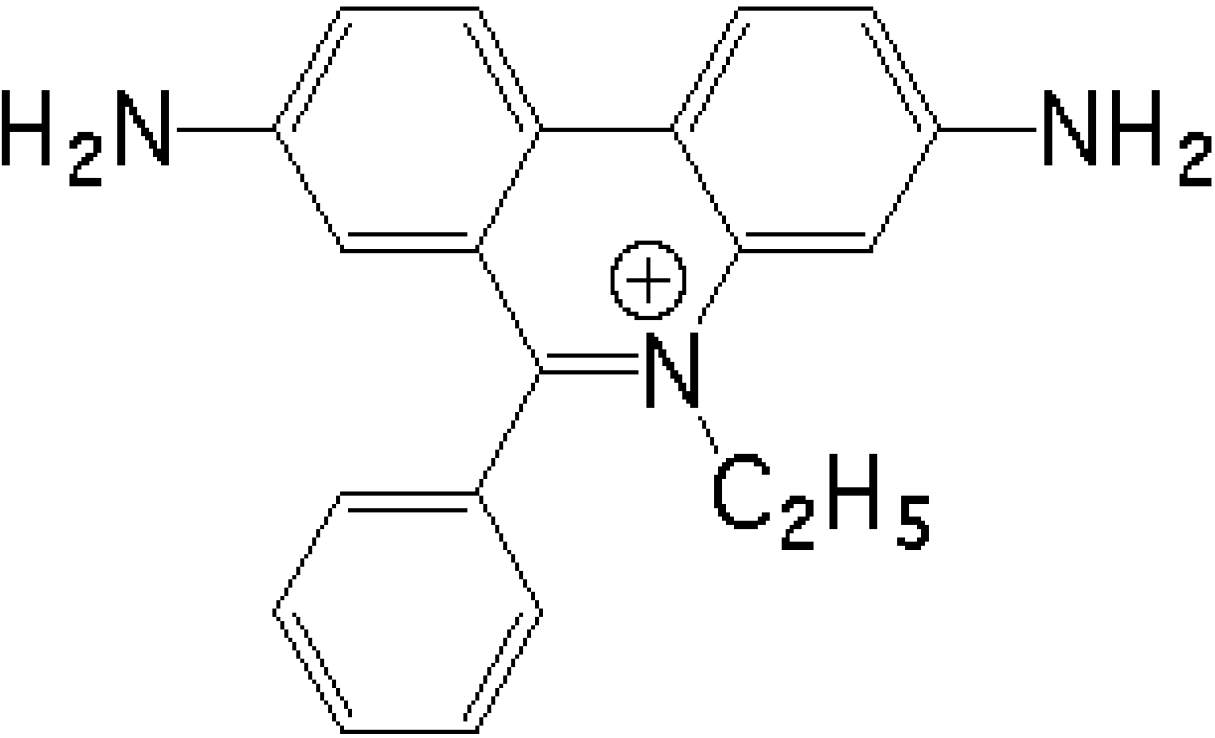
Thermal Denaturation of DNA Aptamers
The thermal denaturation of each aptamer (1.0 μM) was examined by optical melting studies.
