, M; Adjakidje, V; Chalchat, JC; Figuérédo, G. Essential oil chemical composition of Annona muricata L. leaves from Benin. J. Essent. Oil Res 2007, 19, 307–309. [Google Scholar]
- Rupprecht, JK; Hui, YH; McLaughlin, JL. Annonaceous acetogenins: a review. J. Nat. Prod 1990, 53, 237–278. [Google Scholar]
- Cavé, A; Figadère, B; Laurens, A; Cortes, D. Acetogenins from Annonaceae. Fortschr. Chem. Org. Naturst 1997, 70, 281–288. [Google Scholar]
- Wu, FE; Gu, ZM; Zeng, L; Zhao, GX; Zhang, Y; McLaughlin, JL; Sastrodihardjo, S. Two new cytotoxic monotetrahydrofuran Annonaceous acetogenins, annomuricins A and B, from the leaves of Annona muricata. J. Nat. Prod 1995, 58, 830–836. [Google Scholar]
- Wu, FE; Zeng, L; Gu, ZM; Zhao, GX; Zhang, Y; Schwedler, JT; McLaughlin, JL; Sastrodihardjo, S. Muricatocins A and B, two new bioactive monotetrahydrofuran Annonaceous acetogenins from the leaves of Annona muricata. J. Nat. Prod 1995, 58, 902–908. [Google Scholar]
- Wu, FE; Zeng, L; Gu, ZM; Zhao, GX; Zhang, Y; Schwedler, JT; McLaughlin, JL; Sastrodihardjo, S. New bioactive monotetrahydrofuran Annonaceous acetogenins, annomuricin C and muricatocin C, from the leaves of Annona muricata. J. Nat. Prod 1995, 58, 909–915. [Google Scholar]
- Wu, FE; Zhao, GX; Zeng, L; Zhang, Y; Schwedler, JT; McLaughlin, JL; Sastrodihardjo, S. Additional bioactive acetogenins, annomutacin and (2,4-trans and cis)-10R-annonacin-A-ones, from the leaves of Annona muricata. J. Nat. Prod 1995, 58, 1430–1437. [Google Scholar]
- Zeng, L; Wu, FE; Oberlies, NH; McLaughlin, JL; Sastrodihadjo, S. Five new monotetrahydrofuran ring acetogenins from the leaves of Annona muricata. J. Nat. Prod 1996, 59, 1035–1042. [Google Scholar]
- Kim, GS; Zeng, L; Alali, F; Rogers, LL; Wu, FE; McLaughlin, JL; Sastrodihardjo, S. Two new mono-tetrahydrofuran ring acetogenins, annomuricin E and muricapentocin, from the leaves of Annona muricata. J. Nat. Prod 1998, 61, 432–436. [Google Scholar]
- Kim, GS; Zeng, L; Alali, F; Rogers, LL; Wu, FE; Sastrodihardjo, S; McLaughlin, JL. Muricoreacin and murihexocin C, mono-tetrahydrofuran acetogenins, from the leaves of Annona muricata. Phytochem 1998, 49, 565–571. [Google Scholar]
- Chang, FR; Liaw, CC; Lin, CY; Chou, CJ; Chiu, HF; Wu, YC. New adjacent Bis-tetrahydrofuran Annonaceous acetogenins from Annona muricata. Planta Med 2003, 69, 241–246. [Google Scholar]
- Luna, JS; Carvalho, JM; Lima, MR; Bieber, LW; Bento, ES; Franck, X; Sant'ana, AX. Acetogenins in Annona muricata L. (Annonaceae) leaves are potent molluscicides. Nat. Prod. Res 2006, 20, 253–257. [Google Scholar]
- Baskar, R; Rajeswari, V; Kumar, TS. In vitro antioxidant studies in leaves of Annona species. Indian J. Exp. Biol 2007, 45, 480–485. [Google Scholar]
- Santos, AF; Sant'Ana, AEG. Molluscicidal properties of some species of Annona. Phytomedicine 2001, 8, 115–120. [Google Scholar]
- Deraedt, R; Jouquey, S; Delevallée, F; Flahaut, M. Release of prostaglandins E and F in an algogenic reaction and its inhibition. Eur. J. Pharmacol 1980, 51, 17–24. [Google Scholar]
- Hunskaar, S; Hole, K. The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar]
- Shibata, M; Ohkubo, T; Takahashi, H; Inoki, R. Modified formalin test; characteristic biphasic pain response. Pain 1989, 38, 347–352. [Google Scholar]
- Yaksh, TL; Rudy, TA. Studies on direct spinal action of narcotics in production of analgesia in rat. J. Pharmacol. Exp. Ther 1977, 202, 411–428. [Google Scholar]
