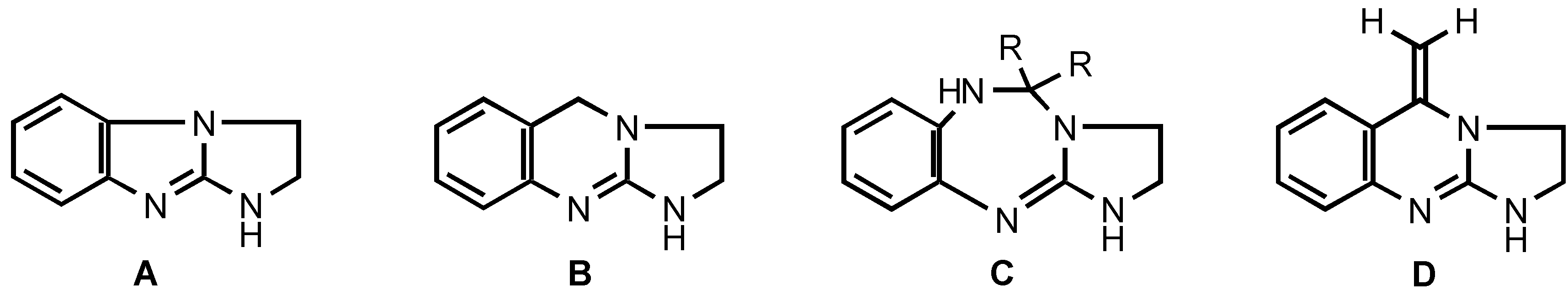
Results and Discussion
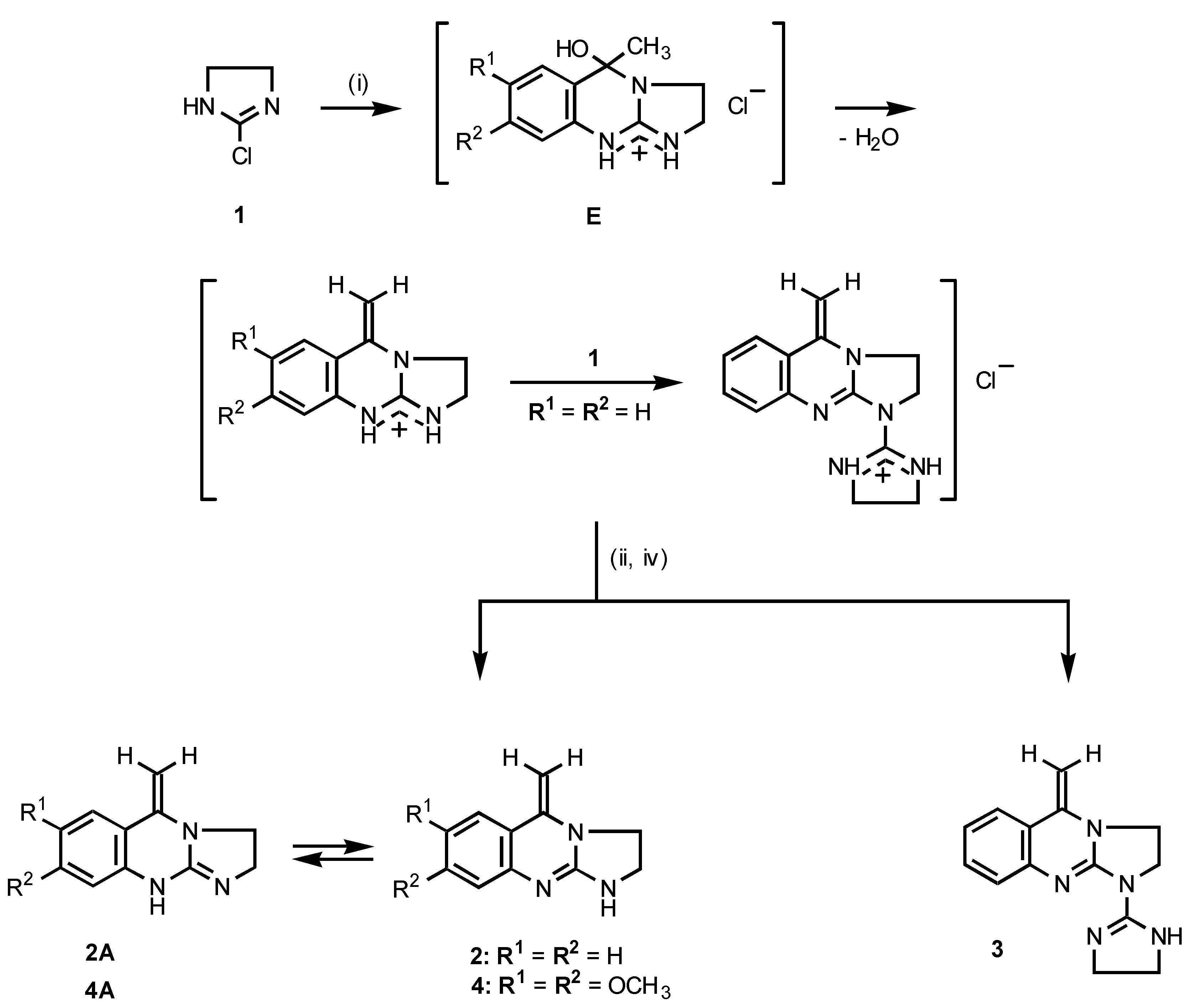
As outlined in
Scheme 1, the title 5-methy1idene-1,2,3,5-tetrahydroimidazo[2,1-
b]quinazolines
2 and
4 were obtained by reacting equimolar amounts of 2-chloro-4,5-dihydroimidazole (
1) with the appropriate 2-aminoacetophenone in dichloromethane at ambient temperature. The reaction apparently proceeds
via the intermediacy of the formed cyclic hemiaminal of type
E, which loses a water molecule with the formation of an enamine moiety. However, when compound
1 (1 equiv) was treated with 2-aminoacetophenone (0.5 equiv) the reaction led to the formation of a mixture of two products which could be separated by fractional crystallization to afford 1-(4,5-dihydroimidazol-2-yl)-5-methy-lidene-1,2,3,5-tetrahydroimidazo[2,1-
b]quinazoline (
3) and the expected compound
2 (
Scheme 1).
The structures of the compounds 2, 3 and 4 were confirmed by elemental analyses as well as IR and NMR spectroscopic data. For example, the 1H NMR spectrum of 2 showed a multiplet in the range of δ 3.59-3.68 originating from CH2-CH2 grouping of the imidazoline ring. Two distinct singlets at δ 3.71 and δ 4.51 were assigned to the nonequivalent olefinic Cβ-H protons.
Scheme 1.
(i) 2-Amino-4-R1-5-R2-acetophenone / CH2Cl2 / r.t.; (ii) 10% NaOH / H2O; (iv) fractional crystallization.