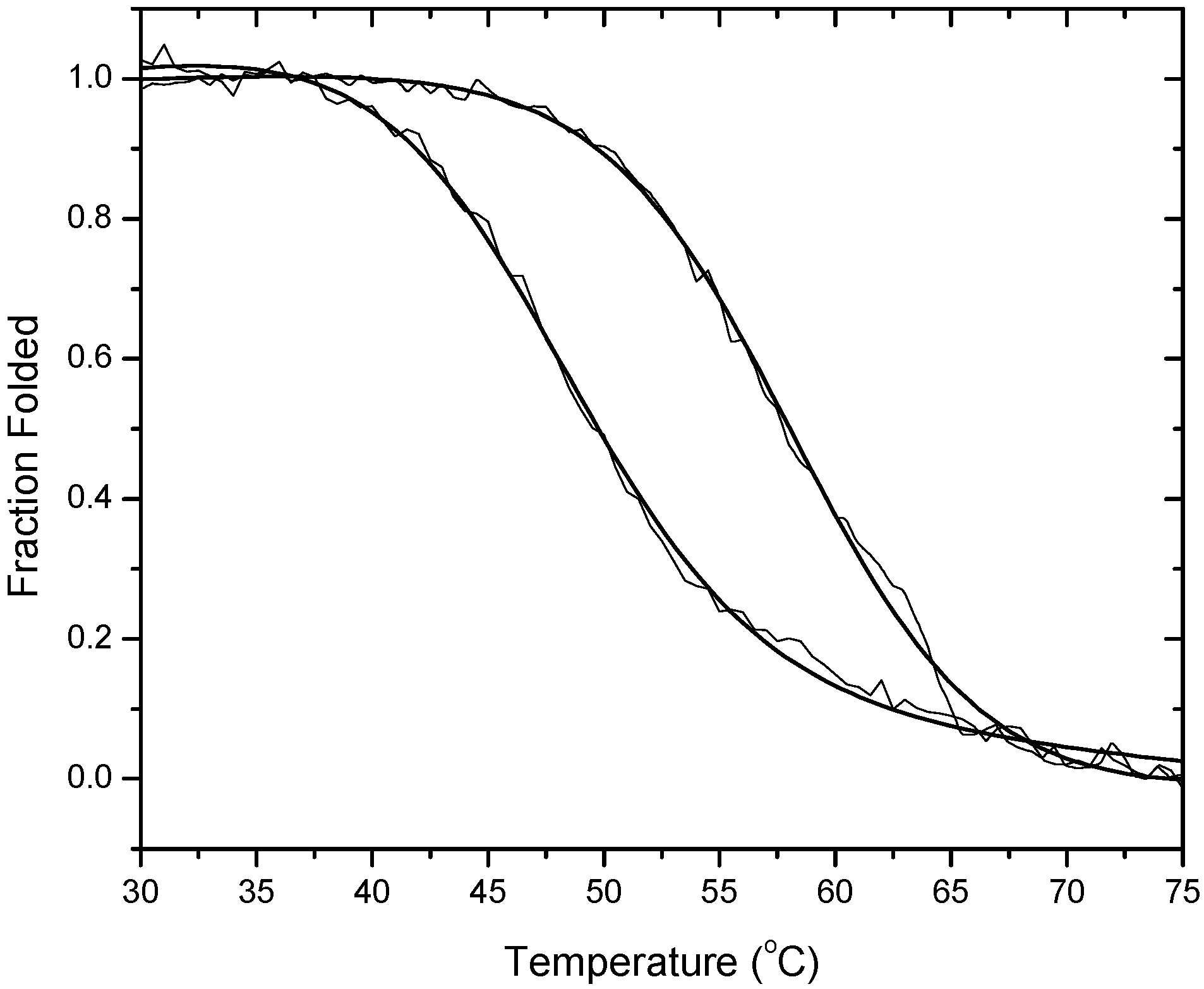
Figure 2 presents results from spectroscopic data obtained at 260 nm. The leftmost curve is that for 1OLD alone. The right most curve is for 1OLD in the presence of a near saturating concentration of ethidium. Each curve is presented as the fraction of total that is folded. The line drawn through each set of raw data is a fit for a single transition model with a correction for a sloping pre- and post-melt baseline (Equation 1).
Figure 2.
Optically detected fractional thermal denaturation of the DNA aptamer 1OLD.
Figure 2.
Optically detected fractional thermal denaturation of the DNA aptamer 1OLD.
The melting data are cast in the form of fraction folded versus temperature for the DNA aptamer 1OLD. Melts were performed in the absence and presence of 10.0 μM ethidium. Similar experiments were performed using 1DB6 and 2ARG (data not shown).
