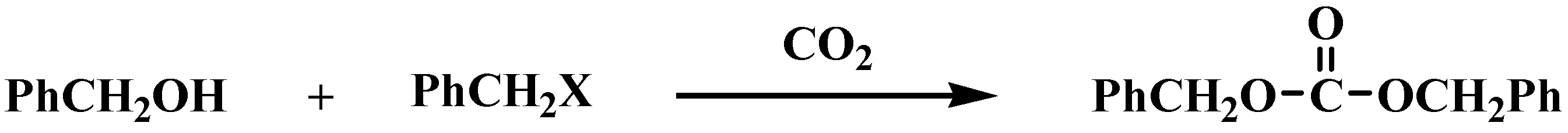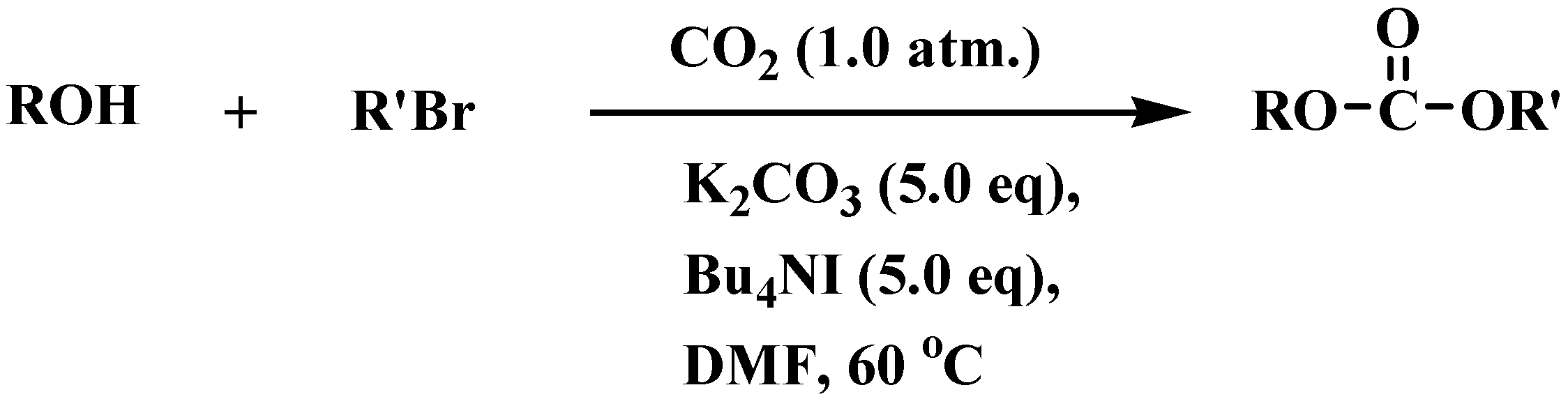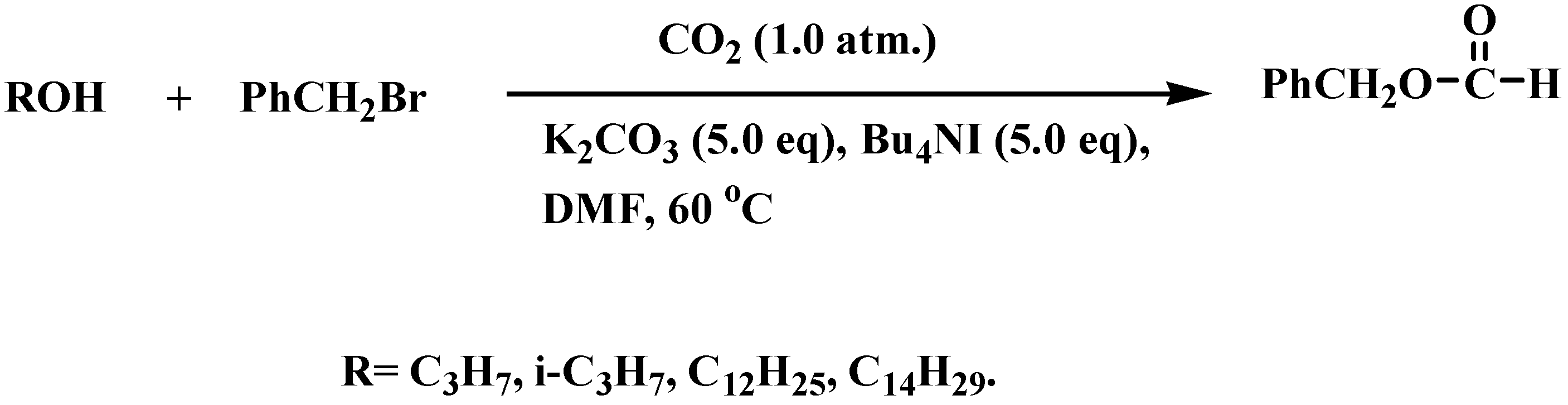General
MPs were obtained with a Yanagimoto micro melting point apparatus and are uncorrected. 1H-NMR spectra were recorded on a Bruker AM-300 spectrometer for solution in CDCl3 with tetramethylsilane (TMS) as internal standard; J-values are in Hz. Mass spectra were recorded with a HP-5989 instrument and HRMS was measured by a Finnigan MA+ mass spectrometer. Organic solvents were dried by standard methods when necessary. Analyzed solid compounds reported in this paper gave satisfactory CHN microanalyses using a Carlo-Erba 1106 analyzer. Commercially obtained reagents were used without further purification. All reactions were monitored by TLC using Huanghai GF254 silica gel coated plates. Flash column chromatography was carried out using 200-300 mesh silica gel.