The fusion of the two ring systems was confirmed by the presence in the
13C-NMR spectrum of two signals at δ 140.17 and δ 154.11, attributable to the C-5 and C=N carbon atoms of the imidazoquinazoline moiety, respectively. On the other hand, a characteristic feature of the
1H-NMR spectrum of the 1-substituted derivative
3 is the presence of two distinct multiplets in the range of δ 3.66-3.71 and δ 4.07-4.09 integrating to six and two protons, respectively, attributable to the two CH
2-CH
2 groupings of the imidazoline rings. In
13C-NMR spectrum, two broad carbon resonances were observed at δ 45.66 and δ 50.92, attributable to C-4' and C-5' of the 2-substituted imidazoline ring, and a signal at δ 148.75, which in the HSQC (heteronuclear single quantum coherence) spectrum did not correlate to any
1H resonances. Analysis of the long-range
1H-
13C coupling pattern obtained from the HMBC (heteronuclear multiple bond coherence) experiment allowed us to assign this signal to the C-2' carbon atom of the imidazoline ring. The structure of
3 was then confirmed unambiguously by X-ray crystallographic analysis (
Figure 2).
Figure 2.
ORTEP drawing showing the asymmetric part of the unit cell of 3 and atom labelling. Hydrogen bonds are shown with dashed lines. Displacement ellipsoids are drawn at 50% probability level.
Figure 2.
ORTEP drawing showing the asymmetric part of the unit cell of 3 and atom labelling. Hydrogen bonds are shown with dashed lines. Displacement ellipsoids are drawn at 50% probability level.
The condensed tricyclic system of
3 is virtually planar with the imidazoline substituent aproximately in its plane. This conformation is stabilized by an intramolecular hydrogen bond N18-H…N1 (H18A…N1A 2.10 Å, <N18A-H18A…N1A 126º; H18A…N1A 2.08 Å, <N18A-H18A…N1A 126º). Whereas bonds C2-N1 and C14-N15 show considerable double-bond character [1.287(3) and 1.277(3) Å, respectively, for the molecule A; 1.280(3) and 1.277(3) Å, respectively, for the molecule B], the bonds C9-N1, C4-N3, N3-C2, C2-N11, N11-C14 and C14-N18 [bond lengths 1.337(3)-1.400(3) Å] are intermediate between single and double bonds. The C4-C18 bond of 1.334(3) Å is longer than the double bond in a disubstituted vinyl group (1.321 Å) [
6] indicating that it is conjugated with the guanidine fragment and the benzene ring. The nitrogen atoms N3 and N11 show sp
2 hybridization, the maximum displacement of the N atom from the plane of its substituents being 0.037 Å.
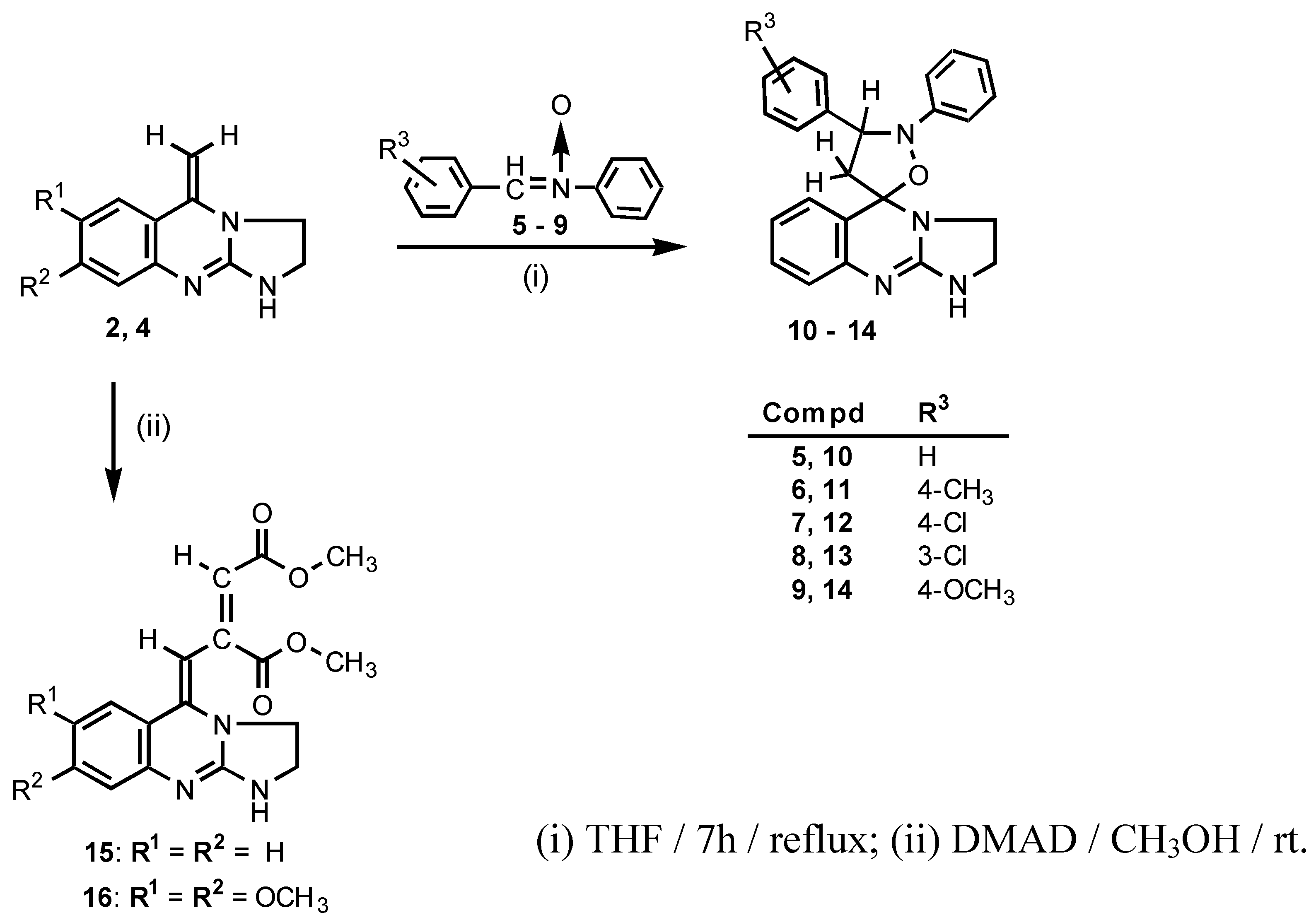
Enamines are known to act as 1,3-dipolarophiles [
8,
9] and the 1,3-dipolar cycloaddition of nitrones to enamines resulting in the formation of isoxazolidine derivatives is well established [
10,
11,
12]. Thus, the 1,2,3,5-tetrahydroimidazo[2,1-
b]quinazoline
2 was subjected to the reaction with nitrones. Upon treatment of
2 with an excess of the appropriate
N-(benzylidene)aniline
N-oxide
5-9 in anhydrous THF under reflux, the desired 1,2,3,5-tetrahydroimidazo[2,1-
b]quinazolin-5,5
′-spiro-2
′-phenyl-3′-(3
″- or 4
″-R
3-phenyl)isoxazolidines
10-14 were obtained (
Scheme 2). For the structural analysis of the compounds
10-14 a complete assignment of all protons was made using HSQC and HMBC spectra. For example, in the
1H-NMR spectrum of
10 the AMX system was observed due to CH
2-CH grouping of the isoxazoline ring formed: δ 3.03 (dd, 1H, part A of AMX system,
JA,M = 13.67 Hz,
JA,X = 10.25 Hz); δ 3.42 – 3.47 (dd, 1H, part M of AMX system); δ 4.88, dd, 1H, part X of AMX system,
JAX = 10.25 Hz,
JMX = 8.18 Hz). In the HMBC spectrum the protons C4'-H correlated to the carbon resonances at δ 71.93 (C3′) and δ 91.11 (C5-O) and the C3′-H resonance showed a long-range correlation to the carbon signal at δ 51.46 (C4′). Hence, the results of NMR experiments supported the presence of 5,5’-spiro-isoxazolidine ring.