Table 2 presents tabulated results from the Tm’s and denaturation enthalpies (ΔH) for each DNA aptamer. In the absence of any ligands, the measured Tm and enthalpy describing the thermal transition both closely agree with predicted values (
Table 1) [
20,
21,
22]. In each of the aptamers, the melting transition in the absence and presence of ethidium is monophasic but does contain a sloping pre- and post-transition baseline which can be corrected (
Figure 2) [
20]. 1DB6 has the highest predicted and measured Tm (
Table 1 and
Table 2). A trend is observed in which the Tm decreases and ΔH increases with increasing aptamer length (
Table 1 and
Table 2).
Ethidium binding shifts the Tm of each aptamer to a higher temperature (
Table 2). The most pronounced Tm shift was for 1OLD. In that case a ΔTm of 10.6
oC was observed (
Table 2:
Figure 2). In 1DB6 and 2ARG the ΔTm’s were 5.2 and 5.3 respectively. Ethidium increased the enthalpic stability of 1DB6 and 1OLD by 2.6 and 2.8 kcal/mole respectively. In contrast ethidium binding to 2ARG decreased the enthalpy by nearly 4 kcal/mole. When melting studies are performed on each aptamer (1.0 μM) in the presence of 10.0 μM ethidium and with added 10.0 mM L-arginiamide, the apparent Tm’s are not appreciably shifted from those values presented in
Table 2 (data not shown) in the presence of ethidium. The ΔH’s for 1DB6, 1OLD and 2ARG were found to be decreased to 47.4
+ 0.3, 49.8
+ 0.8, and 44.4
+ 1.2 kcal/mol respectively.
Table 2.
Effect of ethidium binding on the fitted thermal denaturation parameters for L-argininamide binding DNA aptamers.
Table 2.
Effect of ethidium binding on the fitted thermal denaturation parameters for L-argininamide binding DNA aptamers.
| | Aptamer Minus Ethidiuma | Aptamer Plus Ethidiumb |
|---|
Tmc,d
(oC) | ΔHc,d
(kcal/mol) | Tmc,d
(oC) | ΔHc,d