Representative Experimental Procedure. To benzyl alcohol (216 mg, 2.0 mmol) in anhydrous N,N-dimethylformamide (10 mL) were added potassium carbonate (1.38 g, 10 mmol, 5.0 eq.) and tetrabutylammonium iodide (3.7 g, 10.0 mmol, 5.0 eq.). Carbon dioxide gas was flushed into the reaction mixture three times, and then alkyl bromide (10 mmol, 5.0 equiv.) was added into suspension. The reaction was carried out under carbon dioxide atmosphere for 48 h, during which time the starting material was consumed. The reaction mixture was then poured into water (30 mL) and extracted with EtOAc (3 x 30 mL). The organic layer was washed with water (2 x 30 mL) and brine (30 mL) and dried over anhydrous sodium sulfate. Evaporation of solvent followed by flash column chromatography (eluent: hexane-EtOAc, 20:1) afforded the carbonate as a colorless oil.
Carbonic acid dibenzyl ester (PhCH2OCO2CH2Ph). A colorless liquid; 277 mg, 60%; IR (CHCl3) ν 1745 cm-1 (C=O); 1H-NMR: δ 5.17 (4H, s, CH2), 7.35-7.41 (10H, m, Ph); MS (EI) m/z 197 (M+-45); [Calc. for C15H14O3: requires M, 242.0943. Found: 243.1024 (M+)].
Dibenzyl ether (PhCH2OCH2Ph). A colorless liquid; 217 mg, 16%; IR (CHCl3) ν 1600, 1445 cm-1; 1H-NMR: δ 4.56 (4H, s, CH2), 7.25-7.39 (10H, m, Ph); MS (EI) m/z 197 (M+-1); [Calc. for C14H14O: requires M, 198.1045. Found: 198.1041 (M+)].
Carbonic acid benzyl ester hexadecyl ester (PhCH2OCO2C16H33). A colorless liquid; 543 mg, 76%; IR (CHCl3) ν 1737 cm-1 (C=O); 1H-NMR: δ 0.90 (3H, t, J = 6.9 Hz, CH3), 1.22-1.33 (28H, m, CH2), 1.51-1.60 (2H, m, CH2), 4.17 (2H, t, J = 6.7 Hz, CH2), 5.12 (2H, s, CH2), 7.37-7.42 (5H, m, Ph); MS (EI) m/z 152 (M+-224); [Calc. for C24H40O3: requires M, 376.2979. Found: 376.2966 (M+)].
Carbonic acid benzyl ester butyl ester (n-BuOCO2CH2Ph). A colorless liquid; 221 mg, 56%; IR (CHCl3) ν 1718 cm-1 (C=O); 1H-NMR: δ 0.90 (3H, t, J = 6.9 Hz, CH3), 1.37-1.44 (2H, m, CH2), 1.64-1.69 (2H, m, CH2), 4.12 (2H, t, J = 6.7 Hz, CH2), 5.18 (2H, s, CH2), 7.36-7.42 (5H, m, Ph); MS (EI) m/z 208 (M+); [Calc. for C12H16O3: requires M, 208.1100. Found: 208.1105 (M+)].
Carbonic acid benzyl ester hexadecyl ester [CH3(CH2)5CH2OCO2CH2Ph]. A colorless liquid; 301 mg, 60%; IR (CHCl3) ν 1716 cm-1 (C=O); 1H-NMR: δ 0.89 (3H, m, CH3), 1.29-1.32 (8H, m, CH2), 1.66 (2H, m, CH2), 4.15 (2H, t, J = 6.0 Hz, CH2), 5.16 (2H, s, CH2), 7.35-7.45 (5H, m, Ph); MS (EI) m/z 250 (M+); [Calc. for C15H22O3: requires M, 250.1570. Found: 250.1576 (M+)].