Next, we examined the reaction of 1,2,3,5-tetrahydroimidazo[2,1-
b]quinazolines
2 and
4 with dimethyl acetylenedicarboxylate (DMAD). It is well known, that the reactions of enamines with DMAD, depending on solvent and temperature, afford either the cycloaddition [
13,
14] or Michael addition [
13,
15] products. To identify reactive sites both at the alkene and nitrogen atom incorporated into guanidine moiety, the electronic structure of
2 was studied using
ab initio 6-31G** calculations [
7]. Reactive sites will correspond to regions where either the highest-occupied molecular orbital (orbital-controlled reactions) or negative charge (electrostatically-controlled reactions) is large. As shown in
Scheme 1, the imidazoquinazoline
2 can exist in two possible tautomeric forms, the N1-H tautomer (
2) or the N10-H tautomer (
2A). Calculations of the corresponding energies indicate that 2 is more stable than
2A by 5.1 kcal/mol (
Figure 3).
Figure 3.
Calculated [
7] natural charges (plain), charges from Mulliken population analysis (italic) and atomic charges from electrostatic potential (underlined) for selected atoms of
2 and
2A.
Figure 3.
Calculated [
7] natural charges (plain), charges from Mulliken population analysis (italic) and atomic charges from electrostatic potential (underlined) for selected atoms of
2 and
2A.
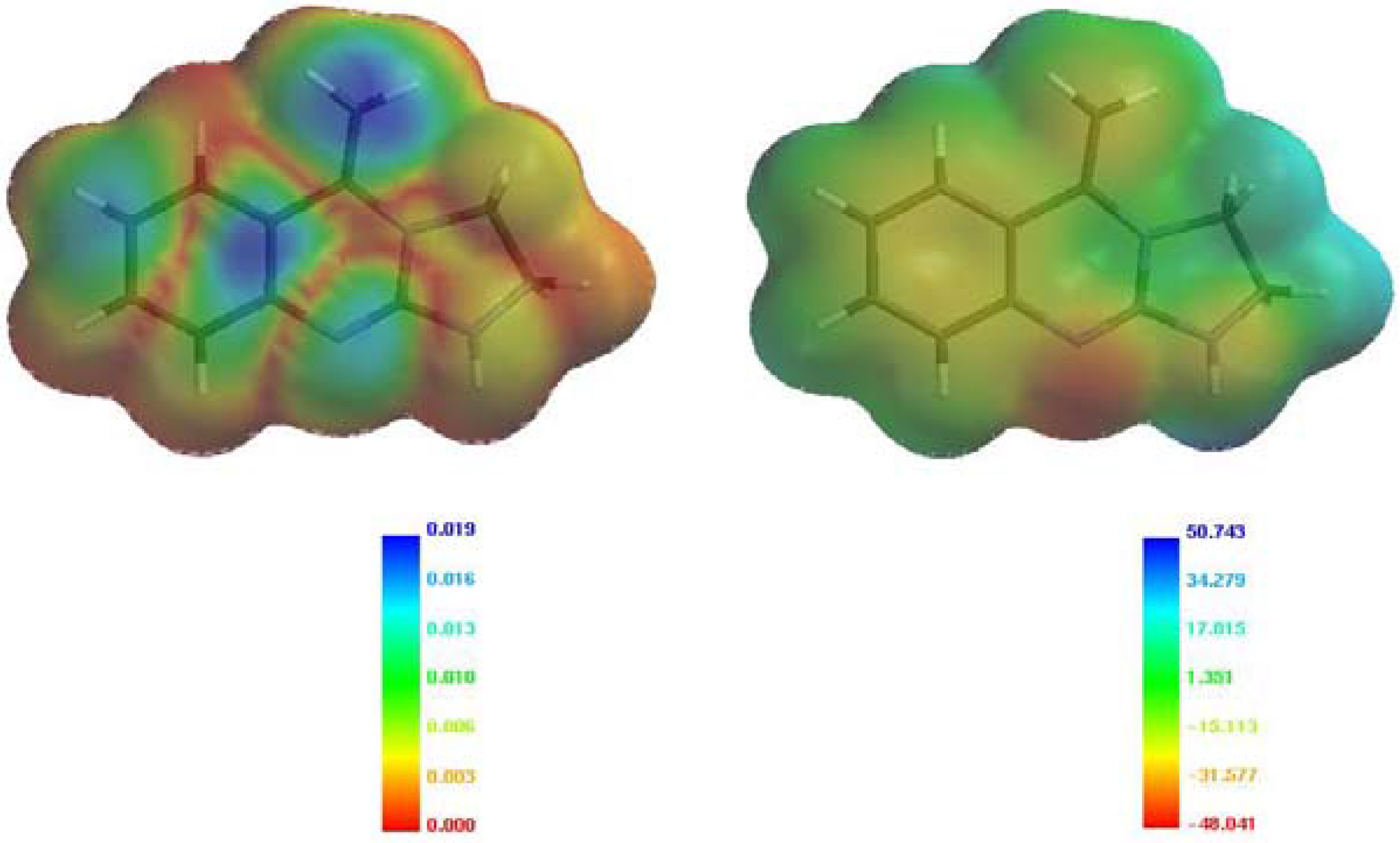
Figure 4.
Absolute values of HOMO (left) and electrostatic potential (right) mapped in color [
7] onto the surface of electron density of
2.
Figure 4.
Absolute values of HOMO (left) and electrostatic potential (right) mapped in color [
7] onto the surface of electron density of
2.
The magnitudes of the calculated charges at N1, N4 and N10 suggest that the electrostatically-controlled reactions of
2 and
2A should give rise to the formation of either the N10- or N1-substituted products. On the other hand, the HOMO (0.0190 a.u.) have the highest contribution for Cβ carbon atom of enamine moiety which should be involved in the orbital-controlled reactions with electrophiles (
Figure 4). Treatment of
2 and
4 with DMAD in methanol at room temperature gave rise to the formation of the Michael addition products dimethyl 2-(2,3-dihydroimidazo[2,1-
b]quinazolin-5(1
H)-ylidenemethyl)but-2-enedioates (
15 and
16) (
Scheme 2). Apparently, the orbital-controlled reaction took place exclusively since no
N-alkenylation was observed. In the
1H- and
13C-NMR spectra of
15 the presence of two methoxycarbonyl groups was confirmed by the resonances at δ
H 3.37 and 3.76 (OC
H3 groups) and δ
C 51.64 and 52.70 (O
CH
3 groups), as well as δ
C 167.41 and 168.77 (
C=O groups). The N1-H proton appeared as a broad singlet at δ 7.58 ppm.