(kcal/mol) |
|---|
| 1DB6 | 59.4
+ 0.2 | 46.2
+ 1.5 | 64.6
+ 0.3 | 48.8
+ 0.2 |
| 1OLD | 47.9
+ 0.1 | 48.9
+ 1.2 | 58.5
+ 0.1 | 51.7
+ 0.5 |
| 2ARG | 47.3
+ 0.4 | 64.1
+ 1.7 | 52.6
+ 0.1 | 60.0
+ 0.7 |
Fluorescence Detected Binding of Ethidium to 1DB6, 1OLD, and 2ARG.
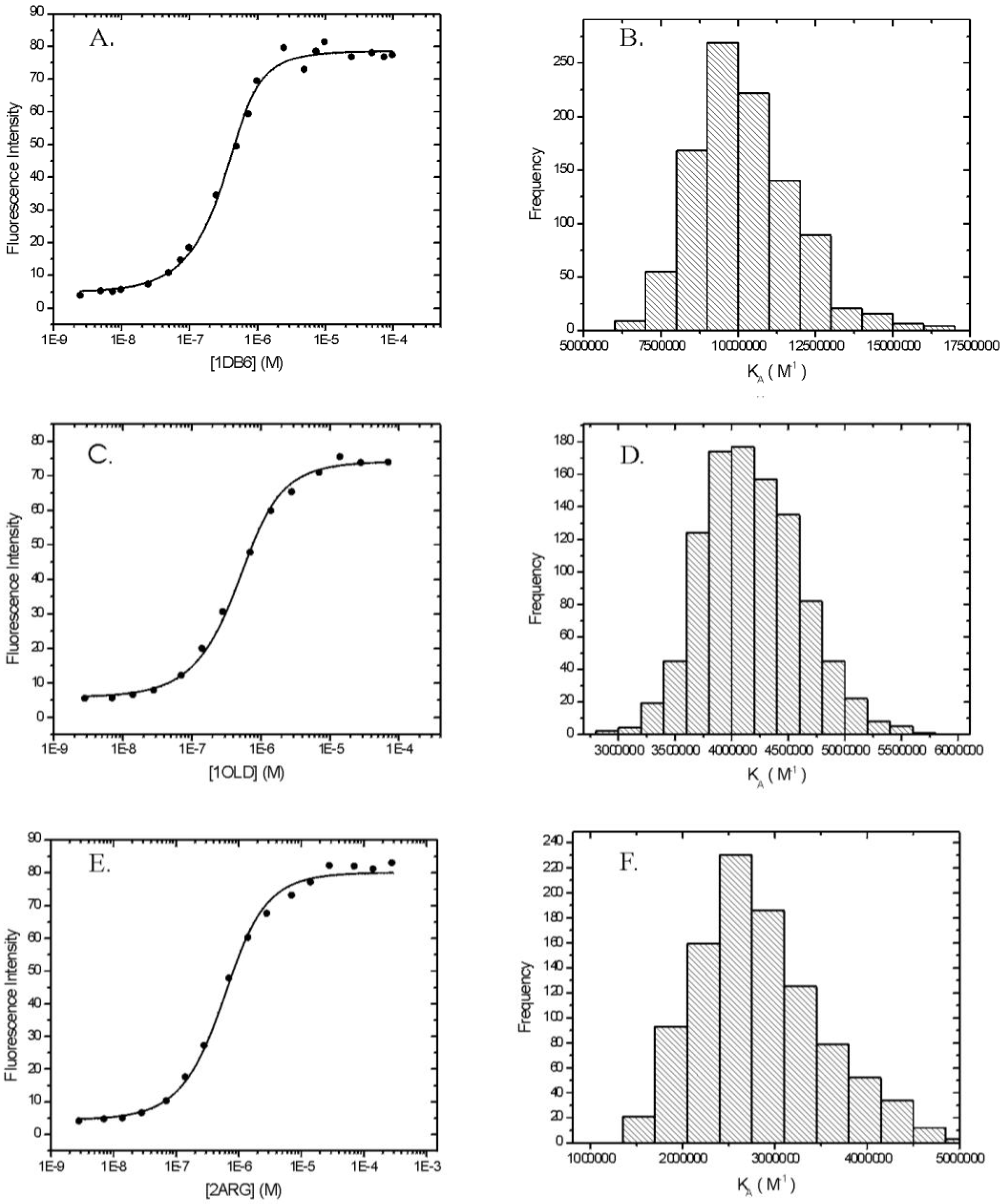
Figure 3 presents the fluorescence emission spectrum of ethidium in the absence (dashed line) and presence (solid line) of a saturating concentration of the DNA aptamer. The emission spectrum of ethidium in the presence of 1DB6 and 2ARG are very similar (data not shown). Ethidium emission intensity increases with increasing aptamer concentration. In
Figure 4 are shown the binding isotherms and Monte Carlo results of fits obtained by plotting the fluorescence intensity at 590 nm extracted from the raw spectral data (e.g.
Figure 3) as a function of the logarithm of the aptamer concentration.
Figure 3.
Fluorescence emission spectra of ethidium in the absence (dashed line) and presence (solid line) of a saturating concentration of the DNA aptamer 1OLD.
Figure 3.
Fluorescence emission spectra of ethidium in the absence (dashed line) and presence (solid line) of a saturating concentration of the DNA aptamer 1OLD.
The binding isotherms for the interaction between ethidium and the DNA aptamers 1DB6, 1OLD, and 2ARG are presented in Panels A, C, and E of
Figure 4, respectively. For each aptamer, the raw fluorescence data are well described by the model for a single-site binding model (Equation 2). When fits are scrutinized by Monte Carlo analysis a rigorous determination of the means and standard deviations of the fitted parameters is possible. The results of these Monte Carlo simulations are shown in
Table 3.
Table 3.
Fitted parameters for the titration of ethidium by L-argininamide binding DNA aptamers.
Table 3.