Carbonic acid benzyl ester cyclopentyl ester [PhCH2OCO2(CH2)5]. A colorless liquid; 167 mg, 40%; IR (CHCl3) ν 1736 cm-1 (C=O); 1H-NMR: δ 1.56-1.93 (8H, m, CH2), 5.01-5.10 (1H, m, CH), 5.12 (2H, s, CH2), 7.31-7.45 (5H, m, Ph); MS (EI) m/z 219 (M+-1); [Calc. for C13H16O3: requires M, 220.1100. Found: 220.1094 (M+)].
Carbonic acid benzyl ester 3-methylbutyl ester [PhCH2OCO2CH2CH2CH(CH3)2]. A colorless liquid; 219 mg, 52%; IR (CHCl3) ν 1742 cm-1 (C=O); 1H-NMR: δ 0.94 (6H, d, J = 6.7 Hz, CH3), 1.59 (2H, q, J = 6.9 Hz, CH2), 1.69-1.76 (1H, m, CH), 4.20 (2H, t, J = 6.9 Hz, CH2), 5.18 (2H, s, CH2), 7.34-7.42 (5H, m, Ph); MS (EI) m/z 222 (M+); [Calc. for C13H18O3: requires M, 222.1256. Found: 222.1244 (M+)].
Carbonic acid benzyl ester octyl ester (n-C8H17OCO2CH2Ph). A colorless liquid; 301 mg, 60%; IR (CHCl3) ν 1745 cm-1 (C=O); 1H-NMR: δ 0.88 (3H, t, J = 6.9 Hz, CH3), 1.27-1.43 (10H, m, 5CH2), 1.62-1.69 (2H, m, CH2), 4.15 (2H, t, J = 6.9 Hz, CH2), 5.16 (2H, s, CH2), 7.33-7.41 (5H, m, Ar); MS (EI) m/z 264 (M+); [Calc. for C16H24O3: requires M, 264.1726. Found: 264.1725 (M+)].
Carbonic acid benzyl ester hexyl ester (n-C6H13OCO2CH2Ph). A colorless liquid; 300 mg, 67%; IR (CHCl3) ν 1744 cm-1 (C=O); 1H-NMR: δ 0.88-0.90 (3H, m, CH3), 1.31-1.41 (6H, m, CH2), 1.61-1.71 (2H, m, CH2), 4.10- (2H, t, J = 6.9 Hz, CH2), 5.16 (2H, s, CH2), 7.33-7.41 (5H, m, Ar); MS (EI) m/z 236 (M+); [Calc. for C14H20O3: requires M, 236.1412. Found: 236.1372 (M+)].
Carbonic acid benzyl ester 3-phenylpropyl ester [Ph(CH2)3OCO2CH2Ph]. A colorless liquid; 291 mg, 69%; IR (CHCl3) ν 1723 cm-1 (C=O); 1H-NMR: δ 1.98-2.07 (2H, m, CH2), 2.71-2.78 (2H, m, CH2), 4.17-4.23 (2H, m, CH2), 5.19 (2H, s, CH2), 7.21-7.40 (5H, m, Ar); MS (EI) m/z 208 (M+-60); [Calc. for C17H18O3: requires M, 270.1256. Found: 270.1245 (M+)].
Carbonic acid benzyl ester heptyl ester (n-C7H15OCO2C4H9). A colorless liquid; 238 mg, 58%; IR (CHCl3) ν 1744 cm-1 (C=O); 1H-NMR: δ 0.86-0.96 (6H, m, CH3), 1.25-1.50 (10H, m, CH2), 1.60-1.68 (4H, m, CH2), 4.10-4.16 (4H, m, CH2); MS (EI) m/z 215 (M+-1); [Calc. for C12H24O3: requires M, 216.1726. Found: 216.1722 (M+)].
Carbonic acid dibutyl ester (n-C4H9OCO2C4H9). A colorless liquid; 231 mg, 70%; IR (CHCl3) ν 1745 cm-1 (C=O); 1H-NMR: δ 0.88 (6H, t, J = 6.9 Hz, CH3), 1.20-1.40 (4H, m, CH2), 1.50-1.65 (4H, m, CH2), 4.05 (4H, t, J = 6.7 Hz, CH2); MS (EI) m/z 175 (M++1); [Calc. for C9H18O3: requires M, 174.1256. Found: 174.1263 (M+)].
Formic acid benzyl ester [PhCH2OC(O)H]. A colorless liquid; 217 mg, 60%; IR (CHCl3) ν 1724 cm-1 (C=O); 1H-NMR: δ 5.12 (2H, s, CH2), 7.33-7.41 (5H, m, Ar), 8.14 (1H, s, CH); MS (EI) m/z 136 (M+); [Calc. for C8H8O2, M 136.1749: requires C, 70.57; H, 5.92. Found: C, 70.12; H, 6.21].