Compound
2 was then subjected to reaction with electrophilic acetyl chloride and benzoyl chloride in pyridine at ambient temperature to afford the corresponding N1-substituted enaminones
17 and
18, respectively (
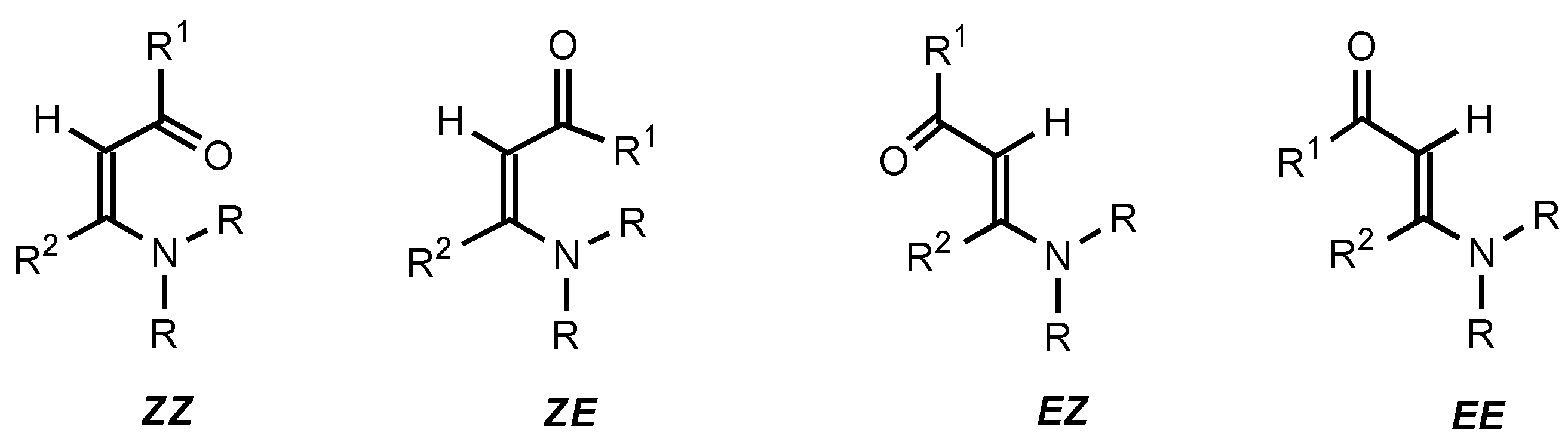
Scheme 3). It is well known that enaminones can exist in the four possible conformations depicted in
Figure 5 due to restricted rotation around the C=C double and the C−C=O single bonds [
16,
17,
18,
19,
20,
21,
22].
Analysis of the
1H-NMR spectra of compound
17 run in CDCl
3 revealed doubled signals indicating the presence of a mixture of
ZZ and
EZ isomers (
Figure 6). The olefinic Cβ-H proton appears at δ 5.22 for
17 (
EZ), and at δ 6.13 for
17 (
ZZ). Deshielding of the olefinic proton of
ZZ isomer results from its relative proximity to the benzene ring. Furthermore, the aromatic proton C6-H of the
EZ isomer (δ 9.03) is downfield of that of the
ZZ isomer (δ 7.84) by 1.19 ppm, while signals of other protons are not duplicated.
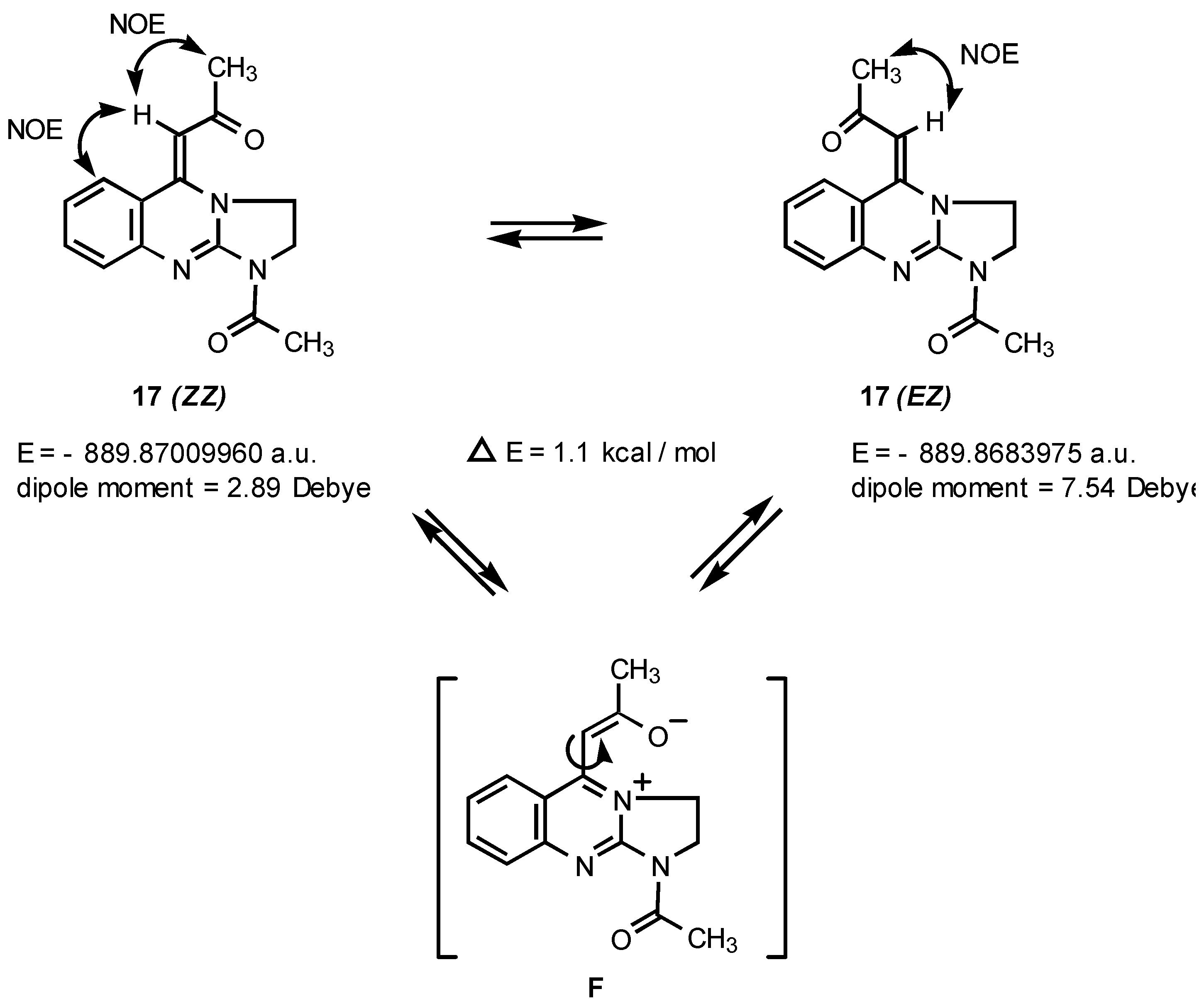
As shown in
Figure 6, the ROESY spectrum of
17 displayed intense NOE cross peaks between the resonances of Cβ-H and both CH
3 and C6-H for
ZZ isomer, whereas for isomer
EZ the corresponding NOE effect was observed between Cβ-H and CH
3 group only. Based on the relative intensities of signals observed in
1H NMR spectrum we conclude that in CDCl
3 the compound
17 exists in form of a mixture of two isomers
ZZ and
EZ in ratio 3.3 : 1.