Fitted parameters for the titration of ethidium by L-argininamide binding DNA aptamers.
| | Ffreea,b,c | Fbounda,b,c | KAa,c
(M-1) | ΔGo d
(kcal/mol) |
|---|
| 1DB6 | 4.91
+ 0.86 | 78.53
+ 0.85 | 10,200,800
+ 1,665,410 | -9.43
+ 1.54 |
| 1OLD | 5.86
+ 0.64 | 74.17
+ 0.76 | 4,180,480
+ 430,510 | -8.91
+ 0.92 |
| 2ARG | 4.55
+ 1.83 | 81.05
+ 2.00 | 2,850,480
+ 710,776 | -8.68
+ 2.16 |
Panels B, D, and F of
Figure 4 are the resulting histograms showing the distribution of possible fitted values of
Keq obtained from Monte Carlo analyses for 1DB6, 1OLD, and 2ARG, respectively. The histograms for the parameters
F0 and
Fb are not shown. In all cases the fitted parameters are nearly Gaussianly distributed and the residuals are randomly distributed indicative of a good fit. The only possible exception is 2ARG which shows some skew in the histogram (Panel F,
Figure 4). When similar data were collected in a different set of experiments, the resulting histogram was normally distributed (data not shown).
Figure 4.
Fluorescence detected binding of ethidium to three L-argininamide recognizing DNA aptamers
Figure 4.
Fluorescence detected binding of ethidium to three L-argininamide recognizing DNA aptamers
Effect of L-Argininamide on the Binding Stoichiometry of Ethidium to 1DB6, 1OLD, and 2ARG
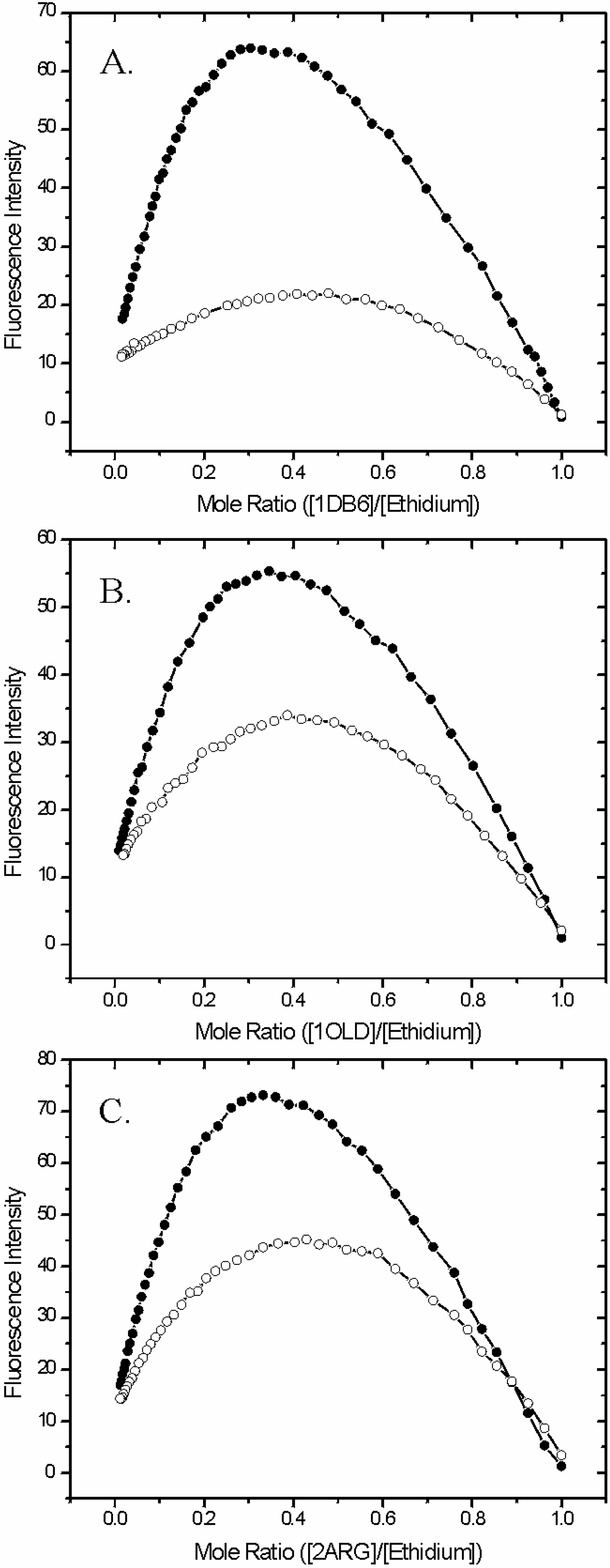
The method of continuous variations was used to assess the quantitative extent of the binding of L-Arm in the loop region of the aptamer and how this influences the binding of ethidium molecules bound in the helical stem region of the same aptamer.