- Sousa, OV; Del-Vechio-Vieira, G; Amaral, MPH; Pinho, JJRG; Yamamoto, CH; Alves, MS. Efeitos antinociceptivo e antiinflamatório do extrato etanólico das folhas de Duguetia lanceolata St. Hil. (Annonaceae). Lat. Am. J. Pharm 2008, 27, 398–402. [Google Scholar]
- Sousa, OV; Del-Vechio-Vieira, G; Kaplan, MAC. Propriedades analgésica e antiinflamatória do extrato metanólico de folhas de Annona coriacea Mart. (Annonaceae). Lat. Am. J. Pharm 2007, 26, 872–877. [Google Scholar]
- Di Rosa, M; Giroud, JP; Willoughby, DA. Studies on the mediators of the acute inflammatory response induced in rats in different sites by carrageenan and turpentine. J. Pathol 1971, 104, 15–29. [Google Scholar]
- Seibert, K; Zhang, Y; Leahy, K; Hauser, S; Masferrer, J; Perkins, W; Lee, L; Isakson, P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc. Natl. Acad. Sci. USA 1994, 91, 12013–12017. [Google Scholar]
- Nantel, F; Denis, D; Gordon, R; Northey, A; Cirino, M; Metters, KM; Chan, CC. Distribution and regulation of cyclooxygenase-2 in carrageenan-induced inflammation. Br. J. Pharmacol 1999, 128, 853–859. [Google Scholar]
- Stochla, K; Maślinśki, S. Carrageenan-induced oedema in the rat paw-histamine participation. Agents Actions 1982, 12, 201–202. [Google Scholar]
- Hwang, SB; Lam, MH; Li, CL; Shen, TY. Release of platelet activation factor and its involvement in the first phase of carrageenin-induced rat foot edema. Eur. J. Pharmacol 1986, 120, 33–41. [Google Scholar]
- De Campos, RO; Alves, RV; Kyle, DJ; Chakravarty, S; Mavunkel, BJ; Calixto, JB. Antioedematogenic and antinociceptive actions of NPC 18521, a novel bradykinin B2 receptor antagonist. Eur. J. Pharmacol 1996, 316, 277–286. [Google Scholar]
- Gilligan, JP; Lovato, SJ; Erion, MD; Jeng, AY. Modulation of carrageenan-induced hind paw edema by substance P. Inflammation 1994, 18, 285–292. [Google Scholar]
- Ammendola, G; Di Rosa, M; Sorrentino, L. Leucocyte migration and lysosomal enzymes release in rat carrageenin pleurisy. Agents Actions 1975, 5, 250–255. [Google Scholar]
- Almeida, AP; Bayer, BM; Horakova, Z; Beaven, MA. Influence of indomethacin and other anti-inflammatory drugs on mobilization and production of neutrophils: studies with carrageenan-induced inflammation in rats. J. Pharmacol. Exp. Therap 1980, 214, 74–79. [Google Scholar]
- Capasso, F; Dunn, CJ; Yamamoto, S; Willoughby, DA; Giroud, JP. Further studies on carrageenan-induced pleurisy in rats. J. Pathol 1975, 116, 117–124. [Google Scholar]
- Vinegar, R; Truax, JF; Selph, JL. Some quantitative temporal characteristics of carrageenin-induced pleurisy in the rat. Proc. Soc. Exp. Biol. Med 1973, 143, 711–714. [Google Scholar]
- Oyemitan, IA; Iwalewa, EO; Akanmu, MA; Olugbade, TA. Antinociceptive and antiinflammatory effects of essential oil of Dennettia tripetala G. Baker (Annonaceae) in rodents. Afr. J. Tradit. Complement. Altern. Med 2008, 5, 355–362. [Google Scholar]
- Dietrich, L. A new approach to practical acute toxicity testing. Arch. Toxicol 1983, 54, 275–287. [Google Scholar]
- Litchfield, JT; Wilcoxon, F. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Therap 1949, 96, 99–113. [Google Scholar]
- Collier, HDJ; Dinnin, LC; Johnson, CA; Schneider, C. The abdominal response and its suppression by analgesic drugs in the mouse. Br. J. Pharmacol. Chemother 1968, 32, 295–310. [Google Scholar]
- Eddy, NB; Leimbach, D. Synthetic analgesics. II. Dithienylbutenyl and dithienylbutilamines. J. Pharmacol. Exp. Ther 1953, 107, 385–393. [Google Scholar]