Quantum chemical calculations [
7] indicate that the
EZ and
ZZ isomers of
17 differ in energy by only 1.1 kcal/mol. Based upon their calculated dipole moments,
EZ (μ = 7.54 D) would be predicted to predominate over
ZZ (μ = 2.89 D) in polar solvents. Indeed, an inspection of the
1H NMR spectrum of
17 run in DMSO-d
6 revealed a change of the
ZZ/
EZ isomer ratio to 1 : 1.1 (see Experimental section). Hence, these data postulate that the isomerism observed is due to a restricted rotation around the C=C double bond and the interconversion between
ZZ and
EZ isomers is possible
via a zwitterionic intermediate of type
F (
Figure 6).
It should be pointed out that 2-(1-benzoyl-2,3-dihydroimidazo[2,1-
b]quinazolin-5(1
H)-ylidene)-1-phenyl-ethen-1-one (
18) exists both in solution and solid phase as a single
ZZ isomer, which was confirmed by NMR spectroscopy and X-ray crystallographic analysis (
Figure 7).
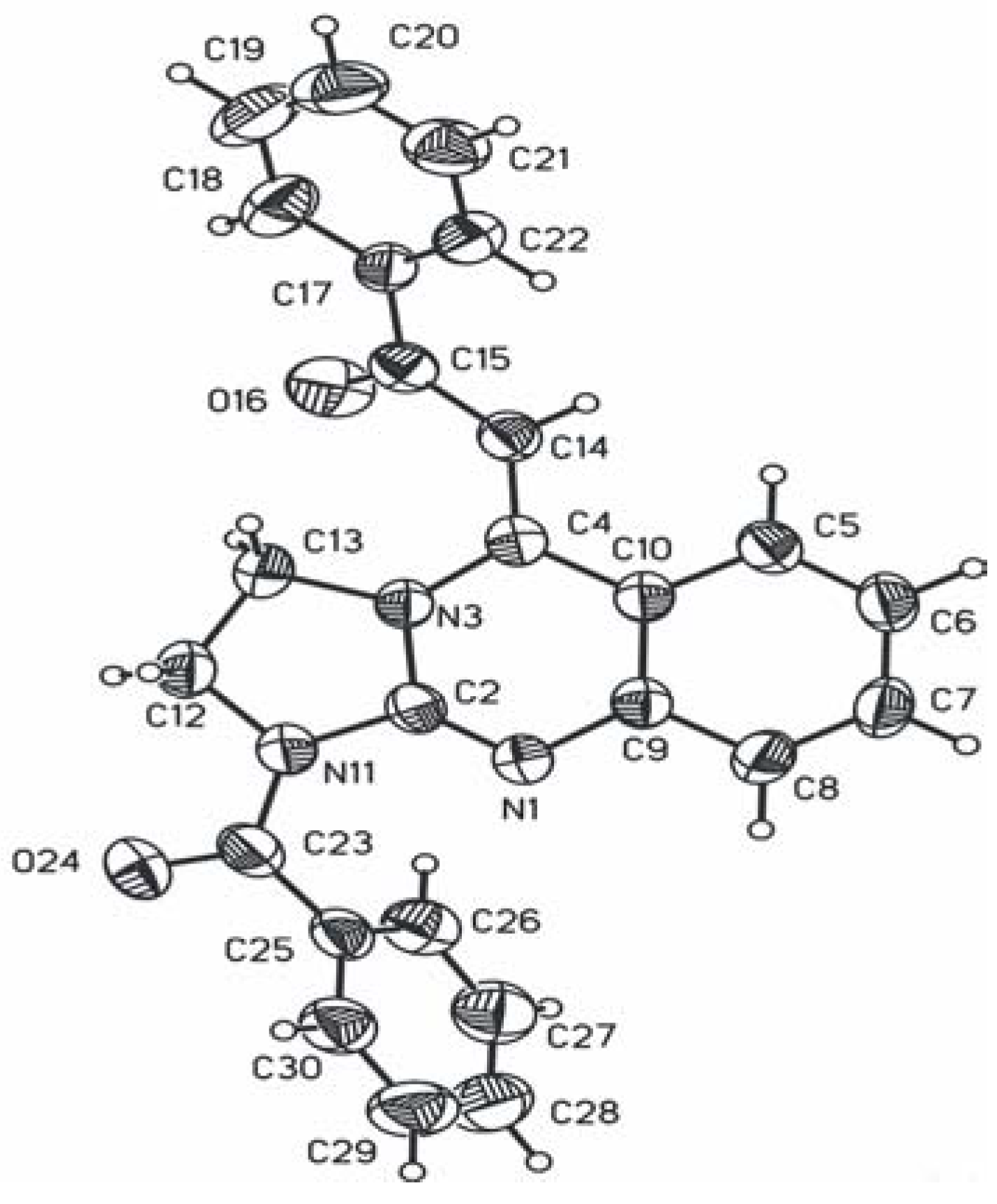
Figure 7.
ORTEP drawing of molecule 18 with atom labelling. Displacement ellipsoids were draw at 50% probability level.
Figure 7.
ORTEP drawing of molecule 18 with atom labelling. Displacement ellipsoids were draw at 50% probability level.
The tricyclic system of
18 is less planar than in
3 (cf.
Figure 2) due to the steric hindrance caused by introduction of the benzoyl substituent at C14. The release of this strain is accomplished by a twist about the formally double C4-C14 and partially double C14-C15 bonds by 21.6º and 31.3º, respectively. In effect the carbonyl O16 is at a distance of 2.24 Å from one of the imidazolidine C13 hydrogen atoms, i.e. at a distance ca. 0.4 Å shorter than the sum of appropriate van der Waals radii [
23]. The formally double bond C4-C14 which is conjugated not only with the quinazoline moiety like in
3 but also with the benzoyl group is significantly longer and equals to 1.367(3) Å. The tertiary amide group is strongly non-planar with atoms C12 and C2 deviating from the plane of C25, C23, O24 and N11 by –0.224 and 0.539 Å, respectively. Contrary to
3, the nitrogen atoms N3 and N11 show hybridization intermediate between sp
2 and sp
3with the displacement of the N atoms from the plane of their substituents equal to 0.099 and 0.146 Å, respectively.
Finally, we have also performed the reaction of
2 with methanesulfonyl chloride (MsCl). The reaction proceeded smoothly in pyridine at ambient temperature and furnished 1-mesyl derivative
19 (
Scheme 3), which, as evidenced by its
1H-NMR spectrum run in DMSO-d
6, exists as a mixture of
ZZ and
EE isomers in a 1 : 9 ratio, respectively.
Experimental
General
Melting points (mp) were determined on a Büchi 535 apparatus and are uncorrected. IR spectra (KBr pellets) were measured on a Perkin Elmer 1600 FTIR spectrophotometer.
1H-NMR and
13C‑NMR were recorded on a Varian Gemini 200 or Varian Unity 500 spectrometer and chemical shifts (δ) are expressed in ppm relative to internal tetramethylsilane. 2D NMR experiments were carried out on a Varian Unity Plus 500 spectrometer. Mass spectra were recorded on a Finnigan MAT 95 spectrometer at 70 eV. The starting 2-chloro-4,5-dihydroimidazole (
1) and
N-oxides
N-(3- or 4-R
3-benzylidene)aniline (
5-9) were prepared according to procedures described previously [
24,
25].