Figure 5 and
Table 4 present results from Job analyses for each of the aptamers under investigation in the absence and presence of L-Arm. In each case the Job plot is shifted to the right which is indicative of fewer ethidiums bound per aptamer unit (
Figure 5). For example, the apex of the curve for 1DB6 in the absence of L-Arm (Panel A of
Figure 5) is 0.30 compared to 0.48 in the presence of a saturating amount of L-Arm. These values equate to binding stoichiometries of ethidium:1DB6 of 2.3:1 and 1.1:1 respectively. In addition, the overall fluorescence intensity taken from the apex of the curve obtained in presence of L-Arm is diminished by a factor of 2.9 of that observed in the absence of the ligand. Taken together, these data are highly suggestive that the binding of the cognate ligand L-Arm is capable of excluding a portion of the ethidium bound to the aptamer. Specifically it is found that approximately 1.2 ethidiums are released from 1DB6 in the presence of L-Arm. Similarily but not as dramatically, the stoichiometry of the ethidium:1OLD complex is found to be 1.9:1 in the absence of L-Arm and 1.6:1 in the presence of the L-Arm ligand. The overall fluorescence intensity observed at the apex of the curve in the presence of L-Arm is, once again, diminished. In this case, however, it is only by a factor of 1.6 compared to that observed in the absence of L-Arm. Thus, the same pattern of ethidium exclusion is observed, albeit less dramatic than that observed for 1DB6. The ethidium:2ARG stoichiometry is similarly found to be 2.0:1 in the absence of L-Arm and 1.3:1 in the presence of the ligand. The fluorescence intensity of the system in the presence of L-Arm is diminished by a factor of 1.6.
Table 4.
Results from the method of continuous variations for the binding of ethidium to the L-argininamide binding DNA aptamers 1DB6, 1OLD, and 2ARG.
Table 4.
Results from the method of continuous variations for the binding of ethidium to the L-argininamide binding DNA aptamers 1DB6, 1OLD, and 2ARG.
| | 1DB6 | 1DB6 +
L-Arm | 1OLD | 1OLD +
L-Arm | 2ARG | 2ARG +
L-Arm |
|---|
| Fluorescence Intensity (Height) | 63.94 | 22.00 | 55.35 | 33.99 | 73.15 | 45.17 |
| Peak Ratio | 2.91 | 1.63 | 1.62 |
| Relative Fluorescence Area | 42.47 | 16.01 | 37.39 | 23.81 | 47.94 | 32.26 |
| Total Fluorescence Area Ratio | 2.65 | 1.57 | 1.49 |
| Width | 0.68 | 0.81 | 0.70 | 0.74 | 0.69 | 0.76 |
| Width Ratio | 0.84 | 0.94 | 0.91 |
| Mole Ratio at Peak | 0.30 | 0.478 | 0.345 | 0.386 | 0.33 | 0.43 |
| Stoichiometry | 2.33 | 1.09 | 1.90 | 1.60 | 2.00 | 1.33 |
| Ethidiums Released | 1.24 | 0.30 | 0.67 |
The original goal of the work presented herein was to probe the structure of DNA aptamers with a known intercalator (ethidium) to determine the helical content present in each aptamer in the absence of any bound ligands. Three main features of the three L-argininamide binding DNA aptamers were identified from the current work. First, the intramolecular folded structures of each aptamer contain a discernable but variable helical stem structure in the absence of any bound ligands. Secondly, this helical region is capable of binding the heterocycle ethidium in a stoichiometry of approximately 2:1 in each of the aptamers studied. Of further note is the finding that the affinity of ethidium for each aptamer is between 3-4 kcal/mol more favorable than the affinity of the cognate ligand L-argininamide for the aptamer 1OLD [
23]. Thirdly, a linkage exists between the binding of ethidium in the helical region and the binding of L-Arm in the disordered loop. Binding of L-argininamide is capable of displacing between 0.3-1.2 ethidiums from the relevant aptamer.