- Winter, CA; Risley, EA; Nuss, GW. Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc. Soc. Exp. Biol. Med 1962, 111, 544–547. [Google Scholar]

| Group | Dose (mg/kg) | Number of writhes | Inhibition (%) |
|---|---|---|---|
| Control | Saline | 67.62 ± 2.03 | - |
| 100 | 67.50 ± 1.74 | - | |
| Ethanol Extract | 200 | 57.87 ± 1.55** | 14.42 |
| 400 | 39.62 ± 1.97*** | 41.41 | |
| Indomethacin | 10 | 18.25 ± 0.80*** | 73.01 |
| Group | Dose (mg/kg) | Duration of paw licking (s) | |||
|---|---|---|---|---|---|
| First phase | Inhibition (%) | Second phase | Inhibition (%) | ||
| Control | Saline | 86.62 ± 3.18 | - | 93.87 ± 2.73 | - |
| 100 | 85.87 ± 2.88 | - | 91.25 ± 3.07 | - | |
| Ethanol Extract | 200 | 66.12 ± 1.54*** | 23.67 | 65.62 ± 1.72*** | 30.09 |
| 400 | 47.62 ± 2.13*** | 45.02 | 46.75 ± 1.68*** | 50.20 | |
| Morphine | 1 | 16.25 ± 1.44*** | 81.24 | 19.37 ± 0.94*** | 79.36 |
| Group | Dose (mg/kg) | Time after drug administration (s) | |||
|---|---|---|---|---|---|
| 0 min | 30 min | 60 min | 90 min | ||
| Control | Saline | 5.50 ± 0.80 | 6.12 ± 0.44 | 6.50 ± 0.50 | 6.75 ± 0.79 |
| 100 | 5.37 ± 0.80 | 6.25 ± 0.62 | 7.12 ± 0.29 | 7.25 ± 0.45 | |
| Ethanol Extract | 200 | 5.50 ± 0.78 | 6.75 ± 0.45 | 8.50 ± 0.50* | 9.25 ± 0.67* |
| 400 | 5.75 ± 0.72 | 7.37 ± 0.80 | 11.87 ± 0.64*** | 13.25 ± 0.84*** | |
| Morphine | 1 | 5.75 ± 0.65 | 9.62 ± 0.82** | 13.75 ± 1.10*** | 16.87 ± 0.93*** |
| Naloxone + Morphine | 1 + 1 | 6.00 ± 0.68 | 7.87 ± 0.69 | 8.00 ± 0.46* | 7.87 ± 0.55 |
| Naloxone + Extract | 1 + 400 | 5.62 ± 0.68 | 7.25 ± 0.75 | 8.75 ± 0,45** | 10.87 ± 0.83** |
| Group | Dose (mg/kg) | Volume of hind paw (mL) | |||
|---|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | ||
| Control | Saline | 0.53 ± 0.06 | 0.72 ± 0.05 | 0.95 ± 0.06 | 0.75 ± 0.06 |
| 100 | 0.52 ± 0.09 | 0.68 ± 0.06 | 0.80 ± 0.06 | 0.63 ± 0.04 | |
| Ethanol Extract | 200 | 0.50 ± 0.10 | 0.65 ± 0.09 | 0.73 ± 0.06* | 0.53 ± 0.03** |
| 400 | 0.48 ± 0.07 | 0.60 ± 0.04 | 0.67 ± 0.04** | 0.47 ± 0.02*** | |
| Indomethacin | 10 | 0.47 ± 0.10 | 0.58 ± 0.05 | 0.62 ± 0.06** | 0.43 ± 0.02*** |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Sousa, O.V.; Vieira, G.D.-V.; De Pinho, J.d.J.R.G.; Yamamoto, C.H.; Alves, M.S. Antinociceptive and Anti-Inflammatory Activities of the Ethanol Extract of Annona muricata L. Leaves in Animal Models. Int. J. Mol. Sci. 2010, 11, 2067-2078. https://doi.org/10.3390/ijms11052067
De Sousa OV, Vieira GD-V, De Pinho JdJRG, Yamamoto CH, Alves MS. Antinociceptive and Anti-Inflammatory Activities of the Ethanol Extract of Annona muricata L. Leaves in Animal Models. International Journal of Molecular Sciences. 2010; 11(5):2067-2078. https://doi.org/10.3390/ijms11052067
Chicago/Turabian StyleDe Sousa, Orlando Vieira, Glauciemar Del-Vechio Vieira, José de Jesus R. G. De Pinho, Célia Hitomi Yamamoto, and Maria Silvana Alves. 2010. "Antinociceptive and Anti-Inflammatory Activities of the Ethanol Extract of Annona muricata L. Leaves in Animal Models" International Journal of Molecular Sciences 11, no. 5: 2067-2078. https://doi.org/10.3390/ijms11052067
APA StyleDe Sousa, O. V., Vieira, G. D.-V., De Pinho, J. d. J. R. G., Yamamoto, C. H., & Alves, M. S. (2010). Antinociceptive and Anti-Inflammatory Activities of the Ethanol Extract of Annona muricata L. Leaves in Animal Models. International Journal of Molecular Sciences, 11(5), 2067-2078. https://doi.org/10.3390/ijms11052067