5-Methylidene-1,2,3,5-tetrahydroimidazo[2,1-b]quinazoline (2) and 1-(4,5-Dihydro-1H-imidazol-2-yl)-5-methylidene-1,2,3,5-tetrahydroimidazo[2,1-b]quinazoline hydrate (3).
2-Aminoacetophenone (1.66 g, 12.3 mmol) was added to a solution of 1 (2.5 g, 24 mmol) in CH2Cl2 (30 mL) and the reaction mixture was stirred at ambient temperature for 17 h. The solid that precipitated was collected by filtration, washed with CH2Cl2 (20 mL), dried and dissolved in water (25 mL). To the solution thus obtained 10% aqueous NaOH (10 mL, 25 mmol) was added and the resulting mixture was stirred for 30 min. The crude product (a mixture of 2 and 3) was filtered off, washed with water (10 mL), dried and heated in toluene (1:5) under reflux for 5 min. Then, the insoluble material was separated from the filtrate (denoted as “A” and stored for further work up), washed with hot toluene (4 mL) and dried to give 2 (0.42 g, 18%). The; mp 194-197°C; IR (KBr) cm-1: 3155, 3110, 3060, 3020, 2860, 1655, 1615, 1595, 1560, 1495, 1460, 1440, 1280; 1H-NMR (200 MHz, DMSO-d6) δ: 3.59-3.68 (m, 4H, 2CH2), 3.71 (s, 1H, CH), 4.51 (s, 1H, CH), 6.86-6.90 (m, 2H, H-7, -9), 7.13-7.24 (m, 2H, H-8 and NH), 7.58 (d, J6,7 = 7.4 Hz, 1H, H-6); 13C-NMR (200 MHz, DMSO-d6) δ: 39.75, 44.14, 75.60, 118.17, 121.09, 122.91, 123.54, 129.88, 140.17, 145.68, 154.11; Anal. Calcd. for C11H11N3: C, 71.33; H, 5.98; N, 22.68; Found: C, 71.02; H, 5.69, N, 22.82.
The filtrate “A” was concentrated under reduced pressure. The precipitate thus obtained was collected by filtration and recrystallized from toluene to give 3 (0.43 g, 13%); mp 118-121°C; IR (KBr) cm-1: 3320, 3290, 2935, 2860, 1655, 1615, 1600, 1505, 1485, 1460, 1435, 1340, 1280; 1H-NMR (500 MHz, CDCl3) δ: 3.66-3.71 (m, 6H, 3CH2), 3.72 (d, J = 2.44 Hz, 1H, CH), 4.07-4.09 (m, 2H, CH2), 4.55 (d, J = 2.44 Hz, 1H, CH), 7.02 (ddd, J7,6 = 7.81 Hz, J7,8 = 7.33 Hz, J7,9 = 1.46 Hz, 1H, H-7), 7.08 (dd, J9,8 = 8.30 Hz, J9,7 = 1.46 Hz, 1H, H-9), 7.24-7.27 (m, 1H, H-8), 7.53 (dd, J6,7 = 7.81 Hz, J6,8 = 1.47 Hz, 1H, H-6), 7.73 (br s, 1H, NH); 13C-NMR (500 MHz, CDCl3) δ: 42.36, 42.72, 45.66, 50.92, 77.90, 119.28, 123.03, 124.10, 125.30, 130.57, 140.05, 142.96, 148.75, 157.60; Anal. Calcd. for C14H15N5·H2O: C, 61.98; H, 5.57; N, 25.81; Found: C, 61.72; H, 5.32, N, 25.64.
7,8-Dimethoxy-5-methylidene-1,2,3,5-tetrahydroimidazo[2,1-b]quinazoline (4).
The reaction of 1 (2.5 g, 24 mmol) with 2-amino-4,5-dimethoxyacetophenone (2.40 g, 12.3 mmol) was carried out according to the procedure described above for 2 and 3. The crude product obtained was recrystallized from dioxane to give 4 (0.8 g, 33%); mp 185-189°C; IR (KBr) cm-1: 3170, 3115, 3075, 3010, 2880, 2835, 1655, 1625, 1615, 1510, 1490, 1450, 1420, 1270, 1225, 1210; 1H-NMR (500 MHz, DMSO-d6) δ: 3.50-3.53 (m, 2H, CH2), 3.55 (s, 1H, CH), 3.60-3.64 (m, 2H, CH2), 3.71 (s, 3H, OCH3), 3.72 (s, 3H, OCH3), 4.29 (s, 1H, CH), 6.41 (s, 1H, H-9), 6.88 (1H, br s, NH), 6.99 (1H, s, H-6); 13C-NMR (500 MHz, DMSO-d6) δ: 39.69, 44.83, 55.90, 56.60, 74.08, 106.45, 107.06, 110.57, 140.87, 141.12, 145.00, 151.59, 154.32; MS (70 eV) m/z: 245 (M+, 100%); Anal. Calcd. for C13H15N3O2: C, 63.67; H, 6.16, N, 17.13; Found: C, 63.35; H, 5.97, N, 17.01.
General Procedure for the Preparation of 1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2'-phenyl-3'-(3''- or 4''-R3-phenyl)isoxazolidines (10-14).
A mixture of 2 (0.45 g, 2.4 mmol) and the appropriate N-(3- or 4-R3-benzylidene)-aniline N-oxide (5-9) (5.3 mmol) in anhydrous THF (15 mL) was refluxed for 7 h, and then, the resulting solution was allowed to stand overnight before workup according to procedures A or B.
Procedure A: The precipitate thus obtained was separated by suction, washed successively with anhydrous THF (6 mL) and anhydrous acetone (10 mL) and dried to give the products 10 and 11, respectively.
1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2',3'-diphenylisoxazolidine (10). Yield 50%; mp 173-174°C (dec.); IR (KBr): 3155, 3100, 3060, 2865, 2830, 1660, 1595, 1570, 1475, 1460, 1280 cm-1; 1H-NMR (500 MHz, DMSO-d6) δ: 3.03 (dd, JA,M = 13.67 Hz, JA,X = 10.25 Hz, 1H, HA-4'), 3.35-3.39 (m, 1H, CH2), 3.42-3.47 (m, 1H, HM-4'), 3.50-3.53 (m, 1H, CH2), 4.09-4.23 (m, 2H, CH2), 4.88 (dd, JX,A = 10.25 Hz, JX,M = 8.18 Hz, 1H, HX-3'), 6.81-6.96 (m, 6H, Ar), 7.10-7.18 (m, 3H, Ar), 7.31-7.43 (m, 3H, 2H aromat. and NH), 7.56-7.61 (m, 3H, Ar); 13C-NMR (500 MHz, DMSO-d6) δ: 39.54, 44.62, 51.46, 71.93, 91.11, 117.09, 119.57, 121.18, 121.89, 122.74, 124.39, 127.16, 127.76, 127.91, 128.57, 129.02, 129.63, 129.81, 140.33, 146.01, 149.28, 156.67; Anal. Calcd. for C24H22N4O: C, 75.37; H, 5.80; N, 14.65; Found: C, 75.18; H, 5.48; N, 14.19.