Thermal denaturation results (
Table 2 and
Figure 2) demonstrate several features regarding the stability of each DNA aptamer studied. First each aptamer exhibits a Tm that is consistent with that predicted by the
Mfold algorithm. The measured Tm’s decrease as aptamer length increases (
Table 2). Of interest is the increase in the ΔH term with increasing aptamer length which is opposed to that predicted for each sequence by
Mfold (
Table 1). This is likely due to contributions from the apparently disordered L-argininamide binding site positioned in the loop region of the aptamer. The correlation of the ΔH with the L-argininamide binding site is consistent with other results collected in our laboratory which demonstrate that L-argininamide binding has little effect on the Tm of the optically observed melting transition of each aptamer but does decrease the ΔH of the thermal transition [
23].
Figure 5.
Effect of L-argininamide on the binding stoichiometry between ethidium and the L-argininamide binding DNA aptamers. Panels A, B, and C are job plots for 1DB6, 1OLD, and 2ARG respectively. In each case, Job plots were determined in the absence (solid circles) and presence (open circles) of 6.0 mM L-argininamide.
Figure 5.
Effect of L-argininamide on the binding stoichiometry between ethidium and the L-argininamide binding DNA aptamers. Panels A, B, and C are job plots for 1DB6, 1OLD, and 2ARG respectively. In each case, Job plots were determined in the absence (solid circles) and presence (open circles) of 6.0 mM L-argininamide.
When melted in the presence of a 10 fold excess of ethidium, the apparent Tm of each aptamer (1DB6, 1OLD, and 2ARG) increases by approximately 5-10
oC (
Table 2). The largest ethidium dependent stabilization is in 1OLD. The other aptamers have more subtle increases in Tm, and this is likely due to the possibility that we may not have saturated all of the ethidium binding sites. This is very likely given the positioning of the 5’-Pyrimidine-Purine-3’ steps in the helical region of each aptamer (underlined in
Table 1). Clearly 1OLD has a high affinity binding site positioned in the middle of the complementary region. In contrast both 1DB6 and 2ARG exhibit each of their predicted higher affinity binding sites near the end and in (or near) the stem-loop junction.
Previously, we have demonstrated that the optically determined transition observable near 50
oC is attributable to the helical stem structure and not the ligand binding loop region in each of the three aptamers analyzed [
23]. These results along with the lack of additional melting transitions in the presence of ethidium suggest that the heterocycle is, in fact, binding to and stabilizing, the helical stem region of the aptamer. These results do not necessarily exclude the possibility of ethidium binding to the loop region [
8,
9], but it does suggest that ethidium binding to the loop region is a minor binding component. The Marky lab has reported binding constants for the interaction of ethidium with DNA hairpins containing variable amounts of successive poly(dT) in the hairpin region. No such sequence exits in any of the aptamers examined here [
8,
9]. In their experiments, binding free energies (ΔG
o) in the range of -8.6 kcal/mol for binding of ethidium to the stem region of a hairpin were observed. Our results are consistent with their findings (
Table 3).Together, these binding free energies (~ -8.6 kcal/mol) are more favorable by nearly 2 kcal/mol than that observed for the interaction of ethidium with calf thymus DNA (-6.7 kcal/mol) [
24]. It is not readily obvious why the binding of ethidium to oligonucleotides containing hairpins and stem-loop structures is so much more favorable than for natural duplex DNA. One certain difference is that the calf thymus DNA experiments were performed in the presence of 200 mM NaCl compared to 50 mM NaCl in our experiments [
24]. Ionic interactions make favorable contributions (i.e., approximately -5 kcal/mol) to the overall free energy for the binding of ethidium to duplex DNA [
24]. Also it is likely that distortions of the helical structure required to accommodate the intercalating ethidium are more unfavorable in duplex polymers. Such distortions contribute unfavorably to the overall binding free energy [
24]. On a related note, since the Tm of 1OLD does not change with total strand concentration, this indicates that the observed Tm is that of the intramolecular helix and is not, therefore, attributable to an intermolecular complex between complementary regions of separate strands (data not shown).
Investigation of the sequences in
Table 1 suggests that there are different potential ethidium binding sites in each aptamer studied. Results from fluorescence titrations and Monte Carlo simulations of fits (
Table 3,
Figure 3 and
Figure 4) demonstrate that ethidium binds rather tightly to each DNA aptamer studied. However, in contrast to our hypothesis that 1OLD would exhibit the tightest binding, it is clear from
Table 3 that 1DB6 exhibits the highest affinity for ethidium. Of considerable interest is the observation that in each aptamer, the affinity for ethidium is larger than the aptamer’s affinity for the ligand L-Arm to which it was selected to bind. In the case of L-Arm binding to 1OLD, the affinity was found to be approximately 5400 M
-1 with a ΔG
0 of -5.0 kcal/mol [
23]. That is 4 kcal/mol less favorable than that observed for the binding of ethidium to 1OLD.
In addition to the above affinity experiments, the stoichiometry of ethidium binding to each DNA aptamer was determined by the method of continuous variations (
Table 4 and
Figure 5) which demonstrated that in each case, approximately 2 ethidiums were bound per aptamer. In those experiments it was observed that the resulting Job plots were very broad for each aptamer studied. This observation holds over a range of total aptamer+ethidium concentrations ranging from 0.5 μM to 20 μM (data not shown). This broadening may be due to binding heterogeneities that are not observable in the binding experiments shown in
Figure 4 where the ethidium concentration is limiting and only high affinity binding sites are populated on the aptamer. As a result of the breadth of the Job plots, it is difficult to determine the top of the curve (i.e., the apex) which is typically determined graphically in Job plots. We tried multiple methods including curve fitting to approximate the top of each curve but found that fits of the data by higher order polynomials had the effect of returning skewed information which tended to either underestimate or overestimate the mole fraction where the top is observed. In the end, we opted to analyze the data by integrating the curve to find its total area height, width, and center (
Table 4). From these we extracted the molar ratio and the stoichiometry for the complex from the center of the curve for each aptamer.
When similar Job plots are generated in the presence of 4-10 mM L-Arm, the maximum overall fluorescence of each plot is significantly diminished and the curve is quantitatively shifted to higher molar ratio of aptamer:ethidium. Combined these data demonstrate that a linkage exists between the binding of ethidium and the binding of L-Arm. It is of some interest that ethidium can be displaced at all by L-Arm given the weak affinity of L-Arm compared to ethidium. However, it may not be suprising when one considers that the binding of L-Arm to 1OLD is accompanied by a large exothermic enthalpy which pays the cost of an unfavorable conformational change that is characterized as a negative entropy [
23]. Thus, in the case of linkage the whole thermodynamic signature must be considered and not just the overall standard state binding free energies (ΔG
o).
Of the three aptamers investigated, 1DB6 shows the largest extent of linkage between ethidium and L-Arm binding with approximately one ethidium being released from the aptamer receptor. This is in contrast to both 2ARG and 1OLD where the number of ethidiums released is fractional. 1OLD shows the smallest number of ethidiums released. This may be explainable since the high affinity ethidium binding site is positioned further away from the stem-loop junction (
Table 1).
In the case of DNA containing hairpins occurring in chromatin, the formation of a hairpin in a single strand of duplex DNA must occur in the presence of its complementary strand. The exact nature of the thermodynamics that link the competitive formation of such intra- and intermolecular complexes is a topic of on-going research [
1,
2,
3,
4,
5]. Recent work by both the Chaires [
25,
26] and Hud [
27] laboratories have separately demonstrated the importance of ligand-induced non-cannonical conformations and structures in model duplex and triplex DNA structures, respectively. Since hairpins have potentially important biological functions, they are interesting and obvious drug targets.