1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2'-phenyl-3'-(4''-methylphenyl-isoxazolidine (11). Yield 54%; mp172-173°C (dec.); IR (KBr): 3165, 3050, 3025, 2970, 2920, 2850, 1650, 1595, 1490, 1475, 1460, 1280 cm-1; 1H-NMR (200 MHz, DMSO-d6) δ: 2.31 (s, 3H, CH3), 3.01 (dd, JA,M = 13.56 Hz, JA,X = 10.34 Hz, 1H, HA-4'), 3.43-3.63 (m, 3H, HM-4' and CH2), 4.10-4.25 (m, 2H, CH2), 4.83 (dd, JX,A = 10.34 Hz, JX,M = 8.52 Hz, 1H, HX-3'), 6.80-7.01 (m, 6H, Ar), 7.12-7.23 (m, 5H, 4H Ar and NH), 7.48-7.59 (m, 3H, Ar); Anal. Calcd. for C25H23N4O: C, 75.93; H, 5.86; N, 14.17; Found: C, 76.21; H, 6.11; N, 13.91.
Procedure B: The insoluble side products were filtered off and then the solvent was distilled off under reduced pressure. The residue was treated with anhydrous acetone (7 mL), and the precipitate thus obtained was collected by filtration, washed with acetone (5 mL) and dried to give the products 12–14.
1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2'-phenyl-3'-(4''-chlorophenyl)isoxazolidine (12). Yield 58%; mp172-175°C (dec.); IR (KBr): 3160, 3100, 3030, 2870, 1660, 1595, 1570, 1490, 1480, 1460, 1280 cm-1; 1H-NMR (200 MHz, DMSO-d6) δ: 3.01-3.09 (m, 1H, H-4'), 3.45-3.58 (m, 3H, H-4' and CH2), 4.11-4.25 (m, 2H, CH2), 4.95-5.00 (m, 1H, H-3'), 6.79-7.23 (m, 9H, Ar), 7.53-7.67 (m, 5H, 4HAr and NH); Anal. Calcd. for C24H21ClN4O: C, 69.14; H, 5.08; N, 13.44; Found: C, 69.27; H, 4.92; N, 13.59.
1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2'-phenyl-3'-(3''-chlorophenyl)isoxazolidine (13). Yield 67%; mp 158-161°C; IR (KBr): 3160, 3095, 3060, 3025, 2885, 2865, 1660, 1595, 1570, 1485, 1475, 1460, 1280 cm-1; 1H-NMR (200MHz, DMSO-d6) δ: 3.04 (dd, JA,M = 13.56 Hz, JA,X = 10.34 Hz, 1H, HA-4'), 3.29-3.56 (m, 3H, HM-4' and CH2), 4.13-4.26 (m, 2H, CH2), 5.00 (dd, J = 10.34 Hz, J = 9.52 Hz, 1H, HX-3'), 6.79-7.01 (m, 5H, Ar), 7.12-7.23 (m, 3H, Ar), 7.39-7.64 (m, 5H, 4H Ar and NH); Anal. Calcd. for C24H21ClN4O: C, 69.14; H, 5.08; N, 13.44; Found: C, 68.96; H, 5.16; N, 13.62.
1,2,3,5-Tetrahydroimidazo[2,1-b]quinazolin-5,5'-spiro-2'-phenyl-3'-(4'-methoxyphenyl)isoxazolidine (14). Yield 68%; mp 158-160°C (dec.); IR (KBr): 3160, 3100, 3030, 2880, 2830, 1650, 1595, 1570, 1510, 1490, 1460, 1285, 1250 cm-1; 1H-NMR (200 MHz, DMSO-d6) δ: 3.04-3.12 (m, 1H, H-4'), 3.40-3.53 (m, 3H, H-4' and CH2), 3.78 (s, 3H, OCH3), 4.13-4.22 (m, 2H, CH2), 4.78-4.87 (m, 1H, H-3'), 6.79-7.00 (m, 7H, Ar), 7.01-7.23 (m, 3H, Ar), 7.39 (s, 1H, NH), 7.50-7.66 (m, 3H, Ar); Anal. Calcd. for C25H24N4O2: C, 72.79; H, 5.86; N, 13.58; Found: C, 72.63; H, 6.11; N, 13.22.
General Procedure for the Preparation of Dimethyl 2-(7-R1-8-R2-2,3-dihydroimidazo[2,1-b]-quinazol-ine-5(1H)-ylidenemethyl)but-2-enedioates (15, 16).
To a suspension of the appropriate 5-methylidene-1,2,3,5-tetrahydroimidazo[2,1-b]quinazoline (2 or 4) (1.4 mmol) in anhydrous methanol (20 mL) DMAD (0.23 g, 1.6 mmoles) was added. The resulting mixture was stirred at room temperature for 15 h, and then the crude product (15 or 16) was separated by suction, washed with methanol (4 mL), dried and purified by crystallization from anhydrous methanol.
Dimethyl 2-(2,3-dihydroimidazo[2,1-b]quinazolin-5(1H)-ylidenemethyl)but-2-enedioate (15). Yield 0.2 g (38%); mp 183-184°C; IR (KBr): 3170, 3115, 3020, 2880, 1715, 1700, 1665, 1600, 1580, 1460, 1250, 1180 cm-1; 1H-NMR (500 MHz, CDCl3) δ: 3.37 (s, 3H, OCH3), 3.76 (s, 3H, OCH3), 3.79-3.82 (m, CH2), 3.99-4.03 (m, CH2), 6.21 (s, 1H, HC=C), 6.29 (d, J = 1.47 Hz, 1H, HC-CO), 6.91-7.00 (m, 2H, H-7, -9), 7.25-7.31 (m, 2H, H-6, -8), 7.58 (br s, 1H, NH); 13C-NMR (500 MHz, CDCl3) δ: 39.76, 45.76, 51.64, 52.70, 93.02, 118.54, 119.18, 121.95, 123.20, 128.09, 131.70, 143.67, 146.16, 146.84, 155.07, 167.41, 168.77; Anal. Calcd. for C17H17N3O4: C, 62.38; H, 5.23; N, 12.84; Found: C, 62.01; H, 5.17; N, 12.60.
Dimethyl 2-(7,8-dimethoxy-2,3-dihydroimidazo[2,1-b]quinazolin-5(1H)-ylidenemethyl)-but-2-ene-dioate (16). Yield 0.31 g (57%); mp 215-216°C (dec.); IR (KBr): 3165, 3110, 3040, 2945, 2890, 2840, 1725, 1700, 1660, 1615, 1565, 1440, 1260, 1210, 1180 cm-1; 1H NMR (200 MHz, CDCl3) δ: 3.43 (s, 3H, OCH3), 3.73-3.81 (m, 5H, CH2 and OCH3), 3.84 (s, 3H, OCH3), 3.90 (s, 3H, OCH3), 4.01-4.09 (m, 2H, CH2), 6.16 (s, 1H, HC=C), 6.24 (s, 1H, HC-CO), 6.60 (s, 1H, H-9), 6.77 (s, 1H, H-6); Anal. Calcd. for C19H21N3O6: C, 58.91; H, 5.46; N, 10.85; Found: C, 58.79; H, 5.28; N, 10.51.
1-(1-Acetyl-2,3-dihydroimidazo[2,1-b]quinazolin-5(1H)-ylidene)acetone (17).
To a suspension of 2 (0.53 g, 3 mmol) in pyridine (10 mL) acetyl chloride (0.71 g, 9 mmol) was added at 0 ºC. After an exothermic reaction had subsided (15 min), the reaction mixture was stirred at rt for 6 h. Then, the precipitate was collected by filtration, dried, and treated with water (10 mL). The mixture thus obtained was made alkaline to pH 9 with 5% aqueous Na2CO3 and filtered off to give the crude product 17 as a mixture of two isomers (ZZ/EZ) which was purified by crystallization from DMF. Yield 0.43 g (56%); mp 202-205 °C; Anal. Calcd. for C15H15N3O2: C, 66.90; H, 5.61; N, 15.60; Found: C, 66.78; H, 5.42; N, 15.62; IR (KBr): 3060, 2920, 1680, 1645, 1635, 1600, 1560, 1520, 1480, 1470, 1410, 1380, 1230, 1150 cm-1; 1H-NMR (500 MHz, CDCl3): ZZ/EZ isomer ratio 3.3:1, respectively: δ: 2.30 (s, 3H, OCH3), 2.77 (s, 3H, OCH3), 3.85-3.89 (m, 0.92H, CH2CH2 imidaz., EZ isomer), 4.07-4.20 (m, 3.08H, CH2CH2 imidaz., ZZ isomer), 5.22 (br s, 0.23H, CH, EZ isomer), 6.13 (s, 0.77H, CH, ZZ isomer), 7.22-7.40 (m, 2H, H-7, -9), 7.49-7.57 (m, 1H, H-8), 7.84 (s, 0.77H, H-6, ZZ isomer), 9.03 (br s, 0.23H, H-6, EZ isomer); ZZ/EZ isomer ratio 1:1.1, respectively: δ: 2.21 (s, 3H, OCH3), 2.64 (s, 3H, OCH3), 3.93-3.97 (m, 4H, CH2CH2 imidaz.), 5.38 (s, 0.52H, CH, EZ isomer), 6.30 (s, 0.48H, CH, ZZ isomer), 7.20-7.31 (m, 2H, H-7, -9), 7.56 (s, 1H, H-8), 8.05 (s, 0.47H, H-6, ZZ isomer), 9.11 (s, 0.53H, H-6, EZ isomer).
2-(1-Benzoyl-2,3-dihydroimidazo[2,1-b]quinazolin-5(1H)-ylidene)-1-phenylethen-1-one (18).
Benzoyl chloride (0.98 g, 7 mmol) was added at 0 ºC to a suspension of 2 (0.43 g, 2.3 mmol) in pyridine (10 mL). After the initial exothermic reaction had subsided (ca. 15 min), the reaction mixture was stirred at rt for 6 h, and then concentrated to dryness under reduced pressure. The residue was treated with 5% aqueous Na2CO3 and extracted with methylene chloride (100 mL). The organic phase was dried over MgSO4 and then the solvent was evaporated under reduced pressure. The dry residue thus obtained was recrystallized from DMF to give 18 (0.42 g, 47%); mp 207-210°C; IR (KBr): 3060, 1666, 1645, 1620, 1595, 1530, 1470, 1400, 1315, 1220 cm-1; 1H-NMR (500 MHz, CDCl3) δ: 4.24-4.29 (m, 4H, 2CH2), 6.74 (br s, 1H, CH), 6.91 (d, J = 7.81 Hz, 1H, Ar), 7.21-7.24 (m, 1H, Ar), 7.40-7.58 (m, 7H, Ar), 7.71-8.04 (m, 5H, Ar); 13C-NMR (500 MHz, CDCl3) δ: 43.27, 46.90, 91.16, 119.07, 123.77, 125.71, 127.01, 127.58, 128.24, 128.35, 129.01, 131.61, 131.70, 132.68, 134.36, 140.33, 143.72, 146.89, 148.28, 169.37, 186.82; MS (70 eV) m/z 393.1 (M+ 100%); Anal. Calcd. for C25H19N3O2: C, 76.32; H, 4.87; N, 10.68; Found: C, 76.05; H, 4.61; N, 10.70.
1-Mesyl-5H-5-mesylomethylidene-2,3-dihydroimidazo[2,1-b]quinazoline (19).
To a suspension of 2 (1 g, 5.4 mmol) in pyridine (10 mL) MsCl (1.85 g, 16.2 mmol) was added at 0 ºC. After the exothermic reaction had subsided (15 min), the reaction mixture was stirred at rt for 72 h. Then, the precipitate was collected by filtration and treated with water (10 mL). The solution thus obtained was made alkaline to pH 9 with 5% aqueous Na2CO3 and the solid that precipitated was filtered off to give the crude product 19 as a mixture of two isomers (Z/E) in a ratio 1:9, which was purified by crystallization from methanol. Yield: 0.7 g (38%); mp 220-221°C (dec.); IR (KBr): 3010, 2920, 1640, 1600, 1565, 1540, 1480, 1420, 1350, 1280, 1260, 1160 cm-1; 1H-NMR (200 MHz, DMSO-d6) δ: 3.17 (s, 3H, CH3), 3.35 (s, 3H, CH3), 3.90-4.08 (m, 3.6H, CH2CH2 imidaz., E isomer), 4.46-4.62 (m, 0.4H, CH2CH2 imidaz., Z isomer) 5.55 (s, 0.9H, CH, E isomer), 6.45 (s, 0.1H, CH, Z isomer), 7.32-7.60 (m, 3H, H-7, -8, -9), 7.98 (d, J = 5.35 Hz, 0.1H, H-6, Z isomer), 8.59 (d, J = 5.45 Hz, 0.9H, H-6, E isomer); Anal. Calcd. for C13H15N3O4S2: C, 45.73; H, 4.40; N, 12.31; Found: C, 45.89; H, 4.26; N, 12.28.
X-Ray Structure Analysis of Compounds 3 and 18 [26].
Crystal data for C14H15N5·H2O (3): monoclinic, space group P21/c, a = 15.952(1), b = 4.995(1), c = 33.246(1) Å, β = 93.61(1), V = 2643.8(6) Å3, Z = 8, dχ = 1.363 g.cm-3, T = 293 K. Data were collected for a crystal with dimensions 0.5 x 0.3 x 0.05 mm on a Kuma KM4 diffractometer using graphite monochromated Cu Kα radiation. Final R indices for 3031 reflections with I>2σ(I) and 368 refined parameters are: R1 = 0.0457, wR2 = 0.1105 (R1 = 0.0867, wR2 = 0.1270 for all 2102 data).
Crystal data for C25H19N3O2 (18): orthorhombic, space group Pna21, a = 11.6591(11), b = 9.8033(9), c = 17.1225(16) Å, V = 1957.1(3) Å3, Z = 4, dχ = 1.335 g.cm-3, T = 293 K. Data were collected for a crystal with dimensions 0.5 x 0.5 x 0.5 mm on a Kuma CCD diffractometer using graphite monochromated Mo Kα radiation. Final R indices for 1879 reflections with I>2σ(I) and 291 refined parameters are: R1 = 0.0376, wR2 = 0.0927 (R1 = 0.0432, wR2 = 0.0969 for all 4437 